Back to Journals » Cancer Management and Research » Volume 12
Radiotherapy for Cervical Cancer in Patients with Systemic Lupus Erythematosus
Authors Ma J , Wang W , Shen J, Hou X , Lian X, Yan J , Sun S, Miao Z, Meng Q, Hu K , Zhang F
Received 28 May 2020
Accepted for publication 28 August 2020
Published 21 September 2020 Volume 2020:12 Pages 8675—8683
DOI https://doi.org/10.2147/CMAR.S264795
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Seema Singh
Jiabin Ma, Weiping Wang, Jing Shen, Xiaorong Hou, Xin Lian, Junfang Yan, Shuai Sun, Zheng Miao, Qingyu Meng, Ke Hu,* Fuquan Zhang*
Department of Radiation Oncology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Ke Hu; Fuquan Zhang Tel/Fax +86-10-69155482
; +86-10-69155485
Email [email protected]; [email protected]
Objective: To determine the toxicity and efficacy of radiotherapy for cervical cancer in patients with systemic lupus erythematosus (SLE).
Methods: Medical records of patients with SLE who received radiation for cervical cancer from January 2011 to January 2019 were reviewed. For definitive radiotherapy, a dose of 50.4 Gy in 28 fractions was delivered by intensity-modulated radiation therapy (IMRT) combined with high-dose-rate brachytherapy of 28 to 30 Gy in 5 fractions. A dose of 45 to 50.4 Gy in 25– 28 fractions was delivered for postoperative radiation. All patients were in remission with or without a low dosage of prednisone or immunosuppressive medication. Survival data were analyzed with the Kaplan–Meier method. The incidence of severe toxicities among patients was compared with the chi-square test or Fisher’s exact test.
Results: Twelve patients with SLE were included in this study. Definitive radiotherapy was delivered in 7 patients with FIGO (2009) stage II–III disease. Five patients with FIGO I disease received postoperative radiation. The median follow-up time was 22.1 months. The 3-year overall survival (OS) and 3-year progression-free survival (PFS) were 77.8% and 83.3%, respectively. One patient of definitive radiotherapy and one patient of postoperative radiation died due to distant metastasis of cervical cancer. Grade 3 or higher acute and chronic reactions occurred in 58.3% and 8.3% patients, respectively. Acute grade 3 or higher toxicity correlated with the presence of chemotherapy (p = 0.045).
Conclusion: Modern radiotherapy for cervical cancer was well tolerated in SLE patients with remission and provided a favorable outcome.
Keywords: systemic lupus erythematosus, SLE, cervical cancer, intensity-modulated radiation therapy, IMRT, radiotherapy, toxicity
Background
Cervical cancer is the fourth most common cancer in women worldwide,1 for both incidence and mortality. Systemic lupus erythematosus (SLE), a typical representative of connective tissue diseases (CTDs), is a systemic autoimmune disease with various pathogenic factors and multiple organ involvement as well as diverse clinical manifestations and a relapsing and remission course. With the continuous improvement of diagnosis and treatment of SLE, the survival time of lupus has been prolonged significantly. The cause of death of many SLE patients is no longer the progression of primary disease, but the occurrence of some complications. A French study identified 1593 deaths related to SLE, the most common causes of death were cardiovascular diseases (35.7%), neoplasms (13.9%), and infectious diseases (10.3%).2 There has been evidence that SLE patients are at higher risk for HPV infection and cervical dysplasia than the general population with a hazard ratio (HR) of 2.12 (95% CI: 1.65, 2.71).3–5 The odds ratio for the risk of high-grade squamous intraepithelial lesions in patients with SLE was 8.66 (95% CI: 3.75–20.00).5 Concurrent chemoradiotherapy has been well established as the standard treatment for advanced cervical cancer. And radiation also plays an important role in the adjuvant treatment after radical surgery of early-stage cervical cancer. However, the use of radiation has been limited in patients with SLE for the concern of poor tolerance and severe toxicity.
