Back to Journals » International Journal of General Medicine » Volume 14
Prevalence of Hepatitis B Virus Among Pregnant Women on Antenatal Care Follow-Up at Mizan-Tepi University Teaching Hospital and Mizan Health Center, Southwest Ethiopia
Authors Asaye Z , Aferu T , Asefa A , Feyissa D , Regasa T , Kebede O , Feyisa D , Feyisa M
Received 13 November 2020
Accepted for publication 7 January 2021
Published 18 January 2021 Volume 2021:14 Pages 195—200
DOI https://doi.org/10.2147/IJGM.S292070
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Zufan Asaye,1 Temesgen Aferu,2 Adane Asefa,3 Desalegn Feyissa,2 Tolcha Regasa,2 Oliyad Kebede,2 Diriba Feyisa,2 Mulugeta Feyisa4
1Department of Statistics, College of Natural Sciences, Mizan-Tepi University, Mizan-Aman, Ethiopia; 2School of Pharmacy, College of Health Sciences, Mizan-Tepi University, Mizan-Aman, Ethiopia; 3School of Public Health, College of Health Sciences, Mizan-Tepi University, Mizan-Aman, Ethiopia; 4Department of Midwifery, College of Health Sciences, Selale University, Fiche, Ethiopia
Correspondence: Temesgen Aferu
School of Pharmacy, College of Health Sciences, Mizan-Tepi University, Mizan-Aman, Ethiopia
Email [email protected]
Background: Hepatitis B virus(HBV) infection is a global public health problem, even though its prevalence is disproportionately high in low- and middle-income countries. Mother-to-child transmission is a major route of HBV transmission in endemic areas. This study aimed to assess the prevalence of HBV and its determinants among pregnant women attending antenatal care at Mizan-Tepi University Teaching Hospital and Mizan Health Center, Southwest Ethiopia.
Methods: A cross-sectional study was conducted between January 13th 2020 and February 5th 2020 among 370 pregnant women. The sample size was proportionally allocated to each health institution according to the total pregnant women on antenatal care at the respective health institution and a consecutive sampling technique was used to select study participants. Serum hepatitis B surface antigen (HBsAg) was tested using a rapid diagnostic test. Data were analyzed using Statistical Package for Social Sciences (SPSS) software version 22. Multiple logistic regression analysis was done to identify the independent predictors of HBsAg serostatus at p-value < 0.05.
Results: Three hundred seventy of the total 375 pregnant women participated in the study resulting in a response rate of 98.7%. Twenty-two (5.9%) of the pregnant women screened were found positive for HBsAg (prevalence=5.9%; 95% CI: 3.9– 8.80%). History of contact with jaundice patients (AOR=9.87; 95% CI: 2.98– 32.65), sharing sharp materials (AOR=3.96; 95% CI: 1.23– 11.08) and history of multiple sexual partners (AOR=6.77; 95% CI: 2.44– 18.78) were significantly associated with Hepatitis B Virus infection.
Conclusion: The endemicity of hepatitis B virus seroprevalence is intermediate in the study settings. Factors associated with hepatitis B virus serostatus were behavioral; hence, modification of these factors may help to prevent the infection.
Keywords: hepatitis, hepatitis B, hepatitis B surface antigen, pregnant women
Introduction
Hepatitis B virus (HBV) infection is a global public health problem. The World Health Organization (WHO) estimated in 2015 that about 257 million people were living with chronic HBV infection worldwide and 900, 000 people had died mostly due to chronic complications such as cirrhosis and hepatocellular carcinoma.1 The burden of chronic HBV infection varies geographically. It is high (>8%) in Asia Pacific and sub-Saharan African regions and intermediate (2–8%) in North Africa and the Middle East, parts of Eastern and Southern Europe, parts of Latin America, and South Asia. Some countries in South America, Australia, Asia, Northern and Western Europe, Japan, and North America have low (<2%) burden of chronic HBV infection.2–4
In endemic areas, mother-to-child transmission of HBV contributed nearly half of the transmission routes of chronic HBV infections.5 Furthermore, most HBV-associated deaths among adults are secondary to infections acquired at birth or in the first five years of life. Individuals above the age of 5 years rarely develop chronic condition if infected.1,6 Thus, prevention of mother-to-child transmission is an essential step in reducing the global burden of chronic HBV.5 Although there are inconsistencies, studies have reported that maternal HBV infection is associated with preterm birth,7 miscarriage,8 gestational diabetes mellitus,9,10 pregnancy-induced hypertension and preeclampsia.11
The prevalence of HBV infection among pregnant women and its associated factors varies across studies12–17 and therefore studies from different settings are crucial to precisely measure the burden of the problem, and to make informed decisions. This study aimed to assess the prevalence of hepatitis B virus infection and its determinants among pregnant women attending antenatal care (ANC) services at Mizan-Tepi University Teaching Hospital and Mizan Health Center, Southwest Ethiopia.
