Back to Journals » Journal of Hepatocellular Carcinoma » Volume 7
Rare Site Hepatocellular Carcinoma Metastasis
Authors Boldo E , Santafe A, Mayol A , Lozoya R, Coret A , Escribano D, Fortea-Sanchis C , Muñoz A, Pastor JC, Perez de Lucia G, Bosch N
Received 17 November 2019
Accepted for publication 25 February 2020
Published 25 March 2020 Volume 2020:7 Pages 39—44
DOI https://doi.org/10.2147/JHC.S238963
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Ahmed Kaseb
Enrique Boldo,1 Ana Santafe,1 Araceli Mayol,1 Rafael Lozoya,1 Alba Coret,1 Diana Escribano,1 Carlos Fortea-Sanchis,1 Andres Muñoz,1 Juan Carlos Pastor,1 Guillermo Perez de Lucia,1 Nuria Bosch2
1Department of Surgery, Consorcio Hospitalario Provincial de Castellon, Castellon, Spain; 2Pathology Department, Consorcio Hospitalario Provincial de Castellon, Castellon, Spain
Correspondence: Enrique Boldo
Servicio de Cirugia, Consorcio Hospitalario Provincial de Castellon, Avenida Dr Clara 19, Castellon 12002 Tel +34-964359700
Fax +34-964354301
Email [email protected]
Introduction: Besides more common sites such as lung or peritoneum, hepatocellular carcinoma (HCC) can metastatize to rare sites. We report herein a new metastatic site of HCC: the nail-bed. We also review other recently reported rare site HCC metastases (RSHM).
Case Report: A 66-year-old woman with a 12-year history of resected-stage IA HCC who later presented lung, spleen and brain metastases treated with surgery, systemic therapies (sorafenib, sunitinib, capecitabine) and radiotherapy. The patient was referred to us because of a painful and rapidly evolving mass in the nail-bed of the left thumb. Biopsy confirmed nail-bed HCC metastasis, and the finger was amputated. The patient died few weeks later.
Conclusion: This case was an opportunity for us to review RSHM. This type of metastasis seems to be an early event, in the context of advanced stage HCC with elevated protein induced by vitamin K absence-II (PIVKA II). The Lee nomogram is useful in detecting patients at high risk of developing RSHM. We would suggest insisting on systemic treatment in these metastatic patients although overall survival after RSHM diagnosis is poor.
Keywords: hepatocellular carcinoma, rare metastasis, surgery, PIVKA II, systemic treatment, nomogram
Introduction
The most frequent hepatocellular carcinoma (HCC) metastases sites are, in descending order, lung, peritoneum, bone, spleen, adrenal gland, brain, pleura and kidneys.1 In addition, unusual locations for HCC metastasis have been reported. In this article, we report a case of HCC metastasis at a location not previously described, the nail-bed. We also aimed to answer the questions raised to us by this case by contrasting it with other rare site HCC metastases cases (RSHM) reported in the last decade.
Case Report
A 66year-old woman was referred from our Medical Oncology department because of a painful subungual mass in the left thumb, which has been growing rapidly over the prior 4 months. On physical exam, an inflammatory mass with hemorrhagic suffusion and nail extrusion was observed (Figure 1).
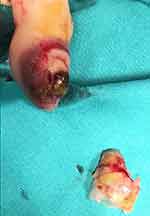 |
Figure 1 Subungual painful mass in the left thumb at presentation. |
The patient's past medical history was relevant for a 10 cm HCC in the right hepatic lobe resected 12 years before. She had no history of transfusions or alcoholic abuse. All hepatitis viral serologies were negative. A pulmonary HCC metastasis was resected two years later. On the path exam, PD-L1 222C3 was negative and MLH1, MSH2, MSH6 and PMS2 were positive. One year later, an unresectable pulmonary local relapse was treated with sunitinib on first line and sorafenib on second line. She subsequently received capecitabine (1800 mg/12h) on progression, with a complete response. Four years later a solitary lung metastasis was resected and the patient received capecitabine as adjuvant therapy. One year later, spleen and brain metastasis were discovered. The patient was treated with regorafenib and whole-brain radiotherapy, with a partial response.
Given her history and the macroscopic appearance the subungual mass (Figure 2), a biopsy was taken after nail resection. Path exam reported HCC metastasis with tumor emboli. The thumb was amputated, and path exam confirmed the diagnosis of HCC metastasis in the nail-bed (Figures 3–5) with intact phalanx bone. The patient died 2 weeks later with seizures. Written informed consent was provided by the patient to have the case details and any accompanying images published. No institutional approval was required to publish the case details, although the Ethics Committee of our center was informed about this submission.
