Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Predictors of Current DSM-5 PTSD Diagnosis and Symptom Severity Among Deployed Veterans: Significance of Predisposition, Stress Exposure, and Genetics
Authors Hu Y , Chu X , Urosevich TG, Hoffman SN, Kirchner HL , Adams RE , Dugan RJ , Boscarino JJ , Shi W, Withey CA , Figley CR , Boscarino JA
Received 27 August 2019
Accepted for publication 7 December 2019
Published 8 January 2020 Volume 2020:16 Pages 43—54
DOI https://doi.org/10.2147/NDT.S228802
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Roger Pinder
Yirui Hu,1 Xin Chu,2 Thomas G Urosevich,3 Stuart N Hoffman,4 H Lester Kirchner,1 Richard E Adams,5 Ryan J Dugan,6 Joseph J Boscarino,7 Weixing Shi,2 Carrie A Withey,6 Charles R Figley,8 Joseph A Boscarino6
1Biomedical and Translational Informatics, Geisinger Clinic, Danville, PA, USA; 2Obesity Institute, Geisinger Clinic, Danville, PA, USA; 3Ophthalmology Service, Geisinger Clinic, Mount Pocono, PA, USA; 4Sleep Disorders Center, Geisinger Clinic, Danville, PA, USA; 5Department of Sociology, Kent State University, Kent, OH, USA; 6Department of Population Health Sciences, Geisinger Clinic, Danville, PA, USA; 7Department of Clinical Psychology, William James College, Newton, MA, USA; 8Department of Social Work, Tulane University, New Orleans, LA, USA
Correspondence: Joseph A Boscarino
Geisinger Clinic, 100 N. Academy Avenue, 44-00, Danville, PA 17822, USA
Tel +1570-214-9622
Email [email protected]
Background: Previously we reported a genetic risk score significantly improved PTSD prediction among a trauma-exposed civilian population. In the current study, we sought to assess this prediction among a trauma-exposed military population.
Methods: We examined current PTSD diagnosis and PTSD symptom severity among a random sample of 1042 community-based US military veterans. Main effects and interaction effects were assessed for PTSD genetic risk by trauma exposure using cross-product terms for PTSD x trauma exposures, including combat, lifetime trauma, and adverse childhood exposures. The PTSD risk variants studied were within genetic loci previously associated with PTSD, including CRHR1, CHRNA5, RORA, and FKBP5 genetic variants, which were used to calculate a total PTSD genetic risk score (range=0–8, mean=3.6, SD=1.4).
Results: Based on DSM-5 PTSD criteria, 7.1% of veterans (95% CI=5.6–8.8) met criteria for current PTSD. The PTSD genetic risk count was significantly higher among PTSD cases vs non-cases (3.92 vs 3.55, p=0.027). Since the PTSD genetic risk score was not significant in the PTSD diagnosis model, we assessed this association using PTSD symptom severity. Because these symptom data were skewed (mean=9.54, SD=12.71, range=0–76), we used negative binomial regression to assess this outcome. This symptom model included a PTSD genetic risk score, demographic factors, trauma exposures, current insomnia, current depression, concussion history, and attention-deficit disorder, expressed as incident rate ratios (IRR), which is an estimate of one-unit increase in PTSD severity, given other variables are held constant. Variables in the final model included age and sex (both p<0.001), PTSD genetic risk (IRR=1.02, p=0.028), warzone tours (IRR=0.94, p=0.003), childhood abuse (IRR=1.50, p<0.0001), current depression (IRR=1.89, p<0.0001), current insomnia (IRR=2.58, p<0.0001), low social support (IRR=1.19, p<0.0001), attention-deficit disorder (IRR=1.51, p<0.0001), agreeable personality (IRR=0.77, p<0.0001), and concussion (IRR=1.38, p<0.0001). Significant interactions were detected for combat and lifetime trauma exposure by PTSD genetic risk (both p<0.0001), suggesting that the impact of trauma exposures on PTSD severity was lower when the PTSD genetic risk was higher.
Conclusion: Both warzone and non-warzone factors predicted current PTSD symptoms among veterans, including a PTSD genetic risk score. Interaction effects were detected for combat exposure and lifetime trauma by genetic risk score for PTSD symptoms, suggesting that PTSD symptom manifestation was more dependent on PTSD risk variants than the level of trauma or combat exposure. This suggests that controlling for other factors, the absence of genetic risk variants may confer PTSD resilience. Further research is planned.
Keywords: posttraumatic stress disorder, veterans, combat exposure, trauma, warzone deployment, genetic risk, resilience
Introduction
While studies suggest that most individuals have experienced lifetime traumatic events, relatively few develop posttraumatic stress disorder (PTSD).1–3 Studies indicate that PTSD is moderately heritable, with approximately 30% of variance accounted for by genetic factors.4 Currently, genetic components for PTSD have been identified, which may explain this risk.5–8 As we describe below, these genetic components include biologic pathways involving the hypothalamic-pituitary-adrenal (HPA), locus coeruleus, noradrenergic, and the limbic system, among others.6,9–11 Recent large-scale genome-wide association studies (GWAS) of PTSD have provided new insights and have suggested overlap with other disorders, such as schizophrenia and bipolar disorder.12,13 In particular, the Psychiatric Genomics Consortium-Posttraumatic Stress Disorder group (PGC-PTSD) combined genome-wide data across multiethnic studies to quantify PTSD heritability, to examine potential shared genetic risk with schizophrenia, bipolar disorder, and major depressive disorder and to identify risk loci for PTSD.12 We review and discuss these GWAS findings in the discussion section of the paper. Recent research suggests that PTSD genetics are complex, multicausal and that the biologic pathways for this disorder are yet to be elucidated.14,15 As described below, the genetic risk factors for PTSD that we assessed were among outpatients, all of whom were formerly deployed military veterans receiving care in a large multi-hospital system in central Pennsylvania.16–19 Although the number of veterans in these settings with PTSD has varied, about 7–10% of them have been diagnosed with current PTSD in recent years,16,18,20 which is consistent with the prevalence rate in the current study.
