Back to Journals » Risk Management and Healthcare Policy » Volume 7
Performance of health product risk management and surveillance conducted by health personnel at sub-district health promotion hospitals in the northeast region of Thailand
Authors Kanjanarach T , Jaisa-ard R, Poonaovarat N
Received 4 July 2014
Accepted for publication 19 August 2014
Published 24 October 2014 Volume 2014:7 Pages 189—197
DOI https://doi.org/10.2147/RMHP.S70653
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Frank Papatheofanis
Tipaporn Kanjanarach,1,2 Raksaworn Jaisa-ard,1,2 Nantawan Poonaovarat3
1Faculty of Pharmaceutical Sciences, Khon Kaen University, Khon Kaen, Thailand; 2Center for Research and Development of Herbal Health Products, Khon Kaen University, Khon Kaen, Thailand; 3Health Consumer Protection, Chaiyapum Health Provincial Office, Chaiyapum, Thailand
Background: Health personnel at sub-district health promotion hospitals (SD-HPHs) are assigned to take responsibility for 15 activities related to health product risk management and surveillance (HP-RM&S). This cross-sectional survey aimed to identify factors that determined their job performance and to record their expressed needs to support HP-RM&S operation. In this study, job performance was defined as completion of all 15 activities.
Methods: Self-administered postal questionnaires were used to collect data from 380 randomly selected health personnel who were in charge of HP-RM&S at SD-HPHs in the northeast of Thailand.
Results: Thirty-six point one percent (n=137) of the respondents were able to perform all 15 of the HP-RM&S activities assigned to SD-HPHs. A logistic regression model identified three factors that statistically significantly determined the completion of all 15 HP-RM&S activities. These were: receiving a high or very high level of support from the community (adjusted odds ratio [OR]: 2.5; 95% confidence interval [CI]: 1.5, 4.1), the responsible persons for HP-RM&S did not hold an administrative position (adjusted OR: 1.7; 95% CI: 1.1, 2.7), and having at least one training session related to HP-RM&S per year (adjusted OR: 1.7; 95% CI 1.1, 2.6). There were 1,536 expressed needs which can be classified into four major categories, ie, training needs (41.6%, n=639), resource support (28.3%, n=435), mechanisms that facilitate HP-RM&S operation (24.1%, n=370) and adjusting of the scope of HP-RM&S (6.0%, n=92). The topics most frequently referred to in training needs were drug law, food law, and cosmetics law.
Conclusion: A strategy for improvement of the job performance in HP-RM&S of health personnel in SD-HPHs should target identifying schemes to encourage the community to proactively participate in HP-RM&S. The district health office as the organization directly controlling and supervising SD-HPHs should also regularly update knowledge base and skills necessary for HP-RM&S operation through training of the responsible health personnel.
Keywords: health products, risk management, surveillance, job performances, sub-district health promotion hospital, training need
Introduction
The Thailand Food and Drug Administration has classified health products into eight types: medicine, food, cosmetics, narcotic drugs used for medical purposes, psychiatric drugs, volatile compounds, medical instruments, and domestic hazardous materials.1 The three products which are most frequently found not meeting standards set by Thai laws, being counterfeit or being advertised inappropriately in local media, are medicines, food, and cosmetic products.2 Protecting consumers from potential dangers posed by these three types of products has therefore been stipulated as one of the major missions of the eleventh National Health Development Plan (2012–2016). In Thailand, especially in rural communities, law enforcement is not effective, surveillance of low quality or counterfeit health products relies heavily on close monitoring from responsible health personnel.
Sub-district health promotion hospitals (SD-HPHs), the front-line health service units of the Ministry of Public Health are located within communities. Health personnel at SD-HPHs are assigned to take responsibility for health product risk management and surveillance (HP-RM&S) within their catchment area. The mission of health personnel in HP-RM&S is divided into five sub-missions (15 activities).3 The five sub-missions are: surveillance for unsafe health products, surveillance for inappropriate advertising, investigation of complaints, strengthening of consumer networks, and maintenance of HP-RM&S databases. In 2011, it was reported that only 55.1% of the SD-HPHs were able to perform 80% or more of the assigned HP-RM&S activities.4 This suggests that despite the intentions of the eleventh National Health Development Plan, there is still a large proportion of Thai communities at risk from unsafe health products.
