Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 14
Quadriceps Endurance Increases Following Cycling Exercise With Non-Invasive Ventilation In Moderate-To-Severe COPD Patients. A Non-Randomized Controlled Study
Authors Labeix P , Berger M , Court Fortune I , Feasson L, Verges S, Costes F
Received 19 May 2019
Accepted for publication 23 September 2019
Published 5 November 2019 Volume 2019:14 Pages 2461—2468
DOI https://doi.org/10.2147/COPD.S216347
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Pierre Labeix,1,2 Mathieu Berger,3 Isabelle Court Fortune,4 Léonard Feasson,2,5 Samuel Verges,6 Frédéric Costes7,8
1Centre VISAS, Service de Physiologie Clinique et de l’Exercice, CHU St Etienne, France; 2Univ Lyon, UJM Saint-Etienne, Laboratoire Interuniversitaire de Biologie de la Motricité, EA 7424, Saint-Etienne F-42023, France; 3Univ Lyon, UJM Saint-Etienne, laboratoire SNA-EPIS, EA4607, Saint-Etienne, France; 4Service de Pneumologie et d’Oncologie Thoracique, CHU St Etienne, France; 5Unité de Myologie, CHU St Etienne, France; 6Université Grenoble Alpes, Inserm, Laboratoire HP2, Grenoble, France; 7Université Clermont Auvergne, INRA, UNH Clermont Ferrand, France; 8Service de Médecine du Sport et des Explorations Fonctionnelles, CHU Clermont Ferrand, France
Correspondence: Frédéric Costes
Service de Médecine du Sport et des Explorations Fonctionnelles CHU Gabriel Montpied, 58 rue Montalembert, F63002 Clermont Ferrand cedex 1, France
Tel +33473751665
Email [email protected]
Purpose: Application of non-invasive ventilation (NIV) during exercise improves exercise tolerance in severe COPD patients; however, the underlying mechanism is only partially unraveled. As part of its known effect to unload the respiratory muscles, we looked for the influence of NIV on post-exercise quadriceps muscle endurance.
Patients and methods: We included 25 severe COPD patients entering an outpatient pulmonary rehabilitation program. They performed, on successive days, three quadriceps endurance tests at 70% of the maximal strength (1RM) to task failure (TlimQ); 1) control condition; 2) following constant load cycling exercise to exhaustion without Inspiratory Pressure Support (TlimQ IPS-); 3) following the same cycling exercise with IPS (TlmQ IPS+).
Results: Dyspnea Borg score was significantly reduced at the end of the constant load cycling exercise with IPS+ compared to IPS- (3.5±2.6 to 4.3±2.3, p<0.05). Compared to controlled condition, TlimQ was reduced from 78.9±22.7 s to 64.7±22.1 s (p<0.01) with IPS+ and to 48.9±13.7 s (p<0.001) with IPS-. Sensitive analysis revealed a positive effect of NIV on TlimQ in only 15 of the 25 included patients (60%) and was unpredictable from exercise tolerance or maximal quadriceps strength.
Conclusion: Using a simple muscle endurance test, we showed the protective effect of NIV on the exercise-induced quadriceps dysfunction. This beneficial effect is inconstant in our small series of patients and could not be predicted by exercise capacity or endurance to exercise. However, measuring quadriceps muscle endurance following a session of exercise could determine in which patient NIV would improve the benefit of pulmonary rehabilitation.
