Back to Journals » Cancer Management and Research » Volume 10
Orchestration of immune checkpoints in tumor immune contexture and their prognostic significance in esophageal squamous cell carcinoma
Authors Zhao JJ, Zhou ZQ, Wang P, Chen CL, Liu Y , Pan QZ, Zhu Q, Tang Y, Weng DS, Xia JC
Received 30 July 2018
Accepted for publication 30 October 2018
Published 28 November 2018 Volume 2018:10 Pages 6457—6468
DOI https://doi.org/10.2147/CMAR.S181949
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Xueqiong Zhu
Jing-Jing Zhao,1,2,* Zi-Qi Zhou,1,2,* Peng Wang,3,4,* Chang-Long Chen,1,2 Yuan Liu,1,2 Qiu-Zhong Pan,1,2 Qian Zhu,1,2 Yan Tang,1,2 De-Sheng Weng,1,2 Jian-Chuan Xia1,2
1Department of Experimental Research, Sun Yat-Sen University Cancer Center, State Key Laboratory of Oncology in South China, Collaborative Innovation Center for Cancer Medicine, Guangzhou, China; 2Department of Biotherapy, Sun Yat-Sen University Cancer Center, Guangzhou, China; 3Department of Emergency Medicine, Sun Yat-Sen Memorial Hospital, Sun Yat-Sen University, Guangzhou, China; 4Department of Medical Research, Guangdong Provincial Key Laboratory of Malignant Tumor Epigenetics and Gene Regulation, Sun Yat-Sen Memorial Hospital, Sun Yat-Sen University, Guangzhou, China
*These authors contributed equally to this work
Introduction: Esophageal squamous cell carcinoma (ESCC) develops in a background of chronic inflammation; therefore, it is a promising candidate for treatment by immunotherapy. Although tumor immunity is critically involved in tumor growth and metastasis in ESCC, important gaps exist in our understanding of its immune microenvironment. This study aimed to investigate the expression and prognostic significance of immune checkpoint proteins in ESCC and the associated T-cell densities.
Materials and methods: We investigated the infiltration of CD8+ T cells and the expressions of immune checkpoint proteins (PD-1, TIGIT, PD-L1, and PD-L2) in 154 primary ESCC patients by immunohistochemistry. The correlation of immune checkpoint proteins’ expression and clinical outcomes was determined by Kaplan–Meier test and multivariate Cox regression analysis.
Results: PD-L1 and PD-L2 expression were detected in 45.5 and 59.7% of the ESCC samples, respectively. The high densities of PD-1+ and TIGIT+ tumor-infiltrating lymphocytes (TILs) were expressed in 47.4 and 49.4% of the ESCC patients, respectively. The number of PD-1+ TILs was significantly positively correlated with CD8+ TILs (P<0.001). Cases displaying high PD-L1 expression exhibited consistently high CD8+ T-cell infiltration (P=0.0157). Increased numbers of PD-1+ and TIGIT+ TILs alone or both, as well as PD-L1 and PD-L2 expression alone or both, were significantly and associated with a shorter overall survival among these patients. The combined analysis of the expression of PD-1, TIGIT, PD-L1, and PD-L2 found that a group of patients with PD-1+/TIGIT+ TILs and PD-L1- and/or PD-L2-positive tumor cells had the worst prognosis in primary ESCC.
Conclusion: These immune profiles of checkpoint proteins expression should guide the selection of ESCC patients to receive suitable immunotherapies.
Keywords: biomarker, PD-1, prognostic significance, immune microenvironment, tumor-infiltrating lymphocytes
Introduction
Esophageal cancer is one of the most aggressive and lethal malignancies among gastrointestinal cancer and is the sixth leading cause of cancer death.1 It has the following two main subtypes: squamous-cell carcinoma and adenocarcinoma. Esophageal squamous cell carcinoma (ESCC) accounts for more than 90% of esophageal cancer cases worldwide. Although advances have been made in the therapy with neoadjuvant chemotherapy or radiochemotherapy, it still remains a major cause of morbidity and mortality in Asia, Africa, and South America.1 Therefore, new clinical parameters for prognosis and new treatment approaches for adjuvant treatment are needed.
