Back to Journals » Journal of Multidisciplinary Healthcare » Volume 7
Overview of guidelines for the prevention and treatment of venous leg ulcers: a US perspective
Authors White-Chu EF, Conner-Kerr T
Received 26 September 2013
Accepted for publication 27 November 2013
Published 11 February 2014 Volume 2014:7 Pages 111—117
DOI https://doi.org/10.2147/JMDH.S38616
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
E Foy White-Chu,1 Teresa A Conner-Kerr2
1Oregon Health and Science University, Portland VA Medical Center, Portland, OR, 2Winston-Salem State University, Department of Physical Therapy, Winston Salem, NC, USA
Abstract: Comprehensive care of chronic venous insufficiency and associated ulcers requires a multipronged and interprofessional approach to care. A comprehensive treatment approach includes exercise, nutritional assessment, compression therapy, vascular reconstruction, and advanced treatment modalities. National guidelines, meta-analyses, and original research studies provide evidence for the inclusion of these approaches in the patient plan of care. The purpose of this paper is to review present guidelines for prevention and treatment of venous leg ulcers as followed in the US. The paper further explores evidence-based yet pragmatic tools for the interprofessional team to use in the management of this complex disorder.
Keywords: venous insufficiency, varicose ulcer, wound, compression bandages
Introduction
Care for patients with chronic wounds, and venous leg ulcers in particular, requires an interprofessional approach due to their etiologic complexity, common recurrence, and sometimes devastating morbidity. Interprofessional care is “care delivered by intentionally created, usually relatively small work groups in health care, who are recognized by others as well as by themselves as having a collective identity and shared responsibility for a patient or group of patients”.1 A Canadian study reviewed the utility of interprofessional teams caring for patients with chronic wounds, including venous leg ulcers, and found improvement in healing rates, pain management, and management of day-to-day dressing issues.2
In the US, patients who present with nonhealing venous leg ulcers are typically referred to a wound care specialist. This specialist may be a certified wound care nurse, physical therapist, surgeon, podiatrist, or internist. Physical and occupational therapists certified in manual lymphatic drainage techniques also contribute to the care of patients with chronic lower extremity swelling associated with venous insufficiency. As many providers recognize the complex needs of these patients, there has been an increasing trend towards utilizing interprofessional teams in chronic wound care.
The purpose of this paper is to review the guidelines for prevention and treatment of venous leg ulcers as followed in the US. Much of the information is gleaned from the guidelines published by the Association of Advanced Wound Care (AAWC) in 2012 and the Wound Healing Society (WHS) in 2006 (with an update in 2012).3–5 This paper further explores evidence-based, yet pragmatic, tools for the interprofessional team to use in the management of this complex disorder. The level of evidence is outlined in Table 1.
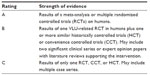  | Table 1 Level of evidence |
Epidemiology
Approximately 1% of the western population suffers from venous leg ulcers.6 One longitudinal study conducted in one US city in 1973 estimated the prevalence of venous leg ulcer to be 400,000–500,000 by extrapolating their data to 1970 US Census data.7 Another study estimated the prevalence of venous leg ulcers in patients over the age of 65 years to be 1.75 million by extrapolating Swedish point prevalence data gleaned from a patient survey. There are no sources of epidemiologic studies for incidence and prevalence specifically in the US.8 These data are sorely needed for us to be able to assess the need for appropriate resources.
One New Mexico study in 2000, looked at Medicaid fee-for-service in patients with wounds and determined that US$19,000–US$24,000 was spent annually on each patient with a venous leg ulcer.9 Another study from 1999 evaluated a retrospective cohort of 78 patients and found that an average total cost of US$9,685 (median US$3,036) was spent per patient. This cost included total outpatient costs (facility and physician), hospitalizations (if any), dressing supplies, medications (topical and oral), and home health care (if any).10 Outside of medical costs are quality of life costs. Patients often suffer from decreased mobility, leading to loss of work, challenges with finances, and isolation.11 Recent and consistent findings have determined that the most common concern for patients is pain, especially surrounding dressing changes. Pain further complicates mobility, sleep, and overall sense of well-being.12
Many studies suggest a higher prevalence of chronic venous insufficiency and venous leg ulcers in women, but this disparity decreases with age.13 Common risk factors for chronic venous insufficiency include family history, multiparity, obesity, and a history of deep venous thrombosis or thrombophlebitis.
