Back to Journals » Cancer Management and Research » Volume 12
Plasma Long Non-Coding RNA RP11-438N5.3 as a Novel Biomarker for Non-Small Cell Lung Cancer
Authors Chen Q, Zhu C, Jin Y, Si X, Jiao W, He W, Mao W, Li M, Luo G
Received 1 November 2019
Accepted for publication 10 January 2020
Published 28 February 2020 Volume 2020:12 Pages 1513—1521
DOI https://doi.org/10.2147/CMAR.S237024
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Antonella D'Anneo
Video abstract presented by Qingjuan Chen.
Views: 123
Qingjuan Chen, 1,* Chenjing Zhu, 2,* Yingying Jin, 3 Xiaomin Si, 4 Wan Jiao, 4 Wenjing He, 4 Wei Mao, 4 Ming Li, 1 Guomin Luo 1
1Department of Oncology, Yongchuan Hospital of Chongqing Medical University, Chongqing 40016, People’s Republic of China; 2Department of Radiation Oncology, Jiangsu Cancer Hospital and Jiangsu Institute of Cancer Research and The Affiliated Cancer Hospital of Nanjing Medical University, Nanjing, Jiangsu 210009, People’s Republic of China; 3Department of Oncology, The Second Affiliated Hospital of Xi’an Jiaotong University, Xi’an, Shaanxi Province 710004, People’s Republic of China; 4Department of Oncology, Xianyang Center Hospital, Xi’an, Shaanxi Province 712000, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Qingjuan Chen
Department of Oncology, Yongchuan Hospital of Chongqing Medical University, Chongqing 40016, People’s Republic of China
Email [email protected]
Background: Lung cancer is one of the most common malignancies around the world. The lack of early diagnosis and effective treatment strategies contributes to the poor prognosis of patients with lung cancer. Recent studies have implied the role of long non-coding RNAs (lncRNAs) in oncogenesis. The purpose of our study was to identify specific lncRNAs which were correlated with non-small cell lung cancer (NSCLC) and their potential functions.
Materials and Methods: The global plasma lncRNA profiling was performed using LncPathTM Human Cancer Array, and 11 lncRNAs were then selected for quantitative reverse transcription PCR (qRT-PCR) validation in 138 plasma samples from 69 NSCLC patients and 69 healthy controls (HCs). A noteworthy lncRNA, RP11-438N5.3, the function of which was previously unknown, was further explored on the aspect of the correlation of its expression level with clinicopathological factors.
Results: The results revealed that plasma level of RP11-438N5.3 was significantly lower in NSCLCs than that in HCs (p < 0.01). Receiver operating characteristic (ROC) analyses showed that the area under the ROC curve (AUC) for plasma RP11-438N5.3 was 0.814 (95% CI, 0.743– 0.885; p< 0.01). High expression of RP11-438N5.3 in plasma correlated with favorable prognosis for NSCLC patients (Hazard ratio = 2.827; 95% CI: 1.036 to 7.718; p = 0.024; Cox regression analysis). Moreover, we found that the plasma level of stromal interaction molecule 1 (STIM1) mRNA was remarkably higher in NSCLC compared with HC (p< 0.01), and the AUC for STIM1 was 0.753 (95% CI, 0.673– 0.833; p< 0.01), RP11-438N5.3 and STIM1 were inversely correlated with each other.
Conclusion: Our results indicated that RP11-438N5.3 and STIM1 might provide a new strategy for NSCLC diagnosis. Furthermore, increased circulating RP11-438N5.3 level holds great potential in indicating a beneficial prognosis in NSCLC patients.
Keywords: long non-coding RNA, biomarker, non-small cell lung cancer, RP11-438N5.3, stromal interaction molecule 1
A Letter to the Editor has been published for this article.
Introduction
Lung cancer, which consists of small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC), is the most common malignancy around the world and the main cause of cancer-related deaths in China.1 NSCLC comprises more than 85% of lung cancer cases.2 Although therapeutic advances have been achieved in surgery, chemotherapy or radiation for patients with lung cancer over the past decades, the overall 5-year-survival rate is still less than 15%.3 The main reason was that the majority of NSCLC patients were in middle or advanced stages at the time of diagnosis, thus missing the opportunities for radical surgery. Therefore, finding reliable markers for the early diagnosis and effective treatment in NSCLC patients was of utmost importance.
