Back to Journals » OncoTargets and Therapy » Volume 12
Pyrotinib treatment on HER2-positive gastric cancer cells promotes the released exosomes to enhance endothelial cell progression, which can be counteracted by apatinib
Authors Gao Z , Song C, Li G , Lin H, Lian X, Zhang N, Cao B
Received 15 November 2018
Accepted for publication 12 March 2019
Published 11 April 2019 Volume 2019:12 Pages 2777—2787
DOI https://doi.org/10.2147/OTT.S194768
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Geoffrey Pietersz
Zhengxing Gao,1 Chunqing Song,2 Guangxin Li,1 Haishan Lin,1 Xiangyao Lian,1 Ninggang Zhang,1 Bangwei Cao1
1Cancer Center, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, People’s Republic of China; 2Department of Oncology, Beijing Daxing District Hopeople’s Hospital, Capital Medical University, Beijing 102600, People’s Republic of China
Aims: Pyrotinib is a newly developed irreversible pan-ErbB receptor tyrosine kinase inhibitor for treatment of human epidermal growth factor receptor 2 (HER2)-positive cancers, and clinic trials of pyrotinib in treatment of HER2-positive gastric cancer (GC) are underway. Exosomes are tiny vesicles secreted by cancer cells and take essential roles in the progression of carcinoma. Whether pyrotinib application has any effect on the cancer cell-released exosomes has not been studied. The aim of our work was to address if pyrotinib treatment impacts the effect of HER2-positive GC cell-derived exosomes on endothelial cell (EC) progression.
Methods: Isolation of exosomes released by HER2-positive NCI-N87 and MKN45 lines after pyrotinib treatment was performed. Then, human umbilical vein endothelial cells (HUVECs) were incubated with different concentrations of exosomes to address their proliferation by 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS). Effect of pyrotinib-treated exosomes at concentration of 10 μg/mL was compared to that without pyrotinib treatment over 96-hr time course. Transwell assay and wound-healing assay were carried out by incubating with exosomes released by NCI-N87 and MKN45 cells with/without pyrotinib treatment over 24-hr time course. The aforementioned experiments were done under same conditions in order to evaluate the combined effect of apatinib and pyrotinib on HUVEC motility and invasive capacity.
Results: We showed that HUVEC proliferation, motility and invasive capacity were further enhanced upon incubation with exosomes released by pyrotinib-treated GC cell lines, compared to those without pyrotinib treatment. Significantly, this effect was counteracted by the vascular endothelial growth factor receptor (VEGFR)-2 inhibitor apatinib which inhibits EC progression.
Conclusion: Our study suggests that pyrotinib application on HER2-positive GC produces stronger exosomes that promote the proliferation and motility of vascular ECs, and combination of pyrotinib with apatinib provides potentially better therapy.