With improvements in medical treatment in recent years, the survival time of SLE is obviously prolonged, while the incidence of tumor has increased due to the use of immunosuppressants in those patients. Therefore, more SLE patients with malignancies are expected to be in need of cancer management including radiotherapy, in the future. Several early studies reported a higher incidence of radiation complications in patients with CTDs.6–10 Whereas recent studies have shown that CTDs did not cause a significant increase in radiation toxicity.11,12 Previous studies on the safety of radiation for patients with CTDs are generally heterogeneous in terms of tumor type, anatomical region, radiation site, radiation dose and therapeutic modalities. Even fewer studies have focused on SLE patients with cervical cancer, except for anecdotal case reports.6 In this study, we reported toxicities and survival outcomes of patients with SLE who received radiotherapy for the treatment of cervical cancer. To our knowledge, no case series have been reported in these patients.
Patients and Methods
Patients
The study reviewed medical records of patients with SLE who received radiotherapy for cervical cancer at Peking Union Medical College Hospital (PUMCH) between January 2011 and January 2019. The PUMCH Institutional Review Board approved this study (Protocol number: S-K1213) and determined that this study was exempt from full IRB review based on the following reason: The study only involved the collection or study of existing data, documents, records, pathological specimens, or diagnostic specimens, and the information was recorded by the investigator in such a manner that subjects could not be identified, directly or through identifiers linked to the subjects. The inclusion criteria were a diagnosis of SLE and treatment with radiation (either definitive or postoperative) for cervical cancer. The diagnosis for SLE was defined by the 1997 American College of Rheumatology (ACR) based on clinical symptoms, serological test results and histopathological findings. The diagnosis of cervical cancer in all cases was confirmed by pathology, and staged by International Federation of Gynecology and Obstetrics (FIGO) 2009. Information on patients’ characteristics, treatment details and survival data was collected.
Radiotherapy
External beam radiotherapy (EBRT) was delivered by intensity-modulated radiation therapy (IMRT). For definitive radiation, whole pelvic radiation using 6-MV photon beam was performed with a dose of 1.8 Gy per fraction five times a week to a total dose of 50.4 Gy, combined with intracavitary brachytherapy at a dose of 28–30 Gy in 5 fractions. Lymph nodes larger than 1 cm in its longest axis were boosted to 60.2 Gy simultaneously. Para-aortic lymph node region was included in patients with metastasis of para-aortic or common iliac lymph nodes. Postoperative radiotherapy to pelvis was prescribed to 45–50.4 Gy in 25 to 28 fractions, with optional brachytherapy at a dose of 10 Gy. Afterloading radiotherapy in all cases was delivered by iridium-192 high-dose-brachytherapy. Image guidance was performed weekly. The detailed treatment approach was described in previous articles.13
Chemotherapy
Weekly cisplatin (40mg/m2) was used as a concomitant chemotherapy regimen for definitive radiation, and chemotherapy was optional in patients with postoperative radiation.
All patients were evaluated for SLE disease activity before the treatment of cervical cancer. Patient may remain medication of prednisone and immunosuppressants. The most frequently used immunosuppressants were hydroxychloroquine, cyclosporine, mycophenolate mofetil and etoricoxib.
Toxicity and Survival Outcomes
Clinical data of treatment-related toxicities were also reviewed. Toxicities were graded retrospectively according to the Common Toxicity Criteria for Adverse Events (CTC-AE) version 4.0. Symptoms that developed within 90 days from the start of radiation were considered as acute toxicities, while those that occurred later than 90 days were considered chronic toxicities. Patients were followed up every 3 months after treatment for 2 years, every 6 months for the next 3 years, and every 1 year thereafter. The follow-up time was calculated from the date of completion of radiotherapy to the date of latest follow-up. Overall survival (OS) was calculated from the completion of radiotherapy to death or the last follow-up. Progression-free survival (PFS) was defined as the time from the completion of radiotherapy to the date of clinically proven disease progression or the date of the last follow-up. Survival curves were constructed using the Kaplan–Meier method. The incidence of severe toxicities among patients with different characteristics was compared with the chi-square test or Fisher’s exact test. Differences were considered significant at p<0.05. All statistical analyses were performed using SPSS version 22.0 (SPSS, Inc., Chicago, IL, USA).
