Back to Journals » International Journal of Women's Health » Volume 6
Placental tumor (chorioangioma) as a cause of polyhydramnios: a case report
Authors Abdalla N, Bachanek M, Trojanowski S, Cendrowski K, Sawicki W
Received 3 August 2014
Accepted for publication 5 September 2014
Published 20 November 2014 Volume 2014:6 Pages 955—959
DOI https://doi.org/10.2147/IJWH.S72178
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Elie Al-Chaer
Nabil Abdalla, Michal Bachanek, Seweryn Trojanowski, Krzysztof Cendrowski, Wlodzimierz Sawicki
Chair and Clinic of Obstetrics, Gynecology and Oncology, Second Faculty of Medicine, Medical University of Warsaw, Warsaw, Poland
Abstract: Placental chorioangioma is the most common type of placental tumor. It is usually symptomless and may be associated with serious maternal and fetal complication when it reaches a large size. We presented a case of an angiomatous type of placental hemangioma diagnosed in the second trimester of pregnancy in a patient with polyhydramnios. A normal volume of amniotic fluid was successfully achieved by three amnioreductions with conservative management. The size of the placental tumor remained the same from the time of diagnosis to the end of pregnancy. A term labor was uncomplicated and a healthy newborn was delivered. Macroscopic and microscopic examination of the placenta confirmed the diagnosis. Despite the rarity of placental tumors, they should be considered as differential diagnosis in cases of polyhydramnios.
Keywords: amnioreduction, pregnancy disorders, ultrasonography placental tumor, chorioangioma
Background
The primary neoplastic diseases of the placenta include a wide range of conditions that can be classified into two main groups: trophoblastic and nontrophoblastic diseases. Nontrophoblastic diseases occur more frequently, always with benign courses. Chorioangioma and teratoma belong to this group of disorders. The pathogenesis of these neoplasms is controversial; however, they can originate from any part of the placenta excluding the trophoblastic tissues.1–3 Chorioangioma is the most common histological type of placental tumors.4 It can be detected in pathomorphological examination in 0.6%–1% of placentas. An increased incidence rate of choroangioma is associated with maternal age, hypertension, diabetes, female sex of the newborn, premature labors, first delivery and multiple pregnancies.5,6 From a histological point of view, chorioangiomas can be classified into angiomatous (capillary), cellular, and degenerative types. The capillary type is the most common histological subtype.7 Chorioangiomas are usually symptomless, often overlooked during routine ultrasound examination. Abnormalities found elsewhere may arouse suspicion of placental structural abnormalities. Small chorioangiomas are usually not diagnosed, usually without fetal or maternal complications. They can be discovered sometimes postnatally.8 Larger masses, especially those more than 5 cm, are more easily diagnosed by ultrasound scan, and they may cause complications.9 Maternal complications include polyhydramnios which can cause: premature uterine contractions; cervical incompetence; premature labor; placental abruption due to sudden fall of the intrauterine pressure after membrane rupture; malpresentation; increased risk of cesarean section performance; and postpartum hemorrhage.10 Fetal complications include fetal heart failure, thrombocytopenia, nonimmunologic fetal hydrops, hemolytic anemia, intrauterine growth restriction, brain infarction, umbilical vein thrombosis, fetal cerebral embolism, and intrauterine fetal and neonatal death.9–15 Doppler ultrasound examination is the gold standard in primary diagnosis of hemangioma. Magnetic resonance imaging is used only in suspicious cases, while the computed tomography technique has a limited role in the diagnosis of the placental angioma, mainly because of the high radiation risk and poor tissue differentiation. Use of computed tomography to detect metastases is also not indicated since hemangioma are always benign.1
Doppler ultrasound examination may estimate the degree of vascularity of the placental tumor. Intense vascularity of the placental mass may indicate more possible risk of fetal complication. Doppler ultrasound with fetal echocardiography can diagnose the fetal cardiac failure. Fetal cordocentesis can be used as diagnostic method to confirm anemia and to diagnose genetic disorders, and at the same time blood transfusion to the fetus can be done to correct fetal anemia.16 Fetomaternal hemorrhage can be diagnosed by demonstrating the elevated level of fetal hemoglobin in the maternal circulation.17 Serum and/or amniotic fluid levels of alpha-fetoprotein (AFP) can be increased in cases of placental angioma. Increased serum levels of AFP is not pathognomonic for placental angioma since AFP is increased physiologically during pregnancy and a pathological increase of its level may attributed to other fetal and maternal medical conditions.18–19