Keywords: pyrotinib, HER2, gastric cancer, GC, exosome, human umbilical vein endothelial cells, HUVEC, apatinib
Introduction
Human epidermal growth factor receptor 2 (HER2) family consists of four main members, namely HER1/erythroblastic leukemia viral oncogene homolog 1(ErbB1), HER2/ErbB2, HER3/ErbB3 and HER4/ErbB4.1 All of them have a cysteine-rich extracellular ligand-binding site, a trans-membrane lipophilic region and an intracellular domain with tyrosine kinase catalytic activity.2 HERs locate to the cell surface as monomers. But upon ligand binding to their extracellular domains, they undergo homo- or heterodimerization and autophosphorylation of tyrosine residues within the cytoplasmic domain and initiates various downstream signaling pathways, resulting in cell proliferation, survival, differentiation, angiogenesis and invasion.3 It has been well studied that HER2 takes essential roles in the pathogenesis of many human cancers.1 Enhanced expression of HER2 occurs in approximately 15–30% of breast cancers and 10–30% of gastric/gastroesophageal cancers, therefore serving as an important prognostic and predictive biomarker.4 To date, targeting HER2 provides an important strategy for HER2-positive cancer therapy, and many anti-HER2 drugs have been developed. They include HER2 antibody and its derivative, such as trastuzumab5 and pertuzumab;6 small molecular tyrosine kinase inhibitors (TKIs), such as lapatinib7 and afatinib;8,9 and antibody–drug conjugates such as trastuzumab-emtansine.10
Recently, a novel 3-cyanoquinoline derivative, pyrotinib, has been developed by Jiangsu Hengrui Pharmaceutical (Lianyungang, People's Republic of China) for treatment of advanced solid tumors with enhanced expression of HER2.11 Pyrotinib is a pan-erbB/TKI inhibitor that can bind to and inhibit EGFR (ErbB1/HER1) and ErbB2/HER2 with a half maximal inhibitory concentration (IC50) of 5.6 and 8.1 nM, respectively.12 China has recently approved the first global conditional use of pyrotinib in combination with capecitabine for the treatment of HER2-positive, advanced or metastatic breast cancer.12 In addition, Phase I and Phase II development for use of pyrotinib in HER2-positive gastric cancer (GC) is underway in China and the USA.13 Evaluation on the efficacy and safety of pyrotinib in other HER2-positive advanced solid tumors is also underway by some other studies (NCT02834936, NCT03480256, NCT02500199).12 Although promising in clinical trials, comprehensive studies on pyrotinib are in urgent need to help its application in target therapy.
Exosomes are small membrane-derived vesicles secreted by various normal or cancer cell types.14 They act as mediators of intracellular communication in both localized and remote microenvironments to condition premetastatic niche and metastasis development, therefore taking essential roles in tumor proliferation and metastasis.15 Exosomes contain mRNAs, microRNAs (miRNAs) as well as proteins that can be transferred to target cells, inducing various genetic and epigenetic alterations, and they are continuously secreted into the surrounding extracellular matrix and circulation through the fusion of multivesicular bodies with the plasma membrane.16,17 Nevertheless, by now our knowledge that how recipient cells recognize specific exosomal cargo involved in cancer progression is rather limited. It has been found that exosomes get involved in cell to cell communication such as morphogen and RNA transport between cells.18 In particular, cancer cells were shown to secrete a large amount of exosomes to affect cancer cell invasion and angiogenesis of the surrounding endothelial cells (ECs).19 In fact, the exosomes released by various cancer cells affect the immunological response, metastasis and niche formation of cancer cells as well as the angiogenesis around the tumor.14
Pyrotinib is currently being tested in clinical trials for HER2-positive GCs.12 Whether pyrotinib application can alter exosomes released by these cancer cells remains an interesting question. In the present study, we show that exosomes derived from pyrotinib-treated HER2-positive human GC cell lines, NCI-N87 and MKN45, have enhanced ability to promote HUVEC proliferation and increase their motility and invasiveness, compared to those without pyrotinib treatment. Significantly, we demonstrate that the increased progression of HUVEC after incubation with these exosomes is counteracted by angiogenesis-targeting apatinib,20 implicating a clinical combination of pyrotinib and apanitib for treatment of HER2-positive GCs.
Materials and methods
Cell culture and chemical treatment
The human GC cell line NCI-N87 was purchased from Procell company (Wuhan, China) and MKN45 was purchased from KeyGEN bioTECH (Jiangsu, China). The other cell lines, including SGC7901, MGC-803, BGC823, AGS and human umbilical vein endothelial cell (HUVEC),were maintained by the laboratory. All cell experiments were approved by the Ethics Committee of Beijing Friendship Hospital, Capital Medical University. All the cell lines were cultured in DMEM containing 10% FBS and grown in a humidified incubator at 37°C with 5% CO2,as described before.21 For pyrotinib treatment, NCI-N87 and MKN45 cells grown at 80% confluence were washed using PBS, and then changed to serum-free medium. The indicated concentration of pyrotinib was added to the cells for another 72-hr incubation before exosome extraction. For apatinib treatment, NCI-N87 and MKN45 cells were incubated with 60 mg/mL apatinib for 24 hrs.
