Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
Pain Prevalence, Localization, and Intensity in Adults with and without COPD: Results from the Danish Health and Morbidity Survey (a Self-reported Survey)
Authors Hansen J , Molsted S, Ekholm O , Hansen H
Received 3 August 2020
Accepted for publication 6 November 2020
Published 9 December 2020 Volume 2020:15 Pages 3303—3311
DOI https://doi.org/10.2147/COPD.S275234
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Jeanette Hansen, 1 Stig Molsted, 2 Ola Ekholm, 3 Henrik Hansen 4
1Department of Public Health, University of Copenhagen, Copenhagen, Denmark; 2Department of Clinical Research, North Zealand Hospital, Hillerød, Denmark; 3National Institute of Public Health, University of Southern Denmark, Odense, Denmark; 4Respiratory Research Unit, Department of Respiratory Medicine, University Hospital Hvidovre, Hvidovre, Denmark
Correspondence: Henrik Hansen
Respiratory Research Unit, Department of Respiratory Medicine, University Hospital Hvidovre, Kettegård alle 30, Hvidovre 2650, Denmark
Tel +45 28946780
Email [email protected]
Introduction: Pain is a clinical complication to chronic obstructive pulmonary disease (COPD) that interferes negatively with physical activity level (PAL), quality of life (QOL) and pulmonary interventions. Yet, research in pain characteristics including prevalence, localization, and intensity in people with COPD are sparsely researched.
Aim: To investigate self-reported pain prevalence, localization and intensity of pain in people with and without COPD, and to investigate the association between pain intensity and PAL among participants with COPD.
Methods: Data were derived from the Danish Health and Morbidity Survey in 2017. The study population was restricted to individuals aged ≥ 35 years. Data included pain intensity assessed on the Numeric Rating Scale (NRS) and localization, PAL, QoL, sleep disturbance, comorbidities, sociodemographic and behavioral factors.
Results: In all, 528 participants with COPD and 8184 participants without COPD (51% females, mean ±SD age 67.1± 11.4 years) were analyzed. Pain prevalence within the past 14 days was significantly higher in participants with COPD vs nonCOPD (72.7% vs 57.7%, p< 0.001) and mainly located in the limbs, thorax, and lower back. COPD was associated with the prevalence of chronic pain (≥ 6 months) (OR: 2.78, 95%CI: 2.32; 3.34, p< 0.001). Participants with COPD reported a higher pain intensity compared to those with nonCOPD with a mean difference of 1.04 points (95%CI: 0.75; 1.32, p< 0.001) on the NRS. In the adjusted multiple logistic regression analysis, pain intensity was negatively associated with odds of being physical active (OR: 0.72, 95%CI: 0.61; 0.85, p< 0.001).
Conclusion: Pain is more prevalent in people with self-reported COPD. After adjustment for age and gender, COPD was associated with an elevated pain intensity. Sleep disturbance and multimorbidity had the most pronounced impacts on pain intensity in the multiple linear regression model. In participants with COPD, increased pain intensity was negatively associated with being physically active.
Keywords: COPD, pain, cross-sectional, physical activity, quality of life
Introduction
COPD is a progressive lung disease characterized by chronic inflammation of the airways and a persistent airflow limitation.1,2 COPD is the third leading cause of death and morbidity worldwide,3–5 and it is associated with impaired quality of life (QOL), dyspnea, fatigue and physical inactivity.6 Previous studies in people with COPD have reported multiple symptoms including sleep disturbances, impaired mood and bodily pain,6,7 symptoms that are not limited to typical COPD related complaints.
Pain prevalence in people with COPD was recently published in a systematic review, and ranged from 21–82% in the included studies with a mean pooled estimate of 66%.8 The causality of the high pain prevalence and persistent pain in people with COPD is complex and poorly understood.8,9 Persistent pain is a clinical issue with significant clinical implications, which adversely impacts QOL, mood, breathlessness, and participation in activities of daily living.8
A sedentary lifestyle as a result of, eg dyspnea and fear of activity-induced discomfort may aggravate common age-related comorbidities, among them osteoarthritis, low back pain and osteoporosis of which pain is a frequently reported manifestation.6,10,11 Other factors related to the manifestations of COPD may also contribute to pain in people with COPD. Firstly, an elevated activation of cytokines may induce an inflammatory pain,6,12 and it is plausible that the systemic inflammatory process in COPD contributes to the generation of more prevalent pain.13 Secondly, it may be anticipated that a relative abnormal hyper-expanded and rigid chest wall limits free range of motion and thereby contributes to several joint pathologies leading to thoracic pain in people with COPD.6,14 Pain and muscle discomfort may arise from medication. Oral corticosteroids induce muscle wasting, LABAs may give painful muscle spasms, and many other treatments such as statins may harm the muscle or change the perception of using them.