Keywords: limb muscle, endurance, non-invasive ventilation, exercise, COPD
Introduction
Exercise tolerance is markedly impaired in severe patients with chronic obstructive pulmonary diseases (COPD) due to dyspnea and peripheral muscle weakness which affects up to one-third of patients.1 In most disabled patients, application of non-invasive ventilation (NIV) was shown to increase exercise endurance and to enhance the benefit of pulmonary rehabilitation.2 By decreasing the work of breathing, NIV alleviates exercise-induced dyspnea and allows the patients to prolong exercise. However, this improved exercise tolerance was observed in only 60% of the patients evaluated by van’t Hul et al.3 It is not clear from the available literature which patients would benefit the most from NIV and what are the mechanisms underlying the improved exercise tolerance. As a potential consequence of respiratory muscle unloading, Borghi Silva et al showed an improved leg muscle oxygenation, measured non-invasively by near-infrared spectroscopy, when COPD patients exercised with NIV compared to a sham ventilation.4 This suggests a peripheral muscle effect of NIV on O2 delivery by reducing the associated hemodynamic consequences of the respiratory work.5
Muscle fatigue is difficult to characterize objectively in humans. Maximal voluntary contraction of the quadriceps is largely influenced by the participation of the subject, especially after completion of a strenuous exercise. By using magnetic stimulation of the femoral nerve, several studies described the development of muscle fatigue after exercise in healthy subjects as well as in COPD patients.6,7 Not all COPD patients develop quadriceps muscle fatigue after symptom-limited cycling exercise, underlying the heterogeneity of the mechanisms limiting exercise performance in this population.7,8
In COPD patients, reducing the work of breathing decreased post-exercise quadriceps fatigue.9 This mechanism could partly explain the beneficial influence of NIV on both acute exercise tolerance and on pulmonary rehabilitation outcomes, by improving both the central cardiorespiratory and the peripheral muscle components of exercise limitation.
Measuring endurance of the quadriceps could represent an alternative to maximal muscle force for assessing muscle function and the development of fatigue following an exhaustive exercise. Quadriceps endurance is more systematically affected than maximal force in COPD patients,10 emphasizing the relevance of evaluating muscle endurance in this population. During such isolated endurance test (e.g., isolated knee extensions), only a small muscle mass is active which prevents a ventilatory or cardiovascular limitation to exercise, and thus makes this kind of evaluation an attractive way to specifically test peripheral muscle function.
Hence, this study aimed to evaluate whether the reduced work of breathing during cycling exercise with NIV in moderate-to-severe COPD patients would limit the importance of muscle fatigue, as assessed by an improved post-exercise quadriceps endurance. We reasoned that the reduced diaphragmatic work with NIV would increase oxygen delivery to peripheral muscle and delayed quadriceps fatigue. Moreover, we looked for correlations between the post-exercise improvements in peripheral muscle function with NIV and pulmonary function or exercise capacity parameters.
Materials And Methods
Patients And Study Design
We included patients with moderate-to-severe COPD, according to the GOLD criteria,11 referred to our center for a pulmonary rehabilitation program. All were in a stable condition. At inclusion in the program, they were evaluated for pulmonary function, exercise capacity and body composition by bio-impedance analysis (Nutriguard, Pöcking, Germany). The protocol was approved by our local ethics committee (CPP Sud Est I 2014-A00145-42). All participants provided written informed consent, and the study was conducted in accordance with the Declaration of Helsinki. The flow chart of the study is given in Figure 1. All the experiments took place in the outpatient rehabilitation center of the University Hospital of Saint-Etienne, France.
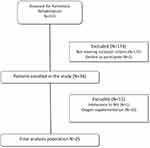 |
Figure 1 Study flowchart. |
Maximal power output (Wmax) and peak oxygen consumption (peak VO2) were determined during an incremental cardiopulmonary exercise test on a computer-electrically braked cycloergometer. After a 2-min resting period, a warm-up workload was set at 10 W, followed by 5 to 10 W increments every minute until exhaustion. Expired gases and volumes were measured breath by breath on an automated metabolic card (Ergocard, Medisoft, Sorinnes, Belgium) and averaged on 20-s periods. Electrocardiogram was recorded continuously with a 12-lead ECG. Oxygen saturation was monitored by pulse oximetry and arterialized ear capillary samples were withdrawn at rest and at peak exercise for blood gas measurements (ABL 800 Radiometer, Copenhagen, Denmark). A ventilatory limitation to exercise was defined at peak exercise as a ventilatory reserve ([VEmax pred – VEmax measured]/VEmax pred) below 20% and/or as a more than 5 mmHg increase of arterial CO2 pressure. Then, the subjects were evaluated during three experimental sessions as illustrated in Figure 2. The patients performed, in the same order and separated by at least 48 hrs, three quadriceps endurance tests to exhaustion (TlimQ): 1) without a preceding cycling exercise (TlimQ control), 2) within 5 mins following a constant work rate cycling (CWR) test to exhaustion without inspiratory pressure support (TlimQ IPS-), 3) within 5 mins following a CWR test with inspiratory pressure support (TlimQ IPS+), stopped at isotime of the first CWR test.