A solid tumor is an intricate and dynamic ecosystem comprising tumor cells, immune cells, fibroblasts, blood and lymphoid vessels, nerves, extracellular matrix proteins, endothelial cells, and pericytes.2 The density and composition of immune cell populations is heterogeneous and is the major factor that determines the fate of cancer, such as the prevention or encouragement of cancer initiation, metastasis and invasion, and angiogenesis.3 In various human solid cancers, tumor-infiltrating lymphocytes (TILs) are considered to play important roles in orchestrating the immune response to cancer. Among TILs, most CD8+ T cells are cytotoxic T lymphocytes that were generally considered as the main force against cancer.4 In the vast majority of cancers, the presence, type, and location of CD8+ T cell infiltrates in the tumor mass are associated with longer patient survival.5
The expression of immunosuppressive proteins (immune checkpoints) on tumor-infiltrating T cells suggests that they help the tumor to evade host immune surveillance. PD-1 and its ligands PD-L1 and PD-L2 are important immune checkpoints.6–8 PD-1 is an inhibitory co-signal on activated lymphocytes, and PD-1⁄PD-L pathway plays a critical role in inactivation of the endogenous antitumor immune defense.9 In ESCC, several studies have shown that the expression of the PD-1 on the immune cells,10 or its ligand PD-L1 and PD-L2 on tumor cells,11,12 is associated with a poor clinical outcome. Interestingly, in a recent Phase III trial, favorable responses and survival outcomes were obtained using nivolumab (an anti-PD-1 monoclonal antibody) in advanced squamous cell non-small-cell lung cancer (NSCLC), which is genetically similar to ESCC.13,14 Furthermore, a favorable response and durable efficacy of anti-PD-1 monoclonal antibodies for ESCC were established in early clinical trials.15 Another inhibitory molecule that has received attention recently is T-cell Ig and ITIM domain (TIGIT), which is co-expressed with PD-1 on CD8+ TILs in melanoma, which regulates T-cell function synergistically with PD-1.16,17
Immune checkpoint blockade has changed the treatment landscape for a variety of cancers, most prominently melanoma, NSCLC, renal cell carcinoma, and cancers.13,18,19 These marked successes have led to an increased interest in evaluating these agents in several other malignancies including ESCC. It is important to define the combination of immune-based biomarkers that will predict a patients’ prognosis and further guide immunotherapeutic approaches. No published study has systematically examined the co-expression of PD-1, TIGIT, PD-L1, and PD-L2 in ESCC patients before. Thus, this study aimed to investigate the expression and prognostic significance of immune checkpoint receptors and their paired ligands on ESCC in relation to CD8+ T-cell densities.
Materials and methods
Patient population and tissue samples
Tissue specimens from 154 patients who underwent surgical resection for ESCC between 2002 and 2005 at the Sun Yat-Sen University Cancer Center (Guangzhou, China) were studied. All specimens were fixed in 10% formalin and embedded in paraffin wax. Patients received no immunotherapy or chemotherapy prior surgery. The demographic characteristics of the cohort are shown in Table S1. The original histological diagnosis was classified according to WHO criteria. All tumors were staged pathologically according to the American Joint Committee on Cancer (AJCC, 2002) TNM staging system. Postoperative follow-up was carried out in our outpatient department and included regular clinical and laboratory examinations as follows: every 3 months for the first 2 years, every 6 months for the following 2 years, and annually for an additional 5 years or until patient death, whichever occurred first. This study was approved by the Ethics Committee of the Sun Yat-Sen University Cancer Center and was performed according to the Declaration of Helsinki. Written informed consents had been obtained from all patients.