People with COPD who live with chronic pain are less active physically than similarly aged people without COPD as well as their peers with COPD and no pain.9 A reduced physical activity level (PAL) may have crucial negative implications, as physical activity has positive effects on QOL and survival in COPD.15,16 The intensity of daily physical activity among people with COPD is associated with the peak magnitude of dynamic lung hyperinflation. Physical activity in COPD may be affected by cardiac dysfunction, indicating that their exercise capacity is restricted by a combination of factors as central hemodynamic, oxygen transport, and peripheral muscle oxygenation capacities.17,18
As pain is a clinical complication in COPD that may negatively impact PAL and QOL, further advance, insight and understanding of the characteristics of chronic pain and its influence on various symptoms and behavior are needed to form approaches for routine assessment and subsequent managing pain in people with COPD. Conflicting findings in the exiting literature underscores the importance of continued research in this field.6,9,19
We hypothesized that COPD is associated with higher pain prevalence in the limbs and the thorax and pain intensity being negatively associated with daily level of physical activity. Thus, the aim of this study was to investigate the pain prevalence, localization, and intensity in people with and without COPD, and to investigate the association between pain intensity and daily level of physical activity reported by people with COPD.
Subject Materials and Methods
Data for this study were derived from the nationwide Danish Health and Morbidity Survey 2017.20
The Danish Health and Morbidity Survey has been conducted regularly since 1987 and assesses trends in health and morbidity in the adult Danish population and investigates factors that are associated with health status and health behavior. The survey in 2017 was based on a random sample of 25,000 adults (16 years or older) from the general Danish population. In all, 14,022 individuals (56.1%) completed a self-administered questionnaire.20 The design and characteristics of the survey are described in detail elsewhere. The present study was restricted to respondents who had stated yes or no to self-reported COPD, and were minimum 35 years of age, as persons below 35 years of age have a low prevalence of COPD. Hence, the study population in the present study consisted of 528 people with COPD and 8184 people with nonCOPD. All data were self-reported, except for sex and age that were obtained from The Danish Civil Registration System.21 Furthermore, cohabitation status was classified by combining self-reported information and data from The Danish Civil Registration System.21
Three questions were used to assess pain prevalence and pain localization during the past 14 days:
Pain or discomfort in the shoulder or neck; pain or discomfort in the back or lower back (thorax/back); and pain or discomfort in the arms, hands, legs, knees, hips or joints (the limbs).20 The three questions had the following answer categories: yes, very bothered; yes, bothered a little; or no. The reported answers of pain were in each of the three questions recoded into the dichotomous answers “yes” (yes, very bothered; yes, bothered a little) and “no”.
To assess pain intensity during the past week the numeric rating scale was used and elucidated at a scale from 0–10, where 0 indicates no pain at all, and 10 indicates worst possible pain.20,22 The question was only asked to respondents who answered positively to the following question: do you have chronic/long-lasting pain lasting six months or more?
The PAL was categorized into the four levels from the Saltin–Grimby Physical Activity Level Scale: inactive (sedentary leisure activities); moderate active (minimum four hours of moderate activity weekly as walking, cycling, etc); medium active (minimum four hours exercise training weekly); and high active (strenuous activity or exercise training several times a week).23,24 In order to stratify the respondents into being “inactive” or “active”, the three highest levels of leisure time PAL (“moderate”, “medium” and “high active”) were combined into one level.
Self-rated health was based on the health status of one single question from the questionnaire SF-12: In general, would you say your health is (1) excellent, (2) very good, (3) good, (4) fair, (5) poor.
The reported answers were recoded into a dichotomous answer with “positive” (excellent, very good, good) and “negative” statements (fair, poor).
Sleep disturbance was based on self-reported sleeping problems or insomnia during the past 14 days and stratified to a dichotomous answer “yes, very, and slightly bothered” or “no”.