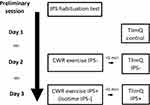 |
Figure 2 Design of the study. Abbreviations: IPS, inspiratory pressure support; TlimQ, quadriceps endurance limit time; CWR, constant work rate cycling exercise. |
Maximal Voluntary Contraction Of The Quadriceps (qMVC)
On the dominant thigh, we measured the quadriceps maximal isometric force on a quadriceps bench with a 90° knee flexion position. Peak force was determined by a fixed strain gauge (Globus, Codogné, Italy); 3 reproducible measurements (±5%) were performed and we recorded the highest value. Then, we determined one maximal repetition (1RM) as previously described.12,13
CWR Cycling Test
Patients performed a CWR test on cycloergometer at 75% of their predetermined Wmax. After 2 mins of unloaded cycling, the load was increased to 75% Wmax and the patients were asked to exercise until exhaustion, without encouragements. Pulse oximetry, transcutaneous carbon dioxide partial pressure (PtcCO2) and heart rate (Sentec, Basel, Switzerland) were monitored continuously during the test. Symptoms (dyspnea and fatigue) were evaluated before starting exercise and at exhaustion, using a 10 points modified Borg scale.
Quadriceps Endurance Test
The quadriceps endurance test was performed on a quadriceps bench with a starting position of 90° knee and hip flexions, with arms crossed over the chest. The test was adapted from Serres et al and consisted of extending the dominant leg against weight corresponding to 70% of 1RM with a pace of 12 movements per minute until exhaustion (duty cycle 40%).14 The test was stopped when the subject could no longer follow the movement amplitude or frequency on two consecutive movements despite strong verbal encouragements. The duration was then recorded as the quadriceps endurance limit time (TlimQ, seconds).
Non-Invasive Ventilation
NIV was applied via a nasal mask in all but one patient (full face mask). We used spontaneous ventilation with an inspiratory assistance mode (AI-VS) (Elisée 150, ResMed Saime, France). Inspiratory and expiratory pressure levels were adjusted during a preliminary habituation test as follows. At rest, we first set inspiratory pressure support (IPS) to 10 cmH2O and positive end-expiratory pressure (PEEP) to 4 cmH2O. Then, PEEP was gradually increased up to the maximum value tolerated by the patient. Thereafter, IPS was adjusted during cycling exercise in order to maintain dyspnea score below 5 on the10-points Borg scale. During the cycling exercise, we also adjusted the ramp and the expiratory trigger according to the patients’ sensations (comfort to breathe and inspiratory time). No oxygen was added to the circuit. The set up of NIV was supervised by the same physiotherapist (PL).
Statistical Analysis
Data are presented as mean ± standard deviation. A positive response to NIV application was set to >10% increase in TlimQ between IPS-and IPS+ CWR tests, according to the reproducibility of similar endurance test reported by Ribeiro et al.15 No previous data were available to calculate precisely a sample size. The sample size estimation was determined according to CONSORT 2010 statement, extension to randomized pilot and feasibility trials16 and Cohen’s recommendations who has defined effect-size bounds. So, with 25 participants, an effect–size around 0.8 (“grossly perceptible and therefore large”) can be highlighted for a two-sided type I error at 5%, a statistical power greater than 90% and an intra-class correlation coefficient at 0.5 to take into account between and within participant variability. TlimQ after each exercise test was compared by ANOVA after verification of the normal distribution of the data by a Shapiro–Wilk test. Symptoms, heart rate, SpO2 and PtcCO2 were compared using paired Wilcoxon signed-rank test. Unpaired t-tests with Welsh correction were used for subgroup analyses. Correlations were performed using least square regression analysis between exercise capacity and muscle function and TlimQ changes with IPS. Differences were considered significant at the level of 5%. Statistical analysis was performed using PRISM V5 software (Graphpad software, La Jolla, CA, USA).
