Back to Journals » International Journal of Nanomedicine » Volume 15
Optimization of Processing Parameters of Nanoemulsion Containing Aripiprazole Using Response Surface Methodology
Authors Samiun WS , Ashari SE , Salim N , Ahmad S
Received 19 December 2018
Accepted for publication 12 November 2019
Published 9 March 2020 Volume 2020:15 Pages 1585—1594
DOI https://doi.org/10.2147/IJN.S198914
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Prof. Dr. Anderson Oliveira Lobo
Wan Sarah Samiun,1 Siti Efliza Ashari,1,2 Norazlinaliza Salim,1,2 Syahida Ahmad3
1Integrated Chemical Biophysics Research, Faculty of Science, Universiti Putra Malaysia, Serdang 43400, Selangor, Malaysia; 2Centre of Foundation Studies for Agricultural Sciences, Universiti Putra Malaysia, Serdang 43400, Selangor, Malaysia; 3Department of Biochemistry, Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, Serdang 43400, Selangor, Malaysia
Correspondence: Wan Sarah Samiun; Siti Efliza Ashari
Integrated Chemical Biophysics Research, Faculty of Science, Universiti Putra Malaysia, Serdang 43400, Selangor, Malaysia
Tel +603-8946 6997
Email [email protected]; [email protected]
Background: Aripiprazole, which is a quinolinone derivative, has been widely used to treat schizophrenia, major depressive disorder, and bipolar disorder.
Purpose: A Central Composite Rotatable Design (CCRD) of Response Surface Methodology (RSM) was used purposely to optimize process parameters conditions for formulating nanoemulsion containing aripiprazole using high emulsification methods.
Methods: This design is used to investigate the influences of four independent variables (overhead stirring time (A), shear rate (B), shear time (C), and the cycle of high-pressure homogenizer (D)) on the response variable namely, a droplet size (Y) of nanoemulsion containing aripiprazole.
Results: The optimum conditions suggested by the predicted model were: 120 min of overhead stirring time, 15 min of high shear homogenizer time, 4400 rpm of high shear homogenizer rate and 11 cycles of high-pressure homogenizer, giving a desirable droplet size of nanoemulsion containing aripiprazole of 64.52 nm for experimental value and 62.59 nm for predicted value. The analysis of variance (ANOVA) showed the quadratic polynomial fitted the experimental values with F-value (9.53), a low p-value (0.0003) and a non-significant lack of-fit. It proved that the models were adequate to predict the relevance response. The optimized formulation with a viscosity value of 3.72 mPa.s and pH value of 7.4 showed good osmolality value (297 mOsm/kg) and remained stable for three months in three different temperatures (4°C, 25°C, and 45°C).
Conclusion: This proven that response surface methodology is an efficient tool to produce desirable droplet size of nanoemulsion containing aripiprazole for parenteral delivery application.
Keywords: aripiprazole, antipsychotic drug, APD, schizophrenia, nanoemulsion, response surface methodology, RSM, palm kernel oil esters, PKOES
Introduction
Schizophrenia is a serious and chronic mental disorder that affects 1% of the world population nowadays.1 This mental disorder is categorized by positive symptoms which are hallucinations, delusions and deranged thoughts, while negative symptoms of schizophrenia are loss of motivation, restricted emotional experience, poverty of speech and cognitive impairment. Dopamine theory is the first hypothesis created that could clarify the pathophysiology of this mental disorder which imputed an important role in dopamine system dysregulation.2–5
Recently, treatment for schizophrenia using aripiprazole has gained growing attention in a current psychiatric hospital. Aripiprazole is an atypical antipsychotic drug which is a third generation antipsychotic drug (APD) with better therapeutic efficacy in managing the symptoms of schizophrenia, a minimum incidence of extrapyramidal side effects (EPS) and weight gain side effects.6–9 Aripiprazole, which is a quinolinone derivative, has been widely used to treat schizophrenia, major depressive disorder, and bipolar disorder. For treatment with a recommended target dose of 10 to 15 mg per day, aripiprazole has been proved to give potent treatment for positive and negative symptoms.10,11
Schizophrenia disorder is mostly connected to the Central Nervous System (CNS) in the body and a well-developed CNS has a blood-brain barrier (BBB).12 The function of BBB is to block any macromolecules from accessing the brain.13 However, the drug deliveries to the brain is a massive challenge due to the existence of the BBB. The BBB is a specialized system of capillary endothelial cells that isolated the brain from the systemic circulation to maintain a stable environment for a proper function of the nervous tissue.14
The BBB has a strict limitation for transportation into the brain through both metabolic (enzymes) and physical (tight junctions) barrier. On the other hand, the peripheral capillaries only allow a relatively free exchange of substances across or between the cells. Thus, the BBB is considered the rate-limiting factor in investigating permeation of therapeutic agents into the CNS.15 Numerous drug delivery and targeting strategies have been developed to overcome this problem. Disruption of the BBB, chemical modification, molecular antibody technology, and various carrier systems have been used as a transport and the distribution of the therapeutic drugs into the brain.16 One of the promising strategies to enhance the drug delivery into the brain is by introducing the drug into the nanoemulsion system.