Immunohistochemical (IHC) staining
Serial 4 µm formalin-fixed paraffin-embedded tissue sections from ESCC were stained for IHC analysis. Deparaffinization was carried out with xylene, and the sections were subsequently hydrated with an ethanol gradient. For antigen retrieval, the tissue sections were immersed in EDTA (1 mmol/L, pH 9.0) and maintained at 100°C for 15 minutes, before cooling at room temperature for 2 hours. The sections were then washed with PBS (pH 7.4) and immersed in 3% H2O2 for 15 minutes to eliminate endogenous peroxidase activity. After incubation in 10% normal goat serum (Thermo Fisher Scientific, Waltham, MA, USA) for 30 minutes at room temperature to block non-specific antigens, sections were then incubated overnight at 4°C with the primary detection antibody against CD8 (Abcam, Cambridge, MA, USA), PD-1 (Abcam), PD-L1 (Abcam), PD-L2 (Cell Signaling Technology, Danvers, MA, USA), and TIGIT (Thermo Fisher Scientific). The sections were then washed with PBS three times. Subsequently, the sections were incubated with horseradish peroxidase-conjugated secondary antibody (EnVision Detection Kit; Dako Denmark A/S, Glostrup, Denmark) at room temperature for 30 minutes. After washing three times with PBS, the sections were stained with 3,3′-diaminobenzidine for 1 minute and nuclei were counterstained with hematoxylin. Slides were dehydrated in an ethanol gradient, mounted with neutral gum, and stored at room temperature for later observation.
Evaluation of immunostaining
IHC of PD-L1 and PD-L2 was scored as 0 (no staining), 1+ (weak membranous staining in <10% of the tumor cells), 2+ (weak-to-moderate membranous staining in ≥10% of the tumor cells), and 3+ (strong membranous staining in ≥10% of the tumor cells). Cases that were scored as 2+ or 3+ were considered to be positive for PD-L1 or PD-L2 expression, respectively. Cases that were scored as 0 or 1+ were considered to be negative for PD-L1 or PD-L2 expression, respectively.
The density of CD8+, PD-1+, and TIGIT+ TILs was determined as lymphocytes that infiltrated into cancer nests. The median TILs’ count was used as a cutoff to categorize each case into either a high (+) TILs group or a low (–) TILs group. Immune cells were identified by their specific markers (CD8, PD-1, and TIGIT). For each section, a minimum of five areas of a representative field of tumor were assessed with a microscopic field of ×200 (0.933 mm2). The average number of immune cells was calculated as the final density of each section (cells/mm2). All scoring and counting were performed independently by two investigators without knowledge of clinical information.
Statistical analyses
Comparisons among the demographic and pathological features, immune marker densities, and PD-L1 and PD-L2 expressions were evaluated using a Chi-squared test or Fisher’s exact test. The difference of TILs density between PD-L1/L2 expression positive and negative was analyzed by the paired t-test. Associations of TILs density between CD8+, PD-1+, and TIGIT+ were examined by calculating Pearson’s correlation coefficient. OS was evaluated using the Kaplan–Meier method, and the differences between survival curves were tested for statistical significance using the log-rank test. The Cox proportional hazards model was used to estimate the independent prognostic factors for OS. All statistical analyses were carried out using SPSS 19.0 (IBM Corporation, Armonk, NY, USA), and a two-sided P-value of <0.05 was considered statistically significant.
Results
Clinical characteristics of patients
A total of 154 (124 males and 30 females) patients were included in the study. The median age was 55 years (range, 37–48 years). The median length of the tumor was 5 cm (range, 2–10 cm). Tumor locations were upper thoracic in 12 patients, middle thoracic in 95 patients, and lower thoracic in 47 patients. The histopathological differentiations were poor in 38 cases, moderate in 73 cases, and well in 43 cases. A total of 108 patients (70.1%) had T3/T4 tumors, and 70 patients (45.5%) had positive lymph nodes. The pathological stages were stage I in eight patients, stage II in 87 patients, stage III in 53 patients, and stage IV in six patients. The estimated 1-, 3-, and 5-year overall survival (OS) rates were 85.1, 61.0, and 51.9%, respectively. The median OS was 41.5 months (range, 1–82 months). Patient characteristics are listed in Table S1.