Exercise
Anything that alters the calf pump muscle function greatly increases the risk of chronic venous insufficiency, which then increases the risk of formation of a venous leg ulcer. The exact mechanism is not completely understood, but multiple theories have been put forward.13
The calf muscle pump has been referred to as the “peripheral heart” because of its role in promoting venous return from the lower extremities.14 The literature describes three components of the calf muscle pump, ie, competent venous valves, functional gastrocnemius and soleus muscles, and unsheathing fascia and skin surrounding the calf muscles. It is thought that the calf muscle pump works much like a “bellows pump” during locomotion and other lower extremity exercises. The gastrocnemius-soleus muscles, contained within a limiting fascia, contract and thereby compress the deep veins and propel venous blood toward the heart through this system. As a result of the increased pressure on the external walls of the deep veins by contraction of the musculature, the blood column is forced upwards through open valve leaflets that are pushed against the walls of the veins. During relaxation periods when the calf muscles are not actively contracting, the hydrostatic pressure of the deep veins drops to near zero and the veins refill. Reflux of the venous column in healthy individuals is prevented by valve closure. At the same time, the negative pressure that results from venous emptying draws fresh arterial blood into skeletal muscle.
Research has shown that the deficiency of the calf muscle pump is significant with regard to the severity of venous ulceration.15 Active ulcerations are associated with greater impairment of the calf-muscle pump. Individuals with impaired calf muscle pumps have significantly lower ejection volumes and fractions. Links have been established between reduced range of motion at the ankle in individuals with venous insufficiency and severity of the disease.16 Impaired calf muscle pump function in the lower limbs is also associated with venous leg ulcers.17
Exercise has been shown to moderate the detrimental effects associated with impaired calf muscle function. A structured program of calf muscle exercise may improve hemodynamic performance and prevent ulcer recurrence (level C).18 Study findings included implementation of walking programs as a practical suggestion for patients who are ambulatory. Higher levels of muscle activity and greater muscle mass may enhance venous emptying in the calf.19 A physical therapist can be instrumental in helping the patient adhere to this care plan.
Nutrition
Multiples studies have found that patients with venous leg ulcers tend to be overweight or obese but nutritionally deficient,20–22 and do not meet their dietary needs for wound healing. There is suboptimal intake of protein, vitamins (especially vitamins C and A), and zinc. It remains to be seen, however, whether these nutritional deficiencies result in delayed wound healing. Older studies (in the 1960s and 1970s) evaluating zinc deficiency suggested that patients with low zinc levels may have faster healing with zinc supplementation.22 Larger trials are warranted before a firm recommendation can be made with regard to zinc or other nutritional supplementation. Guidelines often reference nutritional studies for chronic wounds, but no specific studies with regard to venous leg ulcers. The guidelines still recommend a nutritional assessment when caring for patients with venous leg ulcers (level B).3 Weight loss can be advantageous, given that larger patients tend to be less mobile. This immobility further worsens calf pump muscle function and venous hypertension.22,23 Because these individuals are nutritionally deficient, a weight loss plan, as advised by a dietitian, can assist the patient in losing weight and improving deficiencies.