Circulating biomarkers have shown their potential role in early lung cancer detection,4 including, among the others, circulating tumor DNA,5 microRNAs6 and metabolomics.7 At present, accumulating evidence has implicated that long non-coding RNAs (lncRNAs) are abnormally expressed in many cancers, and are related to carcinogenesis, tumor development, proliferation and invasion, and could be used as important indicators for the early diagnosis, treatment and prognosis of patients.8
A vast majority of non-coding RNAs exist in human genomes, and they once were considered as “transcriptional noise” since they were not able to encode proteins. Non-coding RNAs include microRNAs, siRNAs, long non-coding RNAs (lncRNAs) and so on. Owing to the complexity and diversity of the regulatory mechanisms, lncRNAs’ function has become a hot topic. Previous studies have shown that some lncRNAs could regulate cell differentiation, growth, metabolism and many other biological processes at various levels. Moreover, lncRNAs might also take part in the regulation of oncogenesis, such as H19, HOTAIR and MALAT19 which were abnormally expressed in many cancers. However, studies focusing on the relationship between lncRNAs and NSCLC are lacking. To identify specific lncRNAs in NSCLC, we investigated the expression patterns of lncRNAs in plasma samples of NSCLC patients by lncRNA microarray screening and subsequent qRT-PCR validation, and analyzed the correlation of selected IncRNAs with clinical-pathological factors of NSCLC, hoping to discover a new diagnostic and treatment approach for NSCLC.
Materials and Methods
Patient Samples
This study was carried out from September 2014 to August 2015 at the inpatient service of the Department of Medical Oncology, Xian Yang Center Hospital. A total of 69 patients with newly diagnosed NSCLC who had not received any anti-tumor treatments, except for those patients with secondary metastatic lung cancer, were enrolled. All the selected patients were pathologically confirmed through medical examinations (including fiber bronchoscope, percutaneous needle aspiration biopsy, lymph node biopsy, or surgical resection). During the same period, an additional 69 age- and gender-matched healthy control participants were recruited from the Physical Examination Center from the same hospital. Individuals were excluded if he/she had a history of malignancies in other organs, chronic inflammatory diseases, or recent infections. Written informed consent was obtained from each individual participant and the experimental protocol was approved by the Clinical Research Ethics Committee of Xian Yang Center Hospital. This study was conducted in accordance with the Declaration of Helsinki.
Sample Preparation, RNA Isolation
Venous blood (3~5mL) was collected from each individual participant before treatments and one potion of blood sample was used for preparing individual plasma samples by centrifugation within two hours post the venipuncture. To be brief, the blood samples were centrifuged at 1900×g for 10 mins, followed by centrifugation at 13,000×g for 10 mins to precipitate cell debris and genomic DNAs. The supernatant was taken, aliquoted and frozen at −80°C until the time of assay.
Total RNA was extracted from the plasma samples using Trizol LS Reagent per manufacturer’s instructions (Invitrogen, CA, USA). Briefly, individual plasma samples (250 μL) were mixed with 750 μL of Trizol LS. After centrifugation and washing, the RNA pellets were resuspended in 25 μL of nuclease-free water and stored at −80°C. Total RNA from each sample was quantified with the NanoDrop ND-8000.
lncRNA Profiling
Using paired plasma samples from 3 NSCLCs and HCs, lncRNA profiling was performed with LncPathTM Human Cancer Array. Using Arraystar Flash RNA Labeling protocol (Arraystar), total RNA from each sample was amplified and subsequently transcribed into fluorescent cRNA. The labeled cRNAs were then hybridized onto the LncPathTM Human Cancer Array (6x7K, Arraystar). After washing slides, the arrays were scanned by the Axon GenePix 4000B microarray scanner. We used R software package to perform Quantile normalization and data processing. Volcano Plot filtering was used to identify differentially expressed LncRNAs between two groups. The distinguishable LncRNAs expression pattern among those samples was shown using Hierarchical Clustering.
qRT-PCR Validation of lncRNAs in Plasma
The relative levels of selected lncRNA transcripts were quantified by qRT-PCR using specific primers. RNA was firstly removed from genomic DNA and reverse transcribed to cDNA using a PrimeScript® RT reagent Kit with gDNA Eraser (Takara Code: DRR047, Japan). Then, with the diluted cDNA as a template and β-actin as the internal reference, qRT-PCR was carried out using SYBR® Premix Ex TaqⅡ (Perfect Real Time; TaKaRa Code: DRR081, Japan) on ABI7500. DNA Amplification was performed under the following conditions: 95°C for 30s, followed by 40 cycles of 95°C for 5s, 60°C for 30s and 72°C for 30s. Melting curve analysis: 94°C for 60s; 37°C for 60s; 72°C for 120s. Experiments were performed in triplicates for the same reaction. The relative levels of lncRNA transcripts to the control β-actin were calculated by 2− ΔΔCt.