The expression pattern of PD-L1 and PD-L2 and the infiltration of PD-1+, TIGIT+, and CD8+ TILs in ESCC
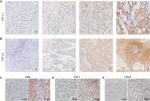
The PD-1+, TIGIT+, and CD8+ TILs in cancer nests and the expression of PD-L1 and PD-L2 in tumor tissues were observed by IHC staining. Representative IHC images of PD-L1, PD-L2, PD-1, TIGIT, and CD8 are presented in Figure 1A–E. PD-L1 and PD-L2 were highly expressed in 45.5 and 59.7% of the ESCC samples, respectively (Table 1 and Figure 1A and B). Among 154 ESCC specimens, the mean number of infiltrating PD-1+, TIGIT+, and CD8+ T cells was 59.21±61.31 TILs/mm2 (median 43.8, range 0–250.0), 41.20±29.90 TILs/mm2 (median 41.5, range 0–133.0), and 323.31±140.47 TILs/mm2 (median 290.0, range 0–790.0), respectively. The high densities of CD8+, PD-1+, and TIGIT+ cells were expressed in 41.6, 47.4, and 49.4% of the ESCC patients, respectively (Table 2 and Figure 1C, D, and E). In addition, a significant positive correlation was found between the densities of PD-1+ and CD8+ T cells (r=0.278, P<0.001), TIGIT+ and CD8+ T cells (r=0.18, P=0.025), and PD-1+ and TIGIT+ cells (r=0.236, P=0.003) in the tumor tissues of the 154 primary ESCC cases (Figure 2A).
  | Table 1 Clinicopathological characteristics of ESCC patients according to PD-L1 and PD-L2 expressions Note: aP<0.05. Abbreviation: ESCC, esophageal squamous cell carcinoma. |
To investigate the relationship between PD-Ls’ expression by tumor cells and immune cell-related parameters, we performed a comparative analysis of PD-L1 and PD-L2 expressions by tumor cells and the number of PD-1+, CD8+, and TIGIT+ TILs. High expression of PD-L1 in ESCC was associated significantly with the number of CD8+ T cells but not with PD-1+ and TIGIT+ TILs (P=0.0157, Figure 2B). However, no significant relationship between PD-L2 expression and the number of PD-1+, TIGIT+, or CD8+ TILs was detected (Figure 2B).
Simultaneous expression of immune checkpoints in primary ESCC identifies patients with poor clinical outcome
Kaplan–Meier survival analysis revealed that the patients carrying a high number of PD-1+ TILs (n=73/154, 47.4%) or TIGIT+ TILs (n=76/154, 49.4%) tended to exhibit a shorter OS (P=0.044, Figure 3A, and P=0.045, Figure 3B). Patients with PD-L1-positive tumors (n=83/154, 53.9%) had a shorter OS than those with PD-L1-negative tumors (n=71/154, 46.1%) (P=0.005, Figure 3C). Patients with PD-L2-positive tumors (n=92/154, 59.7%) displayed a shorter OS compared with those with PD-L2-negative tumor cells (n=62/154, 40.3%) (P=0.002, Figure 3D).
Multivariate analysis of the patients as grouped was performed with other clinicopathological predictors for survival time using the Cox regression model. The results indicated that PD-1+ TILs, TIGIT+ TILs, PD-L1, and PD-L2-positive expressions in cancer were the independent unfavorable prognostic factors in ESCC (Table 3).
  | Table 3 Univariate and multivariate analyses of overall survival in ESCC Note: aP<0.05. Abbreviation: ESCC, esophageal squamous cell carcinoma. |
To investigate the impact of immune checkpoint molecules on the prognostic impact of the CD8+ TILs group, we analyzed the protein expression of PD-1 and TIGIT and co-expression in this group of tumors. Kaplan–Meier survival analysis showed that patients carrying a high number of CD8+ TILs (n=64/154, 41.6%) displayed a longer OS than those carrying a low number of CD8+ TILs (CD8-, n=90/154, 58.4%) (P=0.003, Figure 3E). However, no significant difference in OS was observed between those with high levels of CD8+/PD-1+ TILs (n=42/154, 27.3%), CD8+/TIGIT+ TILs (n=35/154, 22.7%), or CD8+/PD-1+/TIGIT+ (n=27/154, 17.5%) and those with high levels of CD8+ TILs (Figure 3E).