Multimorbidity was defined as reporting at least two of the following conditions: heart diseases, stroke, lung diseases, diabetes, osteoporosis, diseases of the back, cancer, psychiatric disorders, osteoarthritis, and rheumatoid arthritis. For exploratory purposes we created a variable on assumed painful comorbidities, defined as reporting at least one of the following conditions: heart diseases, osteoarthritis, rheumatoid arthritis, osteoporosis, cancer and diseases of the back. Smoking status was categorized as being current smoker or not. High alcohol intake was defined as exceeding the sensible drinking guidelines with a low risk limit (seven standard drinks/week for females, and 14 standard drinks/week for males). Level of highest completed education was categorized into four groups as: <10 years; 10–12 years; and ≥13 years; and other educations (eg foreign educations).
Body mass index (kg/m2) was calculated using self-reported data on height and weight. Cohabitation status was categorized and analyzed as: “living alone”, “living with partner or being married”. Type of COPD (emphysema, chronic bronchitis and bronchiectasis), COPD severity (GOLD I, II, III, IIII) and classification (Group A, B, C and D) were not reported and, thus, data were analyzed as respondents with “COPD” and “nonCOPD”. The study was in accordance with the approval of the local ethical committee and the Danish Data Protection Agency (P-2019-354). The data access and processing agreement was approved by the legal section at National Institute of Public Health, University of Southern Denmark, Denmark.
Statistical Analyses
Descriptive demographic data are presented as numbers and percentages or mean ±SD. Between group differences in continuous variables were analyzed using an unpaired t test or the Mann–Whitney test, and on categorical data using the chi-squared test. Normal distribution of the data was inspected by Q-Q plots and verified by Kolmogorov–Smirnov test. Number of comorbidities are presented as median and IQR.
The association between the exposure variable COPD and pain intensity was examined in a multiple linear regression analysis, and the association between pain intensity and physical activity among participants with COPD was investigated in a multiple logistic regression analysis. Backward stepwise regression was used. The models were adjusted for age, gender, BMI, cohabitation status, educational level, current smoker, high alcohol intake, sleep disturbance and multimorbidity or painful comorbidity to control for confounding. Unadjusted and adjusted models are presented. The results are presented as β coefficient and OR with 95%CIs, respectively. Correlation coefficient for independent variables were provided using Spearman’s rho.
Calibration weighting was applied to reduce the potential impact of nonresponse bias on the estimates in the present study. The weights were computed by Statistics Denmark based on the collected information of variables, among them gender, age, cohabitation status, level of education, income, employment status, country of origin, health-care utilization, and research protection for all individuals who were invited.20 Data were analyzed using IBM SPSS version 26 and p-values of less than 0.05 were considered statistically significant.
Results
Characteristics of the study population are presented in Table 1. The mean age among participants with self-reported COPD were 67.1 ± 11.4 years, 50% were men and 53.4% of the participants with self-reported COPD were married or living with a partner. Among participants with nonCOPD the mean age was 56.5 ± 13.6 years, 51.1% were men and 72.6% were married or living with a partner. A statistically significant greater proportion among participants with COPD had a high alcohol intake, was current smokers, were inactive physically, had more frequent sleep disturbance and more comorbidities (Table 1). Participants with COPD had a statistically significant higher prevalence of all listed comorbidities. A table with prevalence of the comorbidities are available in supplementary material Table S1.
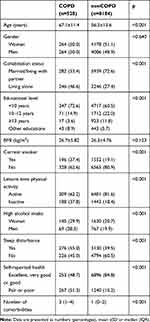 |
Table 1 Characteristics of the Participants with COPD and nonCOPD, Respectively |
The pain prevalence is presented in Figure 1. Pain the past 14 days was more prevalent in participants with COPD compared to nonCOPD in all three locations (eg in the limbs 72.7% vs 57.7%, p<0.001), and was mainly located in the limbs, thorax, or lower back (Figure 1). A gender difference was observed in the pain prevalence in participants with COPD compared to the nonCOPD group, and showed a higher percentage of pain reported by women in all three localization areas; shoulder/neck, thorax/lower back, and limbs (all p<0.001). After adjustment for age and gender, the respondents with COPD had an OR 1.52 (95% CI:1.25; 1.85, p<0.001) for pain in the shoulder/neck area, an OR 1.57 (95% CI:1.28; 1.93, p<0.001) for pain in the limbs, and an OR 2.35 (95% CI:1.92; 2.88, p<0.001) for pain in the thorax/lower back, compared to nonCOPD.