To improve health personnel’s job performance in HP-RM&S, it is imperative to identify key factors that determine their job performance and elicit their needs for support in operation of HP-RM&S.
Based on an integrated model of individual performance, an individual’s performance is determined by a combination of his/her personal attributes, work effort spent on a job, and level of support from the organization.5 Personal attributes which indicated an individual’s capacity to perform are comprised of three categories: demographic characteristics, competency characteristics, and psychological characteristics. A review of the literature identified three demographic characteristics that can influence the performance of health personnel. These were: sex,6,7 work experience,8–12 and administrative position.8,13 The competency characteristics were: understanding of organizational policy,6,10 training,9 and adequate knowledge.8,11 Attitude toward the job was the principal psychological characteristic being identified in the literature.7,10,13 The contribution of work effort to an individual’s performance, which indicates their psychological drive, can be measured through either willingness to perform the job or willingness to accept responsibility for the job.14 Organizational support in the integrated model usually refers to assistance received from one’s own organization. However, proper operation of HP-RM&S at SD-HPHs requires support from four organizations. The first organization is the district health office which directly controls and supports SD-HPHs. The second organization is the local community hospital, specifically the hospital pharmacist, who provides academic assistance and co-operates in the field operations undertaken by health personnel at SD-HPHs. The third organization is the local administration of the area where the HP-RM&S is conducted. The last organization is the community itself, which is the main target of HP-RM&S work.
This study hypothesized that health personnel’s job performance in HP-RM&S may be determined by the following 12 factors: three demographic characteristics (sex, position in SD-HPH, years in charge of HP-RM&S), three competency characteristics (attending training related to HP-RM&S, understanding policies related to HP-RM&S, knowledge necessary to conduct HP-RM&S), two characteristics of psychological drive (attitude toward HP-RM&S, acceptance of responsibilities in HP-RM&S), and support provided by the four organizations responsible for proper operation of HP-RM&S at SD-HPHs (support from district health office, support from community hospitals, support from local authority, and support from the community).
Methods
This study was approved by the Committee for Human Research Ethics, Khon Kaen University.
This study was a cross-sectional survey. The northeast of Thailand was chosen because this region has the highest number of SD-HPHs15 and the highest prevalence for inappropriate use of health products and unsafe health products in the country.16
Data were collected using postal self-administered questionnaires. The questionnaire was divided into five parts. Part 1: demographic information, part 2: competency characteristics, ie, numbers of times attending training related to HP-RM&S, understanding policies related to HP-RM&S and knowledge necessary to conduct HP-RM&S, part 3: psychological factors, ie, attitude toward HP-RM&S and acceptance of responsibilities, part 4: opinion toward support given by related organizations which are responsible for operation of HP-RM&S at SD-HPHs, ie, support from the district health office, support from the community hospital, support from local administration, support from communities, and part 5: suggestions for operational improvement. The questionnaire was written in the Thai language and the validity of the content was assessed by three experts. One of the experts was a public health officer who worked in the district health office and the other two were hospital pharmacists. These three experts have been providing academic assistance and assisting in the HP-RM&S operations undertaken by health personnel at SD-HPHs for more than 10 years. The clarity of the language in the questionnaire was assessed by five public health officers who worked at SD-HPHs. Prior to administering the main survey, a pilot study was conducted in Chaiyapum, a province in the northeast, to estimate the response rate and the parameters required for sample size calculation, and to assess the quality of the research instrument.17
The studied population was the health personnel in charge of HP-RM&S who worked at SD-HPHs in the northeast. In each SD-HPH, there must be one person who was assigned to take charge of HP-RM&S. As there were 3,467 SD-HPHs in the northeast, the total number of studied population was also 3,467.3 The sample size was determined by the formula:18
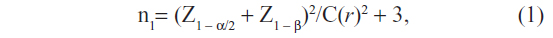
where n1= the minimum number of subjects, α=0.05, β=0.10, Z1 − α/2=1.96, Z1 − β =1.29, C(r) =½ log((1 + r)/(1 − r))
and the correlation coefficient (r), which was obtained from the pilot study, was 0.4. The minimum number of participants needed for this study was 355. This number was adjusted by the response rate which was obtained from the pilot study (30.5%), giving the minimum number of subjects of 1,163.9. The sample size was then rounded up to 1,200.