Methods
Sample
The population for the current study included a sample of community-based veterans of Vietnam, Persian Gulf, Iraq/Afghanistan, and/or other recent conflicts, who were receiving care in a large non-VA multihospital system in Pennsylvania.17,18 We examined post-deployment PTSD status among a cross-sectional survey of 1730 veterans, who were receiving outpatient care in Pennsylvania at a Geisinger Clinic facility. Approximately 95% of the veterans were male (mean age=61.4 [SD=12.1]). For the study, 65 min diagnostic interviews were conducted by telephone, and DNA samples were collected via postage-paid return mail following the survey. To date, approximately 35,000 patients have provided this military information, and this database was used to select a random sample of veterans for the current study.18 Geisinger Clinic serves more than 3 million residents throughout 45 counties in central, south-central and northeast Pennsylvania, which encompasses a 25,000 square mile (40,000 square kilometer) primary service area.21
With patient consent, trained interviewers administered a structured health interview by telephone from February 2016 through February 2017. All veterans recruited had one or more warzone deployments. Veteran status and deployment history were confirmed based on military records provided by the veteran. Among the veterans selected for the surveys, all were under 76 and served in Vietnam or in another post-Vietnam conflict. After 10 telephone calls per household, we were able to complete 1730 interviews for an estimated survey cooperation rate of 55% among those eligible for the survey.22,23 Deceased patients, nursing home patients, institutionalized patients, those who did not serve in Vietnam, Iraq, Afghanistan, Global War on Terrorism, Persian Gulf, or other recent post-Vietnam conflicts were excluded, as were those who were cognitively impaired, and those unavailable during the survey period. Of those who were surveyed, 65% (n=1074) returned usable DNA samples by mail. Oragene DNA saliva kits, manufactured by Genotek (Ottawa, Ontario, Canada), were used to collect the DNA. To avoid confounding results due to genetic admixture,24 non-Caucasian veterans (n=40) were excluded from the analyses, resulting in a final DNA sample of 1042 veterans.
Genetic Methods
Genotyping was performed on an Applied BioSystems 7500 real-time PCR platform (Foster City, CA), using TaqMan kits following the manufacturer’s protocols. Quality control measures included visual inspection of the allelic discrimination plots, monitoring concordance of cross-plated duplicate pairs, monitoring the overall call rate, and monitoring agreement with Hardy-Weinberg expectations.25 The overall call rate for the current study was approximately 95% and the genotypes for the current study met Hardy-Weinberg expectations.25
PTSD Measures and Study Predictor Variables
To assess PTSD in our study, we used an instrument based on the Diagnostic and Statistical Manual of Mental Disorder, Fifth Edition (DSM-5), the PTSD Checklist for Diagnostic and Statistical Manual of Mental Disorders-Fifth Edition (PCL-5).26,27 To receive a diagnosis of current PTSD, veterans had to meet the DSM-5 diagnostic criteria A through G within the past 12 months.28 In addition to a PTSD diagnosis, we also included a count of current DSM-5 PTSD symptom severity (mean=9.54, SD=12.71, range =0–76). This PTSD scale has been used in recent PTSD studies and appears to produce valid and reliable results (Cronbach Alpha=0.92).18,29–31
Predictor variables in the current study also included demographic variables (age, sex, marital status), number of warzone deployments (one vs more than one), childhood abuse history, combat exposure, lifetime traumatic event exposure, current level of social support, history of attention-deficit symptoms, history of in-service concussion, current depression, current insomnia, and select personality traits.32,33 Childhood abuse was measured by the Adverse Childhood Experiences (ACE) scale, a measure used and validated in numerous studies (Cronbach Alpha=0.84).34–36 Combat exposure was measured by the Combat Experience Scale (CES), which is a widely used measure of combat used in Vietnam veteran studies.37,38 Versions of this scale have been used in studies since the 1970s.39–43 Based on previous research, scale measurement for combat exposure was divided into low vs high cut-points described elsewhere (Cronbach Alpha=0.84).20,44,45 To assess trauma exposure, the Lifetime Traumatic Event Scale was used, which measured the occurrence of 12 lifetime traumatic events (e.g., forced sexual contact, domestic abuse, a serious accident, experiencing a major disaster).46 Based on previous research,20 we collapsed these exposures into high vs low count categories. This traumatic event scale was developed from other trauma studies, used in the previous research, and had good reported reliability and validity.20,46–49
Current social support was also measured by a version of a social support scale used in the Medical Outcomes Study.50 This scale has been used in previous trauma studies and is considered a reliable and valid measure of current social support (Cronbach Alpha=0.84).46,48,51 This scale was used as a categorical measure in the current study, with low social support defined as the lowest quintile.49 Attention-Deficit/Hyperactivity Disorder was measured by a scale included in the World Health Organization Attention Attention-Deficit Disorder instrument (Cronbach Alpha=0.65).52 History of in-service concussion was measured based on reported concussions experienced during military deployments (e.g., ever dazed, confused, saw stars, or knocked out), and for this, we used the Brief TBI Screen,53 a scale used in the previous research.20,54 Personality measures assessed included the “agreeable” personality trait, which was measured by the 10-item Personality Inventory, a widely used and validated personality measure.32,33 This trait was selected because it tended to be protective for the onset of mental health disorders.55,56
Depression was assessed using the Major Depressive Disorder (MDD) measure developed from the DSM-IV,57–59 which was used extensively in previous trauma studies.20,47,49,60,61 Data related to the validity of this depression scale were previously reported and suggest this scale can be used to diagnose depression in population studies (Cronbach Alpha=0.87).47,60 To meet the criteria for MDD in the study, subjects had to meet the full DSM-IV criteria for major depression within the past 12 months. Insomnia was assessed using scale items related to difficulty staying or falling asleep in the past year, which was included in the previous research.62,63
Genetic Risk Factor Approach
Much like our previous study, we used a genetic risk score approach, which included 4 SNP genetic variants.8 The SNPs included in the PTSD genetic risk model are presented in Table 1. These SNP markers were combined into a cumulative risk allele count, as is common in these genetic analyses.62 For the current study, we assessed 4 genetic markers using a cumulative risk-allele model to test for an association with PTSD among outpatients, comparable to what has been undertaken to predict genetic associations in other clinical areas.64,65 Extending previous research,8,10 we specifically genotyped SNPs located within the FK506 binding protein-5 (FKBP5), retinoid-related orphan receptor alpha (RORA), cholinergic receptor nicotinic alpha-5 (CHRNA5), and the corticotropin-releasing hormone receptor-1 (CRHR1) gene clusters and assessed these markers for cumulative risk for current PTSD.