Case presentation
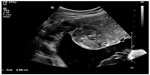
A 26-year-old Caucasian woman gravid at the 28th week of gestation was admitted to the hospital because of premature uterine contraction. She had a successful in vitro fertilization and embryo transfer, and the course of the pregnancy was uneventful apart from anemia and one incidence of one-sided renal colic. The oral glucose tolerance test made at the 24th week of gestation was negative. At admission, the vital signs were in normal range. She experienced an intense pain in the lower abdomen. She was feeling the fetal movements well. The uterus was enlarged to a size corresponding to 34 weeks of gestation. By speculum examination, there was no evidence of premature rupture of membranes or vaginal bleeding. By vaginal examination, the cervix was shortened and closed. Ultrasound examination revealed normal fetal growth regarding gestational age. Neither structural abnormalities nor hydrop of the fetus were confirmed. The amniotic fluid index (AFI) was 45 cm. The placenta was located in the anterior uterine wall. A 5.23×4.45 cm ovoid well-defined heterogenic mass in the placenta was shown (Figures 1 and 2). The Doppler scan showed substantial vascularity of the mass. The length of the cervix was 15 mm and internal cervical os was closed. The ultrasound middle cerebral artery peak systolic velocity was 40 cm/s, which was 1.1 multiple of the median. The patient received intravenous tocolytic drugs (fenoterol and verapamil) and one course of a total dose of 24 mg dexamethasone within 24 hours. The level of the maternal serum AFP was elevated to 304 IU/mL. The intensity of the uterine contractions was increased and an ultrasound-guided amnioreduction in aseptic technique was done, successfully aspirating 1,000 mL of clear amniotic fluid, which resulted in decreasing the intensity of the premature uterine contractions. Serial ultrasound examination at the 30th week of gestation showed a gradual increase of volume of the amniotic fluid and AFI was 43 cm. The ultrasound middle cerebral artery peak systolic velocity was 40.8 cm/s, which was 0.99 multiple of the median for the gestational age. The cervical length was not changed and the internal cervical os was still closed. Neither change of the placental tumor nor fetal abnormalities were confirmed. There was an associated gradual increase of intensity and frequency of the premature uterine contractions. A second ultrasound-guided amnioreduction procedure aspirating 800 mL of clear fluid was performed 9 days after the first ultrasound-guided amnioreduction procedure, resulting in clinical improvement. Within 2 weeks there was gradual increase of the uterine contractions. The serial ultrasound examinations at the 32nd week of gestation again showed a gradual increase of the amniotic fluid volume and the AFI reached 43 cm. The middle cerebral artery peak systolic flow was 49.9 cm/s, which was 1.1 multiple of the median for the gestational age. The cervical length was reduced to 9 mm and the internal cervical os was closed. No change of the size or character of the placental tumor or fetal abnormalities were confirmed. Twelve days after the second amnioreduction, the third one was done, aspirating 1,200 mL of clear amniotic fluid and resulting in clinical improvement. Within 1 month, a spontaneous gradual decrease of the AFI was noticed reaching a normal level for the gestational age at the 36th week of gestation along with gradual cessation of the premature uterine contractions. The size of the placental tumor remained unchanged, with an increasing relative ratio of the placental surface to the placental mass. No fetal abnormalities were confirmed in the ultrasound examinations.
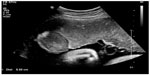  | Figure 2 A gray-scale ultrasound image shows the protruding placental mass into the amniotic cavity from a placenta with the thickest anteroposterior diameter of 5.5 cm. |
After uncomplicated vaginal delivery at the 38th week of gestation, the placenta was delivered completely. A healthy female 3,010 g baby was born with an Apgar score of 10. In macroscopic assessment of the placenta, a single well-circumscribed 5 cm diameter mass was found on the fetal surface of the placenta, with pale yellowish color and with a firm consistency near the edge of the placenta (Figures 3 and 4). The overall placental mass was 600 g. Microscopic examination showed histological features of angiomatous type of chorioangioma. Six weeks after delivery, serum level of AFP was decreased to the normal level of 2.07 IU/mL.