The prevalence of chronic pain was higher in the group with COPD (Figure 1) and COPD was associated with chronic pain with an OR 2.78 (95% CI: 2.32; 3.34, p<0.001), compared to nonCOPD. After adjustment for age and gender, the respondents with COPD had an OR 2.53 (95% CI: 2.11; 3.05, p<0.001) for chronic pain.
Pain intensity from the reported chronic pain was statistically significant elevated in the group with COPD compared to the nonCOPD group with a mean difference of 1.04 points on the NRS (95% CI: 0.75; 1.32, p<0.001) (Figure 2), and after adjustment for age and gender the participants with COPD had an increased NRS pain intensity of 1.03 (95% CI: 0.74; 1.32, p<0.001) points.
 |
Figure 2 Pain intensity of chronic pain in participants with COPD and nonCOPD. Data are unadjusted and presented as mean ±SD. |
Pain in any location were significantly less prevalent within the group with COPD and without any comorbidities (n=65) and were 38.2% in shoulder/neck, 38.2% in limbs and 42.7% in the thorax/low back respectively. The subgroup with COPD and no comorbidities had a statistically significant lower NRS score of 3.56±SD2.34 vs 6.06±SD2.08 in those with COPD and ≥1 comorbidity (p<0.001). Similarly, pain in any location was significantly less prevalent within the group with nonCOPD and no comorbidities (n=2863) respectively 40.8% in shoulder/neck, 39.8% in limbs and 39.1% in thorax/low back. The NRS pain score was statistically significant lower; 4.11±SD2.15 vs 5.07±SD2.23 in those with nonCOPD and ≥1 comorbidity (p<0.001).
Sleep disturbance, multimorbidity, being a current smoker had the most pronounced impacts on pain intensity in the multiple linear regression model (Table 2), whereas COPD was not associated with pain intensity in the regression analysis after confounder adjustments. Multiple linear regression with the exploratory variable of assumed painful comorbidities in replacement of multimorbidity did increase the impact of sleep disturbance (β: 0.91 [95% CI:0.72; 1.09]) on pain intensity, while COPD (β: 0.51 [95% CI: 0.16; 0.87]), being physical inactive (β: 0.53 [95% CI: 0.31; 0.75]) and having ≥1 painful comorbidity (β: 0.76 [95% CI: 0.57; 0.96]) were significantly associated with pain intensity (Table S2).
 |
Table 2 Multiple Linear Regression on the Associations Between COPD and Pain Intensity |
In the multiple logistic regression model elevated pain intensity was associated with reduced odds ratio of being active physically among individuals with COPD (Table 3). In the exploratory multiple logistic regression with painful comorbidity as variable elevated pain intensity was associated with reduces odds ratio of being active physically among people with COPD (OR: 0.72 [95% CI: 0.59; 0.86]) (Table S3). Having ≥1 painful comorbidity was also associated with reduced odds ratio of being active physically, but it was not statistically significant (OR: 0.50 [95% CI: 0.18; 1.41]) (Table S3).
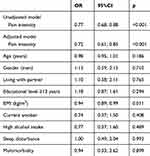 |
Table 3 Multiple Logistic Regression on Pain Intensity and Odds of Being Active Physically Among Individuals with COPD |
Discussion
This is the first study to present novel data on acute and long-term chronic pain in people with self-reported COPD, sleep disturbance, multimorbidity and assumed painful comorbidities in a large population-based sample.
The participants with COPD had a statistically significant higher pain prevalence compared to those with nonCOPD, and it was primarily located in the limbs, thorax, and lower back. Whilst COPD was not associated with pain intensity after confounder adjustments (Table 2), increased pain intensity was associated with reduced odds of being active physically in individuals with COPD.