Exosome isolation
Exosome extraction was carried out as described.21,22 Briefly, after the cells were treated by pyrotinib for 48 hrs, the medium was decanted, spinned at 2,000 ⨰ g for 10 mins to collect cells and then centrifuged again at 10,000 ⨰ g for 30 min to deplete cell debris. Exosomes were further extracted using ultracentrifugation at 120,000 g for 60 mins and the pellets were resuspended using PBS. After repeating two more times, the exosomes were finally resuspended in PBS and stored at −70 ºC for use. Exosome concentration was determined using bicinchoninic acid (BCA) method.
Transmission electron microscopy (TEM)
TEM was used to evaluate the formation of exosomes. Briefly, after isolation, exosomes were first diluted in PBS, then put on copper grids at room temperature (RT) for 1 min, then the grids were fixed with 2% glutaraldehyde at RT for 5 mins, followed by rinsing with PBS three times. The exosome samples were examined immediately using a transmission electron microscope (Hitachi, Japan).
Immunofluorescence
To label exosomes, a PKH67 Green Fluorescent Cell Linker Mini Kit (Sigma-Aldrich, St. Louis, MO, USA) was used following manufacturer's instruction. Then, labeled exosomes were incubated with HUVECs for 12–24 hrs before immunofluorescence was carried out following standard procedure. Briefly, cells were fixed with 4% paraformaldehyde and permeabilized with 0.25% Triton X-100, blocked with 1% BSA in Phosphate-buffered saline Tween 20 (PBST) and probed with primary antibodies against PKH67 (Sigma-Aldrich). Alexa Fluor 488-tagged secondary antibody (Cell Signaling, Cambridge, UK) was used for detection.The nuclei were stained with DAPI (Sigma-Aldrich) and imaged by a Leica confocal microscope.
Nanoparticle tracking analysis
A NanoSight NS500 instrument (Nanosight, Malvern, UK) equipped with 532 nm laser was used to measure particle size distribution and concentration of extracted exosomes. The particle Brownian motion was video-recorded at RT and nanoparticle tracking analysis was done using analytical software version 2.3. For each tracked particle, at least 10 individual measurements for 60 s, with a total of at least 5,000 tracks, were collected.
Western blot analysis
Total protein extraction and western blot were performed as described.21 Briefly, total proteins were extracted and quantified using the BCA assay (Thermo Scientific, Waltham, MA, USA). 20 μg proteins of each sample were separated by 10% SDS polyacrylamide gels and transferred to the nitrocellulose membrane. Then, it was blocked in TBST is the abbreviation for TBS+Tween. TBS is a Tris HCl buffer salt solution adjusted pH to 7.4 with 1N HC1 buffer with 5% fat-free milk for half an hour at RT and incubated with primary antibodies overnight at 4°C. The membranes were then incubated with HRP-conjugated secondary antibody (1:5,000, Immunoway, Cambridge, UK) at RT for 40 mins. The protein signals were detected using chemiluminescence (Millipore, Massachusetts, USA) and exposed to X-ray films. The primary antibodies used in this study are HER2 (1:1,000, Immunoway), CD9 (1:1,000, Abcam, Cambridge, UK), CD63 (1:1,000, Abcam), TSG101 (1:1,000, Abcam) and GAPDH (1:1,000, Abcam).
Cell proliferation assay
Cell proliferation was determined using MTS method described before.21 Briefly, HUVECs were seeded into 96-well plates (3×103 cells per well) and incubated 12 hrs before the medium was changed to serum-free DMEM. Then, the indicated concentration of exosomes was added to the cells. After incubation of the indicated time, the MTS reagents were added and the optical density was measured after 2 hrs.
