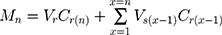Back to Journals » Drug Design, Development and Therapy » Volume 15
Preparation and Characterization of Tacrolimus-Loaded SLNs in situ Gel for Ocular Drug Delivery for the Treatment of Immune Conjunctivitis
Received 30 October 2020
Accepted for publication 14 December 2020
Published 12 January 2021 Volume 2021:15 Pages 141—150
DOI https://doi.org/10.2147/DDDT.S287721
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Anastasios Lymperopoulos
Kexin Sun, Ke Hu
Department of Ophthalmology, The First Affiliated Hospital of Chongqing Medical University, Chongqing Key Laboratory of Ophthalmology, Chongqing Eye Institute, Chongqing, People’s Republic of China
Correspondence: Ke Hu Email [email protected]
Kexin Sun Email [email protected]
Background: The aim of this study is to develop a novel in situ gel of tacrolimus-loaded SLNs (solid lipid nanoparticles) for ocular drug delivery.
Methods: The optimal formulation was characterized by surface morphology, particle size, zeta potential, entrapment efficiency, drug loading and in vitro release behavior. In vivo studies were also conducted to evaluate the pharmacokinetic and pharmacodynamic results.
Results: In this study, TAC-SLNs ISG were prepared using homogenization followed by probe sonication method. The average particle size of TAC-SLNs ISG was observed to be 122.3± 4.3 nm. Compared with TAC-SLNs, in situ gel did not increase particle size, and there was no significant difference between them. The results of viscosity measurement showed that TAC SLNs-ISG were typical of pseudo plastic systems and showed a marked increase in viscosity as temperature increased and ultimately formed a rigid gel (32°C). In vitro and in vivo studies illustrated the sustained release model of the drug from TAC-SLNs ISG. Animal model showed that TAC-SLNs ISG had good pharmacodynamics when compared with eye drops and SLNs.
Conclusion: Our results demonstrated that TAC SLNs-ISG had the potential for being an ideal ocular drug delivery system.
Keywords: tacrolimus, in situ gel, SLNs, ocular drug delivery
Introduction
Immune mediated inflammatory anterior segment diseases (IIAODs) are a kind of common ophthalmic diseases, including conjunctivitis and anterior uveitis. This kind of disease may cause some serious complications and sequelae, which is one of the main causes of blindness. Conjunctivitis requires antibiotic treatment for 5–7 days, which may lead to poor compliance with conventional dosage forms because of the higher frequency of administration, ie 2 drops per day. The reason for that is in the process of ophthalmic administration, due to the physiological constraints of eye protection mechanism, the drug absorption rate is low and the duration of treatment effect is short. When the drug solution drops into the eye, effective tear drainage and blinking can reduce the drug concentration by 10 times in 4–20 minutes.1 We can improve the ocular therapy by improving the precorneal residence time of drugs. In order to slow down drug elimination and to prolong the resident time on the ocular surface, several new preparations have been developed for ocular use, such as liposomes, nanoparticles and nanocapsules.2–8
Many agents available for the treatment of conjunctivitis and tacrolimus (TAC) are the widely used novel macrolide immunosuppressants. However, TAC is a highly lipophilic macrolide lactone and therefore shows very poor water-solubility of 1–2 µg/mL.9–13 Marketed as eye drops, rapid precorneal drainage requires frequent instillation or use of high drug concentration which usually leads to a pulse kinetics pattern of drug concentration. At the same time, another problem is easy hydrolysis, resulting in very low stability in aqueous solutions.14 Therefore, TAC was prepared as an eye drop dispersion containing nearly 100% undissolved TAC. However, drugs, such as TAC, should usually be dissolved to facilitate effective transport or penetration and reach the target site for therapeutic effect.15 It is expected that dissolved TAC will penetrate into the eye tissue more quickly, thus improving bioavailability and reducing treatment failure.15
It can be seen that researchers need to develop a new drug delivery system to meet the better clinical effect, whether from the perspective of human physiology or of the physical and chemical properties of the drug itself. In situ gel is a hot spot in the research of ocular drug delivery system in recent years. It exhibits reversible phase transitions (sol–gel–sol) and pseudoplastic behavior to minimize drainage from blinking.16 In situ gel is formulated as a liquid dosage form which is suitable to be administered as conventional eye drops which, upon exposure to eye physiological conditions, change to the gel phase, thus increasing the precorneal residence time of the delivery system and enhancing ocular bioavailability.
