Back to Journals » HIV/AIDS - Research and Palliative Care » Volume 13
Predictors of Anemia Among HIV-Infected Children on Antiretroviral Therapy in Wolaita Zone, South Ethiopia: A Facility-Based Cross-Sectional Study
Authors Geleta ML, Solomon FB , Tufa EG, Sadamo FE, Dake SK
Received 19 September 2020
Accepted for publication 12 December 2020
Published 8 January 2021 Volume 2021:13 Pages 13—19
DOI https://doi.org/10.2147/HIV.S282845
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Bassel Sawaya
Mesay Lema Geleta,1 Fithamlak Bisetegn Solomon,2 Efrata Girma Tufa,3 Fekadu Elias Sadamo,3 Samson Kastro Dake3
1College of Health Sciences and Medicine, Wolaita Sodo University, Wolaita Sodo, Ethiopia; 2Department of Medical Laboratory, College of Health Science and Medicine, Wolaita Sodo University, Wolaita Sodo, Ethiopia; 3Department of Reproductive Health and Nutrition, School of Public Health, College of Health Science and Medicine, Wolaita Sodo University, Wolaita Sodo, Ethiopia
Correspondence: Samson Kastro Dake
Department of Reproductive Health and Nutrition, School of Public Health, College of Health Science and Medicine, Wolaita Sodo University, Wolaita Sodo, Ethiopia
Tel +251911077516
Email [email protected]
Purpose: Anemia is a global public health problem, and the majority of human immunodeficiency virus (HIV)-positive people become anemic at some point in the course of the disease. We lack adequate evidence on the magnitude of anemia among children on highly active antiretroviral therapy in Ethiopia and particularly in South Ethiopia. Thus, this study aimed at determining the proportion and associated factors of anemia among children on highly active antiretroviral therapy in Wolaita zone, South Ethiopia.
Patients and Methods: A facility-based cross-sectional study was conducted from November to December 2018 on 256 children from 6 months to 14 years of age who were on antiretroviral therapy. Data were collected through an interview with caregivers and review of medical records. CD4+ cell count was analyzed using FACS Calibur, and hemoglobin level was measured with a Hem cue 301 analyzer. Stool samples were examined for the presence of intestinal parasites by direct wet mount technique. Data analyzed with Stata version 14.0 were conveyed in mean and standard deviation of the mean, median and inter-quartile range. Multivariate analysis was carried out to identify independent predictors of the outcome variable. Adjusted odds ratio with 95% confidence interval was reported.
Results: The proportion of anemia was found to be 38.8%. Co-trimoxazole prophylaxis (AOR=0.45; 95% CI: 0.21, 0.95), caregivers not receiving nutritional counseling (AOR=0.90; 95% CI: 0.01, 0.98) and presence of intestinal parasites (AOR=3.10; 95% CI: 1.39, 6.88) were associated with anemia.
Conclusion: The proportion of anemia found in this study is a moderate public health problem. Health education programs in antiretroviral therapy clinics should be targeted at appropriate dietary practice, and appropriate hand washing and other hygienic practices to prevent intestinal parasitic infections. Co-trimoxazole prophylaxis should be given to all eligible children based on the recommendation.
Keywords: anemia, anti-retroviral therapy, HIV, children, Wolaita, Ethiopia
Introduction
Anemia is a condition in which the number of red blood cells is insufficient to meet the body’s physiologic needs, with consequent diminishing of the blood’s oxygen-carrying capacity. Anemia can be caused by iron deficiency, other nutritional deficiencies, acute and chronic inflammation, parasitic infections, inherited or acquired disorders that affect hemoglobin synthesis, and low red blood cell production or survival.1
Children with HIV are exposed to macro- and micro-nutrient related deficiencies; anemia being the commonest.2,3 Anemia is a critical health concern affecting growth and energy levels adversely, and it highly damages immune mechanisms.4,5
Anemia affects both developing and developed countries and thus it is a global public health problem.6 Globally, roughly 1.3–2.2 billion people suffer from anemia and in developing countries 50% of women and children are anemic.7 An estimated 63–95% of people living with HIV become anemic at some point in the course of the disease.8 In India the prevalence of anemia among HIV-infected children was reported to be 47.1%.9 A prevalence level of 69.1% was reported among HIV-infected children in Democratic Republic of Congo.10 In Ethiopia the prevalence of anemia among HIV-infected children ranges between 16.2% and 39.1%.11–13 A combination of environmental and host factors predispose HIV-infected children to anemia.