Results
We identified 12 patients with documented SLE and cervical cancer who received radiotherapy in our institute (Tables 1 and 2). All patients were diagnosed with SLE before cervical cancer with a duration of 8.1 years (range, 2.6–18.6 years). The most commonly involved sites were the skin (60%), kidneys (40%), lungs (40%), nervous system (20%), blood (10%) and joints (10%) (Two patients were excluded from the calculation because of missing information). All patients were in quiescent phases of SLE. Definitive concurrent chemoradiation (CCRT) was delivered in 7 patients (58.3%), with 4 cases of FIGO IIB disease and 3 cases of FIGO IIIB disease. Five patients with FIGO I cervical cancer received radical surgery and postoperative radiation. Two of them had concurrent chemotherapy of cisplatin and one of them received a course of chemotherapy before radiation. During radiotherapy, 8 (66.7%) patients remained at a low dosage of prednisone (2.5–12.5 mg daily) to control SLE. The most frequently used immunosuppressants were hydroxychloroquine (50%), cyclosporine (25%), mycophenolate mofetil (8.3%) and etoricoxib (8.3%). During radiation, 7 (58.3%) patients maintained the usage of immunosuppressants including hydroxychloroquine (41.7%) and cyclosporine (16.6%).
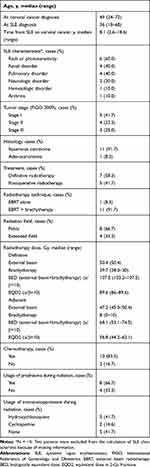 |
Table 1 Characteristics of Patients |
 |
Table 2 Details of Patients |
All patients completed the full course of radiation. The equivalent dose in 2-Gy fractions (EQD2) for tumor (α/β=10) was 89.6 Gy (range, 86.0–89.6 Gy) for definitive radiation and 56.8 Gy (range, 44.2–62.1Gy) for postoperative radiation. Extended-field radiation was delivered in 4 (33.3%) cases.
The median follow-up time was 22.1 months (range, 10.3–102.2 months). The 3-year OS and 3-year PFS were 77.8% and 83.3%, respectively. Two patients died at the last follow-up due to distant metastasis. One of them developed supraclavicular and mediastinal lymph node metastasis after definitive radiation and received an additional course of thoracic radiotherapy. The other patient had pulmonary metastasis after postoperative radiation and received interventional ablation.
A total of 58.3% and 8.3% of the patients had grade 3 or higher acute and chronic reactions, respectively. No grade 5 complications occurred. Ten (83.3%) patients experienced acute hematological toxicity, and 6 (50%) patients had grade 3 or higher toxicity. Grade 3 acute gastrointestinal toxicity occurred in 2 (16.7%) patients. One of them had grade 3 vomiting, and the other developed grade 3 diarrhea. One patient experienced grade 3 acute urinary toxicity. No patient developed grade 4 acute gastrointestinal or urinary effects. Details are listed in Table 3. Chronic hematological toxicity occurred in 10 (83.3%) patients, but only one (8.3%) patient had grade 3 toxicity. No grade 3 or higher chronic gastrointestinal or urinary side effects occurred.
 |
Table 3 Incidence of Acute and Chronic Adverse Events for Patients with SLE |
The association between the incidence of grade 3 or higher acute toxicities and clinical characteristics was analyzed by the chi-square test. Grade 3 or higher acute toxicity correlated with the presence of chemotherapy (p = 0.045). Patients with extended-field radiation had a higher risk of severe acute toxicity than patients with pelvic radiation, though this was not statistically significant (p = 0.081). The associations between other clinical factors (prescribed dose, the diverse organ involvement of SLE, radiation modality) and the risk of severe acute complications were not confirmed in our study (Table 4). Because of the low rate of grade 3 chronic side effects, statistical analysis could not be performed.