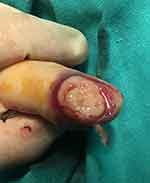 |
Figure 2 Macroscopic appearance the subungual mass: infiltrative soft tumor, paler than normal tissue. |
 |
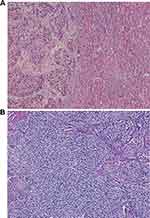
Figure 4 CK7 staining comparison of primary HCC (A) and nail-bed metastasis (B), both negative (10X). |
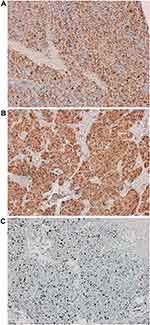 |
Figure 5 Another immunohistochemistry (IHC) features of the nail-bed HCC metastasis: AFP positive (A), glypican 3 positive (B) and Ki67 (C) (10X). |
This case raised several questions: are RSHM observed in initially early-stage HCC cases (like the present one) or in the context of more advanced stage HCCs at presentation? Is PIVKA-II elevated in RSHM? What percentage of these cases have received systemic treatment? Is the interval between primary HCC diagnosis and RSHM long (as in our case) or short? Do RSHM appear in the setting of more common site metastasis? Is the Lee et al nomogram for prediction of extrahepatic HCC metastasis2 useful in these cases? Is the overall survival after RSHM diagnosis always poor? In order to try to answer these questions, we analyzed the RSHM cases published in the last decade together with our case (Table 1).
 |
Table 1 Analysis of the Rare Site Hepatocellular Carcinoma Metastasis (RSHM) Cases Published in the Last Decade |
Discussion
The proposed route for RSHM is the spread of malignant cells via the hepatic veins to caval venous system through the pulmonary circulation and then into arterial vessels.8 Once the malignant cells reach the arterial circulation, HCC metastasis may theoretically be found everywhere. To the best of our knowledge, this is the first report of the nail-bed as a HCC metastatic site. Kim et al17 described a case of a finger HCC metastasis, but it was another cutaneous HCC metastasis report. In their article, Kim et al reviewed six other finger location cases, but they were the more common cutaneous and bone HCC metastasis. Metastasis in the nail-bed are extremely rare. Only three cases can be found in the literature: from a rectal cancer,18 from renal carcinoma19 and from prostate cancer.20
After analyzing the RSHM cases including ours (Table 1), we intended to answer the previous questions.
RSHM are observed mostly in HCCs at advanced stage at presentation (9 out of 14). One possible explanation for the RSHM observed in an early stage (I and II) HCC at presentation might be the presence of histological vascular invasion by the primary tumor. Of the four cases recorded, only Takahashi et al16 reported this finding, whereas it was unknown in the other three.
Elevation of PIVKA II is significantly more frequent in advanced stage HCCs.21 In keeping with that, PIVKA II was elevated in all the RSHM where its value was reported (Table 1).
Less than half of the RSHM reviewed received systemic therapy. Our case suggests that persistence in systemic treatments, even with little expected efficacy (such as capecitabine), may be important for longer survival.
Most of RSHM became clinically apparent within 1 year after the primary tumor diagnosis. In fact, 40% of RSHM were “true” synchronous (present at diagnosis). These facts may suggest that the spread of HCC through this unusual route for the appearance of RSHM seems to be an early event.
Besides, RSHM appear frequently (9 out of 15 cases) in the context of more common site metastasis, especially pulmonary metastasis.
Taken together these facts regarding RSHM presentation (in advanced stage cases, with elevated PIVKA-II, early and when other more usual location metastases are present) suggest a mechanism of early and diffuse systemic metastatic spread. At the same time, these data are contrary to the common notions that RSHM originate from more usual location metastasis in a stepwise fashion. Or that they are the consequence of a longer patient survival.16,22
The Lee nomogram for prediction of extrahepatic metastasis in HCC2 is reliable for RSHM high-risk patients at 1 year (all patients with RSHM). Nevertheless, is less reliable in RSHM low-risk patients (half of them had metastasis at 1 year). At 5 years, its reliability increases, as less RSHM low-risk patients have metastasis and more high-risk patients are affected.
Therefore, we propose that RSHM may be prevented with the earlier use of systemic treatments, especially in patients classified as “high risk” by the Lee nomogram.2
The median overall survival after RSHM diagnosis is only 5 months. RSHM are, as in our case, a final event with an ominous prognosis.
One of the strengths of our report is the awareness created after this new clinical description. All physicians in charge of HCC patients will be much more suspicious for RSHM. Especially for this new possibility, the nail-bed. Another strength is the collection of these rare cases in order to enunciate the main facts of RSHM. The main weakness of our article is the retrospective description and collection, with data lacking for a proper analysis. Obviously, rare cases always prevent from large series that could also power this analysis.