 |
Table 1 Profile of Single Nucleotide Polymorphisms (SNPs) Included in PTSD Genetic Risk Model |
The RORA genetic marker examined (rs8042149) is associated with PTSD in recent research.66,67 The FKBP5 genetic marker studied (rs9470080) regulates glucocorticoid receptor sensitivity, is functionally involved in HPA axis activity, and is also associated with PTSD.5,6,10,68 The CHRNA genetic marker investigated (rs16969968), which encodes components of the nicotinic acetylcholine receptor (nAChR), is associated with nicotine dependence and cigarette smoking,69,70 substance misuse,71 and PTSD.10 The CRHR1 genetic marker studied (rs110402) is a polypeptide hormone and neurotransmitter involved in corticotropin-releasing hormone activity associated with the stress response. Studies suggest that this gene also regulates HPA axis function and is associated with the impact of traumatic stress exposure and PTSD.68,72
PTSD risk alleles were counted for each of the 4 genetic variants included, which resulted in a genetic risk score ranging from 0 to 8 (mean=3.56, SD=1.41), using the PTSD risk alleles variants (A, G, T, respectively) shown in Table 1. This genetic risk method has been described in detail elsewhere8 Assessment of these 4 PTSD genetic markers indicated that all were positively associated with PTSD, with two markers (RORA and FKBP5), significantly associated (both p-values < 0.05). In addition, the cumulated unadjusted PTSD genetic risk score was also significantly associated with PTSD diagnosis (p-value = 0.0275). This genetic risk score was then used as a covariate in our statistical analyses using standard bivariate and multivariate tests.8 PTSD risk alleles counted for each of the 4 genetic variants included 0, 1, 2 risk alleles, resulting in genetic risk score ranging from 0–8 (mean=3.56, SD=1.41), using the risk alleles variants (A, G, T, respectively). The frequencies for these alleles in the current study were CRHR1: A/A = 208 (18.9%); A/G = 535 (48.7%); G/G = 355 (32.3%). CHRNA5: G/G = 446 (40.8%); A/G = 488 (44.6%); A/A = 160 (14.6%). RORA: T/T = 283 (25.6%) G/T = 513 (46.5%) G/G 308 = (27.9%). FKBP5: C/C = 496 (45.2%) C/T = 482 (43.9%) T/T = 119 (10.9%) (see Table 1).
Statistical Analysis
Analyses for the current research included describing the profile of genetic variants studied (Table 1), as well as descriptive statistics depicting the study population and evaluating the impact of both stress exposures and genetic factors on PTSD status, including current diagnosis and symptom status. For descriptive purposes, we present the unadjusted, bivariate baseline characteristics and genetic risk scores of the overall study population in Table 2, with mean (standard deviation) summarized for continuous variables, and frequency (percentage) for categorical variables, by PTSD diagnosis. For multivariate analyses on the PTSD diagnosis outcome, we performed logistic regression, whereby key risk/protective factors (stress exposures, genetic risk score), as well as the interaction term between stress exposures and genetic risk score were included to estimate the odds ratios (ORs), after controlling for age, sex and other factors that might affect the associations by including them in the logistic regression analyses8,62,73 (Table 3). For multivariate analyses on PTSD symptom severity, we fitted a negative binomial count regression model due to the overdispersion of these symptom data (Figure 1), whereby key risk/protective factors (stress exposures, genetic risk score), as well as the interaction term between stress exposures and the genetic risk score were included to estimate the incidence rate ratios (IRRs), controlling for age, sex and other factors that might affect the associations by including them in the regression analyses74 (Table 4). The incident rate ratio is an estimate of one-unit increase in PTSD symptom severity, given the other variables are held constant in the model. For the interaction assessment in the multivariate analyses, significant interaction terms between stress exposures and the genetic risk score are presented in Table 5. Analyses were performed in SAS 9.4 (SAS Institute, Cary NC) using the LOGISTIC procedure for logistic model and GENMOD procedure for negative binomial model.
 |
Figure 1 Distribution of PTSD symptom severity in study sample (range=0–80; mean=9.54; SD=12.71). |
 |
Table 2 Descriptive Statistics of the Study Sample, Overall and by PTSD Diagnosis (N=1042) |
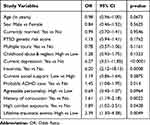 |
Table 3 Logistic Regression Results for Predicting PTSD Diagnosis (N=1042) |
 |
Table 4 Negative Binomial Regression Results for Predicting PTSD Symptom Severity (N=1042) |
 |
Table 5 Interaction Effects from Negative Binomial Regression* |
This study was approved by the Institutional Review Boards of the Geisinger Clinic (IRB #2015-0441) and the US Department of Defense. All patients provided their verbal and written informed consent to participate in this study and were offered small monetary incentives for participation. This study was conducted in accordance with the Declaration of Helsinki.
Results
Current PTSD Diagnosis and PTSD Symptom Severity
Based on the full DSM-5 criteria, 75 cases were diagnosed with PTSD for an estimated prevalence of 7.1% in the past 12 months (95% CI = 5.6–8.8%). Figure 1 presents the distribution of PTSD symptom severity among the veterans, which was highly skewed. As seen, the unconditional mean of this outcome measure is much lower than its variance (mean score = 9.5 [SD=12.8]), prompting use a negative binomial regression model for our analysis of this outcome.74
Descriptive Statistics
Using the psychometric and demographic data collected in the study interview, we present the descriptive statistics using mean with standard deviation for continuous variables and frequency with percentage for categorical variables. Baseline characteristics and PTSD genetic risk scores of the study population are examined by PTSD diagnosis outcome in Table 2. Veterans diagnosed with PTSD tended to be younger (54 vs 60 years old, t-test = 48.3, p<0.001) and have a higher PTSD genetic risk score (3.92 vs 3.55, p=0.027). Veterans diagnosed with PTSD also are more likely to have high combat exposure (46% vs 20%, p<0.0001), high childhood abuse and neglect (33% vs 16%, p=0.0002), high lifetime trauma event exposure (42% vs 17%, p<0.0001), probable attention deficit (55% vs 17%, p<0.0001), history of concussion (67% vs 25%, p<0.0001), current depression disorder (50% vs 5%, p<0.0001), current insomnia (95% vs 53%, p<0.0001), and more likely to have low social support (37% vs 17%, p<0.0001). Finally, the PTSD-positive veterans are less likely to have an agreeable personality trait (11% vs 31%, p=0.0001), suggesting this trait is protective for PTSD.