Nanoemulsion is an emulsion from one immiscible liquid dispersed as droplets within another liquid which are oil-in-water (O/W) or water-in-oil (W/O) emulsion where the average droplet diameter is between 20–200 nm.17 Nanoemulsion system is one of the potential strategies for efficient delivery of lipophilic active across the BBB owing to their nano-sized, biocompatible, biodegradable, superior physical stability, and massive scale production convenience.18
In recent years, Response Surface Methadology (RSM) has been used as one of the multivariate statistical techniques for numerous optimization studies.19 RSM was first introduced by Box and collaborators in the 1950s.20 It can be used to investigate the relationship and interaction of independent variables on the response variables by involving mathematical models and statistical techniques.19 Utilizing RSM for optimization studies will require fewer experiments.21 RSM was employed in the enhancement of CNS therapeautic drugs such as valproic acid-loaded nanoemulsion, sorafenib-encapsulated nanoemulsion, and chloramphenicol-encapsulated nanoemulsion.22–24
From previous work, nanoemulsions containing aripiprazole were successfully developed utilizing high pressure homogenizer with a homogenization pressure of 1000 bar for 14 cycles by optimizing the compositions of formulation nanoemulsion.25,26 In this work, RSM was utilized to identify the optimum processing parameters of encapsulation nanoemulsion containing aripiprazole by using the same optimum formulation from previous work. The influences of four independent variables, including overhead stirring time, shear time, shear rate, and the cycle of high-pressure homogenizer were studied on a response variable, namely droplet size by using CCRD. This design is deployed to reduce the number of experiments with the desirable result under optimum process parameters conditions and also enhance the drug encapsulated efficiency of the nanoemulsion. The main purpose of this experiment is to produce an optimum process parameter condition for formulating the parenteral nanoemulsion containing aripiprazole by RSM to treat the schizophrenia illness utilizing high energy emulsification methods.
Materials and Methods
Materials
Palm kernel oil esters (PKOEs) were synthesized using enzymatic transesterification from palm kernel oil and oleyl alcohol in the lab. Polysorbate 80 (Tween 80) was purchased from Fluka (Germany). Glycerol was acquired from Numedica, JT Baker (USA). Soybean Lecithin (Lipoid S75) was obtained from GmbH (Germany). The aripiprazole was purchased from Laboratory & Scientific Enterprise (Malaysia). Deionized water was obtained using a Milli-Q filtration system (USA).
Identification of the Solubility of Aripiprazole in Oil
The solubility of aripiprazole with different types of oil such as coconut oil, sesame oil, MCT oil, olive oil, sunflower oil, soybean oil, corn oil, castor oil, palm kernel oil esters (PKOEs) and safflower seed oil with presence of 2.0 w/w% lecithin were studied to find the most solubilizing capacity as a carrier. 0.1 w/w% of aripiprazole was added into the oil phase. The total volume of the mixture was 5 mL. The mixture was stirred for 24 h with a speed of 350 rpm at 60 °C. Centrifugation process was made using EBA200, Hettich Zentrifugen, Germany at 4500 rpm for 15 min. The phase separation was observed and recorded.
Preparation of Nanoemulsion Containing Aripiprazole
PKOEs (3.0 w/w%) and lecithin (2.0 w/w%) were stirred using magnetic stirrer at 60 °C. Aripiprazole (0.1 w/w%) was added and continued stirring for 24 h. After the aripiprazole was completely dissolved, Tween 80 (1.0 w/w%) acts as a co-surfactant was then dropped into the oil phase. The oil mixture was dropped wisely into the aqueous phase containing 2.25 w/w% of glycerol and 91.75 w/w% of deionized water.25,26 The mixture was then stirred by using overhead stirrer (IKA®RW 20 Digital, Nara, Japan). High energy emulsification methods, which are high shear homogenizer (Kinematica AG, Luzern, Switzerland) and high pressure homogenizer (Emulsiflex, Canada) were used to prepare the samples.
