Back to Journals » Journal of Pain Research » Volume 13
Preoperative Administration of Extended-Release Dinalbuphine Sebacate Compares with Morphine for Post-Laparoscopic Cholecystectomy Pain Management: A Randomized Study
Authors Lee SO, Huang LP, Wong CS
Received 28 May 2020
Accepted for publication 20 August 2020
Published 9 September 2020 Volume 2020:13 Pages 2247—2253
DOI https://doi.org/10.2147/JPR.S263315
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor E Alfonso Romero-Sandoval
Sing-Ong Lee,1 Li-Ping Huang,1 Chih-Shung Wong1– 3
1Department of Anesthesiology, Cathay General Hospital, Taipei, Taiwan; 2Graduate Institute of Medical Science, National Defense Medical Center, Taipei, Taiwan; 3School of Medicine, Fu-Jen Catholic University, New Taipei, Taiwan
Correspondence: Chih-Shung Wong Department of Anesthesiology
Cathay General Hospital, #280, Renai Road, Section 4, Taipei, Taiwan
Tel +886-2-27082121 Ext 3510
Fax +886-2-87924835
Email [email protected]
Purpose: Perioperative pain management plays a critical role in the effort to promote enhanced recovery after surgery (ERAS). Pain is also the most concern for patients after laparoscopic cholecystectomy (LC). Naldebain (extended-release dinalbuphine sebacate, DS) is an oil-based formulation for intramuscular injection that has been designed for extended release and can be used for preoperative analgesia over a 7-day period. This study was aimed to compare the efficacy of DS injection with that of regular postoperative morphine administered when necessary for the management of post-laparoscopic cholecystectomy pain.
Patients and Methods: Forty-four patients scheduled for elective laparoscopic cholecystectomy were included in this prospective study. The patients were allocated randomly into two groups, with equal numbers receiving preoperative DS versus post-operative morphine. A total of 21 and 22 patients completed the study within the preoperative DS and post-operative morphine group, respectively.
Results: There were no statistically significant differences between two treatment groups with respect to length of surgery, anesthetics used during operation, or the average visual analog scale pain score in the post-operative anesthesia care unit (PACU), and at 4, 24, 48, and 72 hours post-procedure. Morphine was required only during the first postoperative day among those in the DS group. Safety was comparable in both DS and morphine groups.
Conclusion: A single preoperative dose of DS provides sufficient analgesia along with a manageable safety profile and no interference with surgical anesthetics when compared to control cases that underwent surgery without preoperative DS treatment. This pilot study suggests that preoperative administration of DS is safe and may decrease the need for postoperative opioid use after laparoscopic cholecystectomy.
Registration: ClinicalTrials.gov Identifier: NCT03713216.
Keywords: nalbuphine, enhanced recovery after surgery, multimodal analgesia, preventive analgesia
Introduction
Appropriate perioperative pain management is critical factor in the effort to achieve enhanced recovery after surgery (ERAS).1 Although various analgesics had been studied for postoperative pain management, opioids remain the mainstay in most clinical settings.2 Opioid administration results in numerous adverse effects, including pruritus, constipation, and respiratory depression.3 Multimodal opioid-sparing regimens have been devised to address concerns associated with high dose-opioid use; these typically involve combinations multiple drugs to promote pain relief using different pathways and mechanisms of action and achieve more effective analgesia while limiting opioid consumption.4
Laparoscopic procedures are in wide use and present many advantages for the surgeon and the patient including minimized surgical complications and decreased pain intensity; these factors all contribute positively to patient recovery.5,6 One growing concern after laparoscopic procedure is chronic postoperative pain (CPSP) formation; this is because most of the CPSP developed when postoperative acute visceral pain persisted after discharge.7 In recent years, a significant effort has been made towards the identification of optimal combinations of medications for preoperative analgesia; these are widely used to facilitate postoperative outcomes following laparoscopic procedures.8,9 As one example, 8 mg of dexamethasone provided significantly better pain relief when administered preoperatively rather than postoperatively.10 Likewise, Wilson et al demonstrated that intramuscular diclofenac after laparoscopic cholecystectomy (LC) was effective in reducing postoperative pain.11 However, there is as yet no clear understanding of the optimal timing of interventions or regarding specific multimodal regimens for analgesia for LC.12