Meanwhile solid lipid nanoparticles (SLNs) have been used for ocular drug delivery amongst different lipid nano carriers. The biggest advantage of SLNs is that it has the capability to encapsulate lipophilic molecules inside the lipid matrix and improve the poor solubility or permeability of the drug.17,18 Previously, SLNs have been used as an ocular delivery platform for the delivery of various drugs, including natamycin,19 triamcinolone,20 indomethacin,21 atorvastatin,22 a platform that is more beneficial than other conventional preparation systems (solutions and suspensions). However, TAC loaded SLNs in situ gel (TAC-SLNs-ISG) is still an unexplored area in the field of intraocular transport.
The objective of the present study is to develop an ocular system combining SLNs and in situ gel for TAC, and to compare the system with plain eye drops and SLNs formulations. The optimal formulation was characterized by surface morphology, particle size, zeta potential, entrapment efficiency, drug loading and in vitro release behavior. In vivo studies were also conducted to evaluate the pharmacodynamic results.
Materials and Methods
Materials
TAC was purchased from Kerui Pharmaceutical Co., Ltd (Fujian, China). Compritol® 888 ATO (glyceryl behenate) and glyceryl monostearate (GMS) were purchased from Gattefossé. Pluronic® F-68 (Poloxamer 188), Pluronic® F-127 (Poloxamer 407) and Tween® 80 were obtained from Sinopharm Chemical Reagent (Shanghai, China). All the other reagents used were of analytical grade and were used without further purification.
Preparation of TAC SLNs-ISG
TAC SLNs-ISG were prepared by using homogenization coupled with ultra-probe sonication method.23 Briefly, Compritol® 888 ATO (0.25%, w/v) and GMS (2%, w/v) were dissolved in the dichloromethane (2 mL) and were heated to 80 °C, after that TAC (0.1%, w/v) was added under magnetic stirring until a clear drug-lipid phase was obtained. The organic solvent was evaporated in vacuum at 45 °C to form the lipid film, which was further dried in the vacuum drying oven at 37 °C to remove the residual organic solvent. Simultaneously, the aqueous phase, consisting of Tween-80 (0.5%, w/v) and glycerin (2%, w/v) in distilled water, was heated (30 °C) and transferred to the molten drug-lipid mixture under constant stirring, to form a pre-mixture. The mixture was homogenized with a probe-type ultrasonicator (S220, Covaris, USA) in the ice bath for 10 cycles to get the TAC SLNs. Then TAC SLNs-ISG was obtained by mixing TAC SLNs and Poloxamer 188 (12%, w/v)/Poloxamer 407 (26%, w/v) solution in the ice bath.
Characterization of System
Particle Size and Zeta Potential
Intensity-mean particle size, zeta potential and polydispersity index of TAC SLNs-ISG were determined using a Zetasizer NanoZS (Malvern Instruments Ltd.). All measurements were performed under automatic mode at 25°C. The hydrodynamic diameter was calculated from autocorrelation function of the intensity of light scattered from particles with the assumption that the particles have a spherical form.
Entrapment Efficiency
The entrapment efficiency (% EE) was determined by measuring the concentration of unentrapped drug in the lipid dispersion. Briefly, the SLNs-ISG dispersion was subjected to centrifugation for 30 min, 4 °C at 6000 rpm and the amount of TAC in supernatant was determined by dissolving supernatant in acetone and 0.1% acetic acid (1:1) mixture by HPLC. The amount of free drug in the supernatant was determined spectrophotometrically at 210 nm. The entrapment efficiency was determined in triplicate and calculated as follows.
EE%=WTAC/(WSLNs-ISG+WTAC)×100%
WTAC represents the amount of TAC loaded in the SLNs-ISG, WSLNs-ISG represents the weight of the TAC SLNs-ISG.
