Back to Archived Journals » Robotic Surgery: Research and Reviews » Volume 2
Outcomes of robot-assisted laparoscopic radical prostatectomy in high-risk prostate cancer patients: experience in 34 patients with oncologic and functional outcomes
Authors Canda AE , Atmaca AF, Cakici OU, Gok B, Arslan ME, Altinova S, Akbulut Z, Balbay MD
Received 10 November 2014
Accepted for publication 9 January 2015
Published 16 February 2015 Volume 2015:2 Pages 29—35
DOI https://doi.org/10.2147/RSRR.S66645
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Masoud Azodi
Supplementary video showing surgical differences comparatively in high-risk versus low-risk disease during performing of RARP
Views: 793
Abdullah Erdem Canda,1 Ali Fuat Atmaca,1 Ozer Ural Cakici,2 Bahri Gok,2 Muhammed Ersagun Arslan,2 Serkan Altinova,2 Ziya Akbulut,1 Mevlana Derya Balbay3
1School of Medicine, Yildirim Beyazit University, Department of Urology, Ankara Ataturk Training and Research Hospital, Ankara, Turkey; 2Department of Urology, Ankara Ataturk Training and Research Hospital, Ankara, Turkey; 3Department of Urology, Memorial Sisli Hospital, Istanbul, Turkey
Introduction: In this retrospective study, we report outcomes of robot-assisted laparoscopic radical prostatectomy (RARP) in high-risk prostate cancer (HRPC) classified according to a D'Amico risk group with minimum 1-year follow-up.
Methods: A total of 34 patients who had at least one preoperative HRPC feature and who underwent RARP were included. Mean patient age and preoperative serum prostate-specific antigen levels were 62.6±6.4 years and 12.2±9.1 ng/mL, respectively. Preoperatively, two (5.8%), one (2.9%), eleven (32.3%), three (8.8%), and 17 (50%) patients had prostate biopsy Gleason scores of 5+4, 4+5, 4+4, 3+5, and <8, respectively. Bilateral neurovascular bundle (NVB)-sparing, unilateral NVB-sparing, and non-NVB-sparing surgery was performed in 16 (47%), five (15%), and 13 (38%) patients, respectively.
Results: Mean console time, intraoperative blood loss, duration of hospital stay, and urethral catheter removal time were 162.1±64.4 minutes, 232.2±255.1 cc, 4.1±2.1 days, and 12.6±6.2 days, respectively. During the perioperative period (0–30 days), three minor and five major complications occurred as classified using the modified Clavien classification. No complication was detected during postoperative 31–90 days. Postoperative pathologic stages included pT0, pT2a, pT2b, pT2c, pT3a, and pT3b disease in two (5.8%), five (14.7%), three (8.8%), six (17.6%), ten (29.4%), and eight (23.5%) patients, respectively. Positive surgical margin rate was 32.3%. Mean lymph node yield was 11.8±8.3 (range three to 37). Mean follow-up was 27.8±11.1 months. Biochemical recurrence was detected in nine (26.4%) patients. Of the patients, 17 (50%) were fully continent (zero pads/day), six (17.7%) wore a safety pad/day, six (17.7%) wore one pad/day, three (8.8%) wore two pads/day, and two (5.9%) wore more than two pads/day. Of the 24 patients with no preoperative erectile dysfunction, 15 (44.1%) had no erectile dysfunction at a mean follow-up of 1 year. Trifecta and pentafecta rates were 38% and 26%, respectively.
Conclusion: Based on our experience, RARP in HRPC is a safe procedure with satisfactory oncologic and functional outcomes.