Similar to previous studies we found a higher prevalence of pain in the participants with self-reported COPD compared to the general population without self-reported COPD.6,7,9 Several factors explain the noticeable higher prevalence of pain among our participants with COPD. In this study, the participants with COPD were older than the participants with nonCOPD. Pain is associated with a number of degenerative conditions that occur in middle-aged individuals as a natural consequence of ageing.7 However, after adjustment for age and gender, COPD was still associated with an elevated odds ratio. Medical conditions such as peripheral neuropathy and claudicatio intermittens together with other comorbidities, are associated with pain intensity in people with COPD.6,19 Indeed, our participants with COPD reported a significant higher frequency of comorbidities compared to the nonCOPD group, and the presence of multimorbidity or exploratory assumed painful comorbidities among the participants with COPD were associated with an elevated pain intensity (Table 2 and Table S2). In addition, participants with and without COPD, and the presence of no comorbidities both presented lower pain prevalence and pain intensity compared to those with ≥1 comorbidity. The higher pain prevalence registered in the thorax and the lower back in people with COPD can be a result of vertebral deformities and costovertebral arthropathy connected to long-term pharmacologic treatments with inhaled or oral corticosteroids and change in breathing pattern.12,25,26 In our study this group in particular reported the comorbidities osteoarthritis, osteoporosis, and diseases of the back, which were associated with pain.
A gender difference was found in the present study and female participants with COPD reported higher pain prevalence and pain intensity, and this is consistent with results from other studies.9 The gender difference may be due to different perceptions of pain and pain adaptations in the brain, as well as hormonal, social, cultural, psychological and neuroanatomical aspects.27
A high pain intensity among people with COPD can be a result of previous periods of exacerbations with dyspnea and coughing, which are likely to aggravate fatigue and pain intensity in the thorax and back.6,9,28 In the present study sleep disturbance was strongly associated with pain intensity (Table 2 and Table S2). Sleep disturbance is a well-known problem that interrelates and has a negative impact on pain perception and pain intensity due to increased pro-inflammatory cytokine secretion, and possible reinforce physical inactivity.9,29
This study emphasizes the importance of the impact of other surrounding factors than COPD per se has on pain intensity. As COPD is frequently complicated by the presence of pain, our findings emphasize the importance of incorporating routine assessment of pain, sleep quality and comorbidities at the physician consultation and subsequent management in rehabilitation, for example. Additional studies are warranted to determine the exact etiologies and mechanism of the pain, as well as strategies for pain management among this patient group in clinical practice. Pain management is best undertaken using psychological, physical, behavioral, and pharmacological strategies, and may be a goal of the multidisciplinary rehabilitation teams.9
Different findings in the literature in estimates of pain prevalence and intensities among people with COPD, are likely related to the context of the sample (eg stable, acutely unwell, or end of life), study populations (eg outpatients, pulmonary rehabilitation, and population), assessment instruments, and reference period (eg past week, month, and year). A study by Lee et al reported daily chronic pain for more than three months as more prevalent in individuals with COPD compared to healthy age and gender matched individuals.9
People with COPD are less active physically compared to healthy sex and age matched individuals,9,19 which our study also confirmed. A low level of physical activity is associated with several risks, eg metabolic syndrome, cardiovascular diseases, and deterioration of physical function, and in this study increased pain intensity was negatively associated with being active physically among participants with COPD (Table 3). People with COPD have an elevated TNF-α level in the blood, a probable factor for the loss of muscle mass and muscle strength, which may be enhanced by simultaneous physical inactivity.30 Pain may reduce the internal motivation to increase the PAL in people with COPD and thereby contribute to reduced QOL and decreased physical capacity.9 In findings from other studies, an increase in pain intensity in participants with COPD was linked to negative effects on physical activity, QOL and multimorbidity compared to people with COPD with minimal or no pain.9,19 In addition, dyspnea and fatigue are symptoms that can aggravate pain negatively, and cause anxiety and fear-avoidance behavior with reduced compliance and motivation for being active physically. Dyspnea, fatigue, impaired QOL, and low PAL and their relationship with pain in people with COPD is unclear, and probing the possibility of both one-way or two-way associations.9 The consequences of continued physical inactivity in people with COPD are pivotal and consequences may lead to irreversible functional impairments, impaired QOL, risks of hospitalization and mortality. Thus, a more nuanced understanding of how to increase the PAL in patients with COPD in clinical practice is needed.