Due to limitations in accessing the list of names of health personnel in charge of HP-RM&S, SD-HPHs were used as the sampling units. A simple random sampling technique was used to recruit SD-HPHs. A letter and a package were sent to the director of each selected SD-HPH. In the letter, the director was asked to pass the package on to the person in charge of HP-RM&S. The package contained an invitation to participate in the survey, the questionnaire, and a prepaid return envelope.
Variables and measurement
The dependent variable in this study was the self-reporting of job performance in HP-RM&S which was comprised of 15 activities in total. In the questionnaire, it was asked “Do you perform the following activities?” The response options were “yes” and “no”. Each activity being performed received a score of one and each activity not being performed received a score of zero.
Three competency characteristics were measured as followed. Attending training related to HP-RM&S was measured with a question “Please specify the number of training sessions related to HP-RM&S you attended during the fiscal year 2010 through 2012 (October 2009–September 2012)”. Understanding of policies related to HP-RM&S was measured using five items. Knowledge necessary to conduct HP-RM&S was measured with 14 items. Participants were asked to rate their level of understanding of policies and their level of knowledge. The response format was a 5-point rating scale ranging from 1 (very low) to 5 (very high).
The psychological factors; attitude toward HP-RM&S (three positively phrased items and four negatively phrased items) and acceptance of responsibilities (two positively phrased items and three negatively phrased items) were measured using a 5-point rating scale ranging from 1 (strongly disagree) to 5 (strongly agree). In the pilot study, factor analysis was used to extract valid items of the scaled questions of these two cluster variables. Cronbach’s alpha was used to assess reliabilities of the scales. The reliability of attitude toward HP-RM&S was 0.78 and of acceptance of responsibilities was 0.69.17
The questions about level of support were based on the roles and responsibilities that each organization had for SD-HPHs. Lists of support were provided. Participants were asked to rate their opinion toward the level of support being given by each organization. Levels of support were measured using a 5-point rating scale ranging from 1 (very low) to 5 (very high). Support from the district health office comprised 14 items (three items about provision of personnel to assist field activities, three items about budgetary support, four items about provision of materials and equipment necessary for the operation, and four items about management mechanisms). Support from the community hospital comprised three items (academic support, provision of necessary materials, and co-operation in field activities). Support from local administration comprised three items (budgetary support, co-operation in activity planning, and co-operation in field activities). Support from communities comprised four items (co-operation in routine health product surveillance, participation in problem identification, priority setting, and program implementation).
The needs for support and suggestions for operational improvement were collected using open-ended questions.
An example of the questions in the questionnaire is presented in the Supplementary material.
Data analysis
Demographic data and the analysis of job performance rate by activity item are presented with descriptive statistics. For cluster variables, the response scores were averaged and divided equally into five intervals.19 For understanding of policies, level of knowledge, and level of support, the average scores which ranged between 1.00–1.80 were interpreted as very low, 1.81–2.60 as low, 2.61–3.40 as moderate, 3.41–4.20 as high and 4.21–5.00 as very high. For attitude toward HP-RM&S, the average scores which ranged between 1.00–1.80 were interpreted as very negative, 1.81–2.60 as negative, 2.61–3.40 as neutral, 3.41–4.20 as positive and 4.21–5.00 as very positive. For acceptance of responsibilities, the average scores which ranged between 1.00–1.80 were interpreted as not at all accept, 1.81–2.60 as slightly accept, 2.61–3.40 as moderately accept, 3.41–4.20 as highly accept and 4.21–5.00 as completely accept.
Backward stepwise logistic regression model was used to identify the factors that statistically significantly predict job performance in HP-RM&S ie, the completion of all 15 activities being assigned. Alpha level was set at 0.05. Prior to this analysis, job performance was dichotomized into groups which either completed all 15 activities (100% achievement) or did not. Although independent variables in logistic regression can be any type of measurement scales, for convenience of result interpretation, all of the cluster variables measured with 5-point rating scales were dichotomized into a group with mean scores <3.41 and with mean scores ≥3.41.