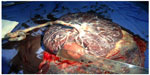  | Figure 3 Macroscopic appearance of the placenta after normal vaginal delivery. |
  | Figure 4 The atypical marginal location of the placental chorioangioma near the placental edge. |
Discussion
Ultrasound examination is the gold standard in the diagnosis of placental chorioangioma during pregnancy. In the presented case, the placental tumor was diagnosed during searches for causes of polyhydramnios in a patient with signs and symptoms of premature labor. According to the ultrasound documentations, the tumor was not diagnosed earlier. It might not be diagnosed earlier due to its small size or may the tumor was not found at all. No earlier ultrasound photo documentations were available. Management of chorioangioma is usually conservative. No specific treatment is needed in case of asymptomatic cases; however, a careful monitoring with serial ultrasound examinations is needed to predict early complications. In our case, because of the premature uterine contractions along with clinical and ultrasonographical sings of cervical incompetence, a steroid administration was performed to induce fetal lung maturation. In the presented case, the amnioreduction procedures were helpful in reducing the amount of the amniotic fluid, resulting in decreases in intrauterine pressure, risk of the premature uterine contractions, and risk of premature labor. Fetoscopic laser ablation of the feeding vessel is considered as a method of successful treatment of even large chorioangiomas. In our case the amnioreduction procedures were able to reduce the symptoms, and the ultrasound examination showed a complex structure of multiple feeding vessels entering the mass from different sites. The fetoscopic laser ablation of the feeding vessels in our case has a theoretical risk of injuring the non pathological vessels leading to placental insufficiency. Also, embolizing the feeding vessels with microcoils or with glue material such as Enbucrilate and intratumoral alcohol injection have been reported. In cases of Enbucrilate and alcohol, the procedure is not associated with fetal toxicity.20–22
Polyhydramnios can be defined as an increased volume of the amniotic fluid regarding the gestational age, and it can be mild, moderate, or severe according to the AFI relative to the gestational age.23 In our case, the polyhydramnios diagnosed in the second trimester of pregnancy was classified as moderate type. Common causes of polyhydramnios such as diabetes mellitus, congenital fetal abnormalities, and multiple pregnancy were excluded; thus, placental tumor was the only reason for polyhydramnios. The mechanism of polyhydramnios formation in cases of placental chorioangioma is not completely understood and several theories have been postulated to explain this phenomenon. Increased intravascular pressure caused by obstruction of the blood flow by the tumor near the umbilical cord insertion may increase transudation into the amniotic cavity, which is in intimate contact with the tumor. The large surface area of the enlarged vessels of the angioma may also predispose to the increased transudation. Polyhydramnios also can be explained by the partial placental insufficiency caused by shunting of the fetal blood into the vessels of the chorioangioma.24 In our case, the polyhydramnios resolved after conservative therapy combined with three amnioreductions. The overall mass of the placenta was increasing throughout pregnancy without changes of the size of hemangioma; therefore, the relative surface of the placental tumor to the overall surface of the placenta was decreasing, and this may decrease the transudation process and at the same time increase the absorption rate of the amniotic fluid by the more viable placenta.
The placenta was expelled spontaneously. The mass was not seen from the maternal side (Figure 5). Expert microscopic examination was needed to confirm the diagnosis and to exclude other more rare malignant conditions. A few cases of rare chorangiocarcinoma have been reported and are characterized by proliferation of atypical trophoblast with high mitotic activity. In these cases, follow-up of the mother and the infant is needed to exclude malignancy.25
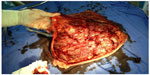  | Figure 5 The complete maternal side of the placenta. |
We present a rare case of placental tumor causing polyhydramnios treated successfully by amnioreductions. Although the placental tumors are rare, they should be investigated and considered in cases of polyhydramnios. Ultrasound scan is the method of choice to detect placental tumors. Serial ultrasound scans were necessary to monitor the amount of the amniotic fluid and to detect possible complication. Histological examination confirmed the benign form of the placental tumor and no more investigations are needed for the mother.
Disclosure
The authors report no conflicts of interest in this work.