Logistic Regression Predicting DSM-5 PTSD Diagnosis
Significant variables in the PTSD diagnosis model (Table 3), and expected based on previous research, included current depression (OR=6.27, 95% CI: 3.31–11.85, p<0.0001), current insomnia (OR=6.20, 95% CI: 2.12–18.13, p=0.0008), history of ADHD (OR=1.45, 95% CI: 1.08–1.95, p=0.014), and history of concussion (OR=1.61, 95% CI: 1.19–2.18, p=0.0022). Inconsistent with our previous research with nonveterans, however, no interaction effects were detected for the genetic risk score by high trauma or high combat exposure, although both high combat and high trauma exposures were significant (Table 3).
Negative Binomial Regression Predicting DSM-5 PTSD Symptom Severity
Significant variables in the final PTSD symptom severity model (Table 4), also predicted PTSD symptoms, expressed as incident rate ratios (IRR), which is an estimate of one-unit increase in PTSD severity, given other variables are held constant. As can be seen in Table 4, significant variables included PTSD genetic risk score (IRR =1.02, 95% CI: 1.00–1.05, p=0.028), multiple warzone tours (IRR=0.94, 95% CI: 0.90–0.98, p=0.003), the latter being protective, childhood abuse/neglect (IRR =1.50, 95% CI: 1.33–1.70, p<0.0001), current depression (IRR=1.89, 95% CI: 1.79–1.99, p<0.0001), current insomnia (IRR=2.58, 95% CI: 2.44–2.73, p<0.0001), low social support (IRR=1.19, 95% CI: 1.14–1.25, p<0.0001), history of ADHD (IRR=1.51, 95% CI: 1.44–1.58, p<0.0001), and history of in-service concussion (IRR=1.38, 95% CI: 1.32–1.44, p<0.0001).
Interaction Terms
Significant interaction effects were detected for traumatic events by genetic risk score. Similar trends were observed for both high combat exposure and high lifetime traumatic event exposure in predicting PTSD symptom severity, whereby the impact on veterans without high combat or high lifetime traumatic events was less than those with combat or lifetime traumatic events, as the genetic risk score increases from 0 to 8.
Table 5 presents the detailed interaction effects from the negative binomial regression model for lifetime trauma exposure (left panel). As can be seen, when the genetic risk score was 1 (relatively low), veterans with high lifetime traumatic events were expected to have 1.72 times in PTSD symptom severity compared to those with low lifetime traumatic events (95% CI: 1.54–1.93, p<0.0001). When the genetic risk score was 4 (median), veterans with high lifetime traumatic events were expected to have 1.56 times in PTSD symptom severity compared to those with low lifetime traumatic events (95% CI: 1.39–1.75, p<0.0001). When the genetic risk score is 7 (relatively high), veterans with high lifetime traumatic events were expected to have 1.41 times in PTSD symptom severity compared to those with low lifetime traumatic events (95% CI: 1.23–1.62, p<0.0001).
Finally, the right panel in Table 5 shows the interaction effects from the negative binomial regression model for combat exposure and PTSD symptom severity. As can be seen, when the genetic risk score was 1 (relatively low), veterans with high combat exposure were expected to have 1.58 times in PTSD severity score compared to those with low combat exposures (95% CI: 1.42–1.76, p<0.0001). When the genetic risk score was 4 (median), veterans with high combat exposures were expected to have 1.43 times in PTSD symptom severity compared to those with low combat exposures (95% CI: 1.27–1.61, p<0.0001). When the genetic risk score was 7 (relatively high), veterans with high combat exposures were expected to have 1.29 times in PTSD symptom severity score compared to those with low combat exposures (95% CI: 1.11–1.50, p<0.0008).
Table 5 shows the interaction effects from the negative binomial regression model for both trauma and combat exposures and PTSD symptom severity, respectively. Figure 2A and B show the statistical estimates for PTSD symptom severity score with 95% confidence limits for both trauma and combat exposures using these estimates.
Discussion
In the current study, we examined post-deployment PTSD status, including current DSM-5 PTSD diagnosis and PTSD symptom severity, among a sample of 1042 community-based veterans. Interaction effects were also assessed for a genetic PTSD risk score by trauma exposures. As noted, the PTSD risk variants studied were within genetic loci previously associated with PTSD, including CRHR1, CHRNA5, RORA, and FKBP5 genetic variants, which were used to calculate a PTSD genetic risk score (range=0–8, mean=3.6, SD=1.4). All veterans studied received routine health care in a large multihospital system in Pennsylvania. Based on DSM-5 PTSD criteria, 7.1% of veterans (95% CI=5.6–8.8) met the criteria for current PTSD. The unadjusted PTSD genetic risk count was significantly higher among PTSD cases vs non-cases (p=0.027). However, since the PTSD risk score was not significant in the PTSD diagnosis model, we assessed this using DSM-5 PTSD symptom severity. Because these symptom data were skewed (range 0–76, mean=9.5, SD=12.7), as discussed, we used the negative binomial regression model to assess this association.