We have very limited evidence on the prevalence of anemia among children on antiretroviral therapy (ART) in Ethiopia and there is a wide variation in the reported prevalence. Thus the main aim of this study was to estimate the proportion of anemia among HIV-infected children on ART and to identify factors associated with the outcome in ART clinics of Wolaita zone, South Ethiopia.
Patients and Methods
Study Setting
A facility-based cross-sectional study was conducted from November to December 2018 at ART clinics of Wolaita zone. There are 16 health facilities that provide ART services. Wolaita zone has a latitude and longitude of 6°54ʹN 37°45ʹE with an average elevation of 1850 meters above sea level. All health facilities that provide ART services were included in the study.
Population and Sampling
Children, 6 months to 14 years of age, on ART paired with their mothers/caretakers who lived in the study area for at least half a year were included in this study. Since there are 262 children on ART attending ART clinics in Wolaita zone, a sample size was not calculated and all eligible children were included. Children who were on anemia treatment and received blood transfusion were excluded from the study.
Data Collection
We adopted a structured questionnaire from relevant articles and related literatures.14 Medical records were reviewed and presence of opportunistic infections, baseline CD4+ count and World Health Organization (WHO) clinical stage were recorded to complement the primary data. Twenty-four hours recall was used to assess dietary diversity. Nurses and laboratory technicians were recruited for interview and laboratory analysis respectively. BSc nurses were recruited for supervision. Two days training was given for data collectors on the method of interviewing. Trained laboratory technicians were employed and additional training was given. The questionnaire was pre-tested on 5% of the sample size at Shone Primary Hospital which is not part of the actual data collection. Close supervision was undertaken during data collection to avoid bias. Standardized operating procedures were strictly followed during blood sample collection, storage and analytical processes.
Laboratory Analysis
Three milliliters of venous blood sample was collected using EDTA anti-coagulated test tube for CD4+ cell count and hemoglobin analyses. CD4+ cell count was analyzed using BD FACS Calibur 2008, a standard automated analyzer which determines both CD4 and CD8 absolute count and percentage by using whole blood samples collected in liquid EDTA. The whole blood is added to the tubes of sample reagent pairs and the fluorochrome labeled antibodies bind specifically to antigens on the surface of lymphocytes. Hemoglobin level was measured with a HemoCue Hb301 analyzer (HemoCue AB, Ängelholm, Sweden. Stool samples were taken and examined by direct wet mount technique to detect present of intestinal parasites microscopically.
Measurements
Anemia is established if the hemoglobin level is below 11 g/dl for age 6–59 months, <11.5 g/dl for 5–11 years and <12 g/dl for 12–14 years and adjusted for altitude.1
Dietary diversity was determined by using three non-consecutive days recall of consumption of common food groups within the past 24 h. Foods were categorized into 12 groups based on FAO recommendations.15 The number of food and food items consumed in each group were summed and classified as <4 and ≥4.16
Dietary support: Additional food aid given by governmental and non-governmental organizations.
Dietary counseling: Dietary advice to use the available food diversity with the aim of assisting the client to manage or modify food choices and eating behavior.
Meal: Any kind of food consumption within 24 hours of a day.
Statistical Analysis
Data were entered into Epi data version 3.1 and exported to Stata version 14.0 for analyses. The data were conveyed in mean and standard deviation of the mean, median and inter-quartile range. Normality was checked for all continuous variables. Hosmer-Lemeshow goodness-of-fit test and Receiver Operating Characteristics (ROC) test were carried out to have confidence in the regression model. Collinearity was diagnosed, and there was no multicollinearity among the exposure variables included in the model. We used logistic regression analysis to identify exposure variables with association to the outcome variable. Exposures and covariates with p<0.25 during the bivariate analysis were included in multivariate analysis to increase the power of the model. Multivariate analysis was carried out to control for potential confounders and identify independent predictors of the outcome variable. Adjusted Odds Ratio (AOR) with 95% confidence interval (CI) was reported. The level of significance was set at p<0.05.