Statistical Analysis
Data were expressed as means ± standard deviations (SDs). The difference between groups was analyzed by the Mann–Whitney of Kruskal–Wallis tests, ANOVA, Student’s t-test or X2 test where applicable. The sensitivity and specificity of the relative levels of plasma lncRNAs for the diagnosis of NSCLC were analyzed by the receiver operating characteristic (ROC) curves. All statistical tests were performed using SPSS 16.0 software (SPSS Ltd., IBM, Armonk, NY) and GraphPad Prism 5.0 (GraphPad Software, LaJolla, CA). A two-sided P value of <0.05 was considered statistically significant.
Results
Demographic and Clinical Characteristics of NSCLC Patients
A total of 138 participants, including 69 NSCLC patients and 69 age- and gender-matched HCs were recruited. The demographic and clinical characteristics of the 69 NSCLC patients are summarized in Table 1. There were no significant differences in the distribution of age or gender between the two groups.
 |
Table 1 Basic Characteristics of Non-Small Cell Lung Cancer (NSCLC) Patients and Healthy Controls (HCs) |
Microarray Hybridization Data
A case-control study was designed to identify candidate plasma lncRNAs that were differentially expressed between NSCLC cases and normal controls. Plasma extracted from 3 NSCLC patients and 3 matched controls were subjected for LncPathTM Human Cancer Array which was used for the global profiling of human lncRNAs and protein-coding transcripts. Plasms lncRNA and mRNA expression was compared between the NSCLC patients and the healthy controls and presented in Figure 1A and B. In all, 1728 lncRNAs and 1906 coding transcripts were detected. Scatterplot showed that the distribution and expression pattern of lncRNAs and mRNAs between NSCLC and healthy control groups were similar but with some lncRNAs and mRNAs differentially expressed (Figure 1C and D).
Analysis of Differentially Expressed lncRNAs Associated with NSCLC
We next selected 11 lncRNAs, including 6 elevated lncRNAs (RP11-440I14.2, RP11-164H13.1, TUG1, TIMM8AP1, CD27-AS1 and RP11-131L23.1) and 5 decreased lncRNAs (BC029835, RP11-149I2.4, CTD-2033A16.1, TCONS_00014664 and RP11-438N5.3), for subsequent qRT-PCR validation with 138 plasma samples from 69 pairs of NSCLC and HC samples (Table 2).We found that among the 11 differentially expressed lncRNAs, the plasma level of RP11-438N5.3 in the NSCLC patients was significantly lower than that in the HCs (p<0.01) but the level of stromal interaction molecule 1 (STIM1) was significantly higher (p<0.01) (Figure 2A and B). Correlation analysis indicated that they were inversely correlated with each other (r = −0.558, p=0.026) (Figure 2C).
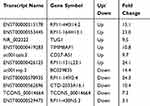 |
Table 2 Differentially Expressed Long Non-Coding RNAs in Plasma Samples |
The Relationship Between Plasma RP11-438N5.3 Expression Level and Clinicopathological Factors in NSCLC Patients
We evaluated correlation between the expression level of RP11-438N5.3 and clinicopathological factors including age (<60 or ≥60), gender (male or female), smoking status (absence or presence), EGFR status (wild type or mutation type), tumor location (left or right lung), TNM staging (I-II or III-IV), histological type (adenocarcinoma or squamous carcinoma), lymph node metastasis (absence or presence) and distant metastasis (absence or presence). We found that high levels of plasma RP11-438N5.3 was positively related to TNM (tumour, node and metastasis) staging (p=0.002) and distant metastasis (p=0.023) of NSCLC. However, no statistically significant difference was found in the distribution of age, gender, smoking status, EGFR status, tumor location, histological type and lymph node metastasis between RP11-438N5.3-high expression and RP11-438N5.3-low expression groups (p>0.05) (Table 3).
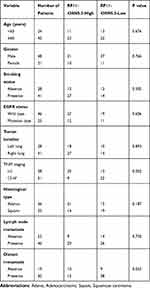 |
Table 3 Correlation of RP11-438N5.3 Expression Level with Clinicopathological Factors in NSCLC Patients |
The Diagnostic and Prognostic Values of RP11-438N5.3 and STIM1 in NSCLC
Receiver operating characteristic (ROC) analyses showed that the area under the ROC curve (AUC) for detecting NSCLC from HC for plasma RP11-438N5.3 was 0.814 (95% CI, 0.743–0.885; p<0.01), and that for STIM1 was 0.753 (95% CI: 0.673–0.833; P<0.01) (Figure 3A and B), indicating the diagnostic values of RP11-438N5.3 and STIM1 in NSCLC. In addition, using the Kaplan–Meier and log-rank methods, we found that high plasma level of RP11-438N5.3 was correlated with longer overall survival (OS) in patients with NSCLC (hazard ratio = 2.827; 95% confidence interval: 1.036 to 7.718; p=0.024), while the plasms level of STIM1 was not associated with OS in patients with NSCLC (hazard ratio = 0.642; 95% confidence interval: 0.886 to 1.750; p=0.726) (Figure 4A and B).