Compression bandaging
Guidelines identify compression bandaging as a central component of the standard of care for individuals with a venous leg ulcer (level A). A 2012 Cochrane review found that venous leg ulcers heal more quickly when treated with compression therapy.6 The AAWC guidelines state that multicomponent, sustained, high compression elastic bandages are more effective than inelastic compression and single component compression systems (Table 2).3 The 2006 WHS guidelines do not distinguish effectiveness between types of compression modalities, but an update in 2012 references an updated Cochrane review that also found increased wound healing with multicomponent systems.4–6 Guidelines also support the use of short stretch compression bandages commonly used by manual lymphatic therapists, the Duke boot (ie, Unna’s boot combined with elastic compression and a hydrocolloid primary dressing), and the Unna’s boot to enhance healing of venous ulcerations.3–5
  | Table 2 Compression comparisons |
Some compression is better than none.6 The team may need to coach the patient through accepting compression bandaging and also lifelong compression garments once the wounds have healed. Efforts to address pain control, bulkiness in the context of footwear, and other patient-centered concerns are important to ally the patient with the treatment plan. After the wound has healed, there is ample evidence to support lifelong use of compression stockings to minimize ulcer recurrence.24–27
Manual lymphatic drainage
Manual lymphatic drainage, ie, use of manual tissue mobilization and stretch, promotes the return of tissue fluid and accompanying protein to the lymph vascular system. The AAWC guidelines support the use of this technique to reduce tissue swelling in individuals with chronic venous insufficiency (level A), but there are no data linking manual lymphatic drainage to wound healing.28 The WHC guidelines do not reference this form of therapy.3–5
Manual lymphatic drainage was shown to decrease foot volumetry and to enhance surgical outcomes as well as quality of life. Studies suggest that chronic venous insufficiency treated with complete decongestive physiotherapy (skin care, manual lymphatic drainage, compression bandaging, and exercise) significantly reduces the volume and percentage of limb swelling and pain associated with this condition.29 Manual lymphatic drainage has been shown to be effective in reducing limb circumference in individuals with edema due to chronic insufficiency in as little as 3 weeks.30,31 The efficacy of manual lymphatic drainage is due in large part to enhancing venous blood flow, most likely in the femoral and great saphenous veins.
In the US, manual lymphatic drainage is provided by physical or occupational therapists in an outpatient rehabilitative setting. Many of these patients with significant chronic venous insufficiency and even lymphedema have mobility issues, so attending an outpatient setting can be challenging. These patients often need a visiting nursing agency to provide dressing changes at home. Because Medicare will only fund in-home care if the patient is “home-bound”, asking the patient to come three times a week to outpatient rehabilitation for manual lymphatic drainage will cancel their in-home care. The patient simply cannot receive both. It is the experience of the authors that wound care specialists can consider training the manual lymphatic drainage therapist in basic dressing changes and wound care, with close supervision, so that the patient can access necessary outpatient manual lymphatic drainage and wound care.
Intermittent pneumatic compression therapy
Intermittent pneumatic compression pumps have been used in the management of chronic venous insufficiency for well over 60 years.32 Initially, this therapy was targeted at the prevention of deep vein thrombosis and pulmonary embolism. Today, intermittent pneumatic compression is also used to reduce tissue swelling by mobilizing excess tissue fluid.
The effects of intermittent pneumatic compression on wound healing and limb volume reduction have been studied in individuals with chronic venous insufficiency. Studies support the use of compression pumps to alleviate symptoms of chronic venous insufficiency and assist with the healing of longstanding chronic ulcerations.33 Sequential gradient compression therapy enhances venous ulcer healing and provides an alternative for patients who cannot tolerate sustained compression.34 The AAWC recognizes that intermittent pneumatic compression heals venous leg ulcers better than no compression (level A).3 The 2006 WHS guidelines (with support in a 2012 update) go further to state that intermittent pneumatic compression can be used with or without compression in patients who are unable to wear an adequate compression system (level A).4,5
Support for the use of intermittent pneumatic compression is important, given the difficulty that caregivers, morbidly obese individuals, and older adults with decreased finger strength and dexterity have in applying daily compression bandaging or definitive compression garments (post volume reduction therapy with compression bandages). However, intermittent pneumatic compression can result in significant collection of tissue fluid and secondary edema in areas immediately adjacent to the edge of the sleeve opening and care must be taken to ensure appropriate device use.
