Back to Journals » Drug Design, Development and Therapy » Volume 15
Qing-Luo-Yin Alleviated Monocytes/Macrophages-Mediated Inflammation in Rats with Adjuvant-Induced Arthritis by Disrupting Their Interaction with (Pre)-Adipocytes Through PPAR-γ Signaling
Authors Wang R, Li DF, Hu YF, Liao Q, Jiang TT, Olatunji OJ , Yang K, Zuo J
Received 17 May 2021
Accepted for publication 9 July 2021
Published 16 July 2021 Volume 2021:15 Pages 3105—3118
DOI https://doi.org/10.2147/DDDT.S320599
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jianbo Sun
Rui Wang,1,2,* Dan-Feng Li,1,2,* Yi-Fang Hu,1,2 Qiang Liao,1,2 Tian-Tian Jiang,1,2 Opeyemi Joshua Olatunji,3 Kui Yang,2,4 Jian Zuo1,2,5
1Department of Traditional Chinese Medicine, the First Affiliated Hospital of Wannan Medical College (Yijishan Hospital), Wuhu, 241000, People’s Republic of China; 2Research Center of Integration of Traditional Chinese and Western Medicine, Wannan Medical College, Wuhu, 241000, People’s Republic of China; 3Faculty of Traditional Thai Medicine, Prince of Songkla University, Hat Yai, 90112, Thailand; 4Department of Pharmacy, the First Affiliated Hospital of Wannan Medical College (Yijishan Hospital), Wuhu, 241000, People’s Republic of China; 5Key Laboratory of Non-coding RNA Transformation Research of Anhui Higher Education Institution, Wannan Medical College, Wuhu, 241000, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Jian Zuo
Department of Traditional Chinese Medicine, the First Affiliated Hospital of Wannan Medical College (Yijishan Hospital), Wuhu, 241000, People’s Republic of China
Email [email protected]
Background: The Chinese herbal formula Qing-Luo-Yin (QLY) has been successfully used in rheumatoid arthritis treatment for decades. It exhibits notable immune and metabolism regulatory properties. Thereby, we investigated its effects on the interplay between (pre)-adipocytes and monocytes/macrophages under adjuvant-induced arthritis (AIA) circumstances.
Methods: Fat reservoir and histological characteristics of white fat tissues (WAT) in AIA rats receiving QLY treatment were examined upon sacrifice. Metabolic parameters, clinical indicators, and oxidative stress levels were determined using corresponding kits, while mRNA/protein expression was investigated by PCR and immunoblotting methods. M1 macrophage distribution in WAT was assessed by flow cytometry. The effects of QLY on (pre)-adipocytes were further validated by experiments in vitro.
Results: Compared with normal healthy controls, body weight and circulating triglyceride were declined in AIA rats, but serological levels of free fatty acids and low-density lipoprotein cholesterol were increased. mRNA IL-1β and iNOS expression in white blood cells and rheumatoid factor, C-reactive protein, anti-cyclic citrullinated peptide antibody, MCP-1 and IL-1β production in serum/WAT were up-regulated. Obvious CD86+CD11b+ macrophages were enriched in WAT. Meanwhile, expression of PPAR-γ and SIRT1 and secretion of adiponectin and leptin in these AIA rats were impaired. QLY restored all these pathological changes. Of note, it significantly stimulated PPAR-γ expression in the treated AIA rats. Accordingly, QLY-containing serum promoted SCD-1, PPAR-γ, and SIRT1 expression in pre-adipocytes cultured in vitro. AIA rats-derived peripheral blood mononuclear cells suppressed PPAR-γ and SCD-1 expression in co-cultured pre-adipocytes, but serum from AIA rats receiving QLY treatment did not exhibit this potential. The changes on PPAR-γ expression eventually resulted in varied adipocyte differentiation statuses. PPAR-γ selective inhibitor T0070907 abrogated QLY-induced MCP-1 production decline in LPS-primed pre-adipocytes and reduced adiponectin secretion.
Conclusion: QLY was potent in promoting PPAR-γ expression and consequently disrupted inflammatory feedback in WAT by altering monocytes/macrophages polarization and adipocytes differentiation.
Keywords: fat metabolism, Qing-Luo-Yin, rheumatoid arthritis, macrophages polarization, adipokine, (pre)-adipocytes
Introduction
Rheumatoid arthritis (RA) is well recognized as a CD4+ lymphocytes-driven disease. Generally, RA conditions are favorable for Th1/17 cells differentiation. These cells cripple the development of Th2 and Treg cells, leading to T cells subsets imbalance. This phenomenon profoundly alters cytokines network profile. Accumulated pro-inflammatory cytokines such as IL-1, IL-17 and IL-22 further exacerbate immune abnormality and activate RA-related effector cells like osteoclasts and synovioblast. At the same time, the deficiency of immunosuppressive cytokines impairs the ability to curb the progression of inflammation and tissue degradation.1,2 Therefore, modulating T cells differentiation and related cytokines production should be the priority for RA treatments. The successful clinical application of IL-6 neutralizing antibodies, JAKs inhibitors and many other biologic disease–modifying anti-rheumatic drugs (bDMARDs) have solidly confirmed this.3 Under this context, innate immunity in RA is usually overlooked. In fact, it is similarly important for the development of RA immune milieu. Taking monocytes/macrophages for example, they are potent in promoting lymphocytes development, and have the potential to transform into osteoclasts. Furthermore, they provoke persistent inflammation and bone erosion in joints.4 High levels of M1 phenotype markers like IL-12 and IL-18 have been extensively detected in RA patients.5 Thus, diminishing certain monocytes/macrophages subsets could be another effective anti-rheumatic approach.