Results
At the completion of the data collection periods, 425 persons returned the questionnaire, giving the response rate of 35.4% (425/1,200). Of the 425 returned questionnaires, 45 were discarded due to incomplete answers. The remaining 380 were used for data analysis.
For the demographic characteristics, the proportion of male and female respondents was similar, the median time of being in charge of HP-RM&S in SD-HPHs was 10 years (interquartile range [IQR]: 4, 20; range 1 to 35) and the majority of respondents were directors of SD-HPHs (Table 1).
It was found that 36.1% of the respondents (n=137) performed all 15 of the HP-RM&S activities and 2.6% (n=10) did not perform any of the 15 activities. The median overall performance score was 12 (IQR: 8, 15; range 0 to 15). Low performance rates were found in organizing knowledge management among consumers’ network (64.7%) and monitoring health product advertisements (65.0%) (Table 2).
In the past 3 fiscal years prior to the survey (October 2010–September 2012), the majority of the respondents had attended training related to HP-RM&S (81.8%, n=311). The remaining 18.16% (n=69 respondents) reported that they had received no training related to HP-RM&S. The median number of times personnel attended training during the past 3 years was two (IQR: 1, 3; range: 0 to 10). A majority of the respondents rated their understanding of policies (mean: 3.87; standard deviation [SD]: 0.59) and their knowledge (mean 3.54; SD: 0.72) at a moderate level.
The mean attitudinal score was 3.34 (SD: 0.57), indicating that a majority of the respondents had neutral attitudes toward HP-RM&S. The mean score of acceptance of responsibility was 3.27 (SD: 0.63) indicating that a majority of the respondents were moderately accepting of their responsibilities. In terms of organizational support, the majority of respondents rated support from all four organizations at a moderate level. The mean level of support from the district health office was 2.92 (SD: 0.58), from community hospitals 3.37 (SD: 0.80), from local authorities 2.83 (SD: 0.95), and from the community 2.85 (SD: 0.89).
Logistic regression analysis showed that three of the 12 factors being studied were statistically significantly associated with job performance in HP-RM&S ie, completion of all 15 activities. These three factors were: receiving a high or very high level of support from the community, that the responsible person for HP-RM&S was not also a director of the SD-HPH, and that the responsible person had at least one training session related to HP-RM&S per year (Table 3).
Three hundred and thirty one respondents provided 1,536 suggestions for operational improvement. These expressed needs can be classified into four major categories: training needs, resource support, mechanisms to facilitate HP-RM&S operation, and scope of the HP-RM&S under the responsibility of SD-HPHs.
Most of the responses (41.6%, 639/1,536) were about the need for updating knowledge and improving skills through workshop training. The topics most frequently requested were drug law (245 responses) followed by food law (177 responses), cosmetics law (142 responses), and techniques in using screening test kits (75 responses).
Three hundred and seventy responses (28.3%) were about the need for resources to operate HP-RM&S. The resources requested were either for materials to be used in field activities, such as screening test kits and ready-to-use media for educational or promotional campaigns (208 responses), or for financial support from district health offices (162 responses).
Another 24.1% of responses (n=435) were about mechanisms to facilitate HP-RM&S operation. Two hundred and ninety one responses were for pharmacists to participate in health product inspection and complaint investigation and 141 were for policy adjustment to address HP-RM&S as one of the key health policies at a provincial level.
In addition, 92 of the responses (6.0%) concerned adjusting the scope of HP-RM&S responsibilities. There were 52 suggestions to transfer health product inspection and complaint investigation to pharmacists. The remaining 40 responses were to take responsibility for monitoring health product advertisements on local radio away from HP-RM&S personnel.
Discussion
This study found that the proportion of health personnel who were able to carry out all HP-RM&S activities was particularly low, indicating that there is an urgent need to identify strategies to facilitate SD-HPHs in the operation of HP-RM&S.
Of the three demographic characteristics being studied, position in SD-HPH was the only factor statistically significantly associated with better job performance of HP-RM&S. Responsible HP-RM&S personnel who did not also hold an administrative position (ie, not a director of SD-HPH) tended to perform more activities than health personnel who hold an administrative position. This is likely to be because directors of SD-HPHs need to oversee all jobs at SD-HPHs and may therefore have difficulty devoting sufficient time to undertake HP-RM&S activities.