Strength and Limitations and Future Perspectives
This study presents data that were collected from a nationally representative survey with relatively great number of participants. The prevalence of COPD in the nationwide Danish Health and Morbidity Survey 2017 was approximate 6% and is representative for the prevalence of COPD in Denmark,31 which is a strength for the external validity. The short reference periods for the measurements of the pain status probably reduced the risk of recall bias. The use of calibration weighting to reduce the potential impact of nonresponse bias was also a strength in this study, where the probability of selection bias was reduced. Reporting of both acute and chronic pain among participants with COPD in this study is a strength and may contribute to the upcoming evidence in this field. The study was limited by the following reasons: data were self-reported, and they did not include data from spirometry, GOLD stages, medication, dyspnea, fatigue and mood disorders. Subsequently, it is a risk that COPD is underreported, and thus the prevalence of COPD underestimated in this study. Secondly, since spirometry was not performed, it is possible that some of the participants in the nonCOPD group had early stages of COPD without clear symptoms. Furthermore, the data were collected in a cross-sectional study, which does not allow conclusions about causality. The future perspectives from our findings are that more frequent use of pain and sleep quality measurements in clinical research and clinical practice are needed, together with classification and focus on the impact of multimorbidity in this group. Pain prevalence and pain management should be mentioned in future COPD guidelines and respiratory societal statements.
Conclusion
In conclusion, participants with self-reported COPD were more likely to report pain and had an elevated pain intensity compared to participants without self-reported COPD, and the pain was primarily located in the limbs, thorax, and lower back. After adjustment for age and gender, COPD was associated with an elevated pain intensity. Sleep disturbance and multimorbidity had the most pronounced impact on pain intensity in the multiple linear regression model. In participants with COPD, increased pain intensity was associated with reduced odds of being active physically. This study suggests that the treatment of COPD should include a routine overall assessment of pain, sleep quality, and comorbidities and subsequent a pain management plan in the rehabilitation programs to improve the odds of an increased level of physical activity.
Disclosure
The authors report no conflict of interest in this work.
References
1. Global Initiative for Chronic Obstructive Lung Disease I. Global strategy for the diagnosis, management, and prevention of Chronis Obstructive Pulmonary Disease; 2020. Available from: https://goldcopd.org/wp-content/uploads/2019/11/GOLD-2020-REPORT-ver1.0wms.pdf.
2. Rabe F, Klaus WH. Chronic obstructive pulmonary disease. Lancet. 2017;389(10082):1931–1940. doi:10.1016/S0140-6736(17)31222-9
3. Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2095–2128. doi:10.1016/S0140-6736(12)61728-0
4. Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2163–2196. doi:10.1016/S0140-6736(12)61729-2
5. Mannino DM, Buist AS. Global burden of COPD: risk factors, prevalence, and future trends. Lancet (London, England). 2007;370(9589):765–773. doi:10.1016/S0140-6736(07)61380-4
6. HajGhanbari B, Holsti L, Road JD, Darlene Reid W. Pain in people with chronic obstructive pulmonary disease (COPD). Respir Med. 2012;106(7):998–1005. doi:10.1016/j.rmed.2012.03.004
7. Bentsen SB, Rustøen T, Miaskowski C. Prevalence and characteristics of pain in patients with chronic obstructive pulmonary disease compared to the Norwegian general population. J Pain. 2011;12(5):539–545. doi:10.1016/j.jpain.2010.10.014
8. Lewthwaite H, Williams G, Baldock KL, Williams MT. Systematic review of pain in clinical practice guidelines for management of COPD: a case for including chronic pain? Healthc (Basel, Switzerland). 2019;7(1).
9. Lee AL, Goldstein RS, Brooks D. Chronic pain in people with Chronic Obstructive Pulmonary Disease: prevalence, clinical and psychological implications. Chronic Obstr Pulm Dis (Miami, Fla). 2017;4(3):194–203.
10. Puhan M, Gimeno-Santos E, Scharplatz M, Troosters T, Walters E, Steurer J. Pulmonary rehabilitation following exacerbations of chronic obstructive pulmonary disease (Review). Cochrane Database Syst Rev. 2011;Art.No.: C(10):CD005305.