Patients and Methods
Study Setting and Study Design
Health facility-based cross-sectional study was conducted in Mizan-Tepi University Teaching Hospital (MTUTH) and Mizan Health Center (MHC) between January 13th 2020 and February 5th 2020. The facilities are located in Mizan-Aman town 585 kilometers away from Addis Ababa, the capital city of Ethiopia.
Population
The source population was all pregnant women who were attending the ANC clinic of MTUTH and MHC. All pregnant women who were attending the ANC clinic of MTUTH and MHC and fulfilled the inclusion criteria were considered as the study population. Those women who were unable to communicate or were not volunteer to participate were excluded from the study.
Sample Size Determination and Sampling Procedure
The sample size required for this study was calculated using a single population proportion formula ( ) taking prevalence (p) of hepatitis B virus among pregnant women to be 9.2% (from a study conducted in Gambella, Southwest Ethiopia),17 3% margin of error (d) and
) taking prevalence (p) of hepatitis B virus among pregnant women to be 9.2% (from a study conducted in Gambella, Southwest Ethiopia),17 3% margin of error (d) and 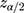 =1.96 at 95% confidence level. Adding 5% for none response, the final sample size for the study was calculated to be 375. A consecutive sampling technique was used to select the study participants. Care was taken not to reinterview pregnant women who were interviewed on the previous visit but came to the facility again during data collection.
=1.96 at 95% confidence level. Adding 5% for none response, the final sample size for the study was calculated to be 375. A consecutive sampling technique was used to select the study participants. Care was taken not to reinterview pregnant women who were interviewed on the previous visit but came to the facility again during data collection.
Data Collection Procedure
Data regarding socio-demographic characteristics (age, marital status, educational status, occupation), reproduction-related factors (parity, gravidity, history of abortion and home delivery), behavioral and medical-related factors (history of blood transfusion, history of contact with jaundice patient, sharing sharp materials, and history of multiple sexual partners) were collected using interviewer administered structured questionnaire.
Measurements
Serum hepatitis B surface Antigen (HBsAg) was checked using a rapid diagnostic test. Approximately 5 ml venous blood samples were collected from each participant in tubes not containing anticoagulant after disinfecting the puncture site. The skin was disinfected with alcohol skin wipe and allowed to get dry. The collected samples were stored at 2–8°C. Samples were centrifuged at 2000–3000 rpm for 5 minutes to separate the serum part of the whole blood. The separated serum with clear and good fluidity was tested for the presence of HBsAg. The HBsAg in serum was detected using HBsAg Rapid Test Cassette (ACON® Laboratories, Inc. San Diego, CA 92121, USA) as per the instructions of the manufacturer. Nitrocellulose membrane is pre coated with anti-HBsAg antibody on the test line region of the strip. After adding three drops of serum to sample pad, the serum migrates chromatographically on the membrane by capillary action to reach the test and control areas and forms a colored line. Results were interpreted after 15 minutes as positive (pink line on both test and control line) and negative (pink line on only control line).
Data Quality Control
The questionnaire was first prepared in English and translated to Amharic and retranslated back to English to ensure consistency of the message. Before the actual data collection, pretest was conducted on 5% of the sample size on nearly similar population outside the study area (at Chena Primary Hospital) to ensure validity of the data collection tool and to standardize the questionnaire. The collected data were checked by principal investigator daily for completeness and all the data were double-checked to ensure data quality. A standard procedure was also followed to collect blood samples and to process them for testing.