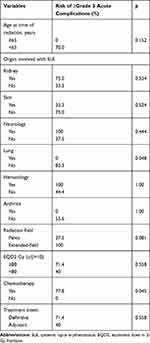 |
Table 4 Incidence of Grade 3 or Higher Acute Toxicities Associated with Clinical Characteristics |
Discussion
The treatment of patients with SLE or other CTDs using radiotherapy has been a challenge for radiologists, in terms of the potentially increased risk of severe radiation toxicity. Radiotherapy might trigger the onset of CTDs by increasing the expression of self-antigens, diminishing regulatory T-cell activity, and activating immunity through Toll-like receptor-dependent mechanisms, all of which could potentially lead to breaks in immune tolerance. The acute and chronic effects of radiation on the skin, soft tissues, and internal organs, including inflammation, edema, vascular changes and fibrosis development, might be additive to the pathophysiologic changes of CTDs, which could lead to significant toxicities14.
A number of anecdotal reports have documented the occurrence of severe complications in patients with CTDs who received radiation.15 An early study by Olivotto documented a case of a patient with SLE who developed fatal pelvic necrosis after radiotherapy for cervical cancer.6 In another study, a patient with SLE who received radiation for mixed Mullerian uterine sarcoma developed radiation-induced bowel necrosis and died of septic complications.11 In contrast to prior studies, a number of other studies published more recently have reported controversial results. In the matched-pair analyses of Phan and Ross, the authors found no significant difference in the incidence of acute or late radiation toxicity of patients with CTDs compared to the general population.11,12 A single-institution retrospective study from Diao et al showed patients with CTDs may be at higher risk of radiation pneumonitis after thoracic radiation for treating intrathoracic malignancy.16 The existing literature has been generally limited by small sample sizes, heterogeneous CTD subtypes, sites of irradiation, treatment modalities, various dosages of radiation and outdated radiation techniques, which have prevented oncologists from forming a firm conclusion about the safety of radiotherapy for patients with CTDs. To reduce the heterogeneity of the above interfering factors, our study chose a homogeneous patient cohort. All patients had received external beam irradiation through IMRT rather than outdated techniques.
As the main prototype of CTDs, SLE is characterized by multiple autoantibodies production, immune complex deposits and multiple organ damage, involving skin, bone, heart, brain, kidney, lung, intestine, liver and blood system injury. For pelvic radiation, common effects of radiotherapy include intestinal toxicity and myelosuppression. Meanwhile, SLE can cause mesenteric vasculitis, intestinal pseudo-obstruction, protein-losing enteropathy, as well as leukopenia, which may be additive with radiation toxicities.17,18 This potential risk has raised the concern about whether patients with SLE can tolerate pelvic radiation well.
The acute grade 3 or higher toxicity occurred in 58.3% of SLE patients, and no acute grade 5 toxicity occurred in our study. Phan reported an analysis of 38 patients with CTDs who were treated with radiation for various malignancies, including 21 patients with SLE. The incidences of grade 2 and grade 3 acute reaction were 49% and 7%, respectively.12 In the study by Pinn including 21 patients with SLE, 21% of patients had acute grade 3 or higher toxicity.19 Compared to those in the literature, we reported a higher incidence of severe acute toxicities among these patients. However, most grade 3 or higher toxicities involved the blood. Except for the hematological effects, the incidence of acute complications was consistent with previous studies. This could possibly be explained by the single radiation site of our study. All patients received pelvic radiation, which strongly affected the hematopoietic function of the bone marrow. Additionally, previous studies commonly included a wide variety of tumor types and a diversity of radiation fields. Concurrent chemotherapy was another contributing factor to hematological toxicity. The incidence of chronic grade 3 or higher toxicity was 8.3% in our study. In previous studies, the incidence was 6.9% to 40%.12,19 The risk of severe chronic toxicities in our study was similar to that in the literature.