To evaluate the possibility that a high level of both PD-1+ and TIGIT+ T-cell infiltrations in cancer might correlate with unfavorable patient prognosis, the patients were classified into the following four groups: PD-1+/TIGIT+ TILs (n=42/154, 27.3%), PD-1+/TIGIT- TILs (n=31/154, 20.1%), PD-1-/TIGIT+ TILs (n=34/154, 22.1%), and PD-1-/TIGIT- TILs (n=47/154, 30.5%). The Kaplan–Meier survival results showed that PD-1+/TIGIT+ TILs in cancer demonstrated significantly lower survival rates than patients with PD-1-/TIGIT- TILs (P=0.005, Figure 3F). However, no significant difference in OS was observed between PD-1+/TIGIT- and PD-1-/TIGIT+ TILs in patients with cancer and patients with PD-1-/TIGIT- TILs (P=0.205 and 0.250, respectively, Figure 3F). The median survival time in four groups is 24.5, 42, 39, and 59 months. Similarly, to evaluate the possibility that a high level of both PD-L1 and PD-L2 expressions in cancer might correlate with unfavorable patient prognosis, the patients were classified into the following four groups: PD-L1 positive/PD-L2 positive (n=54/154, 35.1%), PD-L1 positive/PD-L2 negative (n=30/154, 19.5%), PD-L1 negative/PD-L2 positive (n=39/154, 25.3%), and PD-L1 negative/PD-L2 negative (n=31/154, 20.1%). The Kaplan–Meier survival results showed that PD-L1-positive/PD-L2-positive patients demonstrated significantly lower survival rates than PD-L1-negative/PD-L2-negative patients (P<0.001, Figure 3G). However, no significant difference in OS was observed between PD-L1-positive/PD-L2-negative or PD-L1-negative/PD-L2-positive patients and PD-L1-negative/PD-L2-negative patients (P=0.195 and 0.065, Figure 3G). The median survival time in four groups is 25, 59.5, 42, and 66 months.
To identify the good and poor prognosis of the patients, the combined expression of PD-1, TIGIT in TILs, and PD-L1 and PD-L2 in tumor was analyzed. According to the above prognosis results, we found that patients with PD-1+/TIGIT+ TILs and PD-L1- and/or PD-L2-positive tumor cells (n=34/154, 22.1%) had a markedly shorter OS than patients with PD-1-/TIGIT- TILs and PD-L1- and PD-L2-negative tumor cells (n=10/154, 6.5%) (P=0.003, Figure 3H). The median survival time in two groups is 20 and 72.5 months, respectively. In addition, no significant differences were found between PD-1+ or TIGIT+ alone and both PD-1+/TIGIT+ TILs in the patients with PD-L1- and/or PD-L2-positive tumor cells (Figure 3H). Similarly, no significant differences were found between PD-1- or TIGIT- alone and both PD-1-/TIGIT- TILs in the patients with PD-L1- and PD-L2-negative tumor cells (Figure 3H).
Multivariate analysis of the patients as grouped was performed with other clinicopathological predictors for survival time using the Cox regression model. The results indicated that PD-1+/TIGIT+ TILs, PD-L1-positive/PD-L2-positive and combined PD-1+/TIGIT+ TILs, and PD-L1 positive and/or PD-L2 positive in cancer were the independent unfavorable prognostic factors in ESCC (Table 3).
Discussion
The exploration of immune-based biomarkers involved in tumor microenvironment is becoming a useful strategy to predict patient’s outcomes and guide immunotherapy. However, the intricate and dynamic immune microenvironment is heterogeneous in different tumor types. Emerging studies have suggested that tumor may evade host immune response through the expression of immune checkpoints such as PD-1, TIGIT, PD-L1, and PD-L2. A comprehensive detection and assessment of immune checkpoint factors influencing prognosis are important for improving patient management of ESCC. However, there is no previous study on the expression of PD-1, TIGIT, PD-L1, and PD-L2 simultaneously in ESCC tissues. Our results showed that PD-1, TIGIT, PD-L1, and PD-L2 were aberrantly overexpressed in ESCC tissues and these proteins expression alone or together had a significantly poorer prognosis than the negative patients. We also compared and identified the patients’ cohort with worst prognosis according the combined expression of PD-1, TIGIT, PD-L1, and PD-L2 in ESCC.