Results
Study Population
We included 25 COPD patients. Their characteristics, pulmonary function, exercise capacity and body composition are given in Tables 1 and 2. Two of them were on long-term oxygen therapy and one other patient was treated with nocturnal NIV and supplemental O2. According to GOLD classification, 5 were in stage II, 9 in stage III and 11 patients in stages IV. BODE index was on average 4.6±1.5 points. Seven patients presented with muscle wasting (FFMI lower than 17 or 15 kg.m−2, in males or females, respectively) and six were obese. The incremental exercise test revealed a reduced exercise tolerance with an obvious ventilatory limitation in most of the patients (n= 18).
 |
Table 1 Patients’ Characteristics And Pulmonary Function |
 |
Table 2 Parameters Measured During The Incremental Exercise Test At Peak Exercise And Maximal Quadriceps Force |
Effect Of NIV During CWR Test
During CWR test, mean pressure support was 13±2 cmH2O (mean PEEP of 4±1 cmH2O). At isotime with IPS, dyspnea score decreased significantly compared to IPS- (3.5±2.6 versus 4.3±2.3, p<0.05) but fatigue was unchanged (3.2±2.3 versus 3.3±2.0, p=0.8). The changes in SpO2, HR and PtcCO2were not significantly different between both CWR tests (−2.0±4.2 versus −3.4±4.6%, 25.6±15.2 versus 28.4 ±14.8 min−1 and 1.2±7.4 versus 0.2±4.6 mmHg, for IPS+ and IPS- CWR tests, respectively, all non-significant).
Post Exercise Quadriceps Function
TlimQ control was on average 78.9±22.7 s, while TlimQ IPS- was significantly reduced to 48.9±13.7 s (p<0.001). TlimQ IPS+ was decreased compared to control to 64.7±22.1 s (p<0.01), but to a lesser extent than IPS- (p<0.01). The larger the decrease in TlimQ after cycling without IPS, the larger the recovery with IPS (r=0.59, p=0.002). We found no significant correlations between TlimQ control and maximal power output, qMVC or CWR duration in ambient air. The effect of IPS on TlimQ is illustrated in Figure 3.
The significant differences between TlimQ under the different conditions were maintained in subgroups of patients when we excluded those with less severe disease (GOLD stage 2, n=5), those with Long-Term Oxygen Therapy and/or nocturnal NIV (n=3), those with preserved inspiratory muscle force (MIP>60 cmH20, n=8) or those with muscle wasting (low FFMI, n=7).
Responders And Non-Responders To NIV
Individuals data demonstrated a beneficial effect of IPS on TlimQ in 15 patients (60%, responders group, with an averaged change between conditions of 25.4±11.4%), while IPS did not influence TlimQ in the 10 others (non-responders group) with a mean change 1.4±9.1%. Both groups differed for carbon monoxide diffusing capacity, maximal inspiratory pressure and resting arterial oxygen partial pressure (PO2) (all p<0.05). All other pulmonary function and exercise capacity parameters were similar between groups (see Tables 1 and 2). Tlim cyclo (p=0.6) and TlimQ (p=0.07) tended to be shorter in non-responders group compared to responders group. In responders group only, we found a positive correlation between TlimQcontrol and the difference in TlimQ with IPS (r=0.80, p=0.0003).
Discussion
NIV Reduces Quadriceps Muscle Endurance Impairment
The main result of this study is the reduced quadriceps muscle endurance impairment following CWR exercise in COPD patients when inspiratory pressure support is applied, which occurs in 60% of the patients studied.