Ethics Approval and Consent to Participate
Ethical clearance for the study was obtained from the Ethical review committee at College of Health Science and Medicine in Wolaita Sodo University in Ethiopia. The ethical clearance letter was written to Wolaita zone health department which is the highest government body in the area for health and health research. Permission was granted from Wolaita zone health department and its lower health administrative structures to conduct the study. Finally, informed written consent for participation was obtained from parents/caregivers since we cannot obtain consent from children under 18 years of age. The protocols and ethical principles are all in line with Helsinki declarations.
Results
Socio-Demographic Characteristics
Overall, 256 children on ART participated in the study with a response rate of 97.7%. More than half, 140 (54.7%) were girls. The median age was 12 (10–14) years, and 144 (56.2%) of the children were between 12–14 years of age. About three in four, 192 (75.0%) were rural dwellers. One-third, 85 (33.2%) of the caregivers did not attend formal education and 74 (28.9%) were employed. About two-thirds, 171 (66.8%) of the households use either pipe or public tap water for drinking (Table 1).
 |
Table 1 Socio-Demographic Characteristics of Children and Caregivers Involved in This Study at Wolaita Zone in South Ethiopia, December 2018 |
Clinical and Immunological Characteristics
One hundred and seventy-one children (66.8%) were on ART for more than 60 months. A little higher than half, 132 (51.6%) were in WHO stage 3 at baseline and nearly all, 251 (98.0%) were in WHO stage T1. Forty-two (16.4%) switched their drug regimen and 44 (17.2%) had some form of opportunistic infection. Seventy-three (28.5%) were taking co-trimoxazole prophylaxis. The median CD4+ count was 746.5 (516–1007) cells/mm3, and the majority of them, 182 (85.8%) had a CD4+ count > 350 cells/mm3 (Table 2).
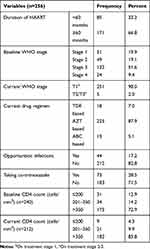 |
Table 2 Clinical and Immunological Characteristics of Children Involved in This Study at Wolaita Zone in South Ethiopia, December 2018 |
Nutritional and Parasitological Characteristics
The majority of caregivers, 210 (82%) affirmed getting dietary advice. Nearly two-thirds, 167 (65.2%) of the children often eat fewer than three meals a day and 115 (44.9%) had dietary diversity of less than 4. Most, 210 (82.0%) of the children had no intestinal parasitic infection (Table 3).
 |
Table 3 Dietary, Hygienic and Parasitological Characteristics of Children and Caregivers Involved in This Study at Wolaita Zone in South Ethiopia, December 2018 |
Proportion and Factors Associated with Anemia
The mean ± standard deviation hemoglobin level of the study participants was 12.2 g/dl ± 1.4. The proportion of anemia was 38.8%. Children taking co-trimoxazole prophylaxis had 55% lower likelihood of being anemic than those who were not taking co-trimoxazole (AOR=0.45; 95% CI: 0.21, 0.95). Children whose caregivers were given nutritional counseling had 91% less likelihood of having anemia (AOR=0.90; 95% CI: 0.01, 0.98). The odds of being anemic was 3 times higher for children who had an intestinal parasitic infection than their counterparts (AOR=3.10; 95% CI: 1.39, 6.88) (Table 4).
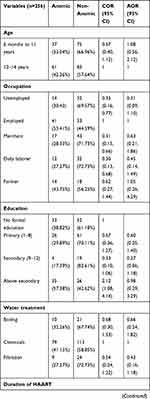 | 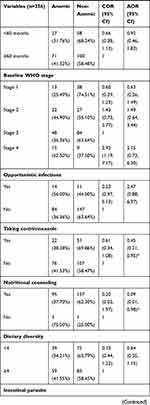 | 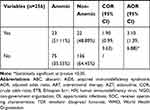 |
Table 4 Factors Associated with Anemia Among Children Involved in This Study at Wolaita Zone in South Ethiopia, December 2018 |
Discussion
In this study, the proportion of anemia among children on ART was found to be 38.8%. The prevalence of anemia in this study is a moderate public health problem according to the WHO classification of anemia.1 This finding is comparable with a similar study conducted in different parts of Ethiopia (35%) and 39.1%.15,17 However, it is lower compared with a study conducted in southwest Ethiopia (53.1%).18 Lower prevalence was reported in different parts of Ethiopia (16.2%), (10%) and (22.2%).11,12,19 This divergence might be due to variation in the culture, dietary practice and socio-economic status across different segments of the population within the country.