RSM Experimental Design
CCRD of RSM was used to optimize the process parameter conditions using high energy processing of four independent variables namely overhead stirring time (80–240 min, A), shear time (10–30 min, B), shear rate (800–5600 rpm, C), and the cycle of high-pressure homogenizer (8–20 cycles, D). This design is used to learn the effects of these variables with droplet size (Y) of nanoemulsion containing aripiprazole as the response. A total number of 30 experimental runs based on the CCRD were employed to identify the optimized levels of significant independent variables and their correlation between independent variables in a process generated by the Design Expert version 7.0 software (Stat-Ease Inc., Minneapolis, USA). At five different levels, these four independent variables were studied for every individual variable. The coded independent variables and their levels utilized in the CCRD are tabulated in Table 1.
 |
Table 1 Variables and Their Coded Levels Utilized in the CCRD Matrix |
Statistical Analysis
Statistical analysis was carried out by using an analysis of variance (ANOVA). By applying ANOVA, it is a more reliable alternative to assess the feature of the model fitted. The importance of ANOVA analysis is to study the significance of second-order polynomial in this model. The p-value is a component that was employed to control each independent variable’s condition. It also exhibits the interactions intensity between every independent variable.
Physicochemical Characterization
Droplet Size, Zeta Potential and Polydispersity Index (PDI) Measurement
The droplet size, zeta potential, and polydispersity index (PDI) of nanoemulsion formulations by experimental design were measured with diffusion method by dynamic light scattering (DLS) droplet analyzer (Zetasizer Nano ZS, Malvern Instruments, Malvern, UK) at temperature 25 °C. The measurement was measured using the Photon Correlation Spectroscopy (PCS) principle.27,28 Nanoemulsion sample was diluted with deionized water until it reached the desired concentration. The count rate for all diluted sample is in range of 150 to 300 kcps. All samples were measured in triplicate and the recorded values were calculated as mean values.
pH Measurement
A pre-calibrated pH Tutor Bench meter (Eutech Instrument, UK) was used to measure the pH values of the optimized nanoemulsions at 25 °C. Three pH standard buffer solutions (pH 4.01, pH 7.00, pH 10.01) were used for calibration. Three measurements were recorded and the average pH value was used.
Viscosity Measurement
The viscosity of the optmized nanoemulsion was measured using viscometer (Brookfield DV-II, Brookfield, US) with SC4-18 spindle at a controlled temperature of 25 °C. The spindle was immersed in the test samples. As the spindle rotated, the measurement of the viscous drag of the sample counter the spindle was taken by observing the deflection of the calibrated spring. The measurements of viscosity were taken three times.
Osmolality Measurement
The osmolality of optimized nanoemulsion was measured by the freezing-point depression method using a semi-micro osmometer (Model 3320, Advanced Instruments, Inc., USA). The instrument was calibrated with reference standards of water for injection and calibration solution NaCl to give values of 0 and 400 mOsm/kg, respectively. The measurements of osmolality were taken three times.
Stability Study
After sample preparation, the centrifugal tests were conducted to study emulsion gravitational stability. The samples were centrifuged for 15 min at 4500 rpm, at room temperature (25 °C). The optimum nanoemulsion containing aripiprazole was kept for three months storage at different temperatures (4 °C, 25 °C, and 45 °C) to study its thermal stability. The droplet sizes of each sample were measured and recorded every month.23
Coalescence rate of nanoemulsion containing aripiprazole was studied to investigate the cause of particle sizes changed over time. The coalescence rate was calculated with the collected data of particle size of nanoemulsion within the given time for all temperatures using the following equation:
(1)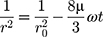
where r is the average radius after time,  is the frequency of rupture per unit of the film surface and r0 the value at time t=0. A graph of 1/r2 against time was plotted in order to determine the rate of coalescence and a linear relationship was expected.
is the frequency of rupture per unit of the film surface and r0 the value at time t=0. A graph of 1/r2 against time was plotted in order to determine the rate of coalescence and a linear relationship was expected.