Cell migration and invasion assays
Transwell assay was used to assess cell migration and wound-healing assay was done to assess cell invasion as described.21 For transwell assay, 1×105 HUVECs grown in serum-free medium (0.5 mL) were seeded in the upper chamber, then exosomes, apatinib or both were added. After 36 h the lower chamber was loaded with 0.8 ml medium supplemented with 10% FBS. After another 36 hrs of incubation at 37°C with 5% CO2, the migrated cells were stained by DAPI and then photographed and counted. For wound-healing assay, 5×105 HUVECs per well were plated on uncoated 6-well culture dishes. After incubating 24 hrs at 37°C, the samples were scratched using a pipette tip. Then, PBS was used to wash and remove the cell debris. The wounded cell samples were then covered by serum-free medium before addition of exosomes, apatinib, or both. After incubating 0 hrs and 24 hrs, images were taken and the migration rate was calculated. These assays were performed three times.
Statistical analysis
All statistical analyses were performed using SPSS 13.0 software. Data were shown as mean ± SD, and the significance of differences between two groups was determined by Student’s t test. All the tests were carried out in two-sided way.
Results
Pyrotinib treatment changes exosomes secreted by HER2-positive human GC cells thereby enhancing EC proliferation
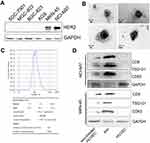
Pyrotinib is a novel irreversible pan-ErbB receptor tyrosine kinase inhibitor developed for treatment of HER2-positive cancers, and clinical trials are underway.11 Despite promising antitumor activity, little is known for the side effect of pyrotinib on HER2-positive cancers, especially GCs. To address this issue, we studied whether exosomes released by pyrotinib-treated HER2-positive GC cells have distinctive effect on HUVEC progression. First, using western blot, we confirmed two GC cell lines, NCI-N87 and MKN45, have higher HER2 protein level than other gastric cell lines, including SGC7901, MGC-803, BGC823 and AGS (Figure 1A). Then, we extracted exosomes released by NCI-N87 and MKN45 cells after pyrotinib treatment by differential ultracentrifugation. The isolated exosomes were then confirmed under TEM, and round, alveolate-shaped structures with diameter of 30−100 nm were observed (Figure 1B). We also performed nanoparticle tracking analysis study on the isolated exosomes, and the results indicated the quality in both size and concentration was good for experiments (Figure 1C). To further verify the exosome identity, we checked the exosomal markers including CD9, TSG101 and CD63, with GAPDH as loading control, using western blot. As shown in Figure 1D, CD9, TSG101 and CD63 were detected in the isolated exosomes from both NCI-N87 and MKN-45 cells. In addition, we could not detect these proteins in intact HUVECs, but after incubation with exosomes, expression of these markers became detectable (Figure 1D). These data support that extraction of exosomes is successful.
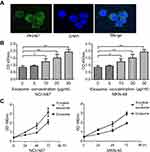
Next, we investigated if pyrotinib treatment changes the exosome ability of enhancing HUVEC proliferation capacity. To ensure successful intake of exosomes by HUVECs, we labelled the NCI-N87 exosomes using PKH67 green fluorescence linker, and then we used them to transfect HUVECs and checked under microscope; the results showed exosomes (green) accumulated in the cytoplasm surrounding the nucleus (blue), indicating successful transfection and intake of isolated exosomes by HUVECs (Figure 2A). We incubated of 2,000 HUVECs per well with different concentrations of exosomes from both GC cells on a 96-well plate. Then, we determined the proliferation of endothelial HUVECs by MTS assay. HUVEC proliferation was significantly increased upon their incubation with pyrotinib-treated exosomes at concentration of 10 µg/mL or more, in contrast to ECs with no exosome incubation (Figure 2B). Moreover, HUVEC proliferation increased in a time-dependent manner over 96 hrs of incubation with exosomes and was further boosted by incubating with exosomes from pyronitib-treated NCI-N87 and MKN45 cells (Figure 2C). Collectively, these data suggest that pyrotinib affects the cargo quality of GC cell-derived exosomes.