Although a majority of health personnel rated their overall level of HP-RM&S knowledge at a moderate level, the high number of expressed needs for more information about drug, food, and cosmetics law indicated that there was a significant gap in knowledge in these areas. The association between attendance of more than one training session and improved HP-RM&S performance indicates that the topics already covered in the training sessions do assist health personnel to perform their work more effectively. Although this study did not collect data on the contents of training sessions, it is known that training usually covers current topics, new regulations, and innovative solutions. Therefore, incorporating topics on food, drug, and cosmetic law into these training sessions could improve HP-RM&S performance.
Less than half of the health personnel who responded to the survey had positive attitudes toward HP-RM&S. Furthermore, only one third of respondents “highly” or “completely” accepted their responsibilities in HP-RM&S. These findings are consistent with recent studies which reported that some health personnel at SD-HPHs believed that operation of HP-RM&S brought negative consequences and some personnel did not conduct health product post-marketing surveillance or failed to take the required legal action for fear of conflict.20,21 Although attitude toward HP-RM&S and acceptance of HP-RM&S responsibilities was not shown to determine job performance in our study, several publications have suggested that negative attitudes and unwillingness to accept responsibility can lower quality of job performance.22–24
In this study, a considerable amount of needs were expressed about reducing the scope of HP-RM&S by removing responsibility for health product inspection, investigation of complaints, and monitoring of health product advertisements. Notably, all of these activities require skill in interpretation of the laws governing drugs, food, and cosmetics. Pharmacists in community hospitals, whose responsibility is to provide academic and technical support, could therefore assist HP-RM&S personnel at SD-HPHs by producing a step-by-step manual for personnel to assess health product compliance and advertisements with respect to the particular laws and regulations covering them. The pharmacists should also respond to the expressed need for increased participation in field investigations. This will enable coaching in application of the law in real situations in addition to establishing good relationships between pharmacists and health personnel at SD-HPHs.
Although health personnel evaluated support from all four related organizations at a moderate level, support from communities was the only factor that showed a statistically significant association with better job performance of HP-RM&S activities. This was not surprising, as most HP-RM&S activities rely on community participation. A low level of support from communities is also likely to negatively affect job performance in building and strengthening of consumer networks. Therefore, strategies to gain sustainable co-operation from communities in HP-RM&S must be sought.
One of the strengths of this study is that the overall HP-RM&S performance was evaluated by individual activity items. This enabled the identification of activities which need urgent attention. In addition, as a low response rate is a common drawback of collecting data by postal self-administered questionnaires, a pilot study was conducted to estimate the response rate and to inform sample size estimation. This process allowed us to obtain sufficient responses for logistic regression analysis; increasing the reliability of the study results. However, in this study, the representativeness of the sample to the studied population was not assessed as the researchers were not able to gain access to the data base of health personnel demographics. It is possible that the respondents may perform more activities than the non-respondents. Therefore interpretation of the findings of this study should be done with caution.
Conclusion
In conclusion, the findings of this study indicate that performance of HP-RM&S conducted by health personnel at SD-HPHs is influenced by demographic characteristics, competency characteristics, and support from related organizations. Of the three factors that were found to have a statistically significant association with improved job performance, support from the community was shown to have the strongest association. Therefore, identifying strategies to build and maintain community participation in HP-RM&S should be given priority. In addition, district health offices should consider implementing regular targeted training and updating knowledge of the applicable drug, food, and cosmetics laws necessary to conduct HP-RM&S as part of their role to directly supervise and support the activities of SD-HPHs.
Acknowledgments
The study was partially funded by the Faculty of Pharmaceutical Sciences Khon Kaen University and the Center for Research and Development of Herbal Health Products Faculty of Pharmaceutical Sciences Khon Kaen University, Thailand. The authors would like to thank Dr Glen N Borlace for English editing.
Disclosure
The authors report no conflicts of interest in this work.
