Back to Archived Journals » Orphan Drugs: Research and Reviews » Volume 5
Profile of lumacaftor/ivacaftor combination: potential in the treatment of cystic fibrosis
Received 11 June 2015
Accepted for publication 14 July 2015
Published 14 August 2015 Volume 2015:5 Pages 61—68
DOI https://doi.org/10.2147/ODRR.S78413
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Lise Aagaard
Alexandria M Arends,1 Rebecca S Pettit2
1Department of Pharmacy, Indiana University Health, 2Department of Pharmacy, Riley Hospital for Children, Indianapolis, IN, USA
Objective: To review the cystic fibrosis transmembrane conductance regulator (CFTR) modulator ivacaftor in combination with lumacaftor for the treatment of cystic fibrosis (CF).
Data sources: Literature was accessed through MEDLINE (1977–June 2015) and national conference abstracts. Search terms included ivacaftor, VX-770, lumacaftor, VX-809, CFTR modulator, and CF.
Data synthesis: CF is an autosomal recessive genetic disease that affects chloride transport into and out of cells, causing the body to produce a thick, sticky mucus. There are over 1,800 CFTR mutations that have been identified and classified into six groups. CFTR modulators lumacaftor and ivacaftor are being studied in combination for the treatment of CF patients. This combination has proved beneficial for CF patients who are homozygous for the F508del mutation. In patients 12 years of age and older, the lumacaftor/ivacaftor combination demonstrated modest improvements in lung function with decreases in pulmonary exacerbation rate. There were clinically significant reductions in exacerbations, hospitalizations, and intravenous antibiotic requirement. Body mass index and Cystic Fibrosis Questionnaire-Revised scores were also positively impacted by therapy. A Phase I study evaluated the pharmacokinetics, safety, and tolerability of lumacaftor/ivacaftor in CF patients 6–11 years old. The combination is being further studied in this age group for clinical efficacy. The combination was generally well tolerated among all age groups, with the most common adverse events including headache and cough.
Conclusion: The use of lumacaftor in combination with ivacaftor in CF patients homozygous for the F508del mutation has shown improvement in percent predicted forced expiratory volume in 1 second and a decrease in the number of exacerbations. The combination was recently approved by the US Food and Drug Administration for homozygous F508del patients ≥12 years old. Studies are ongoing with this combination in younger patients as well as other CFTR modulators.
Keywords: cystic fibrosis, ivacaftor, lumacaftor, orkambi, VX-770, VX-809
Introduction
Cystic fibrosis (CF) is an autosomal recessive genetic disease that affects multiple organ systems throughout the body.1–3 Approximately 30,000 people in the United States and 70,000 people worldwide have been diagnosed with CF; however, about one in 20 Americans are unaffected carriers of this disease.1 CF is caused by a mutation in the gene that encodes for the CF transmembrane conductance regulator (CFTR) protein found on chromosome 7.4,5 Patients with CF have a shorter life expectancy due to disease-related complications; however, with the increase in research and development of new medications, the length and quality of life for CF patients have greatly improved.1
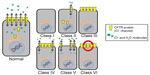
The CFTR protein functions as a chloride channel and is normally found throughout the body on epithelial cells.1–3 Mutations in the CF gene result in a reduction or elimination of CFTR protein production or loss of protein function. The decrease in chloride transport into and out of cells creates an imbalance of water movement throughout the tissues, causing the body to produce thick, sticky mucus.1,6 Primarily resulting in pulmonary and pancreatic disease, CF also affects the gastrointestinal tract, liver, and reproductive system.1–3 Due to the multiple organ systems affected throughout the body, patients with CF must take multiple medications to target each system individually, as there is currently not a single medication that addresses the underlying CFTR dysfunction.1 To complicate things even further, there are over 1,800 CFTR mutations that have been identified and classified into six groups based on the way the mutations affect the CFTR protein.4
The most common mutation affecting approximately 88.5% of the CF patients in the Cystic Fibrosis Patient Registry is F508del.4,7 This mutation falls under mutation class II, which primarily results in trafficking defects. Normal CFTR protein production includes a process of RNA transcription, splicing to form messenger (m)RNA, relocation from the nucleus to the endoplasmic reticulum (ER), translation into a protein and folding, and finally transport into the cell membrane.4,6 Class II mutations result in the CFTR protein not being properly folded and staying within the ER, where it will eventually degrade. The other mutation classes include the presence of premature termination codons resulting in defective or absent protein synthesis (class I), abnormal channel gating resulting in open time reduced due to the lack of CFTR protein activation (class III), presence of cell surface proteins but decreased transport of chloride ions (class IV), promoter or splicing abnormalities resulting in decreased synthesis and surface expression of CFTR proteins (class V), and accelerated turnover due to C-terminus mutations (class VI) (Figure 1).4,6
Lumacaftor ([VX-809]; Vertex Pharmaceuticals Inc., Boston, MA, USA) and ivacaftor (Kalydeco [VX-770]; Vertex Pharmaceuticals Inc.,) are two medications targeted toward CFTR defects. This paper reviews the combination of these two CFTR-modulating drugs and their potential role in therapy specifically for CF patients with the F508del mutation. Research from the North American Cystic Fibrosis (NACF) conference as well as relevant primary studies and review articles addressing primary evaluative studies were included in this review.