Pathway Analysis and KEGG Analysis of the Differentially Expressed mRNAs
Pathway analysis was used to identify significant pathways according to the Kyoto Encyclopedia of Genes and Genomes, Biocarta and Reatome databases. A total of 19 pathways were identified which showed significant differences due to differential gene expression. The upregulated genes were involved in 9 pathways while the downregulated genes were involved in 10 pathways. Predominant pathways are shown in Figure 5A and B. The top 3 upregulated pathways were acute myeloid leukemia-homo sapiens pathway, endometrial cancer and chronic myeloid leukemia pathway. The top 3 downregulated pathways were cell cycle, pathogenic Escherichia coli infection and shigellosis pathways.
 |
Figure 5 Pathway analysis and KEGG analysis of plasma RP11-438N5.3 in NSCLC patients. The upregulated genes were involved in 9 pathways (A) while downregulated genes (B) were involved in 10 pathways. |
Discussion
The discovery of long non-coding RNAs is a crucial frontier in the post-genome era. LncRNAs, although not exhibiting protein coding potential, are involved in a wide range of vital biological activities including embryonic development, stress response, alternative splicing and chromatin remodeling.10 They also have an important impact on many cellular processes including cell proliferation, cell cycle regulation, survival and metabolism.11 LncRNA dysregulation has been reported to play important roles in tumorigenesis, tumor progression and metastasis.12
With the rapid development of genomic technologies including high throughput sequencing and gene microarray, substantial efforts have been made to identify cancer-related lncRNAs. However, research on lung cancer-related lncRNAs is still in its infancy. In our study, we utilized LncPathTM Human Cancer Array to profile the differentially expressed plasma IncRNAs between NSCLC patients and normal healthy controls. Among the 11 aberrantly expressed lncRNAs, we noted a previously not reported lncRNA, RP11-438N5.3, which was considerably down-regulated in NSCLC. RP11-438N5.3, also called heterogeneous nuclear ribonucleoprotein A1 pseudogene 76 (HNRNPA1P76), consists of 509 bp. We further confirmed the expression level of RP11-438N5.3 in another cohort of 69 NSCLC patients and 69 healthy participants by qRT-PCR assay and analyzed its correlation with clinical and pathological features. Based on qRT-PCR data, we found that the expression level of RP11-438N5.3 was correlated with TNM staging and distant metastasis of NSCLC. ROC analyses showed that the AUC for plasma RP11-438N5.3 was 0.814 (95% CI, 0.712–0.905; P<0.01). Importantly, the expression of RP11-438N5.3 was also a biomarker for overall survival.
Additionally, we found a mRNA, STIM1, the plasma level of which was remarkably higher in NSCLC (p<0.01) compared with HC, indicating poor clinical outcomes. RP11-438N5.3 and STIM1 were inversely correlated with each other. The protein of STIM1 is an endoplasmic reticulum (ER)-membrane protein that acts as an activator of store-operated calcium entry (SOCE). It responds to ER Ca2+ store depletion by forming multimers at ER–plasma membrane junctions,13 where it activates Orai1 ion channels and therefore regulates cellular Ca2+ balance and Ca2+ signaling. The role of STIM1 in carcinogenesis has been implicated in previous studies. Overexpression of STIM1 has been found in a variety of cancer cell lines and tissue specimens including colorectal cancer (CRC), hepatocarcinoma, and osteosarcoma cells and was associated with poor prognosis of patients.14 STIM1 is critical for viability, migration and invasion in hepatocarcinoma, osteosarcoma and breast cancer cells.15 Knockdown of STIM1 or pharmacological inhibition of STIM1 suppressed tumor metastasis in breast cancer xenograft models16 and sensitized NSCLC cells to cytotoxic drugs such as Cisplatin.17
In conclusion, our study indicated that RP11-438N5.3 and STIM1 might provide a new strategy for NSCLC diagnosis. Compared with healthy controls (HCs), plasma RP11-438N5.3 expression was significantly lower while plasma STIM1 expression was significantly higher. The expression of RP11-438N5.3 was negatively correlated with STIM1. What’s more, elevated circulating RP11-438N5.3 level might hold a great potential in indicating a beneficial prognosis in NSCLC patients. These lines of evidence indicate that RP11-438N5.3 and STIM1 might be potential biomarkers and targets of gene therapy for NSCLC. Nevertheless, further experimental approaches are warranted to explore how the dysregulation of their expression would prompt tumorigenesis and the mechanisms by which these lncRNAs function.