Dinalbuphine sebacate (DS) injection (Naldebain ER Injection, Lumosa Therapeutics, Taiwan) was designed as a prodrug form of nalbuphine with similar analgesic effects as morphine. Intramuscular DS injection slowly releases into the blood vessel and maintain relatively low nalbuphine concentration covered early recovery period that decreases postoperative pain intensity and threshold of pain feeling. The formulation includes a sesame oil-based solution with 150 mg DS in a 2 mL volume and has been approved for moderate to severe pain relief by the Taiwan Food and Drug Administration (TFDA). Recommended administration time is at least 12 hours prior to surgery. Previous studies have revealed that pre-operative administration of DS resulted in effective pain control for 7 days and was associated with a significant reduction in postoperative ketorolac consumption after hemorrhoidectomy.13
The postoperative analgesic effect of morphine has been studied in association with many types of laparoscopic surgery.14–16 Most studies focus on pain relief or the use of rescue medication in the first 24 hours after surgery. Blichfeldt-Eckhardt et al7 found that early visceral pain was associated with chronic pain at 12 months after LC. Likewise, Richebe et al17 noted that the incidence of chronic pain reached 3–56% among cases in which there was no effective strategy to limit persistent postoperative pain. We recently reported combination use with ultrasound-guided peripheral nerve block, epidural anesthesia, and the extended-release DS provided additional postoperative analgesia and longer duration of time reported as pain-free after surgery.18,19 These studies underscore a potential role for extended-release DS as a component of perioperative multimodal analgesia.20 At this time, there are limited data that propose the utility of extended-release DS for the management of acute postoperative pain in early and intermediate recovery periods in patients undergoing LC. In this study, we aimed to evaluate the effect of preoperative treatment with extended-release dinalbuphine sebacate for postoperative pain relief after elective LC compared with responses to conventional postoperative intravenous morphine treatment.
Patients and Methods
Patients
This was a prospective, randomized-controlled clinical study that was registered on ClinicalTrials.gov (Identifier: NCT03713216) prior to enrolling study participants. This study protocol was approved by the Institutional Review Board of Cathay General Hospital (CGH-P107007) and conducted in accordance with the Declaration of Helsinki. The study flow diagram is presented in Figure 1. Patients were enrolled at Cathay General Hospital, Taipei from October 2018 to October 2019. Written informed consent was obtained from all patients prior to screening for enrollment. All data for background information, efficacy, and safety were collected anonymously and in compliance with the Declaration of Helsinki. Eligible patients were aged 20 years or older and scheduled for elective LC under general anesthesia. Patients meeting the following criteria were excluded from the study: those not willing to adhere to the study visit schedule; those with a history of hypersensitivity or allergy to opioids, NSAIDs or sesame oil, or any clinically significant condition that may interfere with study assessments; those who were pregnant or breastfeeding; those with a medical history that may predispose them to abnormal intracranial pressure; or any history of narcotic dependency, addiction, and withdrawal.
 |
Figure 1 The consort flow diagram. |
Procedure
All patients were randomly assigned (half in each group) to receive either a 150 mg DS injection (DS group) preoperatively or regular morphine after surgery (Morphine group). Random numbers and assignment treatment were generated by computer program before this study initiated. Once the patient was eligible, the random number was assigned and allocated to pre-defined treatment. Patients in the DS group received a single dose of 150 mg DS was injected intramuscularly at least 12 hours prior to surgery. Patients in the Morphine group received intravenous morphine as needed for postoperative pain. Anesthesia was induced in both treatment groups with 2 mg/kg propofol, 2 µg/kg fentanyl, 0.6–1 mg/kg rocuronium bromide; anesthesia was maintained with 1–1.2 minimum alveolar concentration (MAC) of sevoflurane and 100–300 µg fentanyl during the procedure as needed. Anesthesiologists were blinded to the patient treatment group. Either ketorolac or morphine was prescribed to patients in both groups as rescue analgesics by surgeons’ orders when needed; rescue analgesics (5 mg morphine or 30 mg ketorolac per time) were recommended for patients with a VAS above 3.0. The quantity and frequency of rescue analgesics were recorded.