The model for PTSD symptom severity included the genetic score, demographic factors (age and sex) trauma exposures, current insomnia, current depression, history of concussion, and history of attention-deficit disorder. Significant variables in the final PTSD symptom severity model included age, sex, PTSD genetic risk score, combat exposure, multiple warzone tours, childhood abuse and neglect, current depression, current insomnia, lifetime traumatic events, social support, history of attention-deficit disorder, and history of in-service concussion, previously identified in the past research.18,20,44 Significant interaction effects were also detected for both high combat and high lifetime traumatic event exposures by genetic risk score, which suggested the impact of these trauma exposures on PTSD symptom severity manifestation was lower when the genetic risk was higher. Thus, our study appears to have replicated previous results with a trauma-exposed civilian population.8 After deployment, both warzone and non-warzone factors predicted current PTSD among veterans seen in Geisinger Clinic outpatient facilities, including a genetic risk score for PTSD. Interaction effects were detected for combat exposure and lifetime traumatic events by genetic risk score for PTSD symptom severity. One noteworthy finding is the protective effect found for multiple warzone deployments vs only one deployment (Table 4), which may reflect high resilience in these veterans, but further research is required to confirm this.
Study limitations include the fact that our interview data were based on self-report, our sample was limited, and that our study participants were mostly male and drawn from a hospital population in central Pennsylvania. These factors may have biased our results and could limit generalization. The fact that our sample is 95% male is also a potential limitation, since the differences in PTSD biology likely vary significantly by sex.13,75 Our previous PTSD-genetic study was mostly female.62 Also, the total number of currently diagnosed PTSD cases in our study was limited (n=75). Thus, our findings will require further replication.
Conclusion
Previously we reported a lower PTSD prevalence among non-military personnel with no or few PTSD genetic variants, irrespective of trauma exposure,8 which is consistent with the current findings. In the present study, we show that PTSD symptom severity decreases by level of trauma exposure (for both combat and lifetime trauma exposures) when the PTSD genetic risk score goes up (see: Figure 2A and B), suggesting that PTSD symptom severity may be more dependent on genetic risk variants than level of trauma exposure, per se. We suspect that the absence of these PTSD risk genetic variants may also be associated with resilience to other psychological disorders, such as addiction,24,71,76,77 but further research is required to confirm this hypothesis. Nevertheless, while the impact of the genetic markers studied was statistically significant, they still were relatively small. Noteworthy, however, is that not only veterans but those employed in law-enforcement, public safety, and emergency medical work, as well as those exposed to disasters and accidents may also be similarly impacted,78 so these genetic findings may have wider implications.
In summary, our study replicates previous results with a trauma-exposed population. Both warzone and non-warzone factors predicted current PTSD among US veterans, including a PTSD genetic risk score. Interaction effects were detected for combat and lifetime trauma exposures by genetic risk score for PTSD symptom severity, suggesting that PTSD symptom manifestation may be more dependent on having the PTSD genetic risk variants than the level of lifetime trauma or combat exposure, indicating that absence of these risk variants may also confer PTSD genetic resilience.8 Recently, large-scale genome-wide association studies (GWAS) of PTSD have reported genetic overlap with other major disorders, such as schizophrenia and bipolar disorder,12,13 suggesting that PTSD genetics are very complex. Other complications include the diagnostic criteria for PTSD, the diversity of study populations, and the contingency of PTSD on the occurrence of trauma exposure.13 Consequently, given these factors, we may have neither properly controlled for nor have identified all potential risk factors in the current study.
To our knowledge, however, our team was the first to report the association between the cholinergic receptor nicotinic alpha-5 gene and PTSD. This is interesting because PTSD has been associated with increased cigarette smoking post-trauma,79 which is presumed due to the psychoactive effect of nicotine on PTSD symptom reduction.10,80 However, this genetic risk may be on both behavioral and biologic causal pathways associated with PTSD.71 Additionally, it is interesting to note that contemporary discussions of PTSD psychobiology now often include recognition of immunologic and metabolic involvement in PTSD psychopathology,12,13 something we reported some time ago.43,81–83 Again, noteworthy, is that nicotine addiction is typically also implicated in both the immunologic and metabolic disease pathways,81 suggesting additional causal confounding.14,15 Our plan is to also continue these investigations in the future.
Acknowledgments
The study team acknowledges the efforts of the veterans who took part in this study. The study Principal Investigator (JAB) dedicates this research to his twin brother, now deceased, a Vietnam combat veteran who never found peace, but motivated the PI to conduct trauma research. This paper was presented at the 25th Annual Health Care Services Research Network, Meeting, Portland, OR, April 8–10, 2019.
Disclosure
Dr Joseph A Boscarino reports grants from Geisinger Auxiliary Fund, the Kline & Ditty Health Fund, the National Institute of Mental Health (Grant No. R21-MH-086317), the Wounded Warrior Project, and US Department of Defense (Contract No. W81XWH-15-1-0506). Dr H Lester Kirchner reports grants from the US Department of Defense. The authors report no other conflicts of interest in this work.
References
1. Kessler RC, Sonnega A, Bromet E, Hughes M, Nelson CB. Posttraumatic stress disorder in the National Comorbidity Survey. Arch Gen Psychiatry. 1995;52(12):1048–1060. doi:10.1001/archpsyc.1995.03950240066012
2. Boscarino JA, Adams RE. PTSD onset and course following the World Trade Center disaster: findings and implications for future research. Soc Psychiatry Psychiatr Epidemiol. 2009;44(10):887–898. doi:10.1007/s00127-009-0011-y
3. Breslau N, Kessler RC, Chilcoat HD, Schultz LR, Davis GC, Andreski P. Trauma and posttraumatic stress disorder in the community: the 1996 Detroit Area Survey of Trauma. Arch Gen Psychiatry. 1998;55(7):626–632. doi:10.1001/archpsyc.55.7.626
4. Stein MB, Jang KL, Taylor S, Vernon PA, Livesley WJ. Genetic and environmental influences on trauma exposure and posttraumatic stress disorder symptoms: a twin study. Am J Psychiatry. 2002;159(10):1675–1681. doi:10.1176/appi.ajp.159.10.1675
5. Binder EB, Bradley RG, Liu W, et al. Association of FKBP5 polymorphisms and childhood abuse with risk of posttraumatic stress disorder symptoms in adults. JAMA. 2008;299(11):1291–1305. doi:10.1001/jama.299.11.1291
6. Koenen KC. Genetics of posttraumatic stress disorder: review and recommendations for future studies. J Trauma Stress. 2007;20(5):737–750. doi:10.1002/jts.20205
7. Nemeroff CB, Bremner JD, Foa EB, Mayberg HS, North CS, Stein MB. Posttraumatic stress disorder: a state-of-the-science review. J Psychiatr Res. 2006;40(1):1–21. doi:10.1016/j.jpsychires.2005.07.005
8. Boscarino JA, Erlich PM, Hoffman SN, Zhang X. Higher FKBP5, COMT, CHRNA5, and CRHR1 allele burdens are associated with PTSD and interact with trauma exposure: implications for neuropsychiatric research and treatment. Neuropsychiatr Dis Treat. 2012;8:131–139. doi:10.2147/NDT.S29508
9. Broekman BF, Olff M, Boer F. The genetic background to PTSD. Neurosci Biobehav Rev. 2007;31(3):348–362. doi:10.1016/j.neubiorev.2006.10.001
10. Boscarino JA, Erlich PM, Hoffman SN, Rukstalis M, Stewart WF. Association of FKBP5, COMT and CHRNA5 polymorphisms with PTSD among outpatients at risk for PTSD. Psychiatry Res. 2011;188(1):173–174. doi:10.1016/j.psychres.2011.03.002
11. Rauch SL, Drevets WC. Neuroimaging and neuroanatomy of sress-induced and fear circuitry disorders. In: Andrews G, Charney DS, Sirovatka PJ, Regier DA, editors. Stress-Induced and Fear Circuitry Disorders: Refining the Research Agenda for DSM-V. Washington, DC: American Psychiatric Association; 2009:215–254.