Ethics Approval and Consent to Participate
Written informed consent was obtained from each individual participant and the experimental protocol was approved by the Clinical Research Ethics Committee of Xian Yang Center Hospital. This study was conducted in accordance with the Declaration of Helsinki.
Data Sharing Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Funding
This research was supported by the Natural Science Basic Research Program of Shaanxi Province (ID: 2017JM8180).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Chen W, Zhang S, Zou X. Evaluation on the incidence, mortality and tendency of lung cancer in China. Thorac Cancer. 2010;1:35–40. doi:10.1111/j.1759-7714.2010.00011.x
2. Rafei H, El-Bahesh E, Finianos A, Nassereddine S, Tabbara I. Immune-based therapies for non-small cell lung cancer. Anticancer Res. 2017;37:377–387. doi:10.21873/anticanres
3. Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59:225–249. doi:10.3322/caac.20006
4. Seijo LM, Peled N, Ajona D, et al. Biomarkers in lung cancer screening: achievements, promises, and challenges. J Thorac Oncol. 2019;14:343–357. doi:10.1016/j.jtho.2018.11.023
5. Abbosh C, Birkbak NJ, Swanton C. Early stage NSCLC - challenges to implementing ctDNA-based screening and MRD detection. Nat Rev Clin Oncol. 2018;15:577–586. doi:10.1038/s41571-018-0058-3
6. Montani F, Marzi MJ, Dezi F, et al. miR-Test: a blood test for lung cancer early detection. J Natl Cancer Inst. 2015;107:djv063. doi:10.1093/jnci/djv063
7. Singhal S, Rolfo C, Maksymiuk AW, et al. Liquid biopsy in lung cancer screening: the contribution of metabolomics. Results of a pilot study. Cancers (Basel). 2019;11:1069. doi:10.3390/cancers11081069
8. Wang L, Chen Z, An L, et al. Analysis of long non-coding RNA expression profiles in non-small cell lung cancer. Cell Physiol Biochem. 2016;38:2389–2400. doi:10.1159/000445591
9. Dang Y, Wang Y, Ouyang X, Wang L, Huang Q. High expression of IncRNA-PCNA-AS1 in human gastric cancer and its clinical significances. Clin Lab. 2015;61:1679–1685. doi:10.7754/Clin.Lab.2015.150312
10. Qiu YR, Zhao MY, Sun L, et al. Expression of IncRNA UCA1 in ovarian cancer and its clinical significance. Eur J Gynaecol Oncol. 2017;38:191–195.
11. Zhang Y, Wu H, Wang F, Ye M, Zhu H, Bu S. Long non-coding RNA MALAT1 expression in patients with gestational diabetes mellitus. Int J Gynaecol Obstet. 2018;140:164–169. doi:10.1002/ijgo.2018.140.issue-2
12. Botti G, Marra L, Malzone MG, et al. LncRNA HOTAIR as prognostic circulating marker and potential therapeutic target in patients with tumor diseases. Curr Drug Targets. 2017;18:27–34. doi:10.2174/1389450117666151209122950
13. Zhang DM, Lin ZY, Yang ZH, et al. IncRNA H19 promotes tongue squamous cell carcinoma progression through beta-catenin/GSK3beta/EMT signaling via association with EZH2. Am J Transl Res. 2017;9:3474–3486.
14. Tripathi V, Ellis JD, Shen Z, et al. The nuclear-retained noncoding RNA MALAT1 regulates alternative splicing by modulating SR splicing factor phosphorylation. Mol Cell. 2010;39:925–938. doi:10.1016/j.molcel.2010.08.011
15. Klattenhoff CA, Scheuermann JC, Surface LE, et al. Braveheart, a long noncoding RNA required for cardiovascular lineage commitment. Cell. 2013;152:570–583. doi:10.1016/j.cell.2013.01.003
16. Zhang H, Chen Z, Wang X, Huang Z, He Z, Chen Y. Long non-coding RNA: a new player in cancer. J Hematol Oncol. 2013;6:37. doi:10.1186/1756-8722-6-37
17. Qi P, Du X. The long non-coding RNAs, a new cancer diagnostic and therapeutic gold mine. Mod Pathol. 2013;26:155–165. doi:10.1038/modpathol.2012.160
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