Upon infection/inflammatory stimulus, monocytes/macrophages are turned into inflammatory phenotype to fulfill the defensive functions. However, excessive M1 monocytes/macrophages usually serve as a secondary factor in inflammatory diseases like RA. As such, identifying factors driving M1 polarization would benefit the development of novel anti-rheumatic therapies. Mounting evidences have demonstrated that aside classic immune mediators, energy metabolism is another decisive factor controlling the status of monocytes/macrophages.6 Metabolic regulators such as NAMPT, SIRT1 and PPAR-γ are deeply involved in monocytes/macrophages polarization.7–9 M1 and M2 cell subsets have distinctly different metabolic profiles.6 More importantly, high prevalence of metabolic complication is observed in inflammatory diseases, while metabolic disorders are always associated with low-grade inflammation.10,11 Because monocytes/macrophages are vital factors linking the two types of diseases, it is reasonable to assume that certain metabolic interventions could improve the clinical manifestation of RA.
Properties of Traditional Chinese Medicine (TCM) are typically summarized based on the reactions observed on treated subjects. As the basic concept of medicinal properties, four natures (cold, heat, warm and cool) indicate different body temperature changes after treatments. It is apparent that this definition is closely related to basal metabolism alteration. That is, drugs with cold and cool natures would slow down energy metabolism, while warm and hot natured drugs exert the opposite effect. Qing-Luo-Yin (QLY) is a representative herbal formula used for hot syndrome-related RA treatment. It was prescribed by a famous contemporary TCM master Jiren Li, and has been successfully applied in clinical practice for over four decades.12–14 Consistent to the notion mentioned above, QLY generally suppressed energy metabolism. Most energy metabolism-related metabolites especially those involved in fat catabolism were decreased during QLY treatment.15 Unfortunately, clinical implication of these metabolic changes is poorly understood. The network pharmacology prediction suggested that macrophages could be a main target of QLY.15 It inspired us to think over the anti-rheumatic mechanism of QLY based on the feedback between immunity and metabolism. By using adjuvant-induced arthritis (AIA) models, we attempted to clarify how QLY-induced fat metabolism alteration contributed to the improvement of inflammatory monocytes/macrophages-mediated arthritic manifestation.
Materials and Methods
Chemicals and Reagents
Incomplete Freund’s Adjuvant (IFA) and Bacillus Calmette-Guérin (BCG) were purchased from Chondex (Redmond, WA) and Rebio Scientific (Shanghai, China), respectively. ELISA kits for the quantitative determination of rheumatoid factor (RF), C-reactive protein (CRP), anti-cyclic citrullinated peptide antibody (anti-CCP antibody), IL-1β, MCP-1, IL-6, adiponectin and leptin were supplied by Multi-Science (Hangzhou, Zhejiang, China). Quantification kits used for the content determination of blood glucose, total cholesterol (T-CHOL), triglyceride (TG), free fatty acids (FFAs), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), reduced glutathione (GSH) and malonaldehyde (MDA) together with the kit for total superoxide dismutase activity (SOD) test were bought from Solarbio (Beijing, China). ReverAid First-Strand cDNA Synthesis kits and Universal qPCR Master Mix were obtained from Thermo Fisher Scientific (Rockford, IL, USA) and New England Biolabs (Ipswich, MA, USA) respectively. Gene-specific primers were synthesized by Sangon Biotech (Shanghai, China). Primary anti-β-actin, NAMPT, SIRT1, PPAR-γ, SCD-1, JNK, and p-JNK antibodies for immunoblotting assays were supplied by ABclonal Technology (Wuhan, China). PE-tagged anti-CD86 and FITC-tagged anti-CD11b antibodies used in flow cytometry assay as well as enhanced chemiluminescence (ECL) detection kit and cell culture reagents including RPMI-1640 medium, DMEM/F12 medium, and fetal bovine serum (FBS) were procured from Thermo Fisher Scientific (Rockford, IL, USA). Lipopolysaccharide (LPS), penicillin–streptomycin, 3-isobutyl-1-methylxanthine (IBMX), hexadecadrol (DEX), TRNzol Universal total RNA extraction reagent, phosphate-buffered saline (PBS), HRP-conjugated secondary antibodies, BCA protein quantitative kit, RIPA lysis buffer and protein-loading buffer were obtained from either Keygen Biotech (Nanjing, Jiangsu, China) or Beyotime Biotech (Nantong, Jiangsu, China). PPAR-γ selective inhibitor T0070907 was purchased from APExBIO Technology LLL (Houston, TX, USA). Ultra-pure water was prepared using a Milli-Q purification system (Millipore, Bedford, MA, USA). All other chemicals used were of analytical grade.
Exact Preparation
QLY exact preparation was according to the procedures reported previously.15 In brief, four herbal components of QLY including Radix Sophorae flavescentis (Kushen), Caulis Sinomenium acutum (Qingfengteng), Cortex Phellodendri chinensis (Huangbo) and Rhizoma Dioscoreae hypoglaucae (Bixie) were mixed in 1.5:1.2: 1:1 ratio. After being soaked in warm water for 2 h, the mixture was decocted for 1.5 h in 8 times volume of water. The extraction was repeated thrice. The filtrates obtained were combined and concentrated to dryness.