Children who were taking co-trimoxazole prophylaxis had a lesser odds of being anemic compared with those who were not. This is in agreement with studies from Ethiopia and Gambia.11,20 One of the most important mechanisms of anemia in HIV infection is failure of erythropoiesis. Co-trimoxazole may reduce the levels of cytokines that impair erythropoiesis, by reducing immune activation as well as by preventing infections.21
Children whose caregivers did not receive nutritional counseling were more likely to be anemic compared with their counterparts. This finding is in agreement with a study conducted in Hawassa referral hospital where children whose caregivers did not receive nutritional counseling had higher odds of being anemic.22 Better information on the key dietary recommendations might have improved the caregiver’s practice of child feeding.
Consistent with studies from Ethiopia and Tanzania, children with intestinal parasitic infections had much higher odds of having anemia compared with those children who were not infected with intestinal parasites.12,23 This might possibly be due to the attachment of the worm in the intestine to the intestinal mucosa causing intestinal necrosis and blood loss. In addition, intestinal parasitosis may contribute to activate the immune system where immune cells respond aggressively and this leads to anemia. Chronic infections may also lead to anemia which results from excessive iron loss.4
Unlike many other studies, this study did not find an association between lower CD4+ cell count and anemia. This finding disagrees with a similar study from Addis Ababa (Central Ethiopia).12 But our finding corroborated with the finding in South Africa where being anemic is independent of CD4+ count. In the current study, this difference might be due to the fewer children with a low CD4+ count involved in this study.
Study Limitations
Regarding dietary habit, a certain level of recall bias was expected; data collectors were aware of the cultural issues and collected the data in a manner to reduce recall bias. Data on the duration of HAART, baseline CD4+ count, WHO stage, drug regimen, taking co-trimoxazole and presence of opportunistic infections were taken from the patients’ charts. Since the health facilities record each patient’s information consistently and accurately, we believe that the data are unbiased. We did not differentiate the types of anemia the children had.
Conclusion
The magnitude of anemia among children on ART in the study area is a moderate public health problem. Anemia was significantly associated with caregivers not receiving nutritional counseling and intestinal parasitic infections. Taking co-trimoxazole prophylaxis was protective against anemia. Health education programs in ART clinics should be given with due focus on appropriate dietary practice, appropriate hand washing practice and other hygienic practices to prevent intestinal parasitic infections. Co-trimoxazole prophylaxis should be given to all eligible children based on the recommendation.
Acknowledgments
We would like to thank the School of Public Health at Wolaita Sodo University for facilitating the research programme. We acknowledge Wolaita zone health department and its lower administrative structures for official permission. We are grateful to the caregivers and children for their consent to participate in the study. We also thank our responsible data collectors.
Disclosure
The authors declare that they have no conflicts of interest in this work.
References
1. WHO Haemoglobin concentrations for the diagnosis of anaemia and assessment of severity. Vitamin and Mineral Nutrition Information System. Geneva: World Health Organization; 2011 (WHO/NMH/NHD/MNM/11.1). Available from: http://www.who.int/vmnis/indicators/haemoglobin.pdf.
2. Sireesha G, Rajani N. Nutritional status of girls through anthropometric and dietary assessment. IOSR J Dent Med Sci. 2015;14(11):73–78.
3. Prendergast AJ, Humphrey JH. The stunting syndrome in developing countries. Paediatr Int Child Health. 2014;34(4):250–265. doi:10.1179/2046905514Y.0000000158
4. WHO. Iron Deficiency Anemia. Assessment, Prevention, and Control. A Guide for Programme Managers. Geneva: World Health Organization; 2001:47–62.