Data Processing and Analysis
The collected data were checked for completeness, coded, entered into Epi-data version 3.1 and exported to SPSS version 22 for analysis. Descriptive statistics were done for different variables as necessary. Simple logistic regression analysis was conducted to select variables for multiple logistic regression at a p-value less than 0.25. Finally multiple logistic regression model was fitted to identify the independent predictors of HBsAg positivity. Variables with a p-value less than 0.05 in the multiple logistic regression model were considered statistically significant predictors of HBsAg positivity. Model fitness was evaluated using the Hosmer-Lemeshow goodness of fit test.
Results
Prevalence of HBsAg Among Pregnant Mothers
Three hundred seventy of the total 375 pregnant women participated in the study resulting in a response rate of 98.7%. Twenty-two (5.9%) of the pregnant women screened for HBsAg were found positive (prevalence=5.9%; 95% CI: 3.9–8.80%) while 12 (7.8%) of the 153 (41.4%) women aged less than 25 years screened for HBsAg were found positive for the antigen. One hundred thirty-six (37.6%) of the study participants did not attend formal school and 9 (6.6%) of them were positive for HBsAg.
Multipara and primipara were experienced by 178 (48.1) and 113 (30.5%) of study participants respectively. The prevalence of hepatitis B among multipara and primipara was 8 (4.5) and 12 (10.6%) respectively. Forty-two (11.4%) of the study participants had experienced abortion, and the prevalence of hepatitis B among women who had a history of abortion was 3 (7.1%). Of the women who participated in the study, 49 (13.2%) had a history of blood transfusion, 25 (6.8%) had a history of contact with jaundice patients and 44 (11.9%) had a history of multiple sexual partners. The prevalence of HBsAg among women who had a history of blood transfusion and contact with jaundice patients was 5 (10.2%), and 8 (32.0%) respectively (Table 1).
 |
Table 1 Socio-Demographic Characteristics of Pregnant Women on ANC Follow-Up at MTUTH and MHC, 2019 |
Factors Associated with Hepatitis B Infection
Simple logistic regression analysis indicated that the age of respondents, occupation, education, history of abortion, history of home delivery and Human Immune Virus (HIV) serostatus had a p-value greater than 0.25 and hence they were excluded from the multiple logistic regression analysis. The result of multiple logistic regression analysis revealed that history of contact with jaundice patients, sharing sharp materials and history of multiple sexual partners were significantly associated with the risk of HBsAg among pregnant women (p< 0.05). Women who had a history of contact with jaundice patients were 9.87 times more likely to be seropositive for HBV infection compared to their counterpart. Respondents who shared sharp materials were 3.69 times more likely to be infected with HBV compared to those who did not share sharp materials with others. Those women who had a history of multiple sexual partners were also 6.77 times more likely to be seropositive for HBV compared those who had no history of multiple sexual partners (Table 2).
 |
Table 2 Factors Associated with Hepatitis B virus Infection among Pregnant Women on ANC Follow-Up at MTUTH and MHC, 2019 |
Discussion
Maternal HBV infection is an implication for the transmission of the virus to a newborn, and mother to child transmission of this virus is the major transmission route of chronic HBV infection. Thus, this study aimed to assess the prevalence of hepatitis B virus infection and factors associated with it among pregnant women attending the ANC clinic of MTUTH.