Exosomes derived from pyrotinib-treated human GC cells stimulates EC migration
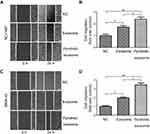
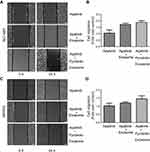
Then, wound-healing and transwell migration assays were carried out to study the effect of exosomes from NCI-N87 and MKN45 cells on the cell motility HUVECs. For the wound-healing assay,5×105 HUVECs per well were used by resuspending them in the migration buffer that contained 10 µg/mL exosomes released by NCI-N87 and MKN45 cells with/without pyrotinib treatment. Cells were photographed at 0 and 24 hrs after incubation under a time-lapse Olympus IX51 microscope (Olympus corporation, Tokyo, Japan). As shown by the micrographs (Figure 3A and C) and quantitative results (Figure 3B and D), the wound-healing assay indicated that incubation of HUVECs for 24 hrs with 10 µg/mL of exosomes released by NCI-N87 and MKN45 cells without pyrotinib treatment significantly promoted the motility of the ECs by 70.23% (P=0.0045) and 68.32% (P=0.0079), compared to HUVEC without exosome incubation. Moreover, after incubation with exosomes originating from pyrotinib-treated GC cells, the increase in HUVEC invasion capacity was further enhanced more than threefolds (P=0.0019 and P=0.0033, respectively). The difference between exosomes with or without pyrotinib treatment is also significant, suggesting pyrotinib incubation stimulated the exosome ability to promote HUVEC mobility.
Results from transwell cell migration assay also confirmed that the motility of HUVECs, 24 hrs after incubation with 10 µg/mL of exosomes secreted by NCI-N87 and MKN45 cells after pyrotinib treatment was significantly enhanced (Figure 4A and C), compared to HUVECs incubated with exosomes released by nontreatment GC cells. Compared to ECs incubated with 10 µg/mL exosomes originating from NCI-N87 and MKN45 cell lines without pyrotinib treatment, HUVECs incubated with the same concentration of exosomes from pyrotinib-treated NCI-N87 and MKN45 cells were migrating through the gluten gel by 30.01% (P=0.0159) and 28.25% (P=0.0135), respectively (Figure 4B and D). Altogether, both the wound-healing and transwell migration assay results suggest that the exosomes derived from pyrotinib-treated GC cells can increase EC motility and migration capacity.
Apatinib counteracts effects of exosomes secreted by pyrotinib-treated GC cells on HUVEC motility and migration
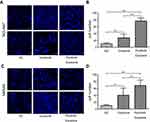
It has been reported that VEGFR-2 selective inhibitor apatinib counteracts the promotive effect of GC cell-released exosomes on EC survival.23 Therefore, we asked whether the apatinib can alter the effect of the pyrotinib-treated GC exosomes to promote HUVEC proliferation and migration capacity. As described before,21 60 µg/mL is the optimal apatinib concentration at which the division of HUVECs incubated with exosomes from treated GC is reduced by half (IC50). Therefore, we treated the HUVECs with 60 µg/mL apatinib for 24 hrs, then incubated them with exosomes derived from either pyrotinib-treated GC cells or those without treatment. We asked if apatinib can negatively affect the invasion properties of ECs incubated with exosomes from pyrotinib-treated GC cell lines. As expected, the difference in the migration capacity of HUVECs between no exosome treatment and exosome treatment was reduced in the presence of apatinib (Figure 5). Importantly, ability of the ECs to proliferate upon incubation with pyrotinib-treated exosomes was hindered, compared to ECs incubated with exosomes extracted from nontreated GC cell lines (Figure 5), as no obvious difference was detected.