References
Elsayes KM, Trout AT, Friedkin AM, et al. Imaging of the placenta: a multimodality pictorial review. Radiographics. 2009;29(5):1371–1391. | |
Prashanth A, Lavanya R, Girisha KM, Mundkur A. Placental Teratoma Presenting as a Lobulated Mass behind the Neck of Fetus: A Case Report. Case Rep Obstet Gynecol. 2012;2012:857230. | |
Miliaras D, Anagnostou E, Papoulidis I, Miliara X. Non-trophoblastic tumor of the placenta with combined histologic features of chorangioma and leiomyoma. Placenta. 2011;32(1):102–104. | |
Jauniaux E, Campbell S. Ultrasonographic assessment of placental abnormalities. Am J Obstet Gynecol. 1990;163(5 Pt 1):1650–1658. | |
Hirata GI, Masaki DI, O’Toole M, Medearis AL, Platt LD. Color flow mapping and Doppler velocimetry in the diagnosis and management of a placental chorioangioma associated with nonimmune fetal hydrops. Obstet Gynecol. 1993;81(5(Pt 2)):850–852. | |
Guschmann M, Henrich W, Entezami M, Dudenhausen JW. Chorioangioma – new insights into a well-known problem. I. Results of a clinical and morphological study of 136 cases. J Perinat Med. 2003; 31(2):163–169. | |
Kirkpatrick AD, Podberesky DJ, Gray AE, McDermott JH. Placental Chorioangioma. Radiographics. 2007;27:1187–1190. | |
Bashiri A, Furman B, Erez O, Wiznitzer A, Holcberg G, Mazor M. Twelve cases of placental chorioangioma. Pregnancy outcome and clinical significance. Arch Gynecol Obstet. 2002;266(1):53–55. | |
Duro EA, Moussou I. Placental chorioangioma as the cause of non-immunologic hydrops fetalis; a case report. Iran J Pediatr. 2011;21(1):113–115. | |
Harman CR. Amniotic fluid abnormalities. Semin Perinatol. 2008; 32(4):288–294. | |
Akercan F, Oncul Seyfettinoglu S, Zeybek B, Cirpan T. High-output cardiac failure in a fetus with thanatophoric dysplasia associated with large placental chorioangioma: case report. J Clin Ultrasound. 2012;40(4):231–233. | |
Sivasli E, Teksam O, Haliloglu M, et al. Hydrops fetalis associated with chorioangioma and thrombosis of umbilical vein. Turk J Pediatr. 2009;51(5):515–518. | |
Imdad A, Sheikh L, Malik A. A large chorioangioma causing intrauterine foetal demise. J Pak Med Assoc. 2009;59(8):580–581. | |
Ghidini A, Locatelli A. Diffuse placental chorioangiomatosis causing multiple fetal cerebral embolism: a case report. J Reprod Med. 2006;51(4):321–324. | |
Witters I, Van Damme MT, Ramaekers P, Van Assche FA, Fryns JP. Benign multiple diffuse neonatal hemangiomatosis after a pregnancy complicated by polyhydramnios and a placental chorioangioma. Eur J Obstet Gynecol Reprod Biol. 2003;106(1):83–85. | |
Gruca-Stryjak K, Ropacka-Lesiak M, Breborowicz G. [Intrauterine blood transfusion in case of placental chorangioma]. Ginekol Pol. 2011;82(4):304–308. Polish. | |
Kawano R, Takemoto S, Shimamatsu K, Hori D, Kamura T. Fetomaternal hemorrhage with intraplacental chorioangioma. J Obstet Gynaecol Res. 2013;39(2):583–587. | |
Androutsopoulos G, Gkogkos P, Decavalas G. Mid-trimester maternal serum HCG and alpha fetal protein levels: clinical significance and prediction of adverse pregnancy outcome. Int J Endocrinol Metab. 2013;11(2):102–106. | |
Højberg KE, Aagaard J, Henriques U, Sunde L. Placental vascular malformation with mesenchymal hyperplasia and a localized chorioangioma. A rarity simulating partial mole. Pathol Res Pract. 1994;190(8):808–813. | |
Babic I, Tulbah M, Kurdi W. Antenatal embolization of a large placental chorioangioma: a case report. J Med Case Rep. 2012;6:183. | |
Ercan CM, Coksuer H, Karasahin KE, Alanbay I, Baser I. Combined approach in a large placental chorioangioma case with intratumoral alcohol injection, cordocentesis, IU transfusion, and amnioreduction. Fetal Pediatr Pathol. 2012;31(6):374–378. | |
Mendez-Figueroa H, Papanna R, Popek EJ, et al. Endoscopic laser coagulation following amnioreduction for the management of a large placental chorioangioma. Prenat Diagn. 2009;29(13):1277–1278. | |
Abele H, Starz S, Hoopmann M, Yazdi B, Rall K, Kagan KO. Idiopathic polyhydramnios and postnatal abnormalities. Fetal Diagn Ther. 2012;32(4):251–255. | |
Kirkpatrick AD, Podberesky DJ, Gray AE, McDermott JH. Best cases from the AFIP: Placental chorioangioma. Radiographics. 2007;27(4):1187–1190. | |
Faes T, Pecceu A, Van Calenbergh S, Moerman P. Chorangiocarcinoma of the placenta: a case report and clinical review. Placenta. 2012;33(8):658–661. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.