Keywords: robotic radical prostatectomy, high-risk prostate cancer, outcomes, minimally invasive surgery, robotic surgery
Introduction
Prostate cancer (PCa) accounts for almost 30% of all newly diagnosed cancers in men in the US and is the second most frequent cause of cancer death in men.1 Almost 20%–30% of patients diagnosed with PCa still have high-risk, nonmetastatic disease.2
D’Amico et al3 proposed a three-group risk stratification system to predict posttreatment biochemical failure following radical prostatectomy (RP) and external-beam radiotherapy. This system classified nonmetastatic PCa into low-, intermediate-, and high-risk PCa according to initial serum prostate-specific antigen (PSA), clinical T-stage, and biopsy Gleason score. High-risk PCa (HRPC) was classified as having any one of the following features: 1992 American Joint Committee on Cancer stage ≥T2c, PSA >20 ng/mL, or Gleason disease score of 8–10.3
Currently, treatment of HRPC that includes a combination of surgery, radiation therapy, and androgen deprivation therapy as a multimodality approach is controversial.4,5
Management of HRPC requires aggressive treatment; otherwise, this disease might progress and cause serious symptoms and complications and eventually patient death.6 The outcomes of the recently published Swedish Registry Study showed that surgery seems to be superior to radiation therapy and longer cancer-specific survival in the surgery group in patients with HRPC as per 15-year cancer-specific survival data7 that suggested a trend toward performing RP in HRPC patients.8
Although open RP is the standard surgical technique in the surgical management of patients with PCa, a robotic approach has become the most common approach in the US.9 However, the number of publications related to the use of robotic surgery in HRPC is very limited.
Herein, we report our experience in RARP and HRPC, including 34 patients classified according to a D’Amico risk group, with minimum 1-year follow-up.
Materials and methods
Between February 2009 and September 2014, we performed almost 600 RARP procedures at our institution. All the data of the patients were recorded prospectively, and this database was used for our study. Of those, 68 patients were classified as having HRPC according to D’Amico risk groups. Of the patients with HRPC, 34 had at least 1-year follow-up who were included in the present retrospective study.
All patients in our series were operated on using a da Vinci Surgical System four-arm surgical robot (Intuitive Surgical, Sunnyvale, CA, USA). Overall, five surgeons performed RARP on HRPC patients (AEC, AFA, SA, ZA, and MDB). We previously reported in detail patient preparation, surgical technique, and postoperative follow-up of patients on whom we performed RARP.10
Pelvic lymph node (LN) dissection was performed in patients who had >5% of LN involvement probability according to Partin’s tables. Mean patient age and preoperative serum PSA were 62.6±6.4 years and 12.2±9.1 ng/mL, respectively.
Biochemical recurrence (BCR) was defined as two consecutive serum prostate-specific antigen (PSA) levels of >0.2 ng/mL.
Statistical analyses were performed with the chi-square test with use of the commercially available software Scientific Package for Social Sciences (SPSS), (version 10.0, Chicago, IL, USA). P-values smaller than 0.05 were considered statistically significant.
Results
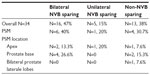
Preoperatively, two (5.8%), one (2.9%), eleven (32.3%), three (8.8%), and 17 (50%) patients had prostate biopsy Gleason scores of 5+4, 4+5, 4+4, 3+5, and <8, respectively. No patient had a Gleason score of 5+5 or 5+3. Bilateral neurovascular bundle (NVB)-sparing, unilateral NVB-sparing, and non-NVB-sparing surgery was performed in 16 (47%), five (15%), and 13 (38%) patients, respectively. Preoperative patient characteristics are presented in Table 1.
Mean console time, intraoperative blood loss, duration of hospital stay, and urethral catheter removal time were 162.1±64.4 minutes, 232.2±255.1 cc, 4.1±2.1 days, and 12.6±6.2 days, respectively. During the perioperative period (0–30 days), three minor (constipation/prolonged ileus [n=1], prolonged anastomotic leakage [n=1], and urinary tract infection [n=1]) and five major complications (intraoperative bladder injury [n=2] and rectum perforation [n=1], which were repaired intraoperatively, intensive care requirement [n=1], and abdominal hematoma [n=1]) occurred according to modified Clavien classification. No complication was detected during the postoperative 31–90 days.
Postoperative pathologic stages included pT0, pT2a, pT2b, pT2c, pT3a, and pT3b disease in two (5.8%), five (14.7%), three (8.8%), six (17.6%), ten (29.4%), and eight (23.5%) patients, respectively. No malignancy was detected in two (5.8%) patients’ pathologic specimens. Positive surgical margin (PSM) rate was 32.3% (2.9% in pT2 and 29.4% in pT3 disease). Of the patients, 17 (50%) underwent extended pelvic LN dissection. Mean LN yield was 11.8±8.3 (range three to 37). One patient had LN metastasis.