Ivacaftor
The CFTR potentiator, ivacaftor (Kalydeco), was initially approved by the US Food and Drug Administration (FDA) in January 2012 for the treatment of CF patients 6 years of age and older who have the G551D mutation (class III).8 This novel medication was the first of its kind to be approved by the FDA and allows for increased chloride transport by increasing the time the CFTR protein channel remains open.8 Two studies that served as the basis for approval, the ENVISION and STRIVE trials, demonstrated improvement in pulmonary function (forced expiratory volume in 1 second [FEV1]), reduction in sweat chloride, increase in body weight, and increase in quality of life scores in patients with the G551D mutation.9,10 Decreased pulmonary exacerbations were also observed in the STRIVE trial.9 Ivacaftor not only improved lung function, but also resulted in longer exacerbation-free periods.9
Despite this breakthrough in CF therapy, the G551D mutation affects only 4% of CF patients. Ivacaftor was also studied in CF patients with the F508del mutation, with hopes to gain approval for a larger patient population.11 There was no significant change in FEV1 or sweat chloride compared to placebo when tested in the F508del patients. This is likely due to the lack of CFTR proteins located on the cell membrane, typical of a class II mutation.
Approved ivacaftor dosing in adults, adolescents, and children ≥6 years old is 150 mg orally twice daily, with fat-containing foods to enhance absorption.8 Ivacaftor is also now FDA approved in patients 2 years of age and older who have one of the following mutations: G551D, G1244E, G1349D, G178R, G551S, S1251N, S1255P, S549N, S549R, and R117H.8,11 The KIWI trial evaluated the use of ivacaftor in children 2–5 years old.12 In children <14 kg, 50 mg every 12 hours orally, and in children ≥14 kg, 75 mg every 12 hours orally had a comparable area under the curve (AUC) and trough serum concentration (Cmin) with adult dosing.12 Available dosage forms include the 150 mg tablet as well as a 50 and 75 mg packet. The packets are to be mixed with 1 teaspoon (5 mL) of soft food or liquid.8
There are no dose adjustments recommended for those with renal impairment; however, ivacaftor should be used with caution in patients with creatinine clearance less than 30 mL/min or end-stage renal disease.8 Dose adjustment to 150 mg daily is recommended in patients with Child–Pugh class B impairment as ivacaftor metabolism is primarily hepatic through CYP3A.8 It is not recommended for use with strong CYP3A inducers.8 When administered with strong CYP3A inhibitors, the dose should be reduced to 150 mg twice a week, and 150 mg daily with moderate inhibitors.8 Grapefruit juice and Seville oranges should also be avoided due to metabolism interactions.8
The most common adverse effects (AEs) observed are headache (24%), oropharyngeal pain (22%), upper respiratory tract infection (22%), nasal congestion (20%), abdominal pain (16%), nasopharyngitis (15%), diarrhea (13%), rash (13%), nausea (12%), and dizziness (9%).8 Serious AEs include transaminase elevations (6%).8 Cataracts have also been reported in pediatric patients up to 12 years of age who received ivacaftor.8 While other risk factors were present in some cases, it is recommended that baseline and follow-up ophthalmological exams are performed in those receiving treatment.