Chemical Composition Characterization
Chemical components in QLY exact were identified based on UPLC/MS2 analysis. The dried extract was dissolved in methanol–water (1:1). The analyte was filtered through a 0.45-μm filter, followed by centrifugation at 12,000 rpm for 10 min. The supernatant was then subjected to the following qualitative analysis. The analytical platform was comprised of a 1290 UPHLC system (Agilent) and a Q Exactive Focus mass spectrometry (Thermo Fisher Scientific). Chromatographic separation was achieved on an ACQUITY UPLC BEH C18 column (2.1×100 mm, 1.7 μm, Waters) at a flow rate of 400 μL/min with the elution program detailed as follows: 0–6.5 min, 5–30% acetonitrile; 6.5–12.5 min, 30–70% acetonitrile; 12.5–18 min, 70–100% acetonitrile; 18–25 min, 100% acetonitrile (in water). Analytical parameters of the mass spectrometry are summarized as below: sheath gas flow rate, 45 Arb; aux gas flow rate, 15 Arb; capillary temperature, 400 °C; full MS resolution, 70,000; MS/MS resolution, 17,500; spray voltage, 4.0 kV (positive) or −3.6 kV (negative). Total ion chromatogram (TIC) was acquired by the software Xcalibur (Thermo Fisher Scientific). A composite score was designated to each signal based on their similarity with MS characteristic of the reference compounds included in an in-house natural compounds MS2 database (Biotree Biotech, Shanghai, China). Those with composite score above 0.6 were taken as reliable, and annotated by corresponding chemical names.16
Induction of AIA in Rats and Treatment
Male Sprague Dawley rats (7 weeks old) supplied by Tianqin Biotechnology (Changsha, Hunan, China) were used in the experiments in vivo. Before the induction of AIA, heat-inactivated BCG was finely grinded and mixed in IFA in a porcelain mortar to prepare Complete Freund’s Adjuvant (CFA, 20 mg/mL). After 7 days of acclimatization, 16 rats received an intracutaneous injection of CFA (0.1 mL) at the plantar skin of left hind paw. Thereafter, they were assigned into two groups (n = 8), and six rats successfully developing secondary inflammation from each group were kept. One group was assigned as QLY-treated group, and orally administered with QLY for 31 days. The other group was designated as AIA control group, and another six healthy rats served as normal controls. Both AIA model and healthy control rats were treated with 0.5% CMC-Na in parallel. Prior to the treatments, QLY extract was firstly dissolved in ethanol, and then dispersed in CMC-Na solution. The final concentration of ethanol was less than 2%. According to body surface area-based equivalent dose calculation, the suitable dose of QLY extract for rats is about 0.3 g/kg. To increase its clinical performance and avoid potential toxicity, the treatment dose was designed as 0.45 g/kg/day. All the animal protocols were approved by the Ethical Committee of Wannan Medical College (LLSC-2020-138), and were in accordance with the Guide for the Care and Use of Laboratory Animals (US National Research Council, 2011).
Clinical Outcomes Evaluation and Sampling
The progress of AIA throughout the experimental duration was continuously evaluated by the arthritis score using a previously defined criterion.15,16 Body weight changes were also periodically recorded. At the end of treatment, the animals were fasted overnight. Next day, they were anesthetized, and blood samples were collected via cardiac puncture. After erythrocyte lysis, white blood cells (WBCs) from anticoagulation blood were obtained after centrifugation, and subsequently lysed in Trizol to prepare total RNA. The serological levels of RF, CRP, anti-CCP antibody, IL-1β, MCP-1, IL-6, adiponectin, leptin, T-CHOL, TG, FFAs, LDL-C, HDL-C, GSH, MDA concentration as well as SOD activity were determined using corresponding kits according to the manufacturers’ instructions. Thereafter, the rats were sacrificed. Hind paws, spleen and left kidney fat pad were dissected from the body and weighted. Portion of the white fat tissues (WAT) were used for histological examination and pre-adipocytes preparation, and the remaining samples were used for immunoblotting, RT-qPCR, ELISA and flow cytometry assays.
Histological Examination of WAT
Fat pads were initially fixed in 4% paraformaldehyde. The fixed specimens were thereafter dehydrated with gradient ethanol solution, and embedded in paraffin. The paraffin blocks were then continuously sectioned. Afterward, the slices were dewaxed, rehydrated with a decreasing concentration of ethanol, and finally stained with hematoxylin/eosin (HE). To perform Oil Red O staining, some other fresh WAT were frozen in liquid nitrogen. The frozen specimens were sectioned at a thickness of 16 μm and mounted on glass slides. The slides were subsequently soaked in Oil Red O staining solution for 20 min, followed by a further 75% isopropanol treatment after extensive washing with water. Morphological observation of histological characteristics including adipocyte size, fat droplet distribution and inflammatory cells infiltration was achieved with the aid of an Olympus BH-2 light microscope (Tokyo, Japan).
Flow Cytometry Analysis
Fresh WAT was digested with type I collagenase. The unicellular suspension was obtained after filtration with steel sieves (200 meshes). After erythrocyte lysis, the remaining cells were harvested by centrifuging at 700 rpm for 5 min. The cells were subsequently washed and resuspended in PBS, and the resulting cell suspension was incubated with PE-CD86 and FITC-CD11b antibodies in the dark for 20 min. Finally, the processed samples were fed to a flow cytometry (FC500, Beckman Coulter, Miami, FL, USA). M1 macrophages within WAT were identified as CD86+CD11b+ cells. The raw data were quantified with Flow Jo V10.2 software (Tree Star, Inc., Ashland, USA).