In conclusion, this new site case has allowed us to analyze the characteristics of the RSHM. This type of metastasis seems to be an early event, in the context of advanced stage HCC with elevated PIVKA II. The Lee nomogram is useful in classifying patients at high risk of developing RSHM. We may suggest insisting in systemic treatment in these metastatic patients although overall survival after RSHC diagnosis is poor.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Abbas A, Medvedev S, Shores N, et al. Epidemiology of metastatic hepatocellular carcinoma, a nationwide perspective. Dig Dis Sci. 2014;59(11):2813–2820. doi:10.1007/s10620-014-3229-9
2. Lee CH, Chang CJ, Lin YJ, et al. Nomogram predicting extrahepatic metastasis of hepatocellular carcinoma based on commonly available clinical data. JGH Open. 2018;3(1):38–45. doi:10.1002/jgh3.12102
3. Takaya H, Kawaratani H, Seki K, et al. A patient with hepatocellular carcinoma with isolated right atrial metastases. Intern Med. 2017;56(19):2589–2594. doi:10.2169/internalmedicine.8568-16
4. Orita K, Sakamoto A, Okamoto T, Matsuda S. Solitary muscle metastasis of hepatocellular carcinoma to the biceps femoris muscle with only elevated serum PIVKA-II: a case report. Am J Case Rep. 2019;20:306–309. doi:10.12659/AJCR.913730
5. Hong YM, Yoon KT, Cho M, et al. Bone marrow metastasis presenting as bicytopenia originating from hepatocellular carcinoma. Clin Mol Hepatol. 2016;22(2):267–271. doi:10.3350/cmh.2015.0017
6. Igawa A, Oka S, Tanaka S, et al. Small bowel metastasis of hepatocellular carcinoma detected by capsule endoscopy. Case Rep Gastroenterol. 2013;7(3):492–497. doi:10.1159/000357302
7. Xue LJ, Mao XB, Geng J, Chen YN, Wang Q, Chu XY. Rare gingival metastasis by hepatocellular carcinoma. Case Rep Med. 2017;2017:3192649. doi:10.1155/2017/3192649
8. Mohammed H, Sheikh R, Rahman W, Sheta S, Dogan Z. Undiagnosed hepatocellular carcinoma presenting as nasal metastases. Case Rep Otolaryngol. 2015;2015:856134.
9. Harada JI, Matsutani T, Hagiwara N, et al. Metastasis of hepatocellular carcinoma to the esophagus: case report and review. Case Rep Surg. 2018;2018:8685371.
10. Kang WD, Kim CH, Cho MK, et al. Hepatocellular carcinoma presenting as uterine metastasis. Cancer Res Treat. 2008;40(3):141–144. doi:10.4143/crt.2008.40.3.141
11. Haruki K, Misawa T, Gocho T, et al. Hepatocellular carcinoma with gastric metastasis treated by simultaneous hepatic and gastric resection: report of a case. Clin J Gastroenterol. 2016;9(5):319–323. doi:10.1007/s12328-016-0677-0
12. Traficante D, Assalone P, Tomei F, et al. A case report of HCC cutaneous metastasis. J Gastrointest Oncol. 2014;5(4):E65–7. doi:10.3978/j.issn.2078-6891.2014.021
13. Yu Y-D, Kim D-S, Jung S-W, Lee J-H, Chae Y-S, Suh S-O. Metastatic hepatocellular carcinoma to the parotid gland: case report and review of the literature. Int J Surg Case Rep. 2013;4(1):76–80. doi:10.1016/j.ijscr.2012.08.018
14. Shen Y, Nie L, Yao Y, Yuan L, Liu Z, Lv Y. Seminal vesicle metastasis after liver transplantation for hepatocellular carcinoma: a case report. Medicine (Baltimore). 2019;98(3):e13770. doi:10.1097/MD.0000000000013770
15. Nagao Y, Cho A, Yamamoto H, et al. Hepatocellular carcinoma with pharyngeal metastasis: report of a case. Surg Today. 2008;38(11):1060–1062. doi:10.1007/s00595-008-3790-y
16. Takahashi K, Putchakayala KG, Safwan M, Kim DY. Extrahepatic metastasis of hepatocellular carcinoma to the paravertebral muscle: a case report. World J Hepatol. 2017;9(22):973–978. doi:10.4254/wjh.v9.i22.973
17. Kim JI, Song CH, Gong HS. Finger skin metastasis from hepatocellular carcinoma: a case report. Hand Surg. 2012;17(1):131–134. doi:10.1142/S0218810412720148
18. Drury BJ. Adenocarcinoma of the rectum with metastasis to the nail-bed of the finger. Calif Med. 1959;91(1):35–36.
19. McClees SF, Duncan JR, Ceilley R, Milless TL, Elewski BE. Isolated nail dystrophy: an unlikely presentation of metastatic renal cell carcinoma. Skin Appendage Disord. 2019;5(3):177–180. doi:10.1159/000495121
20. García-Galaviz R, Domínguez-Cherit J, Caro-Sánchez C, Subungual S-RE. Metastasis of an adenocarcinoma of the prostate in a finger. Skin Appendage Disord. 2018;5(1):46–49. doi:10.1159/000489279
21. Yu R, Tan Z, Xiang X, Dan Y, Deng G. Effectiveness of PIVKA-II in the detection of hepatocellular carcinoma based on real-world clinical data. BMC Cancer. 2017;17(1):608. doi:10.1186/s12885-017-3609-6
22. Ayyadurai P, Badipatla KR, Chime C, et al. Cervical spinal cord compression: a rare presentation of hepatocellular carcinoma. Case Reports Hepatol. 2017;2017:8567695. doi:10.1155/2017/8567695
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.