Lumacaftor
Lumacaftor is another CFTR-modifying drug showing promising results in patients with the F508del mutation. Lumacaftor has shown efficacy in vitro to correct the folding and processing of CFTR, allowing for mature proteins to exit the ER and move to the cell surface.13 The rationale behind this mechanism was that with increased cell surface proteins, there would be an increase in chloride transport, and thus improved lung function, fewer CF exacerbations, and improved quality of life. When used in cultured human bronchial epithelial cells, overall F508del CFTR channel function was restored to approximately 14% of normal levels; however, it was unknown how this would translate within the clinical setting.13 It is also important to note that while improvements in chloride transport were seen in vitro, the underlying defect is not corrected. In contrast, a second in vitro study found no improvement in F508del-CFTR function with lumacaftor therapy.14 These latter results are similar to those seen in clinical trials. Unfortunately, when lumacaftor was studied in adult CF patients with homozygous F508del mutation, the only statistically significant difference observed was in mean changes in sweat chloride.15 There was no difference in pulmonary exacerbation rate, change in FEV1, or change in the Cystic Fibrosis Questionnaire-Revised (CFQ-R) respiratory domain scores.15
Doses of 25, 50, 100, and 200 mg orally daily have been studied in adult CF patients.15 Due to a dose-related decrease in sweat chloride values and no observed plateau at higher doses, it is likely that a dose greater than 200 mg may be needed. Current studies are evaluating doses as high as 600 mg daily. Lumacaftor was generally well tolerated, with no significant difference in the rate of AEs compared to placebo at any dose. Headache and cough were the most common AEs noted.13 In one Phase II trial, two patients experienced increased liver transaminase levels.15
Pharmacokinetic (PK) data showed slow oral absorption, Cmax within 3–4 hours, and a terminal half-life of approximately 24 hours allowing steady state to be achieved in 7 days.15 Lumacaftor is an inducer of cytochrome P450 CYP3A. The majority of lumacaftor is eliminated in the feces.
Lumacaftor plus ivacaftor
Based on the mechanism of action of each medication individually, the combination of both lumacaftor and ivacaftor would allow for increased trafficking of CFTR proteins to the cell surface (lumacaftor) and amplified chloride transport as a result of increased channel opening (ivacaftor). From this theory, it was hypothesized that this combination medication would work not only for patients who are homozygous with the F508del mutation in which there is decreased processing and trafficking of CFTR to the cell surface, but also for patients who are heterozygous with both the F508del and the G551D mutations, in which there is not only a processing and trafficking defect but also a gating defect (low-open probability). In vitro data of the combination therapy demonstrated that when F508del-CFTR channels were exposed to both lumacaftor and ivacaftor, channel expression was increased at the membrane and allowed for maximally potentiated activity.14 This research further supported the addition of lumacaftor to ivacaftor as a viable therapy for patients who have the F508del mutation. The lumacaftor/ivacaftor combination was initially studied within both heterozygous and homozygous F508del populations in Phase II trials. Results showed clinically significant improvements within the homozygous group (see “Outcomes in homozygous F508del patients ≥12 years old” section); however, little benefit was seen within the heterozygous population (Table 1).16 To better understand the potential role of lumacaftor/ivacaftor combination therapy in CF patients, a Phase II study was conducted with the primary purpose of assessing use in those who are heterozygous for the F508del mutation.16
Safety, tolerability, and efficacy were evaluated in a randomized trial where patients received either lumacaftor 400 mg every 12 hours in combination with ivacaftor 250 mg every 12 hours or placebo for 56 days.16 Patients were included if they had the F508del mutation on one allele plus a second allele with a mutation that either resulted in a lack of CFTR production or was expected to be nonresponsive to ivacaftor. In both the treatment and the placebo groups, the absolute change in baseline percent predicted that FEV1 was negative, and there was no significant difference between the two groups at day 56 (−0.6 vs −1.2, respectively; P=0.6). While there was a statistically significant mean decrease from baseline in sweat chloride concentrations observed in the combination therapy group compared to placebo (P<0.0001), there was no significant correlation between absolute change from baseline in percent predicted FEV1 and change in sweat chloride concentrations among individual patients. It is important to note that sweat chloride levels were still >60 mmol/L during and after treatment, which is diagnostic for CF.17 Furthermore, other findings included improvement in the CFQ-R scores for the combination group compared to the placebo group (P=0.01) and no change in body mass index (BMI) or body weight (P=0.26 and 0.37, respectively). AEs were seen in approximately 84% of both populations, with most being categorized as mild or moderate in severity. The AEs that occurred with greater frequency in the combination therapy group included abnormal respiration, dyspnea, and gastroesophageal reflux disease. In contrast, the AEs that occurred with greater frequency in the placebo group included headache, nasopharyngitis, decreased appetite, and sinus congestion. Overall, there was no benefit observed with the lumacaftor/ivacaftor combinations in patients who were heterozygous for the F508del mutation. The improvement in sweat chloride concentrations showed that some correction of the CFTR defect may be taking place despite no improvement in percent predicted FEV1 change; however, this is not clinically significant.