Cell Culture and Rats Serum Preparation
Unicellular suspension from the digestion of WAT was centrifugated for 10 min. Obtained cells were then cultured in DMEM/F12 medium supplemented with 10% FBS and penicillin–streptomycin (100 U/mL) at 37°C in a humid atmosphere containing 5% CO2. When reaching 80% confluence, the cells were passaged. After continuous passage culture, the homogeneous fibroblast-like pre-adipocytes were obtained. Cells from the next three passages were adopted in the in vitro experiments. Peripheral blood mononuclear cells (PBMCs) from anticoagulant blood were prepared by the means of gradient centrifugation using a kit provided by Solarbio (Beijing, China). These cells were maintained in RPMI-1640 medium supplemented with 10% FBS under normal culture conditions, and used directly without further passage culture. The serum from normal healthy, AIA control and QLY-treated rats were prepared using blood obtained from above in vivo experiments. Some other healthy rats were fasted overnight. Half of them received three consecutive doses of QLY by oral gavage. The interval between two doses was 1 h. One hour after the last dose, peripheral blood from the both groups was collected into coagulation-promoting tubes for serum preparation.
Treatments of Pre-Adipocytes
Firstly, pre-adipocytes were treated with the serum obtained from either normal or QLY-treated healthy rats. The cells were seeded in 6-wells plates at the appropriate density beforehand. Twelve hours later, half of the culture medium was replaced with fresh medium containing 10% rat serum. After an additional 12 h, all the cells were harvested. Expression of protein NAMPT, SIRT1, PPAR-γ, and SCD-1 was determined by immunoblotting approach. To figure out if the effects of QLY on WAT were mediated by immune factors, we co-cultured pre-adipocytes with PBMCs from different rats. PBMCs and pre-adipocytes were located in the upper and lower chambers of the transwell, respectively. The co-culture lasted for 12 h, and then pre-adipocytes were collected for immunoblotting and RT-qPCR analyses. To further validate the effects of immune factors on adipocyte differentiation, pre-adipocytes were cultured in DMEM/F12 medium supplemented with 10% FBS, 10 μg/mL insulin, 10 μg/mL NaOH, 1 μM DEX and 0.5 mM IBMX for 2 days after adaptive culture. Thereafter, supernatant from this cell culture system was replaced by medium containing 10% FBS, 10 μg/mL insulin, 10 μg/mL NaOH, and 1 μM DEX. Two days later, the cells were cultured in freshly prepared complete medium by using 10% different rat serums instead of FBS. Some other normal pre-adipocytes seeded in 6-wells plates were challenged by LPS at 500 ng/mL for 1 h, and then treated with 10% normal or QLY-containing rat serum for 12 h in the presence/absence of T0070907. Thereafter, levels of MCP-1 and adiponectin in the medium were determined by ELISA kits, and expression of mRNA PPAR-γ and SCD-1 in the cells were investigated by PCR method.
Western Blot Assay
Total proteins from rat tissues and cells were extracted using pre-chilled RIPA buffer. The lysate was then centrifuged at 12,000 rpm for 10 min at 4°C, and resulting supernatant was denatured by boiling. Samples containing an equal amount of protein (10 μg) were subjected to SDS-PAGE separation. The separated proteins were subsequently transferred onto PVDF membranes. After blocking with 5% skim milk for 2 h, the blotting membranes were incubated with primary antibodies at 4 °C overnight, and further treated with secondary antibodies at room temperature for 2 h. Finally, they were exposed to ECL reagent, and the immunoblots were photographed with an Amersham Imager 600 (GE Healthcare). Semi-quantification of the results was achieved with the aid of Image J software using gray value as reference.
RT-qPCR Assay
Total RNA extracted by Trizol reagent was subjected to reverse transcription using a cDNA synthesis kit. Obtained cDNA was subsequently used as templates for the following PCR procedure with the aid of SYBR Green qPCR Mix on a 7500 Real-Time PCR system (Thermo Fisher Scientific, Rockford, IL, USA). Relative mRNA expression was calculated based on the Comparative-Ct method (2−ΔΔCt) by taking β-actin as the internal reference. Sequences for the primers used in this study are as follows: β-actin, Forward, TGTCCACCTTCCAGCAGATGT, Reverse, AGCTCAGTAACAGTCCGCCTAGA; iNOS, Forward, TGCCTTTGCTCATGACATCG, Reverse, AACACGTTCTTGGCGTGGA; IL-1β, Forward, TCCTCTGTGACTCGTGGGAT, Reverse, TCAGACAGCACGAGGCATTT; IL-10, Forward, AGTGGAGCAGGTGAAGAATGA, Reverse, ATTTGCTGGTTCCCACACTC; SIRT1, Forward, GTGTCATAGGTTAGGTGGCGAG, Reverse, GAAGAGGTGTTGGTGGCAACTC; NAMPT, Forward, GATTCTGGAAATCCGCTCGA, Reverse, TGACTCTAAGGTAAGGTGGCAGC; PPAR-γ, Forward, GGAGATCCTCCTGTTGACCC, Reverse, TGGGTCAGCTCTTGTGAACG; SCD-1, Forward, TCGTCAGCACCTTCTTGAGATA, Reverse, GTTGATGTGCCAGCGGTACT.
Statistical Analysis
Analyses using samples collected from experiments in vitro and in vivo were performed in triplicate and sextuplicate, respectively. Data were expressed as mean ± standard deviation, and analyzed with GraphPad Prism 8.0 software (Cary, NC, USA). Based on the one-way analysis of variance coupled with Tukey post hoc test, statistical differences were considered as significant when p values <0.05 or 0.01.
Results
Alkaloids and Polyphenols are the Major Bioactive Ingredients in QLY
Alkaloids are believed to be the major bioactive components in QLY, because Kushen, Qingfengteng and Huangbo are all rich in these compounds. The current non-targeted UPLC/MS2 analysis supported this notion. In total, 27,987 and 27,620 signals were detected under the negative and positive ion modes, respectively. According to the identification criterion described above, 195 and 267 compounds in the two TICs were identified, respectively. Detailed information about their chemical names/formulas, composite scores, categories, m/z, retention times and relative intensities were included in Supplementary S1. As shown in Figure 1, abundant ion fragments were detected under the positive rather than negative mode, which was consistent to the ionization pattern of alkaloids. Compounds 1–7 annotated in the positive TIC represented cytisin, matrine, sophocarpine, higenamine, oxysophocarpine, isosakuranetin and berberine, respectively. Meanwhile, polyphenols were the dominant compounds detected under negative mode. Most of them belong to flavonoids and phenylpropanoids categories. The representative chemicals with high abundance included aucubin, salidroside, gentisic acid, 3-o-feruloylquinic acid, rosmarinic acid, calycosin, trifolirhizin and kurarinone (compounds 1–8).