Results and Discussion
Screening of Emulsion Compositions
The solubility of aripiprazole in different types of oil with an emulsifier (lecithin) was conducted. The experiments revealed that PKOE was the only oil that can dissolve aripiprazole as shown in Figure 1. The drug and lecithin were not fully dissolved in other types of oils such as coconut oil, sesame oil, MCT oil, olive oil, sunflower oil, soybean oil, corn oil, castor oil, and safflower seed oil. All oil mixtures except PKOE formed precipitation in the test tube. PKOE is a short chain ester which can be a suitable carrier for the delivery of the drug to administer into our body system.29 Lecithin was chosen as an emulsifier to form electrostatic or covalent binding by simulating the biotic system of the brain which aids the transportation of particle through the blood-brain barrier.30 However, the incorporation of lecithin in this nanoemulsion system might cause lysoderivatives’ formation.31 This formation prevented with the addition of Tween 80 as a surface active agents which can improve its physical stability by achieving more flexible interfacial film.32,33
 |
Figure 1 The solubility of aripiprazole in different types of oil containing 2% of lecithin. |
Application of Mathematical Model and Analysis of Variance (ANOVA)
The design matrix of the actual 30 experiments, together with the actual and predicted droplet size are illustrated in Table 2. The coefficients of the regression were obtained based on the CCRD and results of the experiments. The following second-order polynomial equation was accepted to acquire droplet size as in Equation (2):
(2)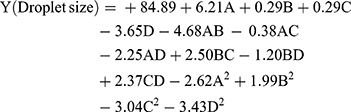
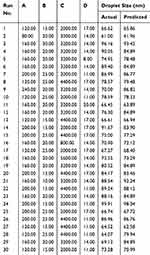 |
Table 2 A CCRD Matrix and Outcome for the Model of Nanoemulsions Containing Aripiprazole |
where A is the symbol of the overhead stirring time, B is the symbol of shear time, C is the the symbol of the shear rate, and D is the symbol of the cycle of the homogenizer. In this equation, the synergistic effect can be shown as a positive sign in a symbol. The negative symbol is shown as an antagonistic effect. A coefficient estimation with negative values is shown as a negative effect of parameters on the droplet size.
The quadratic polynomial model was shown in Table 3. The p-value (0.0003) concluded that this model was statistically significant and acceptable to show the actual relationship between the response (droplet size, nm) and the significant variables. The non-significant “Lack of Fit” (F-value = 0.25) which was comparative to the pure error of the experiments indicated that the quadratic model data was suitable to represent the whole experimental data. The p-value was also deployed to identify the significance of every coefficient. When the p-value has a small value, the corresponding coefficient will be more significant. The table also showed that the coefficients of A, D, AB, A2, B2, C2, and D2 were statistically significant when the p-values below 0.05. The model showed the overhead stirring time (p-value of 0.0003 ≪ 0.05) was the most significant variable, followed by the cycle of high pressure homogenizer. However, the shear time and shear rate did not show any significant effect. Accuracy of the polynomial model is shown to be adequate when the coefficient of determination value (R2) of the model obtained was 0.9238, whereas the adjusted R2 of the model obtained was 0.8268 in Table 4.
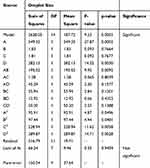 |
Table 3 ANOVA Results of the Fitted Quadratic Equation for Droplet Size of Nanoemulsions |
 |
Table 4 Regression Coefficients of the Final Reduced Model |
To identify the systematic departures from the assumptions used, the residual plot is an important detection method for establishing the regression equations. In Figure 2A, it was confirmed the assumption of normal distribution and the independence of the residuals when the data points on the plot were closed to a straight line.
 |
Figure 2 (A) The residual plot of experimental runs from CCRD; (B) scatter plot of predicted droplet size (nm) versus actual droplet size (nm). |
The correlation between actual and predicted values for the droplet size was shown in Figure 2B. The actual values were the experimental response data for a specific experimental run and the predicted values were the prediction result produced by the model. As can be seen in Figure 2B, the predicted values were corresponded with the actual values by R2 of 0.9238. It was shown that this model predicted accurately to study the relationship between the independent variables and the droplet size.
Mutual Effects of Process Parameter
The relationship between independent variables gave significant influences on the droplet size as shown as in given terms in Equation (2). Instead of studying one variable individually, a study about the relationship between those variables could be carried out to discover their significance for more advanced optimization.