Viscosity Experiments
The sol-gel transition temperature of TAC SLNs-ISG sol in water was measured by the tube transformation method.24 A vial containing 20 mL TAC SLNs-ISG sol was immersed in an oil bath at different temperatures to achieve equilibrium. When the flow rate was no longer visually observable within 30 s by inverting the vial, the temperature was increased by 2°C per step, and the sample was regarded as a “‘gel’”. The viscosity of TAC SLNs-ISG in solution or gel was then measured using a rotating viscometer (60 rpm, rotor 2) with suitable samples (20 mL). The appropriate number of spindles was used for measurement at different speeds. The viscosity was read directly from the viscometer display.
In vitro Release
The in vitro release studies were conducted by a dynamic dialysis method. Briefly, TAC SLNs-ISG containing 20 mg were placed into dialysis bags, which were then placed into an end-sealed vial. The release medium was 2000 mL simulated tear fluid (STF; made with sodium chloride-0.67g, sodium bicarbonate-0.20g, calcium chloride dihydrate −0.008g in distilled water q.s 100 mL). The sealed vials were maintained at 37°C and 75 rpm in a gas bath thermostatic oscillator. Aliquots (1 mL) were withdrawn at intervals of 0.25, 0.5, 1, 2, 4, 6, 8, 12, and 16 h, and the same replacement volume was supplied as the release medium. The collected samples were determined by HPLC. An equal amount of free TAC and TAC SLNs was used for comparison.
Stability Studies
Stability studies were carried out on TAC SLNs-ISG according to ICH (International Conference on Harmonization) Guidelines at 40°C, 75% relative humidity. Three packs of formulations were subjected to these stability studies. The samples were withdrawn at the 0th, 1st and 3rd month. Every time 0.1 mL of samples were withdrawn and analyzed for the assay with HPLC for the drug content. At the same time, some other stability parameters were also determined.
In vivo Kinetics
In vivo ocular disposition studies of TAC SLNs-ISG and TAC SLNs in comparison with TAC eye drops, were carried out in New Zealand rabbits, weighing between 2 to 3 kg. The rabbits were allowed to acclimatize to the new surroundings for one week, and the ocular disposition studies were then performed. A formulation of 50 µL of the TAC SLNs-ISG, TAC SLNs and TAC eye drop (0.1% w/v) was dripped into the cul-de-sac of the right eye while the left eye served as the control. Approximately five microliters of tear were collected from the cul-de-sac of the test eye, using a micropipette, at 15th, 30th min, 1st, 2nd, 4th, 5th, 6th, 8th and 10th h. Ten hours later, the rabbits were euthanized by intravenous injection of pentobarbital. Rinse eyes with cold DPBS and remove them immediately. The eye tissues were carefully separated and stored at −80°C until further analysis. Tear samples were collected in acetone: 0.1% acetic acid = (1:1) and analyzed by HPLC. The ocular tissue samples were extracted using acetone precipitation method and analyzed using LC-MS/MS system. Separation was achieved on a Genesis C18 column with a gradient mobile phase elution. Ammonium-adduct ions formed by a Turbo Ionspray in positive ion mode were used to detect each analyte. The MS/MS detection was traced by monitoring the fragmentation of 807.5 → 772.4 (m/z) for TAC on a triple quadrupole mass spectrometer (Sciex API 3000). The above method was simple, fast, and specific. The calibration curve of the LC assay for TAC in neat solution has good linearity (r2=0.996) over the range of 10–10,000 ng/mL. The lower limit of the quantitation of TAC in the LC-MS/MS assay was found to be 0.1 ng/mL. These results indicated that the assays provided good linearity and sensitivity for their specific applications.