We used the chi-square test to identify the potential risk factors for severe acute toxicities in patients with SLE. The data indicated that patients with chemotherapy combined with radiation were at significantly higher risk of severe acute toxicities (p = 0.045). Regarding the radiation field, 37.5% of patients with pelvic radiation had acute grade 3 or higher complications, whereas all patients with extended-field radiation experienced this. This difference approached but did not reach statistical significance (p = 0.081). This was reasonable since chemotherapy as well as extended-field radiation could induce greater myelosuppression, resulting in more severe hematological complications. Due to the limitation in sample size, we did not find significant associations between the clinical characteristics and risk of grade 3 or higher acute effects in patients with SLE.
Nonetheless, some studies indicated that CTD subtype, different organs involvement, radiation dose and the use of immunosuppressants might be risk factors for severe complications from radiation treatment. A prior study by Chen showed that patients with scleroderma had a significantly increased incidence of complications after breast-conserving surgery and radiation therapy, whereas this significance disappeared in patients with rheumatoid arthritis (RA), SLE, polymyositis and Sjogren’s disease.20 Morris reported that patients with RA did not have an elevated rate of late toxicity, while other CTDs were significantly associated with increased radiation late effects.15 Immunosuppressants might be another contributing factor to the toxicities of radiation. Diao found that patients with intrathoracic malignancy who received methotrexate had a higher rate of radiation pneumonitis regardless of whether therapy was discontinued during radiation.16 Additionally, long-term use of steroids and consequent osteopenia might enhance the risk of bone fractures.21 However, we did not find an association between the use of immunosuppressants and radiation toxicities in the current study. This might be due to the small sample size and the limited dosage of immunosuppressants, resulting from the relatively low disease activity of SLE. Additionally, it might be difficult to differentiate immunosuppressant-induced toxicity from radiation-induced toxicity in some cases. The dosimetric correlation of acute and late toxicities in radiation has been well established. With the availability of modern radiation technology and imaging modalities, IMRT has been widely used in practice and has shown superiority in dose distribution and dose escalation. By decreasing the prescribed dose to the normal tissues, patients are generally well tolerant to modern radiotherapy. Previous studies in CTD patients with radiation were commonly based on conventional radiotherapy. It has been reported that the dose thresholds of lungs, rather than CTD subtypes, were strongly associated with grade 3 or higher radiation pneumonitis.16 This suggested that SLE or other CTD patients might be more tolerant to radiotherapy than expected in the context of a strict limitation in the dose to organs at risk, with the aid of modern radiotherapy techniques. In a case report of a SLE patient diagnosed with FIGO stage II cervical cancer, after the local 45 Gy pelvic irradiation with IMRT, the patient only experienced grade 1 gastrointestinal toxicity and no other greater complications happened.22
By a median follow-up of 22.1 months in the present study, we reported the 3-year OS and 3-year PFS were 77.8% and 83.3%, respectively. Two patients died from distant metastasis of cervical cancer despite good local control. It is still not clear whether patients with SLE have the same response to radiotherapy as the general population. Since SLE is characterized by polyclonal B-cell activation associated with the abnormal expression of cytokines, it has not been concluded how SLE influences radiosensitivity in those patients. Few cases have been reported in this rare group of patients; thus, we had no similar published literature to comparison of treatment efficacy. However, compared to that of the general population of cervical cancer patients, the survival outcome in our study was satisfactory.13 It was encouraging to find that even SLE patients with advanced cervical cancer were able to acquire good disease control through definitive CCRT. Thus, we recommended that definitive radiotherapy should be more frequently considered in patients with cervical cancer and SLE in remission.