Patients were provided with instructions on the use of the 100-mm visual analog scale (VAS; 0 = no pain, 100 mm = the worst pain) and other assessments before the first evaluation. The VAS for pain was recorded by patients themselves in the PACU, and at 4, 24, 48 and 72 hours after surgery. Daily vital signs, any injection site reactions, concomitant medications, and adverse events were recorded by investigators starting from the time of administration of the study medications until discharge. Patients’ satisfaction was evaluated at day 3 using 5-grade score (highly satisfied, satisfied, uncertain, not satisfied, highly unsatisfied).
Statistical Analysis
At least 42 patients were needed in order to detect differences in pain scores within 24 hours after LC with α level of 0.05 (two-tailed) and β level of 0.4 (power 60%). Analgesic interventions were scored using 10 mm VAS as a clinically meaningful improvement. This pilot study included 43 patients; 21 patients received 150 mg DS preoperatively and 22 patients received morphine only as needed for postoperative pain. Differences in average 72-hour VAS pain scores were the primary endpoint. The VAS and use of morphine and ketorolac were analyzed using a Mann–Whitney test. For categorical measures, comparisons were analyzed by X2 or Fisher’s exact test. P values <0.05 were considered as statistically significant.
Results
Study Participants
Forty-four patients were randomly assigned into one of two groups; the DS group received preoperative injection with DS (150 mg; n = 22) and the Morphine group received postoperative intravenous morphine as needed (n = 22; Figure 1). One patient in DS group withdrew from the study prior to the DS injection. Demographics and baseline characteristics are summarized in Table 1. There were no statistically significant differences between the two groups with respect to intraoperative anesthetic use.
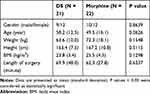 |
Table 1 Demographic Information and Baseline Characteristics |
Efficacy
With respect to total morphine consumption within 72 hours after surgery, patients in the DS group used slightly less opioid analgesics overall than did those in the Morphine group (37 vs 92 mg; Table 2), however, there were no statistically significant differences with respect to mean total morphine consumption after surgery (1.76 ± 3.75 vs 4.18 ± 6.27, for DS vs Morphine groups, p = 0.1448). Patients in the DS group received morphine or ketorolac only while in the PACU, while those in the Morphine group consumed on average 57, 20, and 15 mg morphine per patient on post-operative days 1, 2 and 3, respectively. Fewer patients in DS group used opioids after surgery (23.8 vs 40.9% in the DS vs Morphine groups, respectively). Both groups utilized ketorolac during post-operative day 1 only (5.71 ± 12.07 vs 2.73 ± 8.83, for DS vs Morphine groups, p = 0.4121). Comparison of post-operative pain intensity at consecutive intervals revealed no statistically significant differences between the two groups while in the PACU, or at 4, 24, 48, and 72 hours after the procedure (Figure 2).
 |
Table 2 Frequency and Dose of Rescue Analgesia |
 |
Figure 2 Pain scores over time from PACU to 72 hours after the procedure. |
Safety
A total of 17 patients reported at least one adverse event during the study period, including 13 (61.9%) and 4 (18.2%) patients assigned to the DS and Morphine groups, respectively (Table 3). All adverse events were mild to moderate and required no specific drug treatment.
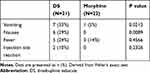 |
Table 3 Incidence of Adverse Events During the Study Period |
The most frequent adverse events were postoperative nausea and vomiting in the DS group (n = 13) and fever in the Morphine group (n = 3). Although more adverse events were reported in the DS group, nearly all were mild and resolved by postoperative day 1. Two patients in DS group reported swelling at the injection site that resolved by days 4 and 8 after DS injection, respectively. In the Morphine group, three subjects experienced moderate fever and one subject vomited twice. Both groups expressed a high degree of satisfaction with postoperative analgesia (100% vs 95% in DS and Morphine groups, respectively).