Pharmacodynamic Evaluation
BALB/c mice were used to investigate the pharmacodynamic effect of TAC SLNs-ISG in this study. The grouping information was as follows: A: Normal (without any treatment, positive control); B: PBS (negative control); C: 0.1% (w/v) TAC eye drop; D: 0.1% (w/v) TAC-SLNs; E: 0.1% (w/v) TAC-SLNs ISG. ①Sensitization stage: BALB/c mice were intraperitoneal injected with Ovalbumin (OVA, 100 µg and 35 µg Al(OH)3 in 200 µL PBS) on immun-day 1. ②Immune intervention stage: on immun-day 7, the eyes were challenged with different formulations three times a day for 5 days of the B, C, D, E group. ③Stimulation stage:
Ova (10 µL, 5 mg/mL) was dissolved in PBS (pH=7.0) after drug immune intervention treatment. The mice in groups B, C, D, E were injected with a micro sampler to induce the clinical symptoms of immune conjunctivitis. The clinical symptoms of immune conjunctivitis were observed and scored once a day for one week. Then the eyes were examined under microscope, and the scoring was performed at the same time every day and carried out once daily from day 1 to day 7. As shown in Table 1, mice were examined biomicroscopically based on 3 independent parameters. Each parameter was ascribed 0 (none) to 3+ points (serious) and was summed to yield a maximum score of 9+. ④After OVA challenge induced immune conjunctivitis, mice in groups B, C, D, and E were treated with PBS, 0.1% TAC eye drop, 0.1% TAC-SLNs, 0.1% TAC-SLNs ISG one time a day for 5 days. Scores of various systems in different groups were calculated and graphed. After pharmacodynamic study, OVA-s IgE, IFN-γ and IL-4 concentration in different treatment groups of mice were determined, respectively.
 |
Table 1 Scoring of Conjunctivitis Signs and Symptoms |
Meanwhile the pathological sections of corneal tissue of all groups were also observed. All animal experiments were performed in accordance with institutional guidelines, following the protocol approved by the Ethics Committees of Chongqing Medical University (19A2040). Strictly follow the National Institutes of health laboratory animal care and use guidelines.
Permeability Studies
Permeability studies were performed on the corneas isolated from rabbit whole eyes. The eyes were stored in Hanks’ balanced salt solution under ice-cold condition and shipped overnight. Immediately upon their receipt, the corneas were carefully separated, and used for the permeability studies. The isolated corneas were washed in ice-cold Dulbecco’s phosphate buffer saline (DPBS) solution, pH 7.4. The tissues were then mounted on Valia-Chien diffusion cells with the epithelial surface towards the donor chamber. The temperature of the diffusion cells was maintained at 32 °C with a circulating water bath, throughout the studies.
TAC concentration in eye drops, SLNs and SLNs-ISG were kept at 0.1% w/v, and about 1 mL of the formulation was added to the donor chamber of the respective diffusion cells. Five milliliters of DPBS with 5% w/v hydroxyl propyl beta cyclodextrin (HPßCD) solution was used as the receiver medium and stirred continuously with magnetic stirrer. Samples (500 µL) were withdrawn from the receiver chamber at the predetermined time points up to 2 h and replaced with an equal amount of DPBS-5% HPßCD solution to maintain sink conditions. The samples were stored at −80 °C until further analysis by HPLC. The analyses for all the samples were carried out in triplicate.
The cumulative amount of TAC was calculated as per the equation:
where n is sampling time point; Vr and Vs are the volume in the receiver chamber (mL) and the volume of the sample collected at the nth time point (mL), respectively; and Cr(n) is the concentration of the drug in the receiver chamber medium at nth time point (µg/mL).
The rate of TAC transported across rabbit cornea was calculated using the slope of the cumulative amount of TAC transported versus time plot. The steady state flux of TAC was determined using the following equation:
Flux(J) = (dM/dt)/A
where M is the cumulative amount of drug transported and A is the surface area of the cornea (0.625 cm2).
The transcorneal permeability of TAC was calculated by the following equation:
Permeability =Steady state flux/Donor concentration.
Statistical Analysis
The results were analyzed by extreme value analysis and ANOVA. SPSS software was used for statistical analysis. The difference was significant when p < 0.05.
Results and Discussion
Preparation and Characterization
The main purpose of the study is to develop SLNs and its corresponding in situ gel to retard the clearance of TAC in the eyes and increase drug absorption. According to the solubility of the drug in the lipid phase (using GMS, and Compritol 888 ATO as the lipid phase), Tween80 and glycerol were selected as the aqueous phase to design, prepare and optimize the SLN formulation.