Vascular surgery
Vascular surgery options to correct vein function have increased in the last decade, bringing with them advanced adjunctive therapies for preventing venous leg ulcer recurrence (level A). Fortunately, these newer options carry less long-term morbidity. To be considered for surgical intervention, patients must have an intact deep venous system. The most common vascular surgical options utilized in the US, along with their indications and risks, are outlined in Table 3. Although subfascial endoscopic perforator vein surgery (SEPS) remains the treatment of choice for perforator vein pathology, it is the authors’ experience that thermal and radiofrequency ablations are starting to replace subfascial endoscopic perforator vein surgery and open procedures.
  | Table 3 Interventions for venous hypertension |
Sclerotherapy appears to be promising for actual venous leg ulcer healing; it is also becoming more common, despite the need for more evidence to support its use.35 Options with level C evidence for wound healing include sclerotherapy with compression, venous valve repair or reconstruction, and stenting of the iliac vein with combined iliac vein obstruction and deep reflux.36–40 All options complement but do not replace evidenced-based compression, and wound, skin, and patient care.
Skin replacement
Innovations in skin replacement and grafting options have also increased in the last decade. Bilayered bioengineered skin (Apligraf®, Organogenesis Inc., Canton, MA, USA) has the strongest evidence to support its use in refractory venous leg ulcer healing (level A). One multicenter study found that longstanding wounds (present for more than a year) healed better with bilayered bioengineered skin replacement over compression alone (63% versus 48.8%, P=0.02). Skin replacement was used with an Unna’s boot and a Coban wrap in this study.41 A Cochrane review of 17 randomized controlled trials also found that artificial skin replacement healed venous leg ulcers better than standard dressings.42
There is not enough evidence to support other forms of skin replacements (eg, split thickness autografts and cultured keratinocyte grafts).42 Despite this, there is conflict between the AAWC and WHS guidelines with respect to using other form of skin replacement. The AAWC recognizes level B and C evidence to support the use of cultured epidermal autografts and allografts, respectively.3 The WHS guidelines state that there is level A evidence showing that these treatments do not improve healing of venous leg ulcers.4 However, the WHS update in 2012 found three studies that contradict this statement.5 Similar to vascular interventions, skin replacements should not be utilized without compression, or wound, skin, and patient care.
The AAWC guidelines encourage providers to consider skin replacement when there is no improvement in a venous leg ulcer after 30 days.3 In the US, insurance policies may place restrictions on when and how often these skin replacements can be used. Therefore, the provider must balance insurance restrictions with progress of healing.
Biophysical interventions
Biophysical interventions are often implemented by physical or occupational therapists. Evidence supports the use of biophysical energy from electromagnetic and acoustic energy spectra for stimulation of wound healing. Numerous small studies were reviewed, with positive effects on venous ulcer healing. Both high frequency (≥1 mHz; periwound application) ultrasound and low frequency (<1 mHz; water bath or noncontact application) ultrasound have been shown to enhance venous ulcer healing.43–48 A 2010 Cochrane review found weak evidence to support high-frequency ultrasound and no evidence to support low-frequency ultrasound.49 The 2006 WHS guidelines state that there is level 1 evidence showing no benefit from ultrasound in venous leg ulcer healing.4 However, the update in 2012 found three studies to refute this claim.5 The 2012 AAWC guidelines found level A evidence in support of this treatment.3
The evidence supports use of electrical stimulation in treating venous ulcers (level A). High-voltage pulsed current or pulse-controlled electrical stimulation applied to chronic leg ulcers may reduce the wound surface area over a 4-week treatment period to approximately one half the initial wound size.50,51 Some effects have also been seen in terms of improved long-lasting pain control and increased perfusion.
Two small studies indicate a potential benefit of electromagnetic therapy for the facilitation of venous ulcer healing.52 However, study design flaws may have produced an overestimation of the beneficial effects.
Several trials provide limited evidence to suggest a positive effect of phototherapy on venous ulcer healing. In a small placebo-controlled, double-blind study, low-energy photon therapy was found to increase healing of venous leg ulcers compared with a sham device.53 A second small, placebo-controlled, double-blind study using broadband (400–800 nm) visible light produced similar effects in leg ulcer patients. Ninety percent of patients receiving active light therapy achieved wound closure compared with 33% in a sham group.54 In a small underpowered, prospective study, venous ulcers treated with infrared and visible light showed improved wound healing and reduced pain.55
The AAWC guidelines gave phototherapy a level C evidence rating with regard to wound healing.3 The WHS guidelines also stated that there is no evidence to support the use of phototherapy for venous leg ulcer wound healing (level A).4 The 2012 update of the WHS guidelines found one randomized controlled trial supporting the use of phototherapy.5,56
Other biophysical agents, such as negative pressure wound therapy and whirlpool and hyperbaric oxygen therapy were assigned level C evidence for wound healing by the AAWC and WHS guidelines.3–5 Preparation of the wound bed for graft placement by negative pressure wound therapy also received a level C rating.