Discussion
To the best of our knowledge, this is the first report describing the analgesic impact of preoperative extended-release DS compared with regular intravenous morphine for the management of post-LC pain. Among our findings, there were no statistically significant differences in average visual analog scale pain score when comparing those in the DS group to those treated postoperatively with intravenous morphine alone. Mean VAS score at each time-point was below 2; this result indicates that both groups received adequate postoperative pain relief. The fact that no opioids were required after 24 hours post-procedure underscores the efficacy of preoperative DS; administration of DS prior to LC resulted in no severe adverse events and was not associated with any opioid antagonism among the patients in this study.
However, our findings contradict those in a previous report in which preoperative injection of DS was associated with a superior level of pain reduction and use of fewer rescue analgesics compared to placebo in patients undergoing hemorrhoidectomy.13 When analyzing outcomes from these two studies, there are important differences that provide clarification. First, the magnitude of postoperative acute pain after LC was much lower (minimum to maximum range: 0.8–1.5) than that associated with hemorrhoidectomy (minimum to maximum range: 2.4~5.4) during the first day after surgery;17,21 as such, the absolute magnitude of pain reduction associated with DS administration was more difficult to evaluate in our study.22 Second, hemorrhoid pain is a mix of visceral and somatic pain and typically persists for several days at the moderate to severe level. However, compared with previous studies that focus on LC, the pain profile is typically at mild intensity (VAS was less than 3) at 24 hours after surgery; as such, the impact of analgesics was not easily recognized. Finally, our study was not double-blinded; as such, it is possible that preoperative treatment with DS may promote some degree of bias based on psychological expectations.23 Interestingly, Riest et al24 found that preoperative analgesia with a single dose of the cyclooxygenase inhibitor, parecoxib, did not lead to universally positive effects on pain score or on the total amount of rescue medication required when compared to use of postoperative analgesia alone.
There were several limitations to our study. First, we did not include a placebo control which would have permitted us to compare our findings to those reported previously. Moreover, patients with gallstones or cholecystitis were eligible for enrollment in this study; as such, tissue inflammation or infection prior to surgery might have an impact on valid postoperative pain and increase incidence of postoperative fever.25 In addition, in accordance with our standard of care, all patients in this study received antiemetics as needed, although this was not mandatory before surgery; as such, it is possible that was associated with the increased incidence of nausea and vomiting in DS group. From our study, we recommend routine administration of antiemetics to be given with preoperative DS treatments.
Due to the fact that nalbuphine is a kappa-receptor agonist and mu-receptor antagonist, it has the potential to increase the risk of opioid-antagonism; this could result in the need for an increased anesthetic dose and/or more severe adverse events. However, our results revealed no significant differences in anesthetic consumption during the procedure. Indeed, more postoperative nausea and vomiting were observed among patients in DS group; these events were mild and resolved in a few hours after appropriate treatment with antiemetics. Most of the rescue morphine used among those in the DS group was for abdominal pain associated with carbon dioxide retention while in the PACU. Carbon dioxide retention in the abdomen, which is typically trapped between liver and right diaphragm, was reported to be a significant cause of abdominal pain and referred shoulder pain.26–28 It is possible that this factor contributes to the consumption of more opioid analgesics at day 1, although this was not observed in the previous hemorrhoidectomy study.
One study has reported that administration of low concentrations of nalbuphine may result in diminished levels of opioid-related side effects, such as pruritus.29 Our results showed that the two treatments had a similar safety profile; none of the patients treated with DS reported postoperative pruritus, even among those who received rescue morphine. Based on pharmacokinetics of DS, the relatively low concentration of nalbuphine provided a background analgesic effect30 which might explain why patients in DS group required no supplemental analgesics after 24 hours, by contrast those in the postoperative morphine group.