Figure 3A shows the interaction between overhead stirring time (A) and shear time (B) as a function of droplet size. Shear rate (C) and cycles of high pressure homogenizer (D) were fixed variables. When the overhead stirring time was minimized, the droplet size of nanoemulsion was tended to decrease. As overhead stirring time and shear time increased, the droplet size of nanoemulsion containing aripiprazole correspondingly increased.
Figure 3B depicts the relationship of overhead stirring time (A) and shear rate (C) as a function of the droplet size. The fixed variables for this interaction were shear time (B) and cycles of high-pressure homogenizer (D). At increasing time of overhead stirring from 120 to 200 min, it caused the droplet size of nanoemulsion to increase correspondingly. However, the obtained result showed that the shear rate did not show a significant effect on the droplet size of nanoemulsions.
The predicted response surface plots for the reciprocation of overhead stirring time (A) and a cycle of high-pressure homogenizer (D) as a function of droplet size of nanoemulsion containing aripiprazole was illustrated in Figure 3C. Shear rate (B) and shear time (C) were fixed variables. Based on the observation, the minimum droplet size of nanoemulsion (62.59 nm) was obtained when the overhead stirring time decreased and the number of cycle of high-pressure homogenizer increased.
The independent variable that successfully contributed to find the smallest droplet size in this design was the number of cycle of high pressure homogenizer. In this research, it showed that the minimum droplet size could be obtained during 120 min of overhead stirring time and 11 cycles of high-pressure homogenizer. The decreasing of droplet size of nanoemulsion could be related to the increasing of mechanical energy. The oil and water mixture was forced to the intense turbulent and shear flow fields in the high-pressure homogenization method. This turbulence would cause the break-up of the dispersed phase into the tiniest droplet size.34 On the other hand, as increases the overhead stirring time, the droplet size of nanoemulsion containing aripiprazole tend to increase. Increasing the time of overhead stirring resulting large droplet size might be due to the coalescence and sedimentation.
Process Parameter Optimization and Model Validation for Nanoemulsion Containing Aripiprazole
The desirable droplet size of nanoemulsion can be obtained by optimum process parameters. Optimum process parameters are used to find the minimum droplet size and have lower overhead stirring time, higher shear rate, lower shear time, and lower number of cycles of the high-pressure homogenizer. To certify the optimum droplet sizes that been predicted by the model, an experiment of recommended optimum process parameter was carried out. Table 5 shows that the residual standard error percentage (RSE%) of optimum conditions for droplet size was 3.08%.
 |
Table 5 Optimal Condition Derived by CCRD Response Surface Methodology |
From Table 5, the optimum process parameter obtained were 120 min of overhead stirring time, 15 min of high shear homogenizer time, 4400 rpm of high shear homogenizer rate, and 11 cycles of the high-pressure homogenizer. The experimental run gave a desirable droplet size of nanoemulsion containing aripiprazole of 64.52 nm. The predicted value from the model was 62.59 nm which is very close to experimental data and confirmed the validity of the model. It can be said that based on the CCRD of RSM was a precise and valid tool to carry out a study about the optimization of the process parameter.
To confirm the validation of the model, three random process parameter conditions were prepared as tabulated in Table 6. The percentage of residual standard error (RSE %) of each validation set was calculated on the droplet size. In order to confirm the adequacy of the model, a comparison between the predicted values and the experimental values was made.
 |
Table 6 Validation Set for Three Different Processing Parameter Conditions of Nanoemulsion Containing Aripiprazole |
pH Measurements
Stable nanoemulsion containing aripiprazole has an initial pH in the range of 3–4. The pH was measured after high pressure homogenization. The low pH value of nanoemulsion containing aripiprazole was expected since aripiprazole was categorized as an acidic drug. A small amount of sodium hydroxide was used to adjust the emulsion pH to 7.4 and no significance effect on physicochemical properties and stability was observed upon addition of sodium hydroxide. The pH was brought to slightly alkaline because there was a reduced in pH during storage that may result from oil oxidation during storage. Further, it has been proposed that lecithin-stabilized emulsions were pronouncedly stable at pH 7 and 8.35
Viscosity and Osmolality Measurement
Viscosity measurement relates to flow characteristic of a system, thus for parenteral deliver product, the formulation should exhibit good syringe ability (easily drawn into a syringe) and inject ability (readily injected from the syringe). Injectability can impact the patient’s adherence even if some studies did not show viscous preparations to increase pain during injection. However, parenteral administration of high viscosity nanoemulsion might increase the blood viscosity which might lead to various cardio- or cerebrovascular events.36,37 A study on parenteral nanoemulsion reported that the acceptable value for parenteral administration was up to 15–20 mPa.s.38 The viscosity value of nanoemulsion containing aripiprazole was found to be 3.72 mPa.s and this value was within the specification for parenteral emulsions. The low viscosity value presented by nanoemulsion containing aripiprazole formulation reflected the safety measure prior to its application.