Summary
Comprehensive care of chronic venous insufficiency and the venous leg ulcers commonly associated with this disease requires a multipronged and interprofessional approach to care. A comprehensive treatment approach includes exercise, nutritional assessment, compression therapy, vascular reconstruction, and advanced treatment modalities. For the most part, US guidelines are consistent in their assessment of the strength of evidence for each of these modalities. The guidelines may be helpful in pulling together team members to help serve patients with challenging venous leg ulcers.
Disclosure
The authors report no conflicts of interest in this work.
References
Interprofessional Education Collaborative Expert Panel. Core competencies for interprofessional collaborative practice: report of an expert panel. Washington, DC, USA: Interprofessional Education Collaborative; 2011. Available from: http://www.aacn.nche.edu/education-resources/ipecreport.pdf. Accessed November 28, 2013. | |
Health Quality Ontario. Community-based care for chronic wound management: an evidence-based analysis. Ont Health Technol Assess Ser. 2009;9:1–24. | |
Association for the Advancement of Wound Care Venous Ulcer Guideline. Malvern, PA, USA: Association for the Advancement of Wound Care; 2012. Available from: http://aawconline.org/wp-content/uploads/2012/03/AAWC-Venous-Ulcer-Guideline-Update+Algorithm-v28.pdf. Accessed November 27, 2013. | |
Robson MC, Cooper DM, Aslam R. Guidelines for the treatment of venous ulcers. Wound Repair Regen. 2006;14:649–662. | |
Tang JC, Marston WA, Kirsner RS. Wound Healing Society venous ulcer treatment guidelines: what’s new in five years? Wound Repair Regen. 2012;20:619–637. | |
O’Meara S, Cullum NA, Nelson EA, et al. Compression for venous leg ulcers. Cochrane Database Syst Rev. 2012;11:CD000265. | |
Coon WW, Willis PW, Keller JB. Venous thromboembolism and other venous disease in the Tecumseh Community Health Study. Circulation. 1973;48:839–846. | |
Nelzen O, Bergqvist D, Lindhagen A. The prevalence of chronic lower-limb ulceration has been underestimated: results of a validated population questionnaire. Br J Surg. 1996;83:255–258. | |
Kumar RN, Gupchup GV, Dodd MA, et al. Direct health care costs of 4 common skin ulcers in New Mexico Medicaid fee-for-service patients. Adv Skin Wound Care. 2004;17:143–149. | |
Olin JW, Beusterien KM, Childs MB, Seavey C, McHugh L, Griffiths RI. Medical costs of treating venous stasis ulcers: evidence from a retrospective cohort study. Vasc Med. 1999;4:1–7. | |
Phillips T, Stanton B, Provan A, Lew R. A study of the impact of leg ulcers on quality of life; financial, social, and psychologic implications. J Am Acad Dermatol. 1994;31:49–53. | |
Green J, Jester R. Health-related quality of life and chronic venous leg ulceration: part 1. Br J Community Nurs. 2009;14:S12, S14, S16–S17. | |
Abbade LP, Lastoria S. Venous ulcer: epidemiology, physiopathology, diagnosis, and treatment. Int J Dermatol. 2005;44:449–456. | |
Meissner MH. Lower extremity venous anatomy. Semin Intervent Radiol. 2005;22:147–156. | |
Araki CT, Back TL, Padberg FT, et al. The significance of calf muscle pump function in venous ulceration. J Vasc Surg. 1994;20:872–879. | |
Dix FP, Brooke R, McCollum CN. Venous disease is associated with an impaired range of ankle movement. Eur J Vasc Endovasc Surg 2003;25:556–561. | |