Outcomes in homozygous F508del patients ≥12 years old
The TRAFFIC and TRANSPORT studies are two of the Phase III randomized controlled trials that evaluated the efficacy and safety of the lumacaftor/ivacaftor combination in CF patients ≥12 years old who were homozygous for the F508del mutation (Table 1).18 In these studies, patients were randomized to receive lumacaftor 600 mg once daily in combination with ivacaftor 250 mg every 12 hours, lumacaftor 400 mg every 12 hours in combination with ivacaftor 250 mg every 12 hours, or matched placebo. Primary end point was the absolute change from baseline in percent predicted FEV1 at 24 weeks. Secondary end points included change in BMI, CFQ-R, proportion of patients with ≥5% and ≥10% increase in relative change from baseline in percent predicted FEV1 at 16 and 24 weeks, and assessment of pulmonary exacerbation. Safety and tolerability were also assessed. Those who completed the study were eligible to participate in the extension study, PROGRESS.18 A statistically significant increase was seen in percent predicted FEV1 within 2 weeks of initiation of both dosing regimens in comparison to placebo (absolute change from baseline: +3.3% for lumacaftor 600 mg daily and ivacaftor 250 mg twice daily, P<0.0001; +2.8% for lumacaftor 400 mg twice daily and ivacaftor 250 mg twice daily, P<0.0001). This improvement was maintained throughout the PROGRESS study up to 48 weeks.18 Furthermore, approximately twice as many patients in the treatment groups achieved ≥5% and ≥10% improvements compared to placebo. Subgroup analyses were performed based on age, sex, and baseline percent predicted FEV1. These data indicated that the improvement observed with both dosing regimens was largely consistent across all patient groups.
Pulmonary exacerbations occurred significantly more frequently in the placebo group compared to both treatment groups.18 The reduction of exacerbations was 30%–39% in the lumacaftor 600 and 400 mg groups, respectively. Not only did the treatment reduce exacerbations, but it also resulted in a decrease in hospitalizations of 39% for lumacaftor 600 mg and 61% for lumacaftor 400 mg, and requiring treatment with intravenous (IV) antibiotics (45% and 56% reduction, respectively). The mean absolute BMI continued to increase for both treatment groups throughout the course of the study and was statistically significant compared to placebo results. CFQ-R scores at 24 weeks were increased by 3.06 points versus placebo for lumacaftor 600 mg (P=0.0071) and by 2.22 points for lumacaftor 400 mg (P=0.0512). The main adverse events noted were pulmonary exacerbations and cough. Elevated liver enzymes (LFTs [liver function tests]) were observed in 5.2% of active drug patients compared to 5.1% of placebo patients. No serious AEs related to abnormal liver tests occurred in the placebo group; however, of seven patients who received active drug, six patients’ LFTs returned to baseline upon discontinuation of therapy.
The findings of these studies demonstrated modest improvements in lung function with decreases in pulmonary exacerbation rate for both doses of the lumacaftor/ivacaftor combination in CF patients ≥12 years old who are homozygous for the F508del mutation.18 The extension study demonstrated maintained response up to 48 weeks. There were also clinically significant reductions in exacerbations, hospitalizations, and IV antibiotic requirement. BMI and CFQ-R scores were also positively impacted by therapy. All of these results occurred in combination with the standard of care treatments for CF patients.