QLY Altered Monocytes Status in AIA Rats
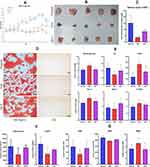
As anticipated, QLY alleviated AIA in rats with high efficacy. Since the onset of secondary inflammation, arthritis scores of QLY-treated AIA rats were constantly and significantly lower than AIA models (Figure 2A). At the end of observational period, arthritic manifestation was still vividly noticed in AIA rats. Extensive redness and severe edema occurred in almost all the paws. Although QLY did not completely cure the inflammation, the severity was significantly alleviated (Figure 2B). This treatment significantly reduced paw edema in AIA rats, but barely affected the spleen hyperplasia (Figure 2C). Serological levels of MCP-1, IL-1β and IL-6 were substantially increased in AIA models, while the increase was subsequently suppressed by QLY (Figure 2D). It preliminarily suggested the inhibition of QLY on monocytes/macrophages-controlled inflammations, which was further supported by PCR analysis. Expression of mRNA iNOS, IL-1β, IL-10 and SIRT1 in WBCs from AIA rats was altered towards to inflammatory phenotype. QLY efficiently suppressed iNOS and IL-1β expression, and promoted SIRT1 and PPAR-γ expression in the treated rats (Figure 2E). The decline in expression of iNOS and IL-1β supported the claim that QLY inhibited M1 polarization of monocytes. Notably, the expression of all metabolic regulators namely NAMPT, SIRT1 and PPAR-γ were greatly affected by QLY (Figure 2E). Therapeutic effects of QLY treatment on AIA were further confirmed by the changes of RA-related diagnostic indicators. The abnormal increase in production of RF, CRP and anti-CCP antibody in AIA rats was substantially inhibited by QLY treatment (Figure 2F).
QLY Reshaped WAT Functions in vivo
Obvious restoration of body weight loss was observed in QLY-treated AIA rats during the later stage of treatment (Figure 3A). The perirenal fat was greatly shrunk in AIA rats, while QLY treatment restored it to certain extent (Figure 3B). The effect of QLY on fat reservoir was further validated by relative fat weight index (Figure 3C). Oil Red O staining-based histological examination found fewer fat distribution in WAT from AIA models, when compared with normal healthy and QLY-treated rats. Examination of HE-stained WAT sections revealed that the decreased fat reservoir was accompanied with shrunk adipocytes size, as well as increased immune cells infiltration. Comparatively, QLY-treated AIA rats exhibited reduced inflammatory infiltration and enlarged adipocytes size (Figure 3D). Subsequently, we conducted serological analysis to confirm the metabolism regulatory properties of QLY. Significant increase in levels of FFAs, LDL-C and T-CHOL was observed in AIA rats. To the opposite, TG was markedly reduced. Meanwhile, HDL-C was unchanged compared to the healthy controls. Generally, QLY treatment restored all these lipids changes. Besides, it reduced abnormally accumulated blood glucose in the treated rats (Figure 3E). These results indicated the disordered energy metabolism in AIA rats, and QLY showed potent capability in controlling these abnormalities. The decreased TG and fat reservoir basically confirmed impaired adipogenesis in AIA rats. Meanwhile, serological levels of adiponectin and leptin fluctuated in accordance with TG. QLY treatment reversed adipokines production decline, suggesting recovered WAT physiological functions (Figure 3F). Altered fat metabolism in AIA rats led to intensified oxidative stress, as observed in the deficiency of GSH and SOD with a concomitant increase in MDA. By easing the metabolic complication, QLY restored changes on these oxidative indicators (Figure 3G).
QLY Inhibited Pro-Inflammatory Macrophages Development in WAT
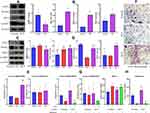
PCR analysis revealed the imbalanced macrophages polarization in WAT of AIA rats. It has been confirmed that SIRT1 and PPAR-γ drive M2 polarization.8,9 Decreased expression of mRNA SIRT1, PPAR-γ and IL-10, and increased expression of IL-1β and iNOS suggest that macrophages resided in AIA WAT tend to differentiate into inflammatory phenotype. QLY effectively promoted PPAR-γ expression in the treated rats, but exerted no obvious effects on NAMPT and SIRT1. It is known that PPAR-γ is predominantly expressed by (pre)-adipocytes, while NAMPT and SIRT1 are extensively distributed in many types of cells.17–19 It implied that QLY mainly targeted (pre)-adipocytes in WAT. Consistent with this hypothesis, the expression of SCD-1 an important PPAR-γ downstream and a regulator of adipogenesis was greatly up-regulated by QLY. Meanwhile, QLY effectively suppressed the expression of mRNA IL-1β and iNOS, although it did not restore the decline of IL-10, (Figure 4A). Similar results were obtained in Western blot assay (Figure 4B). SCD-1 was barely detected in WAT from either normal or AIA rats, but its intense expression was observed in QLY-treated AIA rats. As anticipated, substantial recovery of the decreased PPAR-γ expression in AIA rats was achieved under QLY treatment. Furthermore, the phosphorylation of a key regulator in M1 polarization JNK was inhibited by QLY (Figure 4C). All the uncropped images of Western blots were included in Supplementary S2. This result together with the findings stated in the last section suggest that QLY can promote PPAR-γ expression in both macrophages and adipocytes, and it has the potential to suppress M1 polarization and facilitate adipocyte differentiation.19 High levels of IL-1β and MCP-1 detected in WAT from AIA rats indicated inflammatory immune milieu there. This situation was greatly eased by QLY, and both IL-1β and MCP-1 were decreased after QLY treatment (Figure 4D). Accordingly, compared with normal healthy controls, more inflammatory CD86+CD11b+ macrophages were found in WAT from AIA rats, and QLY treatment significantly reduced their population (Figure 4E).