IPS during exercise has been previously found beneficial in moderate-to-severe COPD patients by increasing exercise tolerance. Its main effect is the reduced burden on respiratory muscle work,17 leveling off minute ventilation and dynamic hyperinflation.18 This noticeably results in reduced dyspnea sensation. Additional effects have also been reported which could affect exercise endurance. The application of IPS could restore normal cardiac output responses to exercise.19 IPS may also switch part of the blood flow from respiratory to limb muscles, which could limit peripheral muscle deoxygenation during exercise.4 Considering the respiratory metaboreflex theory,5 we reasoned that decreasing respiratory work with IPS could improve oxygen delivery through exercise-induced sympatholysis, and consequently increase muscle endurance. In the present study, we did not measure cardiac output or muscle oxygenation which would ascertain the increase in O2 delivery with IPS and could explain the increase in TlimQ. However, a previous study demonstrated a reduced post-exercise quadriceps fatigue with proportional assist ventilation in COPD patients.9 Muscle fatigue is difficult to evidence in a clinical setting since it requires supramaximal nerve stimulation, which is technically demanding.20 By contrast, measurement of the maximal duration a standardized task can be sustained, ie, endurance, is easy to perform for the quadriceps muscle with a resistance training quadriceps bench. As opposed to maximal force capacity, muscle endurance is mainly determined by type I aerobic fibers and oxygen delivery.21 Thus, it is not surprising that muscle endurance was found substantially reduced in COPD patients compared to controls.22 Although a reduced endurance time cannot be interpreted as a proxy of muscle fatigue, its variation after an intervention is directly linked with muscle function and the patient’s perception of effort. We cannot exclude the influence of patient’s motivation on the change in quadriceps endurance following cycling exercise but we believe its role was limited thanks to strong verbal encouragements in each condition.
Responders And Non-Responders To NIV
The number of patients increasing TlimQ when IPS was applied during exercise was similar to those increasing cycle endurance with IPS 10 cmH2O in the van’t Hul‘s study.3 Interestingly, this proportion remains similar in subgroups of patients with more consistent characteristics (i.e., COPD severity stages 3 and 4 of GOLD, no chronic respiratory failure, no muscle wasting or preserved maximal inspiratory force). The intrinsic variability of the quadriceps endurance test used in the present study has not been reported and we defined a cut-off value of 10% for a positive response to IPS, based on Ribeiro et al's study on total isokinetic work at 90°/s in COPD patients.15 The 25% variation in endurance time we observed seems to be large enough to be out of the limits of reproducibility. Exercise capacity (maximal power output or endurance) or quadriceps muscle force could not predict the response of TlimQ to IPS. However, non-responders patients had a lower DLCO, arterial PO2 and maximal inspiratory force compared to responders. This suggests that IPS could not fully compensate for more altered pulmonary gas exchanges, and muscle O2 delivery would not improve to a similar extent than in responders patients. This interpretation deserves to be confirmed by measuring muscle oxygenation since such a difference in gas exchange capacity has never been reported as a predictive factor of NIV response to exercise tolerance.
The level of ventilator support could explain the extent of muscle function improvement. We found no difference in mean inspiratory or expiratory pressures which could have influenced breathing mechanics during CWR test with IPS. Then, the most relevant mechanism underlying the effect of IPS was an improved oxygen delivery to the locomotor muscles. Blood oxygenation (SpO2 and PtcCO2) and heart rate were similar during both CWR tests which could not explain an improved quadriceps muscle function. We did not measure blood lactate following both CWR exercises to demonstrate potential metabolic changes able to explain the better muscle function following exercise with IPS.
Novelty Of The Study And Clinical Implications
In patients entering a pulmonary rehabilitation program, muscle deconditioning represents a common feature. Maximal power output during cycling was low in these patients which may preclude stressing lower limb muscles to their maximal capacity. The isolated quadriceps endurance test allowed to assess quadriceps function while ventilation was not a limiting factor.23 Our results outline an alteration of muscle endurance capacity following maximal cycling exercise in this population of moderate-to-severe COPD despite very low mechanical power output. Measuring the variation of CWR duration with and without IPS gives an overview of exercise tolerance but cannot differentiate between the beneficial effect of NIV on the respiratory system and on the peripheral muscle function. We assessed specifically post-exercise quadriceps endurance after the same duration of cycling exercise which allows to evidence the improvement in muscle function. We set up a simple test which could help to personalize exercise training by increasing the intensity of cycling sessions according to the initial muscle response. For instance, training intensity would be more rapidly increased without discomfort in those who improve TlimQ with IPS, and consequently, training load would be higher compared to non-responders. One could thus expect higher quadriceps adaptations to exercise training which would translate into daily life activities. This remains to be demonstrated in a prospective controlled study.