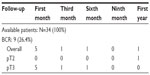
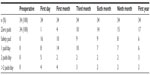
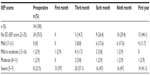
Of the patients, four (11.8%) received adjuvant radiotherapy (ART) alone, four (11.8%) received hormone therapy (HT) alone, and four (11.8%) received ART +HT postoperatively. Mean follow-up was 27.8±11.1 months. BCR was detected in nine (26.4%) patients. Of the 34 patients with 1-year follow-up, 17 (50%) were fully continent (zero pads/day), six (17.7%) wore a safety pad/day, six (17.7%) wore one pad/day, three (8.8%) wore two pads/day, and two (5.9%) wore more than two pads/day. Of the 24 patients with no preoperative erectile dysfunction (ED), 15 (44.1%) had no erectile dysfunction at a mean follow-up of 1-year. Trifecta and pentafecta rates were 38% and 26%, respectively.
Operative parameters (Table 2), NVB sparing and relation to PSM (Table 3), postoperative parameters and complications classified according to modified Clavien–Dindo classification (Table 4), postoperative pathologic outcomes (Table 5), oncologic outcomes (Table 6), postoperative urinary continence outcomes (Table 7), postoperative erectile function outcomes (Table 8), and trifecta and pentafecta outcomes (Table 9) are presented.
  | Table 2 Operative parameters |
  | Table 3 NVB sparing and relation to PSM |
  | Table 4 Postoperative parameters and complications classified according to modified Clavien–Dindo classification |
  | Table 5 Postoperative pathologic outcomes |
  | Table 6 Oncologic outcomes of patients |
  | Table 7 Postoperative urinary continence outcomes of the patients at follow-up |
  | Table 8 Postoperative erectile function outcomes of the patients at follow-up |
Discussion
RARP is increasingly being applied in the surgical management of PCa; however, number of publications related to experience in HRPC is limited. In this paper, we evaluated the outcomes of our RARP experience in 34 HRPC patients.
In our series, mean estimated blood loss was 232.2 cc. Similar to our study, Punnen et al11 reported estimated blood loss as 217 cc in a series of 233 HRPC patients, respectively, who underwent RARP. Mean length of hospital stay was 4.1 days in our series. On the other hand, Punnen et al11 and Gandaglia et al12 reported a shorter length of hospital stay as 1.6 days and 1 day, respectively. In our study, mean length of hospital stay seems to be longer than that in Punnen et al11 and Gandaglia et al.12 We discharge patients when we remove the abdominal drain, generally on postoperative day 3 or 4, and when the patient passes flatus and initiates sufficient oral intake.
In our series, 17 patients (50%) underwent extended pelvic LN dissection and mean LN yield was 11.8. One patient had LN metastasis. We performed bilateral extended pelvic LN dissection in those with an at least 5% risk of pelvic LN involvement by PCa according to Partin’s tables.13 Harty et al14 (n=152), Pierorazio et al15 (n=105), Punnen et al11 (n=233), Busch et al16 (n=110), and Gandaglia et al12 (n=806) performed pelvic LN dissection in 56%, 97.1%, 63%, 94.5%, and 68% of the cases, respectively. Mean LN yield was reported between six and 24.11,15–22
PSM rate was 32.3% (2.9% in pT2 and 29.4% in pT3 disease) in our series. Harty et al14 reported PSM rates as 12% and 79% in pT2 and pT3 disease, respectively. Pierorazio et al15 reported PSM rate as 8.3% in pT2 disease, and Gandaglia et al12 reported PSM rate as 60% in pT2 and pT3a disease. Others reported overall PSM rates between 12% and 48.8%.8,12,14–19 Our PSM rates seem to be similar to those in the published literature. Interestingly, pT0 disease was reported in two patients, and the pathology slides were re-reviewed by the pathology department without any change in the final diagnosis (Table 5).
In our series, at a mean follow-up of 27.8 months, BCR was detected in nine (26.4%) patients. Punnen et al11 reported BCR as 79% at 2-year and 66% at 4-year follow-up. Busch et al16 reported BCR as 41.4% at 3-year follow-up. Of the 34 patients in our study, four (11.8%) received ART alone, four (11.8%) received HT alone, and four (11.8%) received ART+HT postoperatively. Gandaglia et al12 reported that 21.2% of 353 HRPC patients who underwent RARP required additional cancer therapy after surgery. Of those, 15.9% required radiotherapy and 13.9% required androgen deprivation therapy.12 Currently, the mean follow-up time is limited in our series, and the need for additional therapy might change as follow-up increases.