References
Food and Drug Administration. Health Product Acts. Bangkok: Food and Drug Administration; 1994. | |
Food and Drug Administration. Report of Penalty 2012. Nonthaburi: Food and Drug Administration; 2013. Available from: http://www.fda.moph.go.th. Accessed July 2, 2013. | |
Bureau of Primary Care Development Coordination. Operational Guideline of Sub-District Health Promotion Hospitals. 3rd ed. Nonthaburi: Usakarnpim; 2010. | |
Food and Drug Administration Ministry of Public Health. Summary Report of Health Product Consumer Protection Practices in Provincial Areas 2012. Nonthaburi; 2012. | |
Schermerhorn JR, Hunt G, Osborn N. Organizational Behavior. 8th ed. Danver: John Wiley; 2003. | |
Sarirat T. Factor Affecting the Performance of Health Center Officers on Preventing and Controlling of Diabetes Mellitus in Sakon Nakorn Province [master’s thesis]. Mahasarakham: Mahasarakham University; 2004. | |
Jumparway S. Factors Association with Administrative Performances of Primary Care Unit’s Chief in Khon Kaen Province [master’s thesis]. Khon Kaen: Khon Kaen University; 2006. | |
Mungsamak Y. A Study of Factors Affecting the Affectiveness of Roles Performance of the Subdistrict Health Personnel in Chaiyaphum Province [master’s thesis]. Khon Kaen: Khon Kaen University; 1996. | |
Kochsila S. Factors Affecting Health Center Chiefs Performance for Dental Health Surveillance and Promotion among Primary Education Students in Nakonpathom Province [master’s thesis]. Nakhon pathom: Mahidol University; 2001. | |
Limpabendhu P. Factor Affecting Performance on Health Consumer Protection of Sub-District Health Workers Sukhothai Province [master’s thesis]. Pitsanuloke: Naresuan University; 2009. | |
Thongchai T. Motivation Affecting Pharmaceutical Service Performance of Health Personnel at Primary Care Units Nongkhai Province [master’s thesis]. Khon Kaen: Khon Kaen University; 2009. | |
Bouphan J, Bouphan P, Kompor P. Factors Influencing the Professional Working Performance of Dentists at Community Hospitals of the Public Health Region 12. KKU Research Journal. 2011;16(6):679–692. | |