The Effects of QLY on (Pre)-Adipocytes Were Mediated by PPAR-γ Signaling
To enrich bioactive compounds in blood, some healthy rats received repeated oral QLY treatments within 2 h. Compared with the cells cultured by normal rat serum, expression of protein SCD-1, PPAR-γ, SIRT1 in pre-adipocytes incubated by QLY-containing serum was significantly increased, while NAMPT was markedly decreased (Figure 5A). Because short-time stimulus cannot substantially affect immunity in vivo, these changes were mainly elicited by chemicals within QLY. Hence, QLY-brought changes in vivo should be at least partially attributed to the chemical stimulus. It should be noted that the aforementioned changes were not the same with phenomena observed in vivo. QLY-containing serum down-regulated NAMPT expression in adipocytes in vitro, but it did not occur in QLY-treated AIA rats (Figure 5B). This discrepancy could be plausibly caused by the spontaneous alleviation of inflammation during the later stage of AIA, and it remaindered us that when investigating the metabolism regulatory properties of QLY, immune factors should be taken into consideration. Consequently, we co-cultured normal pre-adipocytes with PBMCs from different rats. Obtained results were similar to those observed in vivo (Figure 5C). PBMCs from QLY-treated rats up-regulated expressions of protein SCD-1 and PPAR-γ in the co-cultured pre-adipocytes, but it did not affect NAMPT and SIRT1 (Figure 5D). Expression of mRNA PPAR-γ was significantly increased too (Figure 5E). Together with clues from experiments in vivo, it collectively demonstrates that immune changes brought by QLY treatment were favorable for PPAR-γ expression in (pre)-adipocytes. Since PPAR-γ plays a decisive role in adipogenesis, we observed the changes of adipocyte differentiation afterward. After being cultured in differentiation medium, no obvious fat droplet was observed in AIA rat serum-cultured pre-adipocytes, whereas the differentiation process was accelerated by the serum obtained from QLY-treated AIA rats (Figure 5F). The rats used this assay did not receive QLY treatment on the day of sacrifice, and therefore the concentration of QLY-derived chemicals in serum was very limited after overnight fasting. As such, varied adipocyte differentiation potentials observed should be mainly mediated by immune milieu changes. Furthermore, we confirmed that QLY attenuated pre-adipocytes-mediated inflammation within WAT by up-regulating PPAR-γ using a selective inhibitor T0070907. Expression of mRNA PPAR-γ in pre-adipocytes was impaired by LPS. QLY-containing serum restored this decline, which was subsequently reverted by T0070907. Similar changes about SCD-1 expression were observed, despite the differences were not very significant (Figure 5G). Levels of adiponectin in culture medium fluctuated in a similar pattern with PPAR-γ expression. Of note, QLY-induced MCP-1 production decline was reverted by T0070907 too (Figure 5H).
Discussion
Treatments aiming at easing inflammatory symptoms have been used as the mainstream anti-rheumatic therapies for a long time. By the advance in knowledge about pathogenesis, the importance of immune homeostasis breakdown in RA becomes increasingly obvious. Accordingly, disrupting the malicious autoimmune feedback is deemed as a priority of RA treatments. Many players implicated in the development of immune abnormalities have been identified, including T/B lymphocytes, monocytes/macrophages, granulocytes, DC cells and so on.20 However, exact mechanisms driving their pathological changes are still largely elusive, and many theories have been proposed. Exogenous pathogens, genetic defects, environmental pollution, lifestyle and many other unidentified factors may contribute to the occurrence of RA.20 From this perspective, RA is a heterogeneous disease. Hence, developing an effective and universal anti-rheumatic regimen is very challenging. Almost all DMARDs including the advanced biological reagents cannot achieve satisfying clinical efficacy.21 On the other hand, irrespective of different pathogenesis, immunity as a whole relies on energy metabolism, and any metabolic interference will inevitably affect immune functions.22 It promises a novel anti-rheumatic strategy. Metabolic disorders have been extensively observed in RA patients.23 Although hyperglycemia as a typical metabolic complication of RA is still considered controversial, inflammation is fueled by accelerated aerobic glycolysis without any doubt.24 Thereby, the fact that certain reagents potent in controlling blood sugar possess notable anti-rheumatic potentials is reasonable.25 Fat metabolism alteration is very common in RA patients. Overweight can be noticed in over 30% RA patients, and most of RA patients develop dyslipidemia on the long run.26 Accordingly, antilipemic agents like fibrates have been verified with promising therapeutic effects on RA.27 These facts confirm that metabolic intervention is a feasible strategy to treat RA.