TAC-SLNs ISG were prepared using homogenization followed by probe sonication method. The physicochemical properties including particle size, encapsulation efficiency (EE%) were listed in Table 2. The average particle size of TAC-SLNs ISG was observed to be 122.3±4.3 nm. Compared with TAC-SLNs, in situ gel did not increase particle size, and there was no significant difference between them. Some researchers think that PDI value in the range of 0.01–0.5 represents a sufficiently narrow distribution range. In the case of TAC SLNs ISG, the PDI value of 0.21 was in this range, so it was considered to be a more uniform high-throughput and high permeability formulation, as shown by in vitro release data.25
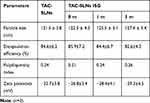 |
Table 2 The Characteristics of TAC-SLNs ISG and TAC-SLNs |
Viscosity Experiments
The residence time of the ophthalmic formulation at the precorneal surface is affected by its viscosity that is of importance for enhancing the residence time. In context to ocular physiology, the range of shear rate experienced during relative movement of eyelids and globe is extremely wide ranging from 0.03 to 0.14 s−1 during inter blinking period to 4250–28,500 s−1 during a blink. Thus the viscosity of formulation should not be such that it disturbs the pseudoplastic behavior of tear film in the eye.26 Hence it is advisable to use a polymer with pseudoplastic character.27 Many literatures have confirmed that the gel prepared by poloxamer has the characteristics of pseudoplastic fluid.28,29 In order to study the effect of temperature on gelation, the viscosity of the gel was determined. The results of viscosity measurement showed that TAC-SLNs-ISG was a typical pseudoplastic system. With the increase of temperature, the viscosity increased obviously, and finally the rigid gel was formed. The gelation temperature of the prepared thermogelling gels was 32°C (Figure 1). At the same time, the viscosity of TAC-SLNs system did not increase with the change of temperature, and it always maintained at a relatively low viscosity level.
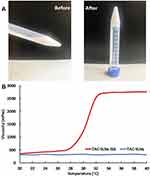 |
Figure 1 (A) Photographs of in situ gels formed before (4°C) and after (37°C) gelation. (B) Mean viscosity-temperature trends profiles of TAC SLNs and TAC SLNs-ISG. (n=3). |
In vitro Release
The in vitro drug release curve of TAC SLNs-ISG, TAC SLNs and free TAC were shown in Figure 2. At the end of the experiment, nearly 80% of the drug was released in TAC SLNs group, while only 56% in TAC SLNs-ISG group did so. The release curve of TAC SLNs-ISG was a two-phase model. TAC SLNs-ISG released quickly at the early stage (about 30% TAC was released within 1 hour), and then released slowly. This phenomenon is mainly caused by the release of drugs dispersed in the gel. In the late stage, the sustained release was due to the fact that with the dissolution and diffusion mechanisms on the lipid matrices and polymer material, the solubilized or dispersed drug can only be released slowly. As was shown in Table 3, the Higuchi equation: Q =9.287t1/2-2.263 (r=0.994) was the best fit well with the release kinetic model of TAC SLNs-ISG. Therefore, it was speculated that the sustained-release characteristics of TAC SLNs-ISG may enhance the ocular absorption of TAC.
 |
Table 3 Release Kinetic of TAC SLNs-ISG |
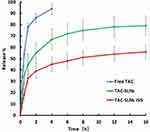 |
Figure 2 The in vitro drug release profiles of TAC eye drops, TAC SLNs and TAC SLNs-ISG. Number represents the percentage of release. (n=6). |
Stability Studies
There were negligible alterations in the initial values of viscosity of the formulations over a storage of 3 months. The samples were also analyzed for drug content by HPLC analysis (data not shown). Again the drug degraded to a negligible extent, and the percentage of drug degradation is < 5%. Other stability parameters are shown in Table 2.