ESCC has been regarded as a proinflammatory neoplasm, where tumor cells produce several cytokines (such as TGF-β, IL-6, IL-8, CCL5, and VEGF) that could lead to the recruitment and activation of polyclonal CD8+ T cells.20–26 CD8+ T cell is a crucial component of cell-mediated immunity as it produces interferon-γ (IFN-γ) upon interaction with tumor targets, and CD8+ TILs have been correlated with a favorable outcome in several tumor types.27–29 However, recent findings demonstrated that high infiltration of CD8+ T cells correlates with poor survival in renal cell cancer,30 diffuse large B-cell lymphoma,31 and Hodgkin lymphoma.32 It has been reported that IFN-γ can induce PD-L1 expression on tumor cells.33,34 In our study, we found that PD-L1-positive tumor had more CD8+ T cells than PD-L1-negative tumor. In addition, a significant positive correlation was found between the densities of PD-1+ or TIGIT+ and CD8+ T cells. Survival analysis showed that patients with high CD8 expression had a significantly better clinical outcome than low CD8 expression, but co-expression of PD-1 or TIGIT with CD8 did not significantly decrease the OS in ESCC patients. Therefore, CD8+ T cells may play an important role in suppressing tumor progression in ESCC.
To the best of our knowledge, this is the first report of the poor prognosis associated with high densities of TIGIT+ cells in ESCC. In addition, we presented evidence that the expression of PD-1 and TIGIT is highly correlated in ESCC. Patients with high expression of PD-1 or TIGIT have significantly shorter OS than low expression of PD-1 and TIGIT patients, and they have similar survival rate, suggesting that TIGIT may have similar effect with PD-1 in immune inhibition in ESCC. Some studies on mouse models have shown a synergistic effect of the inhibition of both pathways in boosting the antitumor immune response.17 Thus, our results suggest that cooperation between infiltrating PD-1+ and TIGIT+ lymphocytes in tumors might be important in the progression of ESCC and support the rationale of dual blockade of these molecules in ESCC.
Our results found that the density of PD-1+ cells and the tumor expression of PD-L1 in primary ESCC were associated with a poor clinical outcome. Furthermore, we described the prognostic significance of PD-L2. PD-L2 seems to be expressed in a higher proportion of tumors (59.7%) than PD-L1 (53.9%), and PD-L2 expression is associated with a poor clinical outcome. We also found that patients with PD-L1-positive/PD-L2-positive tumor had significantly shorter OS than PD-L1-negative/PD-L2-negative patients, and PD-L1 positive/PD-L2 positive was an independent unfavorable prognostic factor in ESCC. This finding might have clinical relevance because anti-PD-L1 treatment alone seems to have a lower response rate than anti-PD-1.35,36 In addition, there are several PD-L1-negative tumors that respond to anti-PD-1 treatment,35 implying that there are other molecules beside PD-L1 that are involved in the PD-1 inhibition axis of ESCC. A few publications have reported PD-L2 expression in other tumors, including NSCLC,37 ovarian cancer, and primary mediastinal large B-cell lymphoma,38,39 where it has shown a restricted impact on patients’ prognosis.
The immunological status in the tumor microenvironment is now well recognized to be a critical determining factor in tumor prevention, development, and progression. Better understanding of the roles of immune cells and molecules in the tumor microenvironment will therefore be essential in the development of histology-specific tumors. Combined analysis of PD-1, TIGIT expression in TILs, and tumorous PD-L1 and PD-L2 expressions in ESCC, we found that only a small proportion of patients (n=10/154, 6.5%) with PD-1-/TIGIT- TILs and PD-L1- and PD-L2-negative tumor cells had best prognosis (median survival time, 72.5 months) and a higher proportion of patients (n=34/154, 22.1%) with PD-1+/TIGIT+ TILs and PD-L1 and/or PD-L2-positive tumor cells had worst prognosis (median survival time, 20 months). Multivariate analysis showed that combined PD-1+/TIGIT+ TILs and PD-L1 positive and/or PD-L2 positive in cancer were the independent unfavorable prognostic factors in ESCC. Our data suggest that patients with PD-1+/TIGIT+ TILs and PD-L1- and/or PD-L2-positive tumor cells may benefit from immune checkpoints blockade therapy but need multiple blockade of these immune molecules or optimize immune checkpoints blockade in ESCC.
Conclusion
Our results evaluated novel prognostic factors in ESCC based on the concomitant quantification of densities of CD8+, PD-1+, and TIGIT+ TILs in addition to PD-L1/PD-L2 expression by tumor cells. These immune profiles should guide the selection of patients to receive suitable immunotherapies and need to be further validated in larger and independent cohorts.