The study showed that the prevalence of HBV among the participants was 5.9%. This finding is almost similar to the reports from studies done in Vientiane Laos (5.44%),18 West Hararghe Zone, Ethiopia (6.1%)19 and Northern Tanzania (5.7%).10 But it is lower than the reports of studies done in the Gambia (9.2%),20 Yirgalem (10.1%)16 and Gambella, Ethiopia (7.9%).17 It is also lower than the findings of studies done in Iran (2.1%),21 and Ethiopia: Gandhi memorial hospital (2.3%),12 Dawuro zone (3.5%),13 Arba Minch hospital (4.3%)14 and Felegehiwot referral hospital (4.7%).15 The variation between the current study and the comparatives might be due to the differences in socioeconomic status, socio-cultural environment, sexual practices, and medical exposure among study settings. The inconsistences could also be due to variations in sample size between the current study (375) and other studies; Gambia (416),20 Yirgalem (475),16 Gambella (253),17 Dawuro zone (289),13 Arba Minch hospital (232)14 and Felegehiwot referral hospital (338).15
Women who had a history of contact with jaundice patients were at a higher risk of being seropositive for HBV infection compared to those who had no such history. This might be due to the transmission of the virus through contact with jaundice that is secondary to HBV infection. Other studies also reported that family history of liver disease was associated with HBV infection.10,19
Pregnant women who shared sharp materials with others were at a higher risk of being infected with HBV compared to those who did not share these materials. This finding agrees with a study done in Brazil that reported sharing of personal objects to be associated with being positive for HBV infection.10
The current study also revealed that women who had a history of multiple sexual partners were at a higher risk of being seropositive for HBV just similar to the findings from other studies.10,19 A meta-analysis conducted in Ethiopia also reported a positive association between multiple sexual partners and HBV infection.22 The association between these two variables shows that women need to take necessary care including limiting the number of sexual partners/one to one sexual practice and increasing condom use behavior so as to minimize/avoid the chance of contracting HBV.
Limitations of the Study
The study design was cross-sectional study and therefore it does not show causal relationship. Social desirability bias from patients’ side may also affect this study.
Conclusion
The seroprevalence of HBV is in intermediate range in the study settings as per WHO classification of HBV endemicity. Contact with jaundice patients, multiple sexual partners, and sharing sharp materials were associated with increased risk of HBV infection. These factors are behavioral and hence, HBV infection can be prevented through modification of these factors.
Data Sharing Statement
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Ethical Approval and Consent to Participate
Ethical clearance was obtained from the Institutional Review Board (IRB) of Mizan-Tepi University. Permission was also obtained from the administrator of MTUTH and MHC to collect the data. Written consent was obtained from all study participants after explaining the purpose of the study (as per Helsinki Declaration of 1975, as revised in 2013). Participation in the study was fully voluntary, and the right to withdraw the interview was also ensured. Confidentiality of the information was ensured throughout the study.
Acknowledgments
The authors are very much grateful to all study participants for their kind cooperation/participation in the study. We are also grateful to Mizan Tepi University Teaching Hospital and Mizan Health Center administration for the contributions they made to this study.
Funding
The authors received no specific funding for this work.
Disclosure
The authors declare that they have no competing interests with regard to publication of this article.
References
1. WHO. Prevention of Mother-to-Child Transmission of Hepatitis B Virus (HBV): Guidelines on Antiviral Prophylaxis in Pregnancy; 2020.
2. MacLachlan JH, Cowie BC. Hepatitis B virus epidemiology. Cold Spring Harb Perspect Med. 2015;5(5):1–12. doi:10.1101/cshperspect.a021410
3. WHO. Global Policy Report on the Prevention and Control of Viral Hepatitis in WHO Member States. Geneva: World Health Organisation; 2013.
4. World Health Organization. Hepatitis B. Geneva, Switzerland [Internet]. 2002. Available from: https://apps.who.int/iris/handle/10665/67746.