The main limitations of the present study included its retrospective nature and small sample size. SLE has been recognized as an uncommon disease, and the prevalence ranges from 9 to 241 per 100,000 person-years, and its incidence ranges from 0.3 to 23.2 per 100,000 person-years.23 The prevalence of SLE in the Chinese population was estimated to be 40–70 cases per 100,000 persons (>100/100,000 among women).24 In view of the low incidence of SLE, the number of patients with SLE complicated with cervical cancer is indeed very small, most of the previous studies are case report or small-scale case series. However, to our knowledge, our study was the largest series of patients with SLE who received IMRT for cervical cancer. Due to the limited number of SLE patients receiving radiation, large-scale patient enrollment is difficult for a single institution and thus requires the cooperation of multiple medical centers. In addition, we did not have a control group for comparison. According to the experience of our hospital, the incidence rates of grade 3 or higher chronic gastrointestinal and genitourinary toxicities of radiation for cervical cancer treatment were 2.3% and 1.3%, respectively.13 These numbers were consistent with our results.
Conclusion
Our study found that definitive radiation and postoperative radiation through IMRT for cervical cancer were generally well tolerated among patients with SLE when they were in the remission period. Although our study lacked the statistical power to be conclusive in establishing that SLE was not a risk factor for radiation complications, these above findings suggested that SLE should not be an absolute contraindication to pelvic radiation, yet that radiation oncologists should be more cautious regarding the dose limitation to normal tissues. A multi-institutional, large-scale, prospective study is needed to identify the risk of radiation complications in patients with SLE as well as other CTDs.
Abbreviations
ACR, American college of rheumatology; BED, biologically equivalent dose; BT, brachytherapy; CCRT, concurrent chemoradiotherapy; CTD, connective tissue disease; CTX, cyclosporine; CTC-AE, Criteria for Adverse Events; EBRT, external beam radiotherapy; EQD2, equivalent dose in 2-Gy fractions; FIGO, International Federation of Gynecology and Obstetrics; HCQ, hydroxychloroquine; HR, hazard ratio; IMRT, intensity-modulated radiation therapy; OS, overall survival; PFS, progression-free survival; PUMCH, Peking Union Medical College Hospital; RA, rheumatoid arthritis; RT, radiotherapy; SLE, systemic lupus erythematosus.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.21492
2. Thomas G, Mancini J, Jourde-Chiche N, et al. Mortality associated with systemic lupus erythematosus in France assessed by multiple-cause-of-death analysis. Arthritis Rheum. 2014;66(9):2503–2511. doi:10.1002/art.38731
3. Santana IU, Gomes Ado N, Lyrio LD, et al. Systemic lupus erythematosus, human papillomavirus infection, cervical pre-malignant and malignant lesions: a systematic review. Clin Rheumatol. 2011;30(5):665–672. doi:10.1007/s10067-010-1606-0
4. Wadstrom H, Arkema EV, Sjowall C, et al. Cervical neoplasia in systemic lupus erythematosus: a nationwide study. Rheumatology (Oxford). 2017;56(4):613–619. doi:10.1093/rheumatology/kew459
5. Zard E, Arnaud L, Mathian A, et al. Increased risk of high grade cervical squamous intraepithelial lesions in systemic lupus erythematosus: a meta-analysis of the literature. Autoimmun Rev. 2014;13(7):730–735. doi:10.1016/j.autrev.2014.03.001
6. Olivotto IA, Fairey RN, Gillies JH, et al. Fatal outcome of pelvic radiotherapy for carcinoma of the cervix in a patient with systemic lupus erythematosis. Clin Radiol. 1989;40(1):83–84. doi:10.1016/S0009-9260(89)80040-6