Conclusion
This study demonstrated that patients receiving preoperative treatment with DS consumed less opioid analgesic after 24 hours with similar satisfactory levels of post-LC pain relief.
Data Sharing Statement
The data collected or analyzed in this study will be available 6 months after publication. Anyone who wishes to access those data could contact the corresponding author by email.
Acknowledgments
The authors are grateful to the participating patients.
Funding
The authors have no sources of funding to declare for this manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Simpson JC, Bao X, Agarwala A. Pain management in enhanced recovery after surgery (ERAS) protocols. Clin Colon Rectal Surg. 2019;32(2):121–128. doi:10.1055/s-0038-1676477
2. Garimella V, Cellini C. Postoperative pain control. Clin Colon Rectal Surg. 2013;26(3):191–196. doi:10.1055/s-0033-1351138
3. Khansari M, Sohrabi M, Zamani F. The usage of opioids and their adverse effects in gastrointestinal practice: a review. Middle East J Dig Dis. 2013;5(1):5–16.
4. Hollmann MW, Rathmell JP, Lirk P. Optimal postoperative pain management: redefining the role for opioids. Lancet. 2019;393(10180):1483–1485. doi:10.1016/S0140-6736(19)30854-2
5. Gerges FJ, Kanazi GE, Jabbour-Khoury SI. Anesthesia for laparoscopy: a review. J Clin Anesth. 2006;18(1):67–78. doi:10.1016/j.jclinane.2005.01.013
6. Alexander JI. Pain after laparoscopy. Br J Anaesth. 1997;79(3):369–378. doi:10.1093/bja/79.3.369
7. Blichfeldt-Eckhardt MR, Ording H, Andersen C, Licht PB, Toft P. Early visceral pain predicts chronic pain after laparoscopic cholecystectomy. Pain. 2014;155(11):2400–2407. doi:10.1016/j.pain.2014.09.019
8. Correll D. Chronic postoperative pain: recent findings in understanding and management. F1000Research. 2017;6:1054. doi:10.12688/f1000research.11101.1
9. Vadivelu N, Mitra S, Schermer E, Kodumudi V, Kaye AD, Urman RD. Preventive analgesia for postoperative pain control: a broader concept. Local Reg Anesth. 2014;7:17–22. doi:10.2147/LRA.S62160
10. Lim SH, Jang EH, Kim MH, et al. Analgesic effect of preoperative versus intraoperative dexamethasone after laparoscopic cholecystectomy with multimodal analgesia. Korean J Anesthesiol. 2011;61(4):315–319. doi:10.4097/kjae.2011.61.4.315
11. Wilson YG, Rhodes M, Ahmed R, Daugherty M, Cawthorn SJ, Armstrong CP. Intramuscular diclofenac sodium for postoperative analgesia after laparoscopic cholecystectomy: a randomised, controlled trial. Surg Laparosc Endosc. 1994;4(5):340–344. doi:10.1097/00019509-199410000-0000
12. Møiniche S, Kehlet H, Dahl JB. A qualitative and quantitative systematic review of preemptive analgesia for postoperative pain relief: the role of timing of analgesia. Anesthesiology. 2002;96(3):725–741. doi:10.1097/00000542-200203000-00032
13. Yeh CY, Jao SW, Chen JS, et al. Sebacoyl dinalbuphine ester extended-release injection for long-acting analgesia: a multicenter, randomized, double-blind, and placebo-controlled study in hemorrhoidectomy patients. Clin J Pain. 2017;33(5):429–434. doi:10.1097/AJP.0000000000000417
14. Alimian M, Pournajafian A, Kholdebarin A, Ghodraty M, Rokhtabnak F, Yazdkhasti P. Analgesic effects of paracetamol and morphine after elective laparotomy surgeries. Anesth Pain Med. 2014; 4(2). doi:10.5812/aapm.12912
15. Wiesel S, Grillas R. Patient-controlled analgesia after laparoscopic and open cholecystectomy. Can J Anaesth. 1995;42(1):37–40. doi:10.1007/BF03010569
16. Bisgaard T. Analgesic treatment after laparoscopic cholecystectomy: a critical assessment of the evidence. Anesthesiology. 2006;104(4):835–846. doi:10.1097/00000542-200604000-00030
17. Richebé P, Capdevila X, Rivat C. Persistent postsurgical pain: pathophysiology and preventative pharmacologic considerations. Anesthesiology. 2018;129(3):590–607. doi:10.1097/ALN.0000000000002238
18. Feng YP, Huang NC, Chang HJ, Wong CS. The role of epidural anesthesia plus ultrasound-guided peripheral nerve block and naldebain in chronic post-surgical pain. Taiwan J Pain. 2018;28(1):22–29.