12. Duncan LE, Ratanatharathorn A, Aiello AE, et al. Largest GWAS of PTSD (N=20,070) yields genetic overlap with schizophrenia and sex differences in heritability. Mol Psychiatry. 2018;23(3):666–673. doi:10.1038/mp.2017.77
13. Nievergelt CM, Ashley-Koch AE, Dalvie S, et al. Genomic approaches to posttraumatic stress disorder: the psychiatric genomic consortium initiative. Biol Psychiatry. 2018;83(10):831–839. doi:10.1016/j.biopsych.2018.01.020
14. Sharma S, Ressler KJ. Genomic updates in understanding PTSD. Prog Neuropsychopharmacol Biol Psychiatry. 2019;90:197–203. doi:10.1016/j.pnpbp.2018.11.010
15. Kendler KS. From many to one to many-the search for causes of psychiatric illness. JAMA Psychiatry. 2019;76:1085. doi:10.1001/jamapsychiatry.2019.1200
16. Adams RE, Urosevich TG, Hoffman SN, et al. Social and psychological risk and protective factors for veteran well-being: the role of veteran identity and its implications for intervention. Mil Behav Health. 2019;7(3):304–314. doi:10.1080/21635781.2019.1580642
17. Boscarino JA, Adams RE, Urosevich TG, et al. Guard/Reserve service members and mental health outcomes following deployment: results from the Veterans’ Health Study. Gen Hosp Psychiatry. 2019;
18. Boscarino JA, Adams RE, Urosevich TG, et al. Mental health impact of homecoming experience among 1730 formerly deployed veterans from the Vietnam war to current conflicts results from the veterans’ health study. J Nerv Ment Dis. 2018;206(10):757–764. doi:10.1097/NMD.0000000000000879
19. Hoffman SN, Urosevich TG, Kirchner HL, et al. Grapheme-color synesthesia is associated with PTSD among deployed veterans: confirmation of previous findings and need for additional research. Int J Emerg Ment Health. 2019;21:1.
20. Boscarino JA, Hoffman SN, Pitcavage JM, Urosevich TG. Mental health disorders and treatment seeking among veterans in non-VA facilities: results and implications from the veterans’ health study. Mil Behav Health. 2015;3(4):244–254. doi:10.1080/21635781.2015.1077179
21. Boscarino JA, Kirchner HL, Pitcavage JM, et al. Factors associated with opioid overdose: a 10-year retrospective study of patients in a large integrated health care system. Subst Abuse Rehabil. 2016;7:131–141. doi:10.2147/SAR.S108302
22. Groves RM, Fowler FJ, Couper MP, Lepkowski JM, Singer E, Tourangeau R. Survey Methodology, Second Edition. New York, NY: Wiley; 2009.
23. American Association for Public Opinion Research. Standard Definitions: Final Dispositions of Case Codes and Outcome Rates for Surveys.
24. Boscarino JA. Genetic epidemiology. In: Figley CR, editor. Encyclopedia of Trauma. Thousand Oaks, CA: Sage Publications; 2012:277–280.
25. Strachan T, Read A. Human Molecular Genetics:.
26. Bovin MJ, Marx BP, Weathers FW, et al. Psychometric properties of the PTSD checklist for diagnostic and statistical manual of mental disorders-fifth edition (PCL-5) in veterans. Psychol Assess. 2016;28(11):1379–1391. doi:10.1037/pas0000254
27. Blevins CA, Weathers FW, Davis MT, Witte TK, Domino JL. The posttraumatic stress disorder checklist for DSM-5 (PCL-5): development and initial psychometric evaluation. J Trauma Stress. 2015;28(6):489–498. doi:10.1002/jts.22059
28. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th Edition. Arlington, DC: American Psychiatric Association; 2013.