When comparing percent predicted FEV1 results seen with the combination product against ivacaftor monotherapy in G551D patients, the improvement in lung function when using the combination product was substantially less (lumacaftor 400 mg twice daily in combination with ivacaftor 250 mg twice daily: +2.8% vs ivacaftor 150 mg twice daily: +10.5%).18,19 While this combination product targets a different patient population, the impact of this combination on percent predicted FEV1 is significantly less than ivacaftor monotherapy in G551D patients.19
Outcomes in homozygous F508del patients 6–11 years old
A Phase I study evaluated the PK, safety, and tolerability of lumacaftor/ivacaftor in CF patients homozygous for the F508del mutation and 6–11 years old (Table 1).20 Patients received lumacaftor 200 mg every 12 hours in combination with ivacaftor 250 mg every 12 hours for 14 days. They were further divided into age subgroups of 6–8 years old and 9–11 years old. Safety and tolerability were assessed through 24 weeks. Among the ten patients enrolled in the study, the concentration vs time profile for both lumacaftor and ivacaftor observed among patients 6–11 years old was similar to that seen in patients ≥18 years of age and older seen in previous studies. The AUC for lumacaftor in the pediatric population was also generally within the range of adult patients; however, the younger age group had a trend toward higher AUC values. The AUC of ivacaftor in the pediatric group was also consistent with previous adult data.
With regard to safety and tolerability, patients had a mean decline of about 10% in predicted FEV1 at 4 hours after administration on day 1.20 It took approximately 7 days to return to baseline. By day 28, the absolute difference of mean predicted FEV1 was +3.5%. AEs were similar among both age groups, with cough and headache being the most frequently reported. Other AEs included nasal congestion, upper abdominal pain, constipation, diarrhea, lymphadenopathy, and pyrexia. No AEs such as wheezing, dyspnea, bronchospasm, or respiratory chest tightness were observed with the initial decline in lung function.
Exposure of lumacaftor 200 mg every 12 hours and ivacaftor 250 mg every 12 hours in patients 6–11 years old who are homozygous for the F508del mutation was generally within range observed in adults.20 While there was an initial decrease in the mean predicted FEV1, patients returned to baseline by day 7. Furthermore, these patients were approximately 3% above baseline at the follow-up visits. These promising study results support the need for further research within the pediatric population. In addition, the combination should be studied in children 2–5 years of age. Starting CFTR-modifying therapy early in the disease process would theoretically decrease the amount of hospitalizations, decline in lung function, and improve quality of life in CF patients compared to starting therapy later in life, as there is not as much organ damage present.
Pharmacokinetic profile, drug interactions, and safety
Steady state plasma concentrations were generally achieved after 7 days of administration.21 When administered with fatty foods, the exposure to lumacaftor is approximately two times higher and that to ivacaftor is three times higher than when taken in a fasting state. Both medications are highly protein bound at 99%. Lumacaftor is not extensively metabolized and is excreted as mostly unchanged drug in the feces. It is, however, an inducer of cytochrome P450 CYP3A, of which ivacaftor is a substrate and also then excreted in the feces. As a result of the metabolism interaction between ivacaftor and lumacaftor, when administered together, the exposure to ivacaftor is reduced by more than 80%. Due to this significant decrease, the dose of ivacaftor for the combination tablet (250 mg orally twice daily) is increased compared to standard monotherapy (150 mg orally twice daily).
A Phase I drug–drug interaction study evaluated the effects of strong and moderate CYP3A inhibitors and a strong inducer of CYP3A on the PK of the lumacaftor/ivacaftor combination.22 This study demonstrated that there is likely no change in dose needed for either lumacaftor or ivacaftor when administered with moderate or strong inhibitors of CYP3A. However, when administered with a strong CYP3A inducer, there was reduced exposure of ivacaftor. Due to a potential reduction in efficacy, it is not recommended to administer lumacaftor/ivacaftor with strong CYP3A inducers. Furthermore, in vitro studies suggest that lumacaftor may also induce several other enzymes including CYP2B6, 2C8, 2C9, and 2C19 as well as inhibit 2C8 and 2C9.21 This is important to keep in mind as this combination therapy may have the potential to increase or decrease the serum concentrations of other medications that CF patients commonly receive, such as antibiotics, antifungals, proton pump inhibitors, and nonsteroidal anti-inflammatory drugs.
Of the 1,108 patients who received the lumacaftor/ivacaftor combination, most of the AEs reported were consistent with the disease.21 One death was reported during the TRAFFIC/TRANSPORT trial.18 This patient was a 24-year old female who developed a pulmonary exacerbation during the extension trial. She eventually required mechanical ventilation followed by extracorporeal membrane oxygenation. Most other serious AEs were related to exacerbations of CF that occurred in approximately 13% of patients on therapy vs 24% on placebo.21 Specific liver safety concerns were raised as a result of the ivacaftor monotherapy trials. There were no differences between the combination treatment group compared to the placebo in overall AEs thought to be liver related (5.4%–6% among all groups).