In our series, during the perioperative period (0–30 days), three minor (constipation/prolonged ileus [n=1], prolonged anastomotic leakage [n=1], and urinary tract infection [n=1]) and five major complications (intraoperative bladder injury [n=2] and rectum perforation [n=1], which were repaired intraoperatively, intensive care requirement [n=1], and abdominal hematoma [n=1]) complications occurred according to modified Clavien classification. We experienced rectal injury in one patient in our series, which we repaired intraoperatively. Postoperative follow-up was uneventful for this patient. No complication was detected during postoperative 31–90 days. Other authors reported complication rates between 4% and 30% in HRPC patients who underwent RARP.17,19,21,23 In these studies, lymphocele, ileus, anastomotic leakage, deep vein thrombosis, and rectal injury were among the reported complications. Ham et al19 reported rectal injury rate as 1.7%. Lymphocele formation was reported between 2.5% and 6.6% in other studies.19,21
Urinary continence and erectile function are the functional outcomes following RARP. Currently, the information about functional outcomes following RARP in HRPC patients is limited in the literature. Yuh et al24 reported 1-year urinary continence (zero to one safety pads/day) rate between 78% and 95% and erectile function recovery rate between 52% and 60%. Yee et al25 reported their 1-year pad-free continence rate as 84% in HRPC patients who underwent RARP. Preoperative erectile function status of the patient, postoperative adjuvant treatment requirement, NVB sparing (unilateral or bilateral), bladder neck preservation, and urethral length should all be considered seriously in the evaluation of postoperative functional outcomes. In our series, bilateral and unilateral NVB sparing was performed on 47% and 15%, respectively. Only 13 patients (38%) did not undergo NVB sparing. Of the 34 patients with 1-year follow-up, 17 (50%) were fully continent (zero pads/day), six (17.7%) wore one pad/day, three (8.8%) wore two pads/day, and two (5.9%) wore more than two pads/day. Of the 24 patients with no preoperative erectile dysfunction, 15 (44.1%) had no erectile dysfunction at a mean follow-up of 1-year. Trifecta and pentafecta rates were 38% and 26%, respectively. Preservation of the NVBs and postoperative adjuvant therapy administration are expected to affect functional outcomes.
Tissue characteristics might be different in HRPC patients compared with low-risk disease during performing of RARP. In addition, possibility of losing tissue of dissection might exist, and sufficient surgical experience might be essential. Therefore, we suggest gaining sufficient surgical experience in low-risk cases initially. Bulky disease and involvement of seminal vesicles or bladder neck could challenge the console surgeon. We demonstrated surgical differences comparatively in high-risk versus low-risk disease during performing of RARP (supplementary video).
Limited sample size, inclusion of more than one surgeon’s experience, and being a retrospective and noncomparative study are the main limitations of our study.
Conclusion
In conclusion, according to our experience, RARP in HRPC is a safe procedure with satisfactory oncologic and functional outcomes in the short term.
Disclosure
This study was presented by Dr Canda as a poster during the European Robotic Urology Symposium (ERUS), which was held on 17–19 September 2014 in Amsterdam, the Netherlands. Dr Canda is a member of the Robotic Urology Working Group of the Young Academic Urologists (YAU) of the European Association of Urology (EAU). The other authors report no conflicts of interest in this work.