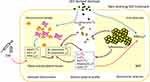
The notion about metabolism-physiological function connection is deeply rooted in the development of TCM theory, and restoring energy metabolism is an ultimate goal of TCM medication. Thus, it is not surprising to find that QLY treatment exerted notable impact on metabolism in rats with experimental arthritis.15 Accelerated fat oxidation has been identified as a key RA-related metabolic change.23 Thereby, we specifically investigated fat metabolism changes in this study. QLY promoted adipocytes differentiation both in vivo and in vitro (Figures 3 and 5). This finding is confusing, because many by-products from fat metabolism are believed to be harmful for the recovery of inflammation, and obesity has been identified a risk factor of RA.18,26 It hinted that the clinical implication of fat metabolism in RA is more complicated than previously thought. Indeed, accumulated fat derivatives could exacerbate inflammation by activating TLR4, promoting inflammatory mediator synthesis and potentiating monocytes/macrophages phagocytosis.28 But RA patients with poor prognosis usually suffer more fat loss, and well preserved fat reservoir promises slighter joint damages.29 The dynamic equilibrium of energy metabolism could be the key to address this lipid paradox. It is certain that fat utilization is accelerated under RA conditions. It meets high resting metabolic rate resulted from intense inflammation and is crucial to replenish accelerated aerobic glycolysis-depleted blood glucose.23,28 From this perspective, increased fat deposition serves as an indicator of reduced energy requirement and inflammatory reactions. According to this theory, QLY-promoted adipocyte differentiation would benefit RA remission by reducing circulating lipids and suppressing gluconeogenesis (Figure 6). This speculation was supported by the declined blood glucose observed in QLY-treated AIA rats (Figure 3E).
NAMPT/SIRT1 controls monocytes/macrophages polarization, and medication targeting this signaling can achieve satisfying anti-rheumatic effects.7,8 Therefore, this pathway was thought to be the main target of QLY bridging its metabolism regulatory and anti-rheumatic properties. As expected, both NAMPT and SIRT1 expressions in WBCs were regulated by QLY; however, there were no changes observed in WAT (Figures 2 and 4). It implied that QLY selectively regulated NAMPT/SIRT1 in immune cells but not in (pre)-adipocytes. In contrast, PPAR-γ expression in both WBCs and (pre)-adipocytes was promoted by QLY. This universal effect on PPAR-γ could have even more profound influence. Indeed, the previous network pharmacology prediction also revealed PPAR-γ as a main target of QLY.15 PPAR-γ activation always attenuates arthritis.30 Interestingly, the hypoglycemic drug with anti-rheumatic potential rosiglitazone has been identified as a PPAR-γ agonist.25 This signaling can cripple the transcriptional activities of NF-κB and AP-1, and thereby hampers M1 polarization.19 This study further confirmed that PPAR-γ activation in (pre)-adipocytes likewise improved RA prognosis. By up-regulating PPAR-γ, QLY facilitated adipogenesis and consequently altered energy metabolism as discussed above. Meanwhile, it reshaped secretion profile of WAT (Figure 6). Unfortunately, we still lack of some essential knowledge about physiopathological functions of WAT-released mediators. Most of adipokines are found to be positively correlated to rheumatic severity, but their role in RA is still controversial. Leptin seems protecting joints from damages.31 Adiponectin is usually defined as an anti-inflammatory factor, and its abnormal increase in RA patients could be also resulted from the immune re-balancing mechanism in the response to inflammation.31,32 Although chemokine MCP-1 is also categorized as an adipocyte nowadays, its production is suppressed by QLY-induced PPAR-γ expression.
Conclusions
QLY inhibited inflammatory polarization of monocytes/macrophages and facilitated adipocytes differentiation by up-regulating PPAR-γ expression in AIA rats. As a result, pathological interplay between inflammatory monocytes/macrophages and (pre)-adipocytes within WAT was disrupted. It consequently led to altered cytokines profile, and restored fat reservoir. Consequently, circulating lipids and pro-inflammatory cytokines were further reduced, which eventually contribute to the improvement situation of AIA.
Acknowledgments
This work was financially supported by National Natural Science Foundation of China (81973828 and 81173596), The Major Project of Natural Science Foundation of the Department of Education of Anhui province (KJ2019A0416 and KJ2020A0868), The Scientific Research Project of Health Commission of Anhui Province (AHWJ2021b061) and The Funding of “Peak” Training Program for Scientific Research of Yijishan Hospital, Wannan Medical College (GF2019J01).
Disclosure
The authors declare no conflicts of interest related to this work.
References
1. Cope AP. T cells in rheumatoid arthritis. Arthritis Res Ther. 2008;10:S1. doi:10.1186/ar2412
2. Toh ML, Miossec P. The role of T cells in rheumatoid arthritis: new subsets and new targets. Curr Opin Rheumatol. 2007;19:284–288. doi:10.1097/BOR.0b013e32805e87e0
3. Levesque D. Biologic rheumatoid arthritis therapies. Biodrugs. 2012;26:65–70. doi:10.2165/11631320-000000000-00000
4. Davignon JL, Hayder M, Baron M, et al. Targeting monocytes/macrophages in the treatment of rheumatoid arthritis. Rheumatology. 2013;4:590–598. doi:10.1093/rheumatology/kes304
5. Petrovic-Rackov L, Pejnovic N. Clinical significance of IL-18, IL-15, IL-12 and TNF-α measurement in rheumatoid arthritis. Clin Rheumatol. 2006;25:448–452. doi:10.1007/s10067-005-0106-0
6. Ali L, Schnitzler JG, Kroon J. Metabolism: the road to inflammation and atherosclerosis. Curr Opin Lipidol. 2018;29:474–480. doi:10.1097/MOL.0000000000000550
7. Travelli C, Colombo G, Mola S, et al. NAMPT: a pleiotropic modulator of monocytes and macrophages. Pharmacol Res. 2018;135:25–36. doi:10.1016/j.phrs.2018.06.022
8. Park SY, Lee SW, Kim HY, et al. SIRT1 inhibits differentiation of monocytes to macrophages: amelioration of synovial inflammation in rheumatoid arthritis. J Mol Med. 2016;94:921–931. doi:10.1007/s00109-016-1402-7
9. Abdalla HB, Napimoga MH, Lopes AH, et al. Activation of PPAR-γ induces macrophage polarization and reduces neutrophil migration mediated by heme oxygenase 1. Int Immunopharmacol. 2020;84:106565. doi:10.1016/j.intimp.2020.106565
10. Afeltra A, Abbate A, Valentini G, et al. Inflammation and dysmetabolism in systemic autoimmune diseases. J Immunol Res. 2019;2019:5438287. doi:10.1155/2019/5438287
11. Kammoun HL, Kraakman MJ, Febbraio MA. Adipose tissue inflammation in glucose metabolism. Rev Endocr Metab Disord. 2014;15:31–44. doi:10.1007/s11154-013-9274-4
12. Zhang B, Wang X, Li S. An integrative platform of TCM network pharmacology and its application on a herbal formula, Qing-Luo-Yin. Evid Based Complement Alternat Med. 2013;2013:456747.