5. Navabakhsh B, Mehrabi N, Estakhri A, Mohamadnejad M, Poustchi H. Hepatitis B virus infection during pregnancy: transmission and prevention. Middle East J Dig Dis. 2011;3(2):92–102. doi:10.15171/middleeastjdi.v3i2.392
6. Shimakawa Y, Yan HJ, Tsuchiya N, Bottomley C, Hall AJ. Association of early age at establishment of chronic Hepatitis B infection with persistent viral replication, liver cirrhosis and hepatocellular carcinoma: a systematic review. PLoS One. 2013;8(7):e69430. doi:10.1371/journal.pone.0069430
7. Liu J, Zhang S, Liu M, Wang Q, Shen H, Zhang Y. Maternal pre-pregnancy infection with Hepatitis B virus and the risk of preterm birth: a population-based cohort study. Lancet Glob Health. 2017;5(6):e624–e632. doi:10.1016/S2214-109X(17)30142-0
8. Cui AM, Cheng XY, Shao JG, et al. Maternal Hepatitis B virus carrier status and pregnancy outcomes: a prospective cohort study. BMC Pregnancy Childbirth. 2016;16(1):1–8. doi:10.1186/s12884-016-0884-1
9. Zhao Y, Chen Y, Song H, et al. Effects of maternal Hepatitis B surface antigen positive status on the pregnancy outcomes: A retrospective study in Xiamen, China, 2011. PLoS One. 2020;15(3):e0229732. doi:10.1371/journal.pone.0229732
10. Reinaldo V, Bueno Z, Wolf JM, et al. Risk factors for Hepatitis B transmission in South Brazil. Mem Inst Oswaldo Cruz. 2017;112(8):544–550. doi:10.1590/0074-02760170043
11. Tan J, Liu X, Mao X. HBsAg positivity during pregnancy and adverse maternal outcomes: a retrospective cohort analysis. J Viral Hepat. 2016;23:812–819. doi:10.1111/jvh.12545
12. Negesse ZA, Debelo DG. Seroprevalence of Hepatitis B surface antigen and associated risk factors among pregnant women in Gandhi Memorial Hospital, Addis Ababa. J Public Health Epidemiol. 2019;11(6):123–129. doi:10.5897/JPHE2019.1153
13. Chernet A, Yesuf A, Alagaw A. Seroprevalence of Hepatitis B virus surface antigen and factors associated among pregnant women in Dawuro zone, SNNPR, Southwest Ethiopia: a cross sectional study. BMC Res Notes. 2017;10:418. doi:10.1186/s13104-017-2702-x
14. Yohanes T, Zerdo Z, Chufamo N. Seroprevalence and predictors of Hepatitis B virus infection among pregnant women attending routine antenatal care in Arba Minch Hospital, South Ethiopia. Hepat Res Treat. 2016;2016. doi:10.1155/2016/9290163.
15. Gedefaw G, Waltengus F, Akililu A, Gelaye K. Risk factors associated with Hepatitis B virus infection among pregnant women attending antenatal clinic at Felegehiwot referral hospital, Northwest Ethiopia, 2018: an institution based cross sectional study. BMC Res Notes. 2019;1–7. doi:10.1186/s13104-019-4561-0
16. Amsalu A, Ferede G, Eshetie S, Tadewos A, Assegu D. Prevalence, infectivity, and associated risk factors of Hepatitis B virus among pregnant women in Yirgalem Hospital, Ethiopia: implication of screening to control mother-to-child transmission. J Pregnancy. 2018;2018:1–8. doi:10.1155/2018/8435910
17. Tanga AT, Teshome MA, Hiko D, Fikru C, Jilo GK. Sero-prevalence of Hepatitis B virus and associated factors among pregnant women in Gambella hospital, South Western Ethiopia: facility based cross-sectional study. BMC Infect Dis. 2019;19:602. doi:10.1186/s12879-019-4220-z
18. Choisy M, Keomalaphet S, Xaydalasouk K, Quet F, Latthaphasavang V, Buisson Y. Prevalence of Hepatitis B virus infection among pregnant women attending antenatal clinics in Vientiane, Laos, 2008 – 2014. Hepat Res Treat. 2017;2017:1–5. doi:10.1155/2017/1284273
19. Belay M, Tesfaye G, Lemessa O. Hepatitis B virus infection and associated factors among pregnant women attending antenatal clinics in West Hararghe public hospitals, Oromia region, Ethiopia. Pan Afri Med J. 2020;35:128. doi:10.11604/pamj.2020.35.128.17645
20. Bittaye M, Idoko P, Ekele BA, Obed SA, Nyan O. Hepatitis B virus sero-prevalence amongst pregnant women in the Gambia. BMC Infect Dis. 2019;19:259. doi:10.1186/s12879-019-3883-9
21. Moghaddasifar I, Lankarani KB, Moosazadeh M, Afshari M. Prevalence of Hepatitis B virus infection among pregnant women in Iran: a systematic review and meta-analysis. Iran J Cancer Prev. 2016;9(6):e3703. doi:10.17795/ijcp-3703.Review
22. Alemu AA, Zeleke LB, Aynalem BY. Hepatitis B virus infection and its determinants among pregnant women in Ethiopia: a systematic review and meta-analysis. Infect Dis Obstet Gynecol. 2020;2020:1–11. doi:10.1155/2020/9418475
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