To date, no strong predictor of the success of NIV to improve exercise tolerance and the benefit on pulmonary rehabilitation has been demonstrated. A reduced maximal inspiratory pressure would predict a beneficial effect of NIV during exercise in COPD patients though this has been recently challenged,24 but the decision to use NIV during pulmonary rehabilitation remains rather empirical. The variation in quadriceps endurance following cycling exercise could evaluate the impact of reducing the work of breathing by NIV during exercise on muscle function and therefore could be used to determine the patients who could benefit from this intervention during pulmonary rehabilitation. This remains, however, to be demonstrated in future studies involving COPD in pulmonary rehabilitation program.
Study Limitations
CWR test with IPS was stopped at the same time sustained without IPS and we cannot demonstrate that IPS improved exercise tolerance in our patients. Therefore, we can only speculate that the longer TlimQ would translate in a longer maximal CWR cycling exercise duration. However, this effect of IPS on exercise endurance would certainly occur in our patients based on the significant reduction in symptoms at the end of CWR IPS+. It was not possible to randomize the order of the tests since determining first the duration of CWR without IPS was required, similar to previous studies with similar study design.9 Therefore, we cannot exclude that part of the effect of IPS on quadriceps muscle endurance was due to some potential training effect, although it is unlikely that repeating twice the exercise session would lead to a 25% improvement in muscle endurance as observed in the present study. Finally, we decided not to use any kind of sham procedure not able to blind appropriately the patients during the CWR test. It has been suggested that a level of inspiratory aid below 10 cmH2O was ineffective to increase exercise tolerance in COPD patients.3 However, the patients could not be totally blinded of the application of IPS since the preliminary habituation test with NIV setting up shows the patient to what extent the symptoms could be alleviated.
Perspectives
These data bring insight into the complex interplay between respiratory system and quadriceps function in COPD patients during exercise. In those patients with a poor exercise tolerance due to ventilatory limitation and candidate for NIV application during pulmonary rehabilitation, we propose to assess post-exercise quadriceps endurance with and without a ventilatory support. Combining the responses to cycling exercise endurance and to post-exercise quadriceps endurance could allow to adjust the training load in order to maximize quadriceps adaptations and would participate in the increased benefit of pulmonary rehabilitation with NIV.
Conclusion
We demonstrated that the application of NIV during exercise improved quadriceps endurance performed immediately after a CWR cycling test. This beneficial occurred in 60% of the patients evaluated and was unpredictable from exercise capacity or endurance to exercise. Therefore, measuring quadriceps muscle endurance with a relatively easy to perform protocol as in the present study could constitute a strategy to assess the benefit of NIV applied during exercise on peripheral muscle function of the patients.
Acknowledgment
This study was promoted by CHU St Etienne, France, and partly supported by a grant from Association Lyonnaise de Logistique Posthospitalière, Lyon, France.
Author Contributions
FC, PL and ICF made substantial contributions to the conception, design, and/or interpretation of data; PL conducted experiment; MB, LF and SV contributed to data analysis. FC and PL analyzed data and wrote the manuscript. All authors contributed to drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflict of interest related to this work.
References
1. Maltais F, Decramer M, Casabury R, et al. An Official American Thoracic Society/European Respiratory Society Statement: update on limb muscle dysfunction in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2014;189:e15–e62. doi:10.1164/rccm.201402-0373ST
2. Menadue C, Piper AJ, Van ’t Hul AJ, Wong KK. Non-invasive ventilation during exercise training for people with chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2014. John Wiley and Sons Ltd. doi:10.1002/14651858.CD007714.pub2