19. Huang WH, Huang NC, Lin JA, Wong CS. Multimodal analgesia for shoulder rotator cuff surgery pain: the role of Naldebain® and ultrasound-guided peripheral nerve blocks combination. J Med Sci. 2020. doi:10.4103/jmedsci.jmedsci_33_20
20. Huang C, Sun WZ, Wong CS. Prevention of chronic postsurgical pain: the effect of preventive and multimodal analgesia. Asian J Anesthesiol. 2018;56(3):74–82. doi:10.6859/aja.201809_56(3).0002
21. Gerbershagen HJ, Aduckathil S, van Wijck AJM, Peelen LM, Kalkman CJ, Meissner W. Pain intensity on the first day after surgery: a prospective cohort study comparing 179 surgical procedures. Anesthesiology. 2013;118(4):934–944. doi:10.1097/ALN.0b013e31828866b3
22. Myles PS, Myles DB, Galagher W, et al. Measuring acute postoperative pain using the visual analog scale: the minimal clinically important difference and patient acceptable symptom state. Br J Anaesth. 2017;118(3):424–429. doi:10.1093/bja/aew466
23. Bingel U, Wanigasekera V, Wiech K, et al. The effect of treatment expectation on drug efficacy: imaging the analgesic benefit of the opioid remifentanil. Sci Transl Med. 2011;3(70):70ra14. doi:10.1126/scitranslmed.3001244
24. Riest G, Peters J, Weiss M, et al. Preventive effects of perioperative parecoxib on post-discectomy pain. Br J Anaesth. 2008;100(2):256–262. doi:10.1093/bja/aem345
25. Indar AA, Beckingham IJ. Acute cholecystitis. BMJ. 2002;325(7365):639–643. doi:10.1136/bmj.325.7365.639
26. Slim K, Bousquet J, Kwiatkowski F, Lescure G, Pezet D, Chipponi J. Effect of CO(2) gas warming on pain after laparoscopic surgery: a randomized double-blind controlled trial. Surg Endosc. 1999;13(11):1110–1114. doi:10.1007/s004649901184
27. Phelps P, Cakmakkaya OS, Apfel CC, Radke OC. A simple clinical maneuver to reduce laparoscopy-induced shoulder pain: a randomized controlled trial. Obstet Gynecol. 2008;111(5):1155–1160. doi:10.1097/AOG.0b013e31816e34b4
28. Tsai HW, Chen YJ, Ho CM, et al. Maneuvers to decrease laparoscopy-induced shoulder and upper abdominal pain: a randomized controlled study. Arch Surg. 2011;146(12):1360–1366. doi:10.1001/archsurg.2011.597
29. Yeh YC, Lin TF, Lin FS, Wang YP, Lin CJ, Sun WZ. Combination of opioid agonist and agonist-antagonist: patient-controlled analgesia requirement and adverse events among different-ratio morphine and nalbuphine admixtures for postoperative pain. Br J Anaesth. 2008;101(4):542–548. doi:10.1093/bja/aen213
30. Tien YE, Huang WC, Kuo HY, et al. Pharmacokinetics of dinalbuphine sebacate and nalbuphine in human after intramuscular injection of dinalbuphine sebacate in an extended-release formulation. Biopharm Drug Dispos. 2017;38(8):494–497. doi:10.1002/bdd.2088
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