13. Li S, Lu AP, Wang YY, et al. Suppressive effects of a Chinese herbal medicine Qing-Luo-Yin extract on the angiogenesis of collagen-induced arthritis in rats. Am J Chin Med. 2003;31:713–720. doi:10.1142/S0192415X03001430
14. Li Y, Li R, Ouyang Z, et al. Herb network analysis for a famous TCM doctor’s prescriptions on treatment of rheumatoid arthritis. Evid Based Complement Alternat Med. 2015;2015:451319.
15. Zuo J, Wang X, Liu Y, et al. Integrating network pharmacology and metabolomics study on anti-rheumatic mechanisms and antagonistic effects against methotrexate-induced toxicity of Qing-Luo-Yin. Front Pharmacol. 2018;2018:1472. doi:10.3389/fphar.2018.01472
16. Zuo J, Tao MQ, Wu XY, et al. Securidaca inappendiculata-derived xanthones protected joints from degradation in male rats with collagen-induced arthritis by regulating PPAR-γ signaling. J Inflamm Res. 2021;14:395–411. doi:10.2147/JIR.S295957
17. Eming SA, Wynn TA, Martin P. Inflammation and metabolism in tissue repair and regeneration. Science. 2017;356:1026–1030. doi:10.1126/science.aam7928
18. Zhang C, Wang K, Yang L, et al. Lipid metabolism in inflammation-related diseases. Analyst. 2018;143:4526–4536. doi:10.1039/C8AN01046C
19. Harmon GS, Lam MT, Glass CK. PPARs and lipid ligands in inflammation and metabolism. Chem Rev. 2011;111:6321–6340. doi:10.1021/cr2001355
20. McInnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Engl J Med. 2011;365:2205–2219. doi:10.1056/NEJMra1004965
21. Woodworth TG, Broeder AD. Treating to target in established rheumatoid arthritis: challenges and opportunities in an era of novel targeted therapies and biosimilars. Best Pract Res: Clin Rheumatol. 2015;29:543–549. doi:10.1016/j.berh.2015.10.001
22. Schuster S, Ewald J, Kaleta C. Modeling the energy metabolism in immune cells. Curr Opin Biotechnol. 2021;68:282–291. doi:10.1016/j.copbio.2021.03.003
23. Yang Y, Wei J, Li J, et al. Lipid metabolism in cartilage and its diseases: a concise review of the research progress. Acta Biochim Biophys Sin. 2021;53:517–527. doi:10.1093/abbs/gmab021
24. Yang Z, Matteson EL, Goronzy JJ, et al. T-cell metabolism in autoimmune disease. Arthritis Res Ther. 2015;17:29. doi:10.1186/s13075-015-0542-4
25. Kwon EJ, Park EJ, Choi S, et al. PPARγ agonist rosiglitazone inhibits migration and invasion by downregulating Cyr61 in rheumatoid arthritis fibroblast-like synoviocytes. Int J Rheum Dis. 2017;20:1499–1509. doi:10.1111/1756-185X.12913
26. Onuora S. Rheumatoid arthritis. How bad is obesity for RA? Nat Rev Rheumatol. 2012;8:306. doi:10.1038/nrrheum.2012.78
27. van Eekeren IC, Clockaerts S, Bastiaansen-Jenniskens YM, et al. Fibrates as therapy for osteoarthritis and rheumatoid arthritis? A systematic review. Ther Adv Musculoskelet Dis. 2013;5:33–44. doi:10.1177/1759720X12468659
28. Fullerton JN, O’Brien AJ, Gilroy DW. Lipid mediators in immune dysfunction after severe inflammation. Trends Immunol. 2014;35:12–21. doi:10.1016/j.it.2013.10.008
29. Baker JF, Billig E, Michaud K, et al. Weight loss, the obesity paradox, and the risk of death in rheumatoid arthritis. Arthritis Rheumatol. 2015;67:1711–1717. doi:10.1002/art.39136
30. Giaginis C, Giagini A, Theocharis S. Peroxisome proliferator-activated receptor-γ (PPAR-γ) ligands as potential therapeutic agents to treat arthritis. Pharmacol Res. 2009;60:160–169. doi:10.1016/j.phrs.2009.02.005
31. Scotece M, Conde J, Gómez R, et al. Beyond fat mass: exploring the role of adipokines in rheumatic diseases. Sci World J. 2011;11:1932–1947. doi:10.1100/2011/290142
32. Senolt L, Pavelka K, Housa D, Haluzík M. Increased adiponectin is negatively linked to the local inflammatory process in patients with rheumatoid arthritis. Cytokine. 2006;35:247–252. doi:10.1016/j.cyto.2006.09.002
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.