3. van ’t Hul A, Gosselink R, Hollander P, Postmus P, Kwakkel G. Acute effects of inspiratory pressure support during exercise in patients with COPD. Eur Respir J. 2004;23:34–40. doi:10.1183/09031936.03.00016903
4. Borghi-Silva A, Oliveira C, Carrascosa C, et al. Respiratory muscle unloading improves leg muscle oxygenation during exercise in patients with COPD. Thorax. 2008;63:910–915. doi:10.1136/thx.2007.090167
5. Dempsey JA, Romer L, Rodman J, Miller J, Smith C. Consequences of exercise-induced respiratory muscle work. Respir Physiol Neurobiol. 2006;151:242–250. doi:10.1016/j.resp.2005.12.015
6. Mador MJ, Deniz O, Aggarwal A, Kufel TJ. Quadriceps fatigability after single muscle exercise in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2003;168:102–108. doi:10.1164/rccm.200202-080OC
7. Saey D, Debigaré R, LeBlanc P, et al. Contractile leg fatigue after cycle exercise: a factor limiting exercise in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2003;168:425–430. doi:10.1164/rccm.200208-856OC
8. Saey D, Michaud A, Couillard A, et al. Contractile fatigue, muscle morphometry, and blood lactate in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2005;171:1109–1115. doi:10.1164/rccm.200408-1005OC
9. Amann M. Exercise-induced metabolic perturbation: all roads lead to Rome. Exp Physiol. 2010;95:765–766. doi:10.1113/expphysiol.2010.053520
10. Coronell C, Orozco-Levi M, Méndez R, et al. Relevance of assessing quadriceps endurance in patients with COPD. Eur Respir J. 2004;24:129–136. doi:10.1183/09031936.04.00079603
11. Vogelmeier C, Criner G, Martinez F, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease 2017 report: GOLD executive summary. Eur Respir J. 2017;49:1700214. doi:10.1183/13993003.00214-2017
12. Levinger I, Goodman C, Hare D, et al. The reliability of the 1RM strength test for untrained middle-aged individuals. J Sci Med Sport. 2009;12:310–316. doi:10.1016/j.jsams.2007.10.007
13. Kraemer WJ, Ratamess N, Fry A, French D. Strength training: development and evaluation of methodology. In: Maud PJ, Foster C, editors. Physiological Assessment of Human Fitness. Champaign: Human Kinetics; 2006:119–150.
14. Serres I, Gautier V, Varray A, Préfaut C. Impaired skeletal muscle endurance related to physical inactivity and altered lung function in COPD patients. Chest. 1998;113:900–905. doi:10.1378/chest.113.4.900
15. Ribeiro F, Lépine P, Garceau-Bolduc C, et al. Test-retest reliability of lower limb isokinetic endurance in COPD: a comparison of angular velocities. Int J of Chron Obstruct Pulmon Dis. 2015;10:1163–1172.
16. Eldridge SM, Chan CL, Campbell MJ, et al. CONSORT 2010 statement: extension to randomised pilot and feasibility trials. Pilot Feasibility Stud. 2016;2. doi:10.1186/s40814-016-0105-8
17. Maltais F, Reissmann H, Gottfried SB. Pressure support reduces inspiratory effort and dyspnea during exercise in chronic airflow obstruction. Am J Respir Crit Care Med. 1995;151:1027–1033. doi:10.1164/ajrccm.151.4.7697226
18. Hussain O, Collins E, Adiguzel N, et al. Contrasting pressure-support ventilation and helium–oxygen during exercise in severe COPD. Respir Med. 2011;105:494–505. doi:10.1016/j.rmed.2010.08.008
19. Oliveira C, Carrascosa C, Borghi-Silva A, et al. Influence of respiratory pressure support on hemodynamics and exercise tolerance in patients with COPD. Eur J Appl Physiol. 2010;109:681–689. doi:10.1007/s00421-010-1408-8
20. Nyberg A, Saey D, Why MF. How limb muscle mass and function should be measured in patients with chronic obstructive pulmonary disease. Ann Am Thorac Soc. 2015;12:1269–1277. doi:10.1513/AnnalsATS.201505-278PS
21. Bassett D, Howley E. Limiting factors for maximum oxygen uptake and determinants of endurance performance. Med Sci Sports Exerc. 2000;32:70–84. doi:10.1097/00005768-200001000-00012
22. Evans S, Watson L, Hawkins M, et al. Respiratory muscle strength in chronic heart failure. Thorax. 1995;50:625–628. doi:10.1136/thx.50.6.625
23. Richardson RS. Skeletal muscle dysfunction vs. muscle disuse in patients with COPD. J Appl Physiol. 1999;86:1751–1752. doi:10.1152/jappl.1999.86.5.1751
24. Koch R, Rapello GVG, de Tarso Müller P. Is inspiratory muscle weakness a determinant of endurance exercise tolerance during NIV-supported exercise in patients with COPD? J Cardiopulm Rehabil. 2018;Prev.38(6):E9–E11.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