Finally, the motility of HUVECs incubated with 20 µg/mL exosomes from NCI-N87 and MKN45 cancer cells without pyrotinib incubation showed no significant difference from the motility of HUVECs in the no exosome control (Figure 6). Despite significant difference being detected between incubation with pyrotinib-treated exosomes and no exosome (P=0.042 and 0.033, respectively), apatinib reduced the motility of the ECs as no significant difference was detected between incubations of exosomes with and without pyrotinib treatment (Figure 6). Together, these results suggest that the VEGFR-2 inhibitor apatinib conteracts pyrotinib-treated GC cell exosome enhancement of the EC motility and invasion capacities.
Discussion
Overexpression of HER2 protein is found in various tumor cells, and the signal transduction pathways it mediates have a close connection with carcinogenesis and with tumor progression and the prognosis.1 HER2 overexpression is also found in GC and associated with a poor prognosis.
Targeted therapy for HER2-positive cancers is deeply rooted in the current practice, and various TKI drugs have been widely used in the context of molecular detection,24 but studies on changes in exosomes before and after TKI treatment are lacking. Pyrotinib is a newly developed irreversible pan-ErbB receptor tyrosine kinase inhibitor for treatment of HER2-positive cancers, and several clinical trials in treating HER2-positive GC are undergoing, but comprehensive studies on pyrotinib effect on cancer cells are quite urgent. In the present study, we examined the changes of exosomes by pyrotinib treatment. We showed exosomes extracted from either pyrotinib-treated GC cells or those without treatment increase similarly EC proliferation and invasion. Strikingly, we find that exosomes derived from pyrotinib-treated GC cell lines obviously stimulate EC proliferation in a dose- and time-dependent manner (Figure 2). Moreover, 10 µg/mL of exosomes released by pyrotinib-treated NCI-N87 and MKN45 GC lines promote the EC motility and the invasiveness after 24 hrs of incubation (Figures 3 and 4). Therefore, pyrotinib treatment stimulates secretion of stronger exosomes to promote proliferation and migration in GC cells, which is consistent to the previous report that exosomes enhance HUVEC invasiveness.21,25 In addition, these results suggest that pyrotinib treatment may generate similar tumor microenvironment that allows cancer cells to survive and proliferate, which has a strong implication in the current clinical trials as well as future application. More attention should be paid to the therapeutic consequences on tumor progression which probably give cancer cells resistance to pyrotinib administration.
However, we also demonstrated this effect can be compensated by combined usage with VEGFR-2 selective inhibitor apatinib. As we showed in Figures 5 and 6, apatinib application attenuates the increased migration and invasiveness of HUVECs after incubation with exosomes secreted by pyrotinib-induced GC cells. Apatinib is the first approved oral targeted agent applied to advanced GC patients.26 It has been proved effective in inhibiting angiogenesis that is well known for its ability to coordinate EC development and regenerate new blood vessels during various cancer progression.16,27,28 In fact, apatinib has been demonstrated to be safe and well-tolerated in many case reports, and our work strongly indicated that a combination of apatinib and pyrotinib provides an improved strategy in target therapy. Therefore, our finding is probably meaningful for decreasing cell death rates and improving disease management, which awaits to be confirmed in future clinical trials.
Conclusion
In the current study, only cytology experiments have been carried out, and it remains unknown whether these effects exist in animals or even in human GCs. To know the underlying mechanism may provide better guidance for clinical application. For instance, determining changes in exosome content may help to understand how GC cells secrete these extracellular vesicles to mediate cell-to-cell transfer from cancer to normal cells. Additionally, the combinatory efficacy of pyrotinib with other developed target therapy method for GC treatment is also worthy of future studies. To conclude, the combination of apatinib with pyrotinib should be further investigated as a new strategy for the treatment of GC as well as other EGFR/HER2-dependent cancers.