In vivo Kinetics
The concentration in tear-time plots, in rabbits after ophthalmic administration of test formulations are shown in Figure 3 and the AUC parameters are tabulated in Table 4. The Tmax was 30 min and the Cmax was 4657.7 ng/mL after ophthalmic administration of TAC eye drops. However, the time to achieve maximum concentration of TAC was delayed in the form of ISG. For TAC-SLNs, highest drug concentration of 1892.6 ng/mL was observed at the 30th min, whereas TAC-SLNs-ISG showed maximum concentration (2132.3 ng/mL) at the second hour. The Cmax of TAC-SLNs and TAC-SLNs-ISG was significantly (p < 0.05) lower than that obtained within the TAC eye drops. The AUC0–t of TAC-SLNs-ISG was 590,355.9 ng·min/mL, 2.65 folds higher than AUC0–t of 222,382.5 ng·min/mL for TAC eye drops, clearly defining performance superiority of in situ gels over drops. SLNs are believed to be actively engulfed by the corneal and conjunctival cells. This is considered to be the main advantage of colloidal dispersion. In addition to passive diffusion out of the SLNs, the lipases in the tear film and in the epithelial cells are also responsible for the controlled release of TAC. Release occurs both in the tear as well as in the corneal matrix. Therefore, in vivo kinetics studies can show the slow release characteristics of drugs from gels.
 |
Table 4 Ocular Tear Pharmacokinetic Parameters of TAC in Tear versus Time Profiles in 600 Min for Various Formulations |
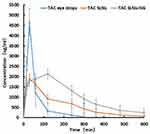 |
Figure 3 Concentration-time curve of TAC in different formulations (TAC eye drops, TAC SLNs and TAC SLNs-ISG). (n=6). |
Pharmacodynamic Evaluation
The establishment of the animal model in this study was completely successful, and there were significant differences between the positive control group and the negative control group (Figure 4).
In the untreated group (Group A), there were almost no symptoms, so the score remained at a relatively low level during the observation period. The symptoms of the positive control group (Group B) were more obvious, and the score was always at a high level. In this experiment, the unified administration time of each group was morning, and the unified time of scoring statistics was afternoon, so on the first day after treatment, there was a significant difference between the three groups. The scores of the three experimental groups (Group C, D, E) showed different degrees of decline from the first day. The effect of group E was the most obvious, and the improvement of symptoms was the most obvious on the fifth day of the experiment. The reason for the relatively short retention time of the eye drops and TAC-SLNs is that they are not completely observed by the naked eye. Through the detection of three biochemical indexes (OVA-s IgE, IFN-γ and IL-4), Group E has significant curative effect on conjunctivitis compared with other control groups (p<0.05, Figure 5). The mechanism of TAC’s action is to inhibit the activation and degranulation of conjunctival mast cells, inhibit the release of inflammatory mediators by mast cells, and down regulate IL-4 in serum, thus inhibiting the antibody response of B cells from IgM to IgE and reducing the synthesis of IgE. Through up regulating IFN - γ in serum, the proliferation of Th2 cells and the function of IL-4 are inhibited, and the transformation of Th1 to Th2 is inhibited, so that Th1 and Th2 are in a dynamic balance state. Through this mechanism, type I allergy can be controlled and immune conjunctivitis can be treated. Through the optimization of dosage forms (SLNs or in situ gel), the therapeutic effect is further expanded. Corneal histopathology of showed that TAC-SLNs-ISG had good safety (Figure 6).
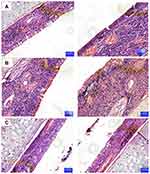 |
Figure 6 Histopathological studies of corneal tissue of different groups. (A) normal; (B) TAC SLNs (C) TAC SLNs-ISG. Left: treatment eye; Right: un-treatment eye. |
Permeability test
The permeability coefficient and flux for TAC in eye drops, SLNs and SLNs-ISG is calculated in Table 5. The transcorneal permeability and flux of TAC from SLNs was significantly higher compared to that of the eye drops. This indicates that the lipid nanoparticles enhance the permeation of the drug through intact corneal tissues. The slightly lower flux and permeability of TAC from the SLNs-ISG compared with SLNs indicates the controlled release of the drug from the higher viscosity formulation.
 |
Table 5 The Result of Permeability and Flux of Different TAC Formulation |
Conclusion
The aim of this study is to develop a novel in situ gel of tacrolimus-loaded SLNs for ocular drug delivery. In this study, TAC-SLNs ISG were prepared using homogenization followed by probe sonication method. The average particle size of TAC-SLNs ISG was observed to be 122.3±4.3 nm. Compared with TAC-SLNs, in situ gel did not increase particle size, and there was no significant difference between them. The results of viscosity measurement showed that TAC SLNs-ISG were typical of pseudo plastic systems and showed a marked increase in viscosity as temperature increased and ultimately formed a rigid gel (32°C). In vitro and in vivo studies illustrated the sustained release model of the drug from TAC-SLNs ISG. Animal model showed that TAC-SLNs ISG had good pharmacodynamics when compared with eye drops and SLNs. Our results demonstrated that TAC SLNs-ISG has potential as an ideal ocular drug delivery system.