7. Hareyama M, Nagakura H, Tamakawa M, et al. Severe reaction after chemoradiotherapy of nasopharyngeal carcinoma with collagen disease. Int J Radiat Oncol Biol Phys. 1995;33(4):971. doi:10.1016/S0360-3016(95)90009-8
8. Varga J, Haustein UF, Creech RH, et al. Exaggerated radiation-induced fibrosis in patients with systemic sclerosis. JAMA. 1991;265(24):3292–3295. doi:10.1001/jama.1991.03460240088033
9. Chon BH, Loeffler JS. The effect of nonmalignant systemic disease on tolerance to radiation therapy. Oncologist. 2002;7(2):136–143. doi:10.1634/theoncologist.7-2-136
10. Lin A, Abu-Isa E, Griffith KA, et al. Toxicity of radiotherapy in patients with collagen vascular disease. Cancer. 2008;113(3):648–653. doi:10.1002/cncr.23591
11. Ross JG, Hussey DH, Mayr NA, et al. Acute and late reactions to radiation therapy in patients with collagen vascular diseases. Cancer. 1993;71(11):3744–3752. doi:10.1002/1097-0142(19930601)71:11<3744::AID-CNCR2820711144>3.0.CO;2-C
12. Phan C, Mindrum M, Silverman C, et al. Matched-control retrospective study of the acute and late complications in patients with collagen vascular diseases treated with radiation therapy. Cancer J. 2003;9(6):461–466. doi:10.1097/00130404-200311000-00005
13. Wang W, Zhang F, Hu K, et al. Image-guided, intensity-modulated radiation therapy in definitive radiotherapy for 1433 patients with cervical cancer. Gynecol Oncol. 2018;151(3):444–448. doi:10.1016/j.ygyno.2018.09.024
14. Giaj-Levra N, Sciascia S, Fiorentino A, et al. Radiotherapy in patients with connective tissue diseases. Lancet Oncol. 2016;17(3):e109–e117. doi:10.1016/S1470-2045(15)00417-9
15. Morris MM, Powell SN. Irradiation in the setting of collagen vascular disease: acute and late complications. J Clinl Oncol. 1997;15(7):2728–2735. doi:10.1200/JCO.1997.15.7.2728
16. Diao K, Chen YH, Catalano PJ, et al. Radiation toxicity in patients with collagen vascular disease and intrathoracic malignancy treated with modern radiation techniques. Radiother Oncol. 2017;125(2):301–309. doi:10.1016/j.radonc.2017.10.002
17. Li Z, Xu D, Wang Z, et al. Gastrointestinal system involvement in systemic lupus erythematosus. Lupus. 2017;26(11):1127–1138. doi:10.1177/0961203317707825
18. Brewer BN, Kamen DL. Gastrointestinal and hepatic disease in systemic lupus erythematosus. Rheum Dis Clin North Am. 2018;44(1):165–175. doi:10.1016/j.rdc.2017.09.011
19. Pinn ME, Gold DG, Petersen IA, et al. Systemic lupus erythematosus, radiotherapy, and the risk of acute and chronic toxicity: the mayo clinic experience. Int J Radiat Oncol Biol Phys. 2008;71(2):498–506. doi:10.1016/j.ijrobp.2007.10.014
20. Chen AM, Obedian E, Haffty BG. Breast-conserving therapy in the setting of collagen vascular disease. Cancer J. 2001;7(6):480–491.
21. Bliss P, Parsons CA, Blake PR. Incidence and possible aetiological factors in the development of pelvic insufficiency fractures following radical radiotherapy. Br J Radiol. 1996;69(822):548–554. doi:10.1259/0007-1285-69-822-548
22. Alsaeed E. Avoidance of radiotherapy-related, gastrointestinal complications in a patient with systemic lupus erythematosus: a case report and review of literature. Saudi J Gastroenterol. 2009;15(4):268–270. doi:10.4103/1319-3767.56103
23. Gergianaki I, Bortoluzzi A, Bertsias G. Update on the epidemiology, risk factors, and disease outcomes of systemic lupus erythematosus. Best Pract Res Clin Rheumatol. 2018;32(2):188–205. doi:10.1016/j.berh.2018.09.004
24. Zhang NZ, Chen R, Darmawan J. Rheumatic diseases in China. J Rheumatol. 1983;10(suppl 10):41–44.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