Back TL, Padberg FT Jr, Araki CT, Thompson PN, Hobson RW 2nd. Limited range of motion is a significant factor in venous ulceration. J Vasc Surg. 1995;22:519–523. | |
Padberg FT Jr, Johnston MV, Sisto SA. Structured exercise improves calf muscle pump function in chronic venous insufficiency: a randomized trial. J Vasc Surg. 2004;39:79–87. | |
Kugler C, Strunk M, Rudofsky G. Venous pressure dynamics of the healthy human leg. Role of muscle activity, joint mobility and anthropometric factors. J Vasc Res. 2001;38:20–29. | |
Tobon J, Whitney JD, Jarrett M. Nutritional status and wound severity of overweight and obese patients with venous leg ulcers: a pilot study. J Vasc Nurs. 2008;26:43–52. | |
Szewczyk MT, Jawien A, Kedziora-Kornatowska K, et al. The nutritional status of older adults with and without venous ulcers: a comparative, descriptive study. Ostomy Wound Manage. 2008;54:34–36. | |
Heinen MM, van Achterberg T, op Reimer WS, van de Kerkhof PC, de Laat E. Venous leg ulcer patients: a review of the literature on lifestyle and pain management interventions. J Clin Nurs. 2004;13:355–366. | |
Yang D, Vandongen YK, Stacey MC. Effect of exercise on calf muscle pump function in patients with chronic venous disease. Br J Surg. 1999;86:338–341. | |
Korn P, Patel ST, Heller JA, et al. Why insurers should reimburse for compression stockings in patients with chronic venous stasis? J Vasc Surg. 2002;35:1–8. | |
Milic DJ, Zivic SS, Bogdanovic DC, et al. A randomized trial of the Tubulcus multilayer bandaging system in the treatment of extensive venous ulcers. J Vasc Surg. 2007;46:750–755. | |
Nelson EA, Jones J. Venous leg ulcers. Clin Evid (Online). 2008; 2008;pii:1902 | |
Moneta GL, Partsch H. Compression therapy for venous ulceration. In: Gloviczki P, editor. Handbook of Venous Disorders. 3rd ed. London, UK: Hodder Arnold; 2009. | |
Molski P, Kruczyński J, Molski A, Molski S. Manual lymphatic drainage improves the quality of life in patients with chronic venous disease: a randomized control trial. Arch Med Sci. 2013;9:452–458. | |
Bakar Y, Ozturk A, Calisal M, Erturk K, Daglar B. Complete decongestive physiotherapy for older people with chronic venous insufficiency. Topics in Geriatric Rehabilitation. 2010;26:164–170. | |
Szewczyk T. The importance of massage in edema of lower limbs in patients with chronic venous insufficiency. Valetudinaria Post py Medycyny Klinicznej i Wojskowej. 2002;7:50–53. | |
Crisóstomo RS, Candeias MS, Ribeiro AM, Martins CD, Armada-da-Silva PA. Manual lymphatic drainage in chronic venous disease: a duplex ultrasound study. Phlebology. August 29, 2013. [Epub ahead of print.] | |
Kelly DG. A Primer on Lymphedema. Upper Saddle River, NJ, USA: Prentice Hall; 2002. | |
Berliner E, Ozbilgin B, Zarin DA. A systematic review of pneumatic compression for treatment of chronic venous insufficiency and venous ulcers. J Vasc Surg. 2003;37:539–544. | |
Smith PC, Sarin S, Hasty J, Scurr JH. Sequential gradient pneumatic compression enhances venous ulcer healing: a randomized trial. Surgery. 1990;108:871–875. | |
Gloviczki P, Gloviczki ML. Evidence on efficacy of treatments of venous ulcers and on prevention of ulcer recurrence. Perspect Vasc Surg Endovasc Ther. 2009;21:259–268. | |
Hamel-Desnos CM, Guias BJ, Desnos PR, et al. Foam sclerotherapy of the saphenous veins: randomised controlled trial with or without compression. Eur J Vasc Endovasc Surg. 2010;39:500–507. | |