Approval status and future research
In May 2015, the FDA’s Pulmonary-Allergy Drugs Advisory Committee (PADAC) voted 12 to one to recommend that the FDA approve the lumacaftor/ivacaftor combination for use in people with CF aged 12 years and older homozygous for the F508del mutation.23 On July 2, 2015, the FDA approved Orkambi (lumacaftor/ivacaftor) as an immediate release fixed drug combination tablet for oral administration in patients with CF 12 years of age and older homozygous for the F508del mutation. The product contains lumacaftor 200 mg and ivacaftor 125 mg with a dose of 2 tablets by mouth every 12 hours (lumacaftor 400 mg and ivacaftor 250 mg).21,24 A reduced dose is recommended in patients with moderate or severe hepatic impairment and during the first week of treatment with concurrent strong CYP3A inhibitors. Warnings and precautions include liver-related events, and monitoring of transaminases and bilirubin should be performed at initiation, every 3 months during the first year, and annually thereafter. Additional monitoring of patients with a percent predicted FEV1 <40 is recommended during the initiation of therapy as chest discomfort, dyspnea, and abnormal respirations have been reported. As with ivacaftor monotherapy, the combination product also carries a cataract warning and baseline, and follow-up examinations are recommended in pediatric patients. The package insert outlines the multiple drug interactions that will need to be considered during regular use. Prescribers should be cognizant of these interactions as some necessitate lumacaftor/ivacaftor dose changes and others require other drug dose modifications.
Ivacaftor/lumacaftor combination is an exciting new treatment for a group of CF patients. However, there is ongoing research on other CFTR modulators that will hopefully treat an even larger number of CF patients. One corrector VX 661 has even greater affinity for the CFTR than lumacaftor, and is currently being studied in combination with ivacaftor in both homozygous and heterozygous F508del patients.25 This corrector as well as other CFTR modulators currently in studies make the treatment of CF an ever changing and exciting area.
Conclusion
Therapy targeted toward underlying mutations of CF is an area that is constantly evolving and expanding. These disease-modifying medications not only have the potential to improve pulmonary function, but also positively affect quality of life. The combination of lumacaftor with ivacaftor in CF patients homozygous for the F508del mutation has been shown to improve percent predicted FEV1 (however, not to the extent of ivacaftor monotherapy in G551D patients); decrease the number of exacerbations, hospitalizations, and use of IV antibiotics; increase body weight and BMI; and improve quality of life in patients ≥6 years old. Furthermore, this combination was well tolerated and associated with minimal adverse events.
Disclosure
The authors report no conflicts of interest in this work.
References
Cystic Fibrosis Foundation. What Is Cystic Fibrosis? 2014. Available from: http://www.cff.org/AboutCF. Accessed June 2, 2015. | |
O’Sullivan BP, Freedman SD. Cystic fibrosis. Lancet. 2009;373: 1891–1904. | |
Rowe SM, Miller S, Sorscher EJ. Cystic fibrosis. N Engl J Med. 2005;352:1992–2001. | |
Rogan MP, Stoltz DA, Hornick DB. Cystic fibrosis transmembrane conductance regulator intracellular processing, trafficking, and opportunities for mutation-specific treatment. Chest. 2011;139: 1480–1490. | |
Riordan JR, Rommens JM, Kerem B, et al. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science. 1989;245:1066–1073. | |
Welsh MJ, Smith AE. Molecular mechanisms of CFTR chloride channel dysfunction in cystic fibrosis. Cell. 1993;73:1251–1254. | |
Cystic Fibrosis Foundation. Patient Registry Annual Data Report 2013. Available from: http://www.cff.org/Our-Research/CF-Patient-Registry/. Accessed June 2, 2015. | |
Kalydeco™ (ivacaftor) [Product information]. Cambridge, MA: Vertex Pharmaceuticals Inc.; 2012. | |