29. Wortmann JH, Jordan AH, Weathers FW, et al. Psychometric analysis of the PTSD Checklist-5 (PCL-5) among treatment-seeking military service members. Psychol Assess. 2016;28(11):1392–1403. doi:10.1037/pas0000260
30. Cox KS, Resnick HS, Kilpatrick DG. Prevalence and correlates of posttrauma distorted beliefs: evaluating DSM-5 PTSD expanded cognitive symptoms in a national sample. J Trauma Stress. 2014;27(3):299–306. doi:10.1002/jts.21925
31. Hoge CW, Riviere LA, Wilk JE, Herrell RK, Weathers FW. The prevalence of post-traumatic stress disorder (PTSD) in US combat soldiers: a head-to-head comparison of DSM-5 versus DSM-IV-TR symptom criteria with the PTSD checklist. Lancet Psychiatry. 2014;1(4):269–277. doi:10.1016/S2215-0366(14)70235-4
32. Gosling SD, Rentfrow PJ, Swann WB. A very brief measure of the big five personality domains. J Res Pers. 2003;37:504–528. doi:10.1016/S0092-6566(03)00046-1
33. Bunevicius A, Katkute A, Bunevicius R. Symptoms of anxiety and depression in medical students and in humanities students: relationship with big-five personality dimensions and vulnerability to stress. Int J Soc Psychiatry. 2008;54(6):494–501. doi:10.1177/0020764008090843
34. Felitti VJ, Anda RF, Nordenberg D, et al. Relationship of childhood abuse and household dysfunction to many of the leading causes of death in adults. The adverse childhood experiences (ACE) study. Am J Prev Med. 1998;14(4):245–258. doi:10.1016/S0749-3797(98)00017-8
35. Chapman DP, Whitfield CL, Felitti VJ, Dube SR, Edwards VJ, Anda RF. Adverse childhood experiences and the risk of depressive disorders in adulthood. J Affect Disord. 2004;82(2):217–225. doi:10.1016/j.jad.2003.12.013
36. Boscarino JA, Rukstalis M, Hoffman SN, et al. Risk factors for drug dependence among out-patients on opioid therapy in a large US health-care system. Addiction. 2010;105(10):1776–1782. doi:10.1111/j.1360-0443.2010.03052.x
37. Laufer RS, Gallops MS, Frey-Wouters E. War stress and trauma: the Vietnam veteran experience. J Health Soc Behav. 1984;25(1):65–85.
38. Frey-Wouters E, Laufer RS. Legacy of a War: The American Soldier in Vietnam. Armonk, NY: Sharpe Pub, Inc.; 1986.
39. Centers for Disease Control. Health status of Vietnam veterans. I. Psychosocial characteristics. The Centers for Disease Control Vietnam Experience Study. JAMA. 1988;259(18):2701–2707. doi:10.1001/jama.1988.03720180027028
40. Kulka RA, Schlenger WE, Fairbank JA, et al. The National Vietnam Veterans Readjustment Study: Tables of Findings and Technical Appendices:. New York, NY: Brunner/Mazel; 1990.
41. Boscarino JA, Forsberg CW, Goldberg J. A twin study of the association between PTSD symptoms and rheumatoid arthritis. Psychosom Med. 2010;72(5):481–486. doi:10.1097/PSY.0b013e3181d9a80c
42. McLeod DS, Koenen KC, Meyer JM, et al. Genetic and environmental influences on the relationship among combat exposure, posttraumatic stress disorder symptoms, and alcohol use. J Trauma Stress. 2001;14(2):259–275. doi:10.1023/A:1011157800050
43. Boscarino JA. Posttraumatic stress disorder, exposure to combat, and lower plasma cortisol among Vietnam veterans: findings and clinical implications. J Consult Clin Psychol. 1996;64(1):191–201. doi:10.1037/0022-006X.64.1.191
44. Adams RE, Urosevich TG, Hoffman SN, et al. Social support, help-seeking, and mental health outcomes among veterans in non-VA facilities: results from the veterans’ health study. Mil Behav Health. 2017;5(4):393–405. doi:10.1080/21635781.2017.1333067
45. Boscarino JA. Post-traumatic stress and associated disorders among Vietnam veterans: the significance of combat exposure and social support. J Trauma Stress. 1995;8(2):317–336. doi:10.1002/jts.2490080211
46. Freedy JR, Kilpatrick DG, Resnick HS. Natural disasters and mental health: theory, assessment, and intervention. J Soc Behav Pers. 1993;8:49–103.
47. Boscarino JA, Galea S, Adams RE, Ahern J, Resnick H, Vlahov D. Mental health service and medication use in New York city after the September 11, 2001, terrorist attack. Psychiatr Serv. 2004;55(3):274–283. doi:10.1176/appi.ps.55.3.274
48. Galea S, Ahern J, Resnick H, et al. Psychological sequelae of the September 11 terrorist attacks in New York city. N Engl J Med. 2002;346(13):982–987. doi:10.1056/NEJMsa013404
49. Boscarino JA, Hoffman SN, Adams RE, Figley CR, Solhkhah R. Mental health outcomes among vulnerable residents after Hurricane Sandy: implications for disaster research and planning. Am J Disaster Med. 2014;9(2):107–120. doi:10.5055/ajdm.2014.0147
50. Sherbourne CD, Stewart AL. The MOS social support survey. Soc Sci Med. 1991;32(6):705–714. doi:10.1016/0277-9536(91)90150-B
51. Boscarino JA, Adams RE, Figley CR. Mental health service use 1-year after the World Trade Center disaster: implications for mental health care. Gen Hosp Psychiatry. 2004;26(5):346–358. doi:10.1016/j.genhosppsych.2004.05.001
52. Kessler RC, Adler L, Ames M, et al. The World Health Organization adult ADHD self-report scale (ASRS): a short screening scale for use in the general population. Psychol Med. 2005;35(2):245–256. doi:10.1017/S0033291704002892
53. Schwab K, Baker G, Ivins B, Sluss-Tiller M, Lux W, Warden D. The brief traumatic brain injury screen (BTBIS): investigating the validity of a self-report instrument for detecting traumatic brain injury (TBI) in troops returning from deployment in Afghanistan and Iraq. Neurology. 2006;66(2):A235.
54. Urosevich TG, Boscarino JJ, Hoffman SN, et al. Visual dysfunction and associated co-morbidities as predictors of mild traumatic brain injury seen among veterans in non-VA facilities: implications for clinical practice. Mil Med. 2018;183(11–12):e564–e570. doi:10.1093/milmed/usy102
55. Taylor MD, Whiteman MC, Fowkes GR, Lee AJ, Allerhand M, Deary IJ. Five factor model personality traits and all-cause mortality in the Edinburgh Artery study cohort. Psychosom Med. 2009;71(6):631–641. doi:10.1097/PSY.0b013e3181a65298
56. Costa PT, McCrae RR. Revised NEO Personality Inventory (NEO PI-R) and NEO Five Factor Inventory (NEO-FFI): Professional Manual:.
57. First MB, Tasman A. DSM-IV-TR Mental Disorders: Diagnosis, Etiology, and Treatment. New York: Wiley; 2004.
58. First MD, Spitzer RL, Gibbon M, Williams JBW. Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I) – Clinician Version. Washinton, DC: American Psychiatric Association; 1997.