Acknowledgments
We wish to express our thanks to Dr. Wei ZHU(Department of Pharmaceutical, School of Pharmacy,Fudan University, Shanghai, China) for his help in the study design.
Disclosure
The authors report no conflicts of interest for this work.
References
1. Maurice DM. Kinetics of topical applied drugs. In: Saettone MS, Bucci P, Speiser P, editors. Ophthalmic Drug Delivery: Biopharmaceutical, Technological and Clinical Aspects. Padova: Liviana Press; 1987:19–26.
2. Pleyer U, Lutz S, Jusko W, et al. Ocular absorption of topically applied FK506 from liposomal and oil formulations in rabbit eye. Invest Ophthalmol Vis Sci. 1993;34(9):2737–2742.
3. Bochot A, Fattal E, Grossiord JL, Puisieux F, Couvereur P. Characterization of a new ocular delivery system based on a dispersion of liposomes in a thermosensitive gel. Int J Pharm. 1998;162:119–127. doi:10.1016/S0378-5173(97)00419-5
4. de Campos AM, Diebold Y, Carvaiho EL, Sanchez A, Alonso MJ. Chitosan nanoparticles as new ocular drug delivery system: in vitro stability, in vivo fate, and cellular toxicity. Pharm Res. 2004;21(5):803–810. doi:10.1023/B:PHAM.0000026432.75781.cb
5. Nayak K, Misra M, Nayak K, et al. A review on recent drug delivery systems for posterior segment of eye. Biomed Pharmacother. 2018;107:1564–1582. doi:10.1016/j.biopha.2018.08.138
6. Bachu RD, Chowdhury P, Al-Saedi ZHF, et al. Ocular drug delivery barriers-role of nanocarriers in the treatment of anterior segment ocular diseases. Pharmaceutics. 2018;10(1):28. doi:10.3390/pharmaceutics10010028
7. De Campos AM, Sánchez A, Gref R, Calvo P, Alonso MJ. The effect of PEG versus a chitosan coating on the interaction of drug colloidal carriers with the ocular mucosa. Eur J Pharm Sci. 2003;20(1):73–81. doi:10.1016/S0928-0987(03)00178-7
8. Lakhani P, Patil A, Majumdar S, et al. Recent advances in topical nano drug-delivery systems for the anterior ocular segment. Ther Deliv. 2018;9(2):137–153. doi:10.4155/tde-2017-0088
9. Ferraboschi P, Colombo D, De Mieri M, et al. Evaluation, synthesis and characterization of tacrolimus impurities. J Antibiot (Tokyo). 2012;65(7):349–354. doi:10.1038/ja.2012.28
10. Honbo T, Kobayashi M, Hane K, Hata T, Ueda Y. The oral dosage form of FK-506. Transplant Proc. 1987;19(5 Suppl 6):17–22.
11. Arima H, Yunomae K, Miyake K, Irie T, Hirayama F, Uekama K. Comparative studies of the enhancing effects of cyclodextrins on the solubility and oral bioavailability of tacrolimus in rats. J Pharm Sci. 2001;90(6):690–701. doi:10.1002/jps.1025
12. Gao S, Sun J, Fu D, Zhao H, Lan M, Gao F. Preparation, characterization and pharmacokinetic studies of tacrolimus-dimethyl-β-cyclodextrin inclusion complex-loaded albumin nanoparticles. Int J Pharm. 2012;427(2):410–416. doi:10.1016/j.ijpharm.2012.01.054
13. Kwon M, Yeom D, Kim NA, et al. Bioequivalence of tacrolimus formulations with different dynamic solubility and in-vitro dissolution profiles. Arch Pharm Res. 2015;38(1):73–80. doi:10.1007/s12272-014-0343-3
14. Skytte DM, Jaroszewski JW, Johansen KT, et al. Some transformations of tacrolimus, an immunosuppressive drug Eur. J Pharm Sci. 2013;48(3):514–522.