Acknowledgments
This study was supported by grants from the CSCO-Hengrui Oncology Research Foundation (No. Y-HR2017-054, to Zhengxing Gao), the Special Fund for Science and Technology Supporting Social Progress of Daxing District Science and Technology Development Project in Beijing (No. KT201902314, to Zhengxing Gao).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Gerson JN, Skariah S, Denlinger CS, Astsaturov I. Perspectives of HER2-targeting in gastric and esophageal cancer. Expert Opin Investig Drugs. 2017;26(5):531–540. doi:10.1080/13543784.2017.1315406
2. Cho HS, Mason K, Ramyar KX, et al. Structure of the extracellular region of HER2 alone and in complex with the Herceptin Fab. Nature. 2003;421(6924):756–760. doi:10.1038/nature01392
3. Spivak-Kroizman T, Rotin D, Pinchasi D, Ullrich A, Schlessinger J, Lax I. Heterodimerization of c-erbB2 with different epidermal growth factor receptor mutants elicits stimulatory or inhibitory responses. J Biol Chem. 1992;267(12):8056–8063.
4. Iqbal N, Iqbal N. Human epidermal growth factor receptor 2 (HER2) in cancers: overexpression and therapeutic implications. Mol Biol Int. 2014;2014:852748. doi:10.1155/2014/852748
5. Pegram MD, Lipton A, Hayes DF, et al. Phase II study of receptor-enhanced chemosensitivity using recombinant humanized anti-p185HER2/neu monoclonal antibody plus cisplatin in patients with HER2/neu-overexpressing metastatic breast cancer refractory to chemotherapy treatment. J Clin Oncol. 1998;16(8):2659–2671. doi:10.1200/JCO.1998.16.8.2659
6. Gianni L, Llado A, Bianchi G, et al. Open-label, phase II, multicenter, randomized study of the efficacy and safety of two dose levels of Pertuzumab, a human epidermal growth factor receptor 2 dimerization inhibitor, in patients with human epidermal growth factor receptor 2-negative metastatic breast cancer. J Clin Oncol. 2010;28(7):1131–1137. doi:10.1200/JCO.2009.24.1661
7. Iqbal S, Goldman B, Fenoglio-Preiser CM, et al. Southwest oncology group study S0413: a phase II trial of lapatinib (GW572016) as first-line therapy in patients with advanced or metastatic gastric cancer. Ann Oncol. 2011;22(12):2610–2615. doi:10.1093/annonc/mdr021
8. Suzawa K, Toyooka S, Sakaguchi M, et al. Antitumor effect of afatinib, as a human epidermal growth factor receptor 2-targeted therapy, in lung cancers harboring HER2 oncogene alterations. Cancer Sci. 2016;107(1):45–52. doi:10.1111/cas.12845
9. Lin NU, Winer EP, Wheatley D, et al. A phase II study of afatinib (BIBW 2992), an irreversible ErbB family blocker, in patients with HER2-positive metastatic breast cancer progressing after trastuzumab. Breast Cancer Res Treat. 2012;133(3):1057–1065. doi:10.1007/s10549-012-2003-y
10. Krop IE, Beeram M, Modi S, et al. Phase I study of trastuzumab-DM1, an HER2 antibody-drug conjugate, given every 3 weeks to patients with HER2-positive metastatic breast cancer. J Clin Oncol. 2010;28(16):2698–2704. doi:10.1200/JCO.2009.26.2071
11. Li X, Yang C, Wan H, et al. Discovery and development of pyrotinib: A novel irreversible EGFR/HER2 dual tyrosine kinase inhibitor with favorable safety profiles for the treatment of breast cancer. Eur J Pharm Sci. 2017;110:51–61. doi:10.1016/j.ejps.2017.01.021