59. Spitzer RL, Williams JB, Gibbon M, First MB. The structured clinical interview for DSM-III-R (SCID). I: history, rationale, and description. Arch Gen Psychiatry. 1992;49(8):624–629. doi:10.1001/archpsyc.1992.01820080032005
60. Kilpatrick DG, Ruggiero KJ, Acierno R, Saunders BE, Resnick HS, Best CL. Violence and risk of PTSD, major depression, substance abuse/dependence, and comorbidity: results from the National Survey of Adolescents. J Consult Clin Psychol. 2003;71(4):692–700. doi:10.1037/0022-006X.71.4.692
61. Acierno R, Kilpatrick DG, Resnick H, Saunders B, De Arellano M, Best C. Assault, PTSD, family substance use, and depression as risk factors for cigarette use in youth: findings from the National Survey of Adolescents. J Trauma Stress. 2000;13(3):381–396. doi:10.1023/A:1007772905696
62. Boscarino JA, Kirchner HL, Hoffman SN, Erlich PM. Predicting PTSD using the New York risk score with genotype data: potential clinical and research opportunities. Neuropsychiatr Dis Treat. 2013;9:517–527. doi:10.2147/NDT.S42422
63. Kessler RC, Ustun TB. The World Mental Health (WMH) survey initiative version of the World Health Organization (WHO) Composite International Diagnostic Interview (CIDI). Int J Methods Psychiatr Res. 2004;13(2):93–121. doi:10.1002/mpr.168
64. Still CD, Wood GC, Chu X, et al. High allelic burden of four obesity SNPs is associated with poorer weight loss outcomes following gastric bypass surgery. Obesity (Silver Spring). 2011;19(8):1676–1683. doi:10.1038/oby.2011.3
65. Lambert SA, Abraham G, Inouye M. Towards clinical utility of polygenic risk scores. Hum Mol Genet. 2019;28:R133–R142. doi:10.1093/hmg/ddz187
66. Lowe SR, Meyers JL, Galea S, et al. RORA and posttraumatic stress trajectories: main effects and interactions with childhood physical abuse history. Brain Behav. 2015;5(4):e00323. doi:10.1002/brb3.2015.5.issue-4
67. Amstadter AB, Sumner JA, Acierno R, et al. Support for association of RORA variant and post traumatic stress symptoms in a population-based study of hurricane exposed adults. Mol Psychiatry. 2013;18(11):1148–1149. doi:10.1038/mp.2012.189
68. Gillespie CF, Phifer J, Bradley B, Ressler KJ. Risk and resilience: genetic and environmental influences on development of the stress response. Depress Anxiety. 2009;26(11):984–992. doi:10.1002/da.v26:11
69. Spitz MR, Amos CI, Dong Q, Lin J, Wu X. The CHRNA5-A3 region on chromosome 15q24-25.1 is a risk factor both for nicotine dependence and for lung cancer. J Natl Cancer Inst. 2008;100(21):1552–1556. doi:10.1093/jnci/djn363
70. Thorgeirsson TE, Geller F, Sulem P, et al. A variant associated with nicotine dependence, lung cancer and peripheral arterial disease. Nature. 2008;452(7187):638–642. doi:10.1038/nature06846
71. Erlich PM, Hoffman SN, Rukstalis M, et al. Nicotinic acetylcholine receptor genes on chromosome 15q25.1 are associated with nicotine and opioid dependence severity. Hum Genet. 2010;128(5):491–499. doi:10.1007/s00439-010-0876-6
72. von Wolff G, Avrabos C, Stepan J, et al. Voltage-sensitive dye imaging demonstrates an enhancing effect of corticotropin-releasing hormone on neuronal activity propagation through the hippocampal formation. J Psychiatr Res. 2011;45(2):256–261. doi:10.1016/j.jpsychires.2010.06.007
73. Hosmer DW, Lemeshow S. Applied Logistic Regression.
74. Hilbe JM. Negative Binomial Regression:. New York, NY: Cambridge University Press; 2007.
75. Lehavot K, Goldberg SB, Chen JA, et al. Do trauma type, stressful life events, and social support explain women veterans’ high prevalence of PTSD? Soc Psychiatry Psychiatr Epidemiol. 2018;53(9):943–953. doi:10.1007/s00127-018-1550-x
76. Boscarino JA. PTSD is a risk factor for cardiovascular disease: time for increased screening and clinical intervention. Prev Med. 2012;54(5):363–364;. doi:10.1016/j.ypmed.2012.01.001
77. Boscarino JA, Figley CR. Understanding the neurobiology of fear conditioning and emergence of posttraumatic stress disorder psychobiology: commentary on Blanchard et al. J Nerv Ment Dis. 2012;200(9):740–744. doi:10.1097/NMD.0b013e318266b5ea
78. Skogstad M, Skorstad M, Lie A, Conradi HS, Heir T, Weisaeth L. Work-related post-traumatic stress disorder. Occup Med . 2013;63(3):175–182. doi:10.1093/occmed/kqt003
79. Vlahov D, Galea S, Ahern J, et al. Consumption of cigarettes, alcohol, and marijuana among New York City residents six months after the September 11 terrorist attacks. Am J Drug Alcohol Abuse. 2004;30(2):385–407. doi:10.1081/ADA-120037384
80. Boscarino JA. Post-traumatic stress disorder and cardiovascular disease link: time to identify specific pathways and interventions. Am J Cardiol. 2011;108(7):1052–1053. doi:10.1016/j.amjcard.2011.07.003
81. Boscarino JA. Posttraumatic stress disorder and physical illness: results from clinical and epidemiologic studies. Ann N Y Acad Sci. 2004;1032:141–153. doi:10.1196/annals.1314.011
82. Boscarino JA, Chang J. Electrocardiogram abnormalities among men with stress-related psychiatric disorders: implications for coronary heart disease and clinical research. Ann Behav Med. 1999;21(3):227–234. doi:10.1007/BF02884839
83. Boscarino JA, Chang J. Higher abnormal leukocyte and lymphocyte counts 20 years after exposure to severe stress: research and clinical implications. Psychosom Med. 1999;61(3):378–386. doi:10.1097/00006842-199905000-00019
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