Ramsey BW, Davies J, McElvaney G, et al. A CFTR potentiator in patients with cystic fibrosis and the G551D mutation. N Engl J Med. 2011;365:1663–1672. | |
Davies JC, Wainwright CE, Canny GJ, et al. Efficacy and safety of ivacaftor in patients aged 6 to 11 years with cystic fibrosis with a G551D mutation. Am J Respir Crit Care Med. 2013;187(11):1219–1225. | |
Flume PA, Liou T, Borowitz D, et al. Ivacaftor in subjects with cystic fibrosis who are homozygous for F508del-CFTR mutation. Chest. 2012;1:1–50. | |
Davies JC, Robertson S, Green Y, Rosenfeld M. An open-label study of the safety, pharmacokinetics, and pharmacodynamics of ivacaftor in patients aged 2 to 5 years with CF and CFTR gating mutation: the KIWI study. Poster presented at: The 28th Annual North American Conference of the Cystic Fibrosis Foundation, October 9–11, 2014, Atlanta, GA. | |
Van Goor F, Hadida S, Grootenhuis PD, et al. Correction of the F508del-CFTR protein processing defect in vitro by the investigational drug VX-809. Proc Natl Am Sci U S A. 2011;108(46):18843–18848. | |
Kopeikin Z, Yukesk Z, Yang H, Bompadre SG. Combined effects of VX-770 and VX-809 on several functional abnormalities on F508del-CFTR channels. J Cyst Fibros. 2014;13:508–514. | |
Clancy JP, Rowe SM, Accurso FJ, et al. Results of a phase IIa study of VX-809, an investigational CFTR corrector compound, in subjects with cystic fibrosis homozygous for the F508del-CFTR mutation. Thorax. 2012;67:12–18. | |
Rowe SM, McColley SA, Rietschel E, et al. Effect of 8 weeks of lumacaftor in combination with ivacaftor in patients with CF and heterozygous for the F508del-CFTR mutation. Poster presented at: The 28th Annual North American Conference of the Cystic Fibrosis Foundation, October 9–11, 2014, Atlanta, GA. | |
Farrell PM, Rosenstein BJ, White TB, et al. Guidelines for diagnosis of cystic fibrosis in newborns through older adults: cystic fibrosis foundation consensus report. J Pediatr. 2008;153(2):S4–S14. | |
Ramsey B, Boyle MP, Elborn S, et al. Effect of lumacaftor in combination with ivacaftor in patients with cystic fibrosis who are homozygous for F508del-CFTR: pooled results from the phase 3 TRAFFIC and TRANSPORT studies. Poster presented at: The 28th Annual North American Conference of the Cystic Fibrosis Foundation, October 9–11, 2014, Atlanta, GA. | |
Accurso FJ, Rowe SM, Clancy JP, et al. Effect of VX-770 in persons with cystic fibrosis and the G551D -CFTR mutation. N Engl J Med. 2010;363:1991–2003. | |
Rosenfeld M, Marigowda G, Liu F, Waltz D. Pharmacokinetics and safety of lumacaftor in combination with ivacaftor in patients aged 6–11 years with CF who are homozygous for F508del-CFTR. Poster presented at: The 28th Annual North American Conference of the Cystic Fibrosis Foundation, October 9–11, 2014, Atlanta, GA. | |
Food and Drug Administration. Clinical Briefing Document for the Pulmonary Allergy Drugs Advisory Committee Meeting. Lumacaftor/Ivacaftor Oral Tablets. Silver Spring, MD: Department of Health and Human Services, Food and Drug Administration; 2015. | |
Pawaskar D, Marigowda G, Waltz D, et al. The effect of ciprofloxacin, itraconazole, and rifampin on the pharmacokinetics of lumacaftor in combination with ivacaftor in healthy individuals. Poster presented at: The 28th Annual North American Conference of the Cystic Fibrosis Foundation October 9–11, 2014, Atlanta, GA. | |
Vertex Pharmaceuticals Incorporated. Food and Drug Administration Advisory Panel Voted 12 to 1 to Recommend Approval of ORKAMBI™ (lumacaftor/ivacaftor) to Treat People with Cystic Fibrosis Ages 12 and Older Who Have Two Copies of the F508del Mutation. Boston, MA: Vertex Pharmaceuticals Incorporated. Available from: http://investors.vrtx.com/releasedetail.cfm?ReleaseID=912730. Accessed May 12, 2015. | |
Orkambi™ (lumacaftor/ivacaftor) [Product information]. Boston, MA: Vertex Pharmaceuticals Inc.; 2015. | |
Clinical Trials.gov VX661 Studies. Available from: https://www.clinicaltrials.gov/ct2/results?term=VX661&Search=Search. Accessed June 8, 2015. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.