15. Loftsson T, Muellertz A, Siepmann J. For the special IJP issue “Poorly soluble drugs”. Int J Pharm. 2013;453(1):1–2. doi:10.1016/j.ijpharm.2013.05.056
16. Xiao P, Dudal Y, Corvini P, Spahr P, Shahgaldian P. Synthesis and characterization of fluoroquinolone-imprinted polymeric nanoparticles. React Funct Polym. 2012;72:287–293. doi:10.1016/j.reactfunctpolym.2012.02.006
17. Müller RH, Mäder K, Gohla S. Solid lipid nanoparticles (SLN) for controlled drug delivery—A review of the state of the art. Eur J Pharm Biopharm. 2000;50(1):161–177. doi:10.1016/S0939-6411(00)00087-4
18. Mehnert W, Mäder K. Solid lipid nanoparticles. production, characterization and applications. Adv Drug Deliv Rev. 2001;47(2–3):165–196. doi:10.1016/S0169-409X(01)00105-3
19. Khames A, Khaleel MA, El-Badawy MF, El-Nezhawy AOH. Natamycin solid lipid nanoparticles - sustained ocular delivery system of higher corneal penetration against deep fungal keratitis: preparation and optimization. Int J Nanomedicine. 2019;14:2515–2531. doi:10.2147/IJN.S190502
20. Tatke A, Dudhipala N, Janga KY, et al. In situ gel of triamcinolone acetonide-loaded solid lipid nanoparticles for improved topical ocular delivery: tear kinetics and ocular disposition studies. Nanomaterials (Basel). 2018;9(1):33. doi:10.3390/nano9010033
21. Balguri SP, Adelli GR, Majumdar S. Topical ophthalmic lipid nanoparticle formulations (SLN, NLC) of indomethacin for delivery to the posterior segment ocular tissues. Eur J Pharm Biopharm. 2016;109:224–235. doi:10.1016/j.ejpb.2016.10.015
22. Yadav M, Schiavone N, Guzman-Aranguez A, et al. Atorvastatin-loaded solid lipid nanoparticles as eye drops: proposed treatment option for age-related macular degeneration (AMD). Drug Deliv Transl Res. 2020;10(4):919–944. doi:10.1007/s13346-020-00733-4
23. Narendar D, Kishan V. Candesartan cilexetil loaded solid lipid nanoparticles for oral delivery: characterization, pharmacokinetic and pharmacodynamic evaluation. Drug Deliv. 2016;23(2):395–404. doi:10.3109/10717544.2014.914986
24. Yu L, Zhang Z, Zhang H, Ding J. Biodegradability and biocompatibility of thermoreversible hydrogels formed from mixing a sol and a precipitate of block copolymers in water. Biomacromolecules. 2010;11(8):2169–2178. doi:10.1021/bm100549q
25. Rakesh K, Sinha VR. Solid lipid nanoparticle: an efficient carrier for improved ocular permeation of voriconazole. Drug Dev Ind Pharm. 2016;42(12):1956–1967. doi:10.1080/03639045.2016.1185437
26. Giannavola C, Bucolo C, Maltese A, et al. Influence of preparation conditions on acyclovir- loaded poly-d, l-lactic acid nanospheres and effect of PEG coating on ocular drug bioavailability. Pharm Res. 2003;20(4):584–590. doi:10.1023/A:1023290514575
27. Srividya B, Cardoza RM, Amin PD. Sustained ophthalmic delivery of ofloxacin from a pH triggered in-situ gelling system. J Control Release. 2001;73(2–3):205–211. doi:10.1016/S0168-3659(01)00279-6
28. Moreira TS, de Sousa VP, Pierre MB. Influence of oleic acid on the rheology and in vitro release of lumiracoxib from poloxamer gels. J Pharm Pharm Sci. 2010;13(2):286–302. doi:10.18433/J34880
29. Perez AP, Mundiña-Weilenmann C, Romero EL, Morilla MJ. Increased brain radioactivity by intranasal P-labeled siRNA dendriplexes within in situ-forming mucoadhesive gels. Int J Nanomedicine. 2012;7:1373–1385. doi:10.2147/IJN.S28261
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.