References
Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71–96. | |
Cooperberg MR, Cowan J, Broering JM, Carroll PR. High-risk prostate cancer in the United States, 1990–2007. World J Urol. 2008;26:211–218. | |
D’Amico AV, Whittington R, Malkowicz SB, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280(11):969–974. | |
Gonzalez JR, Laudano MA, McCann TR, McKiernan JM, Benson MC. A review of high-risk prostate cancer and the role of neo-adjuvant and adjuvant therapies. World J Urol. 2008;26:475–480. | |
Lau WK, Bergstralh EJ, Blute ML, Slezak JM, Zincke H. Radical prostatectomy for pathological Gleason 8 or greater prostate cancer: influence of concomitant pathological variables. J Urol. 2002;167:117–122. | |
Thompson IM, Carroll PR, Carducci MA. Recommendations for defining and treating high risk localized prostate cancer. J Urol. 2006;176(6 Pt 2):S6–S10; quiz S3–S5. | |
Sooriakumaran P, Nyberg T, Akre O, et al. Comparative effectiveness of radical prostatectomy and radiotherapy in prostate cancer: observational study of mortality outcomes. BMJ. 2014;348:g1502. | |
Cooperberg MR, Vickers AJ, Broering JM, Carroll PR. Comparative risk-adjusted mortality outcomes after primary surgery, radiotherapy, or androgen-deprivation therapy for localized prostate cancer. Cancer. 2010;116:5226–5234. | |
Kawachi MH. Counterpoint: robot-assisted laparoscopic prostatectomy: perhaps the surgical gold standard for prostate cancer care. J Natl Compr Canc Netw. 2007;5:689–692. | |
Canda AE, Atmaca AF, Akbulut Z, et al. Results of robotic radical prostatectomy in the hands of surgeons without previous laparoscopic radical prostatectomy experience. Turk J Med Sci. 2012;42(Suppl 1):1338–1346. | |
Punnen S, Meng MV, Cooperberg MR, Greene KL, Cowan JE, Carroll PR. How does robot-assisted radical prostatectomy (RARP) compare with open surgery in men with high-risk prostate cancer? BJU Int. 2013;112(4):E314–E320. | |
Gandaglia G, Abdollah F, Hu J, et al. Is robot-assisted radical prostatectomy safe in men with high-risk prostate cancer? Assessment of perioperative outcomes, positive surgical margins, and use of additional cancer treatments. J Endourol. 2014;28(7):784–791. | |
Partin AW, Mangold LA, Lamm DM, Walsh PC, Epstein JI, Pearson JD. Contemporary update of prostate cancer staging nomograms (Partin Tables) for the new millennium. Urology. 2001;58:843–848. | |
Harty NJ, Kozinn SI, Canes D, Sorcini A, Moinzadeh A. Comparison of positive surgical margin rates in high risk prostate cancer: open versus minimally invasive radical prostatectomy. Int Braz J Urol. 2013;39(5):639–646; discussion 647–648. | |
Pierorazio PM, Mullins JK, Eifler JB, et al. Contemporaneous comparison of open vs minimally-invasive radical prostatectomy for high-risk prostate cancer. BJU Int. 2013;112(6):751–757. | |
Busch J, Magheli A, Leva N, et al. Matched comparison of outcomes following open and minimally invasive radical prostatectomy for high-risk patients. World J Urol. 2014;32(6):1411–1416. | |
Jayram G, Decastro GJ, Large MC, et al. Robotic radical prostatectomy in patients with high-risk disease: a review of short-term outcomes from a high-volume center. J Endourol. 2011;25:455–457. | |
Silberstein JL, Su D, Glickman L, et al. A case-mix-adjusted comparison of early oncological outcomes of open and robotic prostatectomy performed by experienced high volume surgeons. BJU Int. 2013;111(2):206–212. | |
Ham WS, Park SY, Rha KH, Kim WT, Choi YD. Robotic radical prostatectomy for patients with locally advanced prostate cancer is feasible: results of a single-institution study. J Laparoendosc Adv Surg Tech A. 2009;19:329–332. | |
Sagalovich D, Calaway A, Srivastava A, Sooriakumaran P, Tewari AK. Assessment of required nodal yield in a high risk cohort undergoing extended pelvic lymphadenectomy in robotic-assisted radical prostatectomy and its impact on functional outcomes. BJU Int. 2013;111: 85–94. | |
Yuh BE, Ruel NH, Mejia R, Wilson CM, Wilson TG. Robotic extended pelvic lymphadenectomy for intermediate- and high-risk prostate cancer. Eur Urol. 2012;61:1004–1010. | |
Jung JH, Seo JW, Lim MS, et al. Extended pelvic lymph node dissection including internal iliac packet should be performed during robot-assisted laparoscopic radical prostatectomy for high-risk prostate cancer. J Laparoendosc Adv Surg Tech A. 2012;22:785–790. | |
Rogers CG, Sammon JD, Sukumar S, Diaz M, Peabody J, Menon M. Robot assisted radical prostatectomy for elderly patients with high risk prostate cancer. Urol Oncol. 2013;31(2):193–197. | |
Yuh B, Artibani W, Heidenreich A, et al. The role of robot-assisted radical prostatectomy and pelvic lymph node dissection in the management of high-risk prostate cancer: a systematic review. Eur Urol. 2014;65(5):918–927. | |
Yee DS, Narula N, Amin MB, Skarecky DW, Ahlering TE. Robot-assisted radical prostatectomy: current evaluation of surgical margins in clinically low-, intermediate-, and high-risk prostate cancer. J Endourol. 2009;23(9):1461–1465. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