Acknowledgment
This work was supported by grants from the National Natural Science Foundation of China (no 81402560) and the Guangdong Province Science and Technology Plan Project (no 2017A020215029).
Author contributions
JJZ and ZQZ were the major contributors in writing the manuscript. PW analyzed the experimental data. ZQZ collected the tissues and clinical data of patients. CLC and YL drew the figures. QZP did the data interpretation. QZ and YT searched the literature. DSW and JCX designed this study. All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Rustgi AK, El-Serag HB. Esophageal carcinoma. N Engl J Med. 2014;371(26):2499–2509. | ||
Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100(1):57–70. | ||
Giraldo NA, Becht E, Remark R, Damotte D, Sautès-Fridman C, Fridman WH. The immune contexture of primary and metastatic human tumours. Curr Opin Immunol. 2014;27:8–15. | ||
Zitvogel L, Galluzzi L, Kepp O, Smyth MJ, Kroemer G. Type I interferons in anticancer immunity. Nat Rev Immunol. 2015;15(7):405–414. | ||
Fridman WH, Pagès F, Sautès-Fridman C, Galon J. The immune contexture in human tumours: impact on clinical outcome. Nat Rev Cancer. 2012;12(4):298–306. | ||
Dong H, Chen L. B7-H1 pathway and its role in the evasion of tumor immunity. J Mol Med (Berl). 2003;81(5):281–287. | ||
Dong H, Zhu G, Tamada K, Chen L. B7-H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nat Med. 1999;5(12):1365–1369. | ||
Latchman Y, Wood CR, Chernova T, et al. PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat Immunol. 2001;2(3):261–268. | ||
Keir ME, Butte MJ, Freeman GJ, Sharpe AH. PD-1 and its ligands in tolerance and immunity. Annu Rev Immunol. 2008;26:677–704. | ||
Feng Z, Xiang-Lei L, Hai-Tao W, et al. Programmed cell death 1 expression in esophageal squamous cell carcinoma and association with clinical characteristics. Indian J Cancer. 2015;52 Suppl 3:E176–178. | ||
Chen L, Deng H, Lu M, et al. B7-H1 expression associates with tumor invasion and predicts patient’s survival in human esophageal cancer. Int J Clin Exp Pathol. 2014;7(9):6015–6023. | ||
Ohigashi Y, Sho M, Yamada Y, et al. Clinical significance of programmed death-1 ligand-1 and programmed death-1 ligand-2 expression in human esophageal cancer. Clin Cancer Res. 2005;11(8):2947–2953. | ||
Brahmer J, Reckamp KL, Baas P, et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non-Small-Cell Lung Cancer. N Engl J Med. 2015;373(2):123–135. | ||
Song Y, Li L, Ou Y, et al. Identification of genomic alterations in oesophageal squamous cell cancer. Nature. 2014;509(7498):91–95. | ||
Kojima T, Doi T. Immunotherapy for Esophageal Squamous Cell Carcinoma. Curr Oncol Rep. 2017;19(5):33. | ||
Chauvin JM, Pagliano O, Fourcade J, et al. TIGIT and PD-1 impair tumor antigen-specific CD8+ T cells in melanoma patients. J Clin Invest. 2015;125(5):2046–2058. | ||
Johnston RJ, Comps-Agrar L, Hackney J, et al. The immunoreceptor TIGIT regulates antitumor and antiviral CD8(+) T cell effector function. Cancer Cell. 2014;26(6):923–937. | ||
Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711–723. | ||
Motzer RJ, Escudier B, McDermott DF, et al. Nivolumab versus Everolimus in Advanced Renal-Cell Carcinoma. N Engl J Med. 2015;373(19):1803–1813. | ||
Abdel-Latif MM, Duggan S, Reynolds JV, Kelleher D. Inflammation and esophageal carcinogenesis. Curr Opin Pharmacol. 2009;9(4):396–404. | ||
Deans DA, Wigmore SJ, Gilmour H, Paterson-Brown S, Ross JA, Fearon KC. Elevated tumour interleukin-1beta is associated with systemic inflammation: A marker of reduced survival in gastro-oesophageal cancer. Br J Cancer. 2006;95(11):1568–1575. | ||