5. WHO. The World Health Report 2002: Reducing Risks, Promoting Healthy Life. World Health Organization: Geneva; 2002.
6. Haas JD, Brownlie T
7. Stevens GA, Finucane MM, De-Regil LM, et al. Global, regional, and national trends in haemoglobin concentration and prevalence of total and severe anaemia in children and pregnant and non-pregnant women for 1995–2011: a systematic analysis of population-representative data. Lancet Glob Health. 2013;1(1):e16–e25. doi:10.1016/S2214-109X(13)70001-9
8. Mehta S, Jutur S, Gautam D. Hematologic manifestations of HIV/AIDS. Med Update. 2011;9:484–490.
9. Shet A, Bhavani PK, Kumarasamy N, et al. Anemia, diet and therapeutic iron among children living with HIV: a prospective cohort study. BMC Pediatr. 2015;15:164. doi:10.1186/s12887-015-0484-7
10. Mwadianvita CK, Ilunga EK, Djouma J, et al. Study of anemia in HIV positive children naïve to antiretroviral treatment in Lubumbashi, Democratic Republic of Congo. Pan Afr Med J. 2014;17(46). doi:10.11604/pamj.2014.17.46.2046
11. Enawgaw B, Alem M, Melku M, et al. Prevalence and associated risk factors of anemia among HIV infected children attending Gondar university hospital, Northwest Ethiopia: a cross sectional study. BMC Hematol. 2015;15(1):12. doi:10.1186/s12878-015-0032-6
12. Mihiretie H, Taye B, Tsegaye A. Magnitude of anemia and associated factors among pediatric HIV/AIDS patients attending Zewditu Memorial Hospital ART Clinic, Addis Ababa, Ethiopia. Anemia. 2015;2015:1–6. doi:10.1155/2015/479329
13. Teklemariam Z, Mitiku H, Mesfin F. Prevalence of anemia and nutritional status among HIV-positive children receiving antiretroviral therapy in Harar, eastern Ethiopa. HIV/AIDS (Auckland, Nz). 2015;7:191.
14. CSA, ICF. Ethiopia demographic and health survey 2016 key indicators report: final report. Addis Ababa, Rockville: Central Statistical Authority; 2017
15. Ferede G, Wondimeneh Y. Prevalence and related factors of anemia in HAART-naive HIV positive patients at Gondar University Hospital, Northwest Ethiopia. BMC Blood Disord. 2013;13(1):8. doi:10.1186/2052-1839-13-8
16. FAO. Guidelines for Measuring Household and Individual Dietary Diversity. Rome, Italy: FAO; 2013.
17. Zelalem T, Firehiwot HM. Prevalence of anemia and nutritional status among HIV-positive children receiving antiretroviral therapy in Harar, eastern Ethiopa. HIV AIDS. 2015;7:191–196.
18. Abebe M, Alemseged F. Hematologic abormalities among children on Haart, in Jimma university specialized hospital, southwestern ethiopia. Ethiop J Health Sci. 2009;19(2).
19. Mengist HM, Taye B, Tsegaye A. Intestinal parasitosis in relation to CD4+T cells levels and anemia among HAART initiated and HAART naive pediatric HIV patients in a model ART center in Addis Ababa, Ethiopia. PLoS One. 2015;10(1371):1–14.
20. Prendergast A, Walker AS, Mulenga V, et al. Improved growth and anemia in HIV-infeted children taking cotrimoxazole prophylaxis. Clin Infec Dis. 2011;52(7):953–957. doi:10.1093/cid/cir029
21. Kreuzer KA, Rockstroh JK, Jelkmann W, et al. Inadequate erythropoietin response to anemia in HIV patients: relationship to serum levels of tumor necrosis factor-alpha, interleukin-6 and their soluble receptors. Br J Haematol. 1997;96:235–239. doi:10.1046/j.1365-2141.1997.d01-2031.x
22. Wondimu WB. Nutritional status and associated factors in human immunodeficiency virus infected children in Hawassa University Referral Hospital Hawassa City, SNNPR, Ethiopia.
23. Makubi AN, Mugusi F, Magesa PM, et al. Risk factors for anaemia among HIV infected children attending care and treatment clinic at Muhimbili National Hospital in Dar es Salaam, Tanzania. Tanzan J Health Res. 2012;14(1). doi:10.4314/thrb.v14i1.11.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