O’Hare JL, Earnshaw JJ. Randomised clinical trial of foam sclerotherapy for patients with a venous leg ulcer. Eur J Vasc Endovasc Surg. 2010;39:495–499. | |
Perrin M, Hiltbrand B, Bayon JM. Results of valvuloplasty in patients presenting deep venous insufficiency and recurring ulceration. Ann Vasc Surg. 1999;13:524–532. | |
Maleti O, Lugli M. Neovalve construction in postthrombotic syndrome. J Vasc Surg. 2006;43:794–799. | |
Raju S, Fredericks R. Valve reconstruction procedures for non-obstructive venous insufficiency: rationale technique and results in 107 procedures with two to eight year follow up. J Vasc Surg. 1988;7:301–309. | |
Falanga V, Margolis D, Alvarez O, et al. Rapid healing of venous ulcers and lack of clinical rejection with an allogeneic cultured human skin equivalent. Arch Dermatol. 1998;134:293–300. | |
Jones JE, Nelson EA, Al-Hity A. Skin grafting for venous leg ulcers. Cochrane Database Syst Rev. 2013;1:CD001737. | |
Weichenthal M, Mohr P, Stegmann W, et al. Low-frequency ultrasound treatment of chronic venous ulcers. Wound Repair Regen. 1997;5:18–22. | |
Peschen M, Weichenthal M, Schöpf E, et al. Low-frequency ultrasound treatment of chronic venous leg ulcers in an outpatient therapy. Acta Derm Venereol. 1997;77:311–314. | |
Kavros SJ, Schenck EC. Use of noncontact low-frequency ultrasound in the treatment of chronic foot and leg ulcerations: a 51-patient analysis. J Am Podiatr Med Assoc. 2007;97:95–101. | |
Kavros SJ, Liedl DA, Boon AJ, Miller JL, Hobbs JA, Andrews KL. Expedited wound healing with noncontact, low-frequency ultrasound therapy in chronic wounds: a retrospective analysis. Adv Skin Wound Care. 2008;21:416–423. | |
Dyson M, Franks C, Suckling J. Stimulation of healing of varicose ulcers by ultrasound. Ultrasonics. 1976;14:232–236. | |
Roche C, West J. A controlled trial investigating the effect of ultrasound on venous ulcers referred from general practitioners. Physiotherapy. 1984;70:475–477. | |
Cullum N, Al-Kurdi D, Bell-Syer SEM. Therapeutic ultrasound for venous leg ulcers. Cochrane Database Syst Rev. 2010;6:CD001180. | |
Houghton PE, Kincaid CB, Lovell M, et al. Effect of electrical stimulation on chronic leg ulcer size and appearance. Phys Ther. 2003;83:17–28. | |
Jünger M, Arnold A, Zuder D, Stahl HW, Heising S. Local therapy and treatment costs of chronic, venous leg ulcers with electrical stimulation (Dermapulse®): a prospective, placebo controlled, double blind trial. Wound Repair Regen. 2008;16:480–487. | |
Aziz Z, Cullum N, Flemming K. Electromagnetic therapy for treating venous leg ulcers. Cochrane Database Syst Rev. 2013;2:CD002933. | |
Gupta AK, Filonenko N, Salansky N, Sauder DN. The use of low energy photon therapy (LEPT) in venous leg ulcers: a double-blind, placebo-controlled study. Dermatol Surg. 1998;24:1383–1386. | |
Landau Z, Migdal M, Lipovsky A, Lubart R. Visible light-induced healing of diabetic or venous foot ulcers: a placebo-controlled double-blind study. Photomed Laser Surg. 2011;29:399–404. | |
Mercer JB, Nielsen SP, Hoffmann G. Improvement of wound healing by water-filtered infrared-A (wIRA) in patients with chronic venous stasis ulcers of the lower legs including evaluation using infrared thermography. Ger Med Sci. 2008;6:Doc11. | |
Caetano KS, Frade MA, Minatel DG, et al. Phototherapy improves healing of chronic venous ulcers. Photomed Laser Surg. 2009;27:111–118. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