Doyle SL, Donohoe CL, Finn SP, et al. IGF-1 and its receptor in esophageal cancer: association with adenocarcinoma and visceral obesity. Am J Gastroenterol. 2012;107(2):196–204. | ||
Hong S, Lee HJ, Kim SJ, Hahm KB. Connection between inflammation and carcinogenesis in gastrointestinal tract: focus on TGF-beta signaling. World J Gastroenterol. 2010;16(17):2080–2093. | ||
Jenkins GJ, Mikhail J, Alhamdani A, et al. Immunohistochemical study of nuclear factor-kappaB activity and interleukin-8 abundance in oesophageal adenocarcinoma; a useful strategy for monitoring these biomarkers. J Clin Pathol. 2007;60(11):1232–1237. | ||
Liu J, Li F, Ping Y, et al. Local production of the chemokines CCL5 and CXCL10 attracts CD8+ T lymphocytes into esophageal squamous cell carcinoma. Oncotarget. 2015;6(28):24978–24989. | ||
O’Sullivan KE, Phelan JJ, O’Hanlon C, Lysaght J, O’Sullivan JN, Reynolds JV. The role of inflammation in cancer of the esophagus. Expert Rev Gastroenterol Hepatol. 2014;8(7):749–760. | ||
Shankaran V, Ikeda H, Bruce AT, et al. IFNgamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature. 2001;410(6832):1107–1111. | ||
Pagès F, Kirilovsky A, Mlecnik B, et al. In situ cytotoxic and memory T cells predict outcome in patients with early-stage colorectal cancer. J Clin Oncol. 2009;27(35):5944–5951. | ||
Zhang L, Conejo-Garcia JR, Katsaros D, et al. Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N Engl J Med. 2003;348(3):203–213. | ||
Giraldo NA, Becht E, Pagès F, et al. Orchestration and Prognostic Significance of Immune Checkpoints in the Microenvironment of Primary and Metastatic Renal Cell Cancer. Clin Cancer Res. 2015;21(13):3031–3040. | ||
Muris JJ, Meijer CJ, Cillessen SA, et al. Prognostic significance of activated cytotoxic T-lymphocytes in primary nodal diffuse large B-cell lymphomas. Leukemia. 2004;18(3):589–596. | ||
Scott DW, Chan FC, Hong F, et al. Gene expression-based model using formalin-fixed paraffin-embedded biopsies predicts overall survival in advanced-stage classical Hodgkin lymphoma. J Clin Oncol. 2013;31(6):692–700. | ||
Abiko K, Matsumura N, Hamanishi J, et al. IFN-γ from lymphocytes induces PD-L1 expression and promotes progression of ovarian cancer. Br J Cancer. 2015;112(9):1501–1509. | ||
Bellucci R, Martin A, Bommarito D, et al. Interferon-γ-induced activation of JAK1 and JAK2 suppresses tumor cell susceptibility to NK cells through upregulation of PD-L1 expression. Oncoimmunology. 2015;4(6):e1008824. | ||
Brahmer JR, Tykodi SS, Chow LQ, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med. 2012;366(26):2455–2465. | ||
Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366(26):2443–2454. | ||
Zhang Y, Wang L, Li Y, et al. Protein expression of programmed death 1 ligand 1 and ligand 2 independently predict poor prognosis in surgically resected lung adenocarcinoma. Onco Targets Ther. 2014;7:567–573. | ||
Hamanishi J, Mandai M, Iwasaki M, et al. Programmed cell death 1 ligand 1 and tumor-infiltrating CD8+ T lymphocytes are prognostic factors of human ovarian cancer. Proc Natl Acad Sci U S A. 2007;104(9):3360–3365. | ||
Shi M, Roemer MG, Chapuy B, et al. Expression of programmed cell death 1 ligand 2 (PD-L2) is a distinguishing feature of primary mediastinal (thymic) large B-cell lymphoma and associated with PDCD1LG2 copy gain. Am J Surg Pathol. 2014;38(12):1715–1723. |
Supplementary material
  | Table S1 Clinicopathological features of the 154 esophageal cancer patients (at the time of initial diagnosis) |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.