11. Bentsen SB, Rustøen T, Miaskowski C. Differences in subjective and objective respiratory parameters in patients with chronic obstructive pulmonary disease with and without pain. Int J Chron Obstruct Pulmon Dis. 2012;7:137–143. doi:10.2147/COPD.S28994
12. Bordoni B, Marelli F, Morabito B, Sacconi B, Caiazzo P, Castagna R. Low back pain and gastroesophageal reflux in patients with COPD: the disease in the breath. Int J Chron Obstruct Pulmon Dis. 2018;13:325–334. doi:10.2147/COPD.S150401
13. Bordoni B, Marelli F, Morabito B, Castagna R. Chest pain in patients with COPD: the fascia’s subtle silence. Int J Chron Obstruct Pulmon Dis. 2018;13:1157–1165. doi:10.2147/COPD.S156729
14. Heneghan N, Adab P, Jackman S, Balanos G. Musculoskeletal dysfunction in chronic obstructive pulmonary disease (COPD): an observational study. Int J Ther Rehabil. 2015;22(3):119–128. doi:10.12968/ijtr.2015.22.3.119
15. Van Remoortel H, Hornikx M, Demeyer H, et al. Daily physical activity in subjects with newly diagnosed COPD. Thorax. 2013;68(10):962–963.
16. Garcia-Aymerich J, Lange P, Benet M, Schnohr P, Antó JM. Regular physical activity reduces hospital admission and mortality in chronic obstructive pulmonary disease: a population based cohort study. Thorax. 2006;61(9):772–778. doi:10.1136/thx.2006.060145
17. Louvaris Z, Kortianou EA, Spetsioti S, et al. Intensity of daily physical activity is associated with central hemodynamic and leg muscle oxygen availability in COPD. J Appl Physiol. 2013;115(6):794–802. doi:10.1152/japplphysiol.00379.2013
18. Kortianou EA, Aliverti A, Louvaris Z, et al. Limitation in tidal volume expansion partially determines the intensity of physical activity in COPD. J Appl Physiol. 2015;118(1):107–114. doi:10.1152/japplphysiol.00301.2014
19. HajGhanbari B, Garland SJ, Road JD, Reid WD. Pain and physical performance in people with COPD. Respir Med. 2013;107(11):1692–1699. doi:10.1016/j.rmed.2013.06.010
20. Jensen HAR, Ekholm O, Davidsen M, Christensen AI. The Danish health and morbidity surveys: study design and participant characteristics. BMC Med Res Methodol. 2019;19(1).
21. Pedersen CB. The Danish civil registration system. Scand J Public Health. 2011;39(7 S).
22. Thong ISK, Jensen MP, Miró J, Tan G. The validity of pain intensity measures: what do the NRS, VAS, VRS, and FPS-R measure? Scand J Pain. 2018;18(1):99–107. doi:10.1515/sjpain-2018-0012
23. Saltin B, Grimby G. Physiological analysis of middle-aged and old former athletes. Comparison with still active athletes of the same ages. Circulation. 1968;38(6):1104–1115. doi:10.1161/01.CIR.38.6.1104
24. Grimby G, Börjesson M, Jonsdottir IH, Schnohr P, Thelle DS, Saltin B. The “Saltin-Grimby physical activity level scale” and its application to health research. Scand J Med Sci Sport. 2015;25:119–125.
25. Chen Y-W, Coxson HO, Coupal TM, et al. The contribution of thoracic vertebral deformity and arthropathy to trunk pain in patients with chronic obstructive pulmonary disease (COPD). Respir Med. 2018;137:115–122. doi:10.1016/j.rmed.2018.03.007
26. Lee AL, Goldstein RS, Chan C, Rhim M, Zabjek K, Brooks D. Postural deviations in individuals with chronic obstructive pulmonary disease (COPD). Can J Respir Crit Care Sleep Med. 2018;2(2):
27. Nasser SA, Afify EA. Sex differences in pain and opioid mediated antinociception: modulatory role of gonadal hormones. Life Sci. 2019;237:116926. doi:10.1016/j.lfs.2019.116926
28. Borge CR, Wahl AK, Moum T. Pain and quality of life with chronic obstructive pulmonary disease. Heart Lung. 2011;40(3):e90–101. doi:10.1016/j.hrtlng.2010.10.009
29. Cheatle MD, Foster S, Pinkett A, Lesneski M, Qu D, Dhingra L. Assessing and managing sleep disturbance in patients with chronic pain. Sleep Med Clin. 2016;11(4):531–541. doi:10.1016/j.jsmc.2016.08.004
30. Pedersen BK, Saltin B. Exercise as medicine - Evidence for prescribing exercise as therapy in 26 different chronic diseases. Scand J Med Sci Sport. 2015;25:1–72.
31. Flachs EM, Eriksen L, Koch MB, et al. Sygdomsbyrden I Danmark: Sygdomme. Sundhedsstyrelsen; 2015.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