Auiwattanakul P. Practicing and Attitude in Health Consumer Protection of Pharmacy Technicia [master’s thesis]. Khon Kaen: Khon kaen University; 2009. | |
Chirunthorn R, Singpaiboonporn N, Lohanavakul P. Factor Affecting the Role Performance of Pharmacists in Primary Care Units (PCU): A case study in the upper southern region of Thailand. Song Kla Med J. 2006;24(6):505–516. | |
Bureau of Policy and Strategy. Information of service place. Nonthaburi: Bureau of Policy and Strategy; 2012. Available from http://bps.ops.moph.go.th/Healthinformation/index.htm. Accessed April 6, 2012. | |
Food and Drug Administration Ministry of Public Health. Summary Report of Health Product Consumer Protection Practices in Provincial Areas 2010. Nonthaburi; 2010. | |
Poonaovarat N, Kanjanarach T. Job performance and attitudes of health personnel at sub district health promoting hospitals toward health consumer protection: A case study of Chaiyaphum province. Isan Journal of Pharmaceutical Sciences. 2013;9(1):160–165. | |
Sokal RR, Rohlf FJ. Biometry: The Principles and Practice of Statistics in Biological Research. 3rd ed. New York: WH Freeman; 1995. | |
Best WJ, Kahn VJ. Research in Education. 10th ed. Boston: Allyn & Bacon; 2006. | |
Sangownwathana P. The Study of Legitimate Advertising of Health Products Situation in Regional Area. Bulletin of the Department of Medical Sciences. 2009;51(3–4):204–212. | |
Hongsamoot D, Pradubmook P, Kuttimanon P. Problems and Recommendation for Place and Product Surveillance: A Case Study of an Epidemic of Steroid Tablets. Nonthaburi: Health Systems Research Institute; 1998. Available from: http://kb.hsri.or.th/dspace/handle/11228/1586. Accessed October 15, 2014. Thai. | |
Herzberg F. One more time: How do you motivate employees? Harv Bus Rev. 2003;81(1):87–96. | |
Schermerhorn JR, Hunt JG, Osborn RN. Organizational Behavior. 9th ed. New York: Willey; 2005. | |
Lucius B, Rensberg DV. Community Participation in Development: Nine Plagues and Twelve commandments. Community Dev J. 2000;35(1):41–58. |
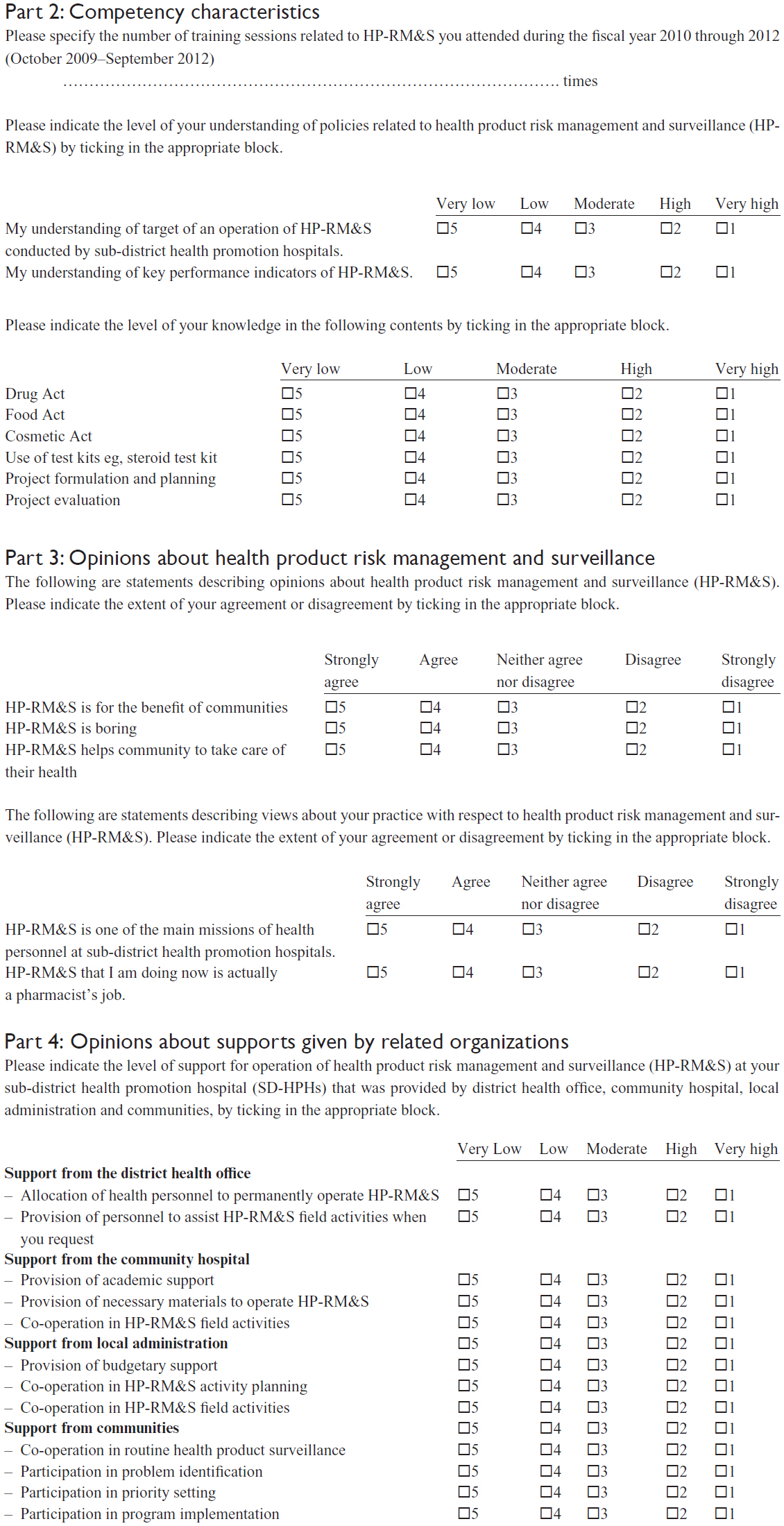
Supplementary material
Example of questions in the survey questionnaire
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