12. Blair HA. Pyrotinib: first global approval. Drugs. 2018;78(16):1751–1755.
13. Ma F, Li Q, Chen S, et al. Phase I study and biomarker analysis of Pyrotinib, a novel irreversible pan-ErbB receptor tyrosine kinase inhibitor, in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer. J Clin Oncol. 2017;35(27):3105–3112. doi:10.1200/JCO.2016.69.6179
14. Shen J, Zhu X, Fei J, Shi P, Yu S, Zhou J. Advances of exosome in the development of ovarian cancer and its diagnostic and therapeutic prospect. Onco Targets Ther. 2018;11:2831–2841. doi:10.2147/OTT.S159829
15. Becker A, Thakur BK, Weiss JM, Kim HS, Peinado H, Lyden D. Extracellular vesicles in cancer: cell-to-cell mediators of metastasis. Cancer Cell. 2016;30(6):836–848. doi:10.1016/j.ccell.2016.10.009
16. Suchorska WM, Lach MS. The role of exosomes in tumor progression and metastasis (review). Oncol Rep. 2016;35(3):1237–1244. doi:10.3892/or.2015.4507
17. Hessvik NP, Llorente A. Current knowledge on exosome biogenesis and release. Cell Mol Life Sci. 2018;75(2):193–208. doi:10.1007/s00018-017-2595-9
18. Lotvall J, Valadi H. Cell to cell signalling via exosomes through esRNA. Cell Adh Migr. 2007;1(3):156–158.
19. Ashiru O, Boutet P, Fernandez-Messina L, et al. Natural killer cell cytotoxicity is suppressed by exposure to the human NKG2D ligand MICA*008 that is shed by tumor cells in exosomes. Cancer Res. 2010;70(2):481–489. doi:10.1158/0008-5472.CAN-09-1688
20. Tian S, Quan H, Xie C, et al. YN968D1 is a novel and selective inhibitor of vascular endothelial growth factor receptor-2 tyrosine kinase with potent activity in vitro and in vivo. CancerSci. 2011;102(7):1374–1380. doi:10.1111/j.1349-7006.2011.01939.x
21. Li G, Lin H, Tian R, et al. VEGFR-2 inhibitor Apatinib hinders endothelial cells progression triggered by irradiated gastric cancer cells-derived exosomes. J Cancer. 2018;9(21):4049–4057. doi:10.7150/jca.25370
22. Muller L, Mitsuhashi M, Simms P, Gooding WE, Whiteside TL. Tumor-derived exosomes regulate expression of immune function-related genes in human T cell subsets. Sci Rep. 2016;6:20254. doi:10.1038/srep20254
23. Meadows KL, Hurwitz HI. Anti-VEGF therapies in the clinic. Cold Spring Harb Perspect Med. 2012;2:10. doi:10.1101/cshperspect.a006577
24. Kumler I, Tuxen MK, Nielsen DL. A systematic review of dual targeting in HER2-positive breast cancer. Cancer Treat Rev. 2014;40(2):259–270. doi:10.1016/j.ctrv.2013.09.002
25. Yukawa H, Suzuki K, Aoki K, et al. Imaging of angiogenesis of human umbilical vein endothelial cells by uptake of exosomes secreted from hepatocellular carcinoma cells. Sci Rep. 2018;8(1):6765. doi:10.1038/s41598-018-24563-0
26. Geng R, Song L, Li J, Zhao L. The safety of apatinib for the treatment of gastric cancer. Expert Opin Drug Saf. 2018;17(11):1145–1150. doi:10.1080/14740338.2018.1535592
27. Peng QX, Han YW, Zhang YL, et al. Apatinib inhibits VEGFR-2 and angiogenesis in an in vivo murine model of nasopharyngeal carcinoma. Oncotarget. 2017;8(32):52813–52822. doi:10.18632/oncotarget.17264
28. Jin Z, Cheng X, Feng H, et al. Apatinib inhibits angiogenesis via suppressing Akt/GSK3beta/ANG signaling pathway in anaplastic thyroid cancer. Cell Physiol Biochem. 2017;44(4):1471–1484. doi:10.1159/000485583
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.