The parenteral product should be isotonic and euhydric (physiological pH) to avoid local damage on vascular endothelium and circulating blood cells.39 Incorporation of a small amount of glycerol assisted in generating emulsion with osmolality value that closes to the normal osmotic pressure at physiological pH (280–320 mOsm/kg). The osmolality value of nanoemulsion containing aripiprazole was 297 mOsm/kg which indicated the isotonic characteristic to blood.
Stability Study
In this formulation study, the stability of nanoemulsion was evaluated. The physical stability and appearance of optimized nanoemulsion containing aripiprazole were observed after centrifugation and after storage at 4 °C, 25 °C, and 45 °C within three months in Table 7. The optimized nanoemulsion was in stable conditions, with no phase separation or flocculation. The observation of incremental of droplet size was made throughout 3-month storage as the temperature of storage increased: 4 °C (<15% droplet size increment), 25 °C (<25% droplet size increment) and 45 °C (>50% droplet size increment) as shown in Figure 4. Therefore, a stability study under coalescence rate was carried out to determine the stability of optimum formulation containing aripiprazole.
 |
Table 7 Physical Stability of Optimized Nanoemulsion |
 |
Figure 4 Graph of droplet size (nm) observed for three months at three different temperatures. |
Coalescence Rate
Coalescence is a process when the breakage of liquid films occured between the droplets and leading to the formation of larger droplets.40 A plotted graph of 1/r2 against storage time depicted in Figure 5. According to Equation (1), a linear straight line should be shown in the plotted graph. Since no linear pattern line shown in this plotted graph, it concluded that there is no coalescence phenomenon occured in this nanoemulsion system. Therefore, the incremental of droplet size over storage time was not due to coalescence phenomenon.
 |
Figure 5 Coalescence rate of optimized nanoemulsion containing aripiprazole at different temperature storage. |
Conclusions
In this study, RSM is a powerful tool in the optimization of process parameters conditions of nanoemulsion containing aripiprazole. The interaction between independent variables and response variables can be studied with RSM. A central composite rotatable design (CCRD) of RSM predicted minimum droplet size based on the given polynomial regression models. Overhead stirring time, high shear time, high shear rate, and number cycle of high-pressure homogenizer showed a linear effect on the droplet size. From the result obtained, the coefficient of determination value (R2) was 0.9238 which is desirable and indicated it was in a good agreement with experimental data. The experimental run gave a desirable droplet size of nanoemulsion containing aripiprazole of 64.52 nm by increasing the overhead stirring time and lowering the cycles of the high-pressure homogenizer. The predicted value from the model was 62.59 nm which is very close to experimental data and confirmed the validity of the model. The optimized processing parameter with 120 min of overhead stirring time, 15 min of high shear time, 4400 rpm of high shear rate and 11 cycles of high-pressure homogenizer provided the most desirable criteria of droplet size for the nanoemulsion containing aripiprazole. The nanoemulsion was physically stable, with no phase separation or flocculation after centrifugation and after storage at 4 °C, 25 °C, and 45 °C within three months. The suitable pH value of 7.4, as well as low viscosity value and suitable osmolality value presented by nanoemulsion containing aripiprazole formulation, reflected the safety measure prior of its application. Thus, nanoemulsion containing aripiprazole showed excellent characteristic in the term of droplet size and could be used as a potential carrier for the parenteral delivery system.
Acknowledgments
The authors would like to convey an acknowledgment to Universiti Putra Malaysia for supporting this project under UPM-IPS.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Compton WM, Helzer JE. New methods in cross-cultural psychiatry: psychiatric illness in Taiwan and the United States. Am J Psychiatry. 1991;148(12):1697.
2. Li H, Luo J, Wang C, et al. Efficacy and safety of aripiprazole in Chinese Han schizophrenia subjects: a randomized, double-blind, active parallel-controlled, multicenter clinical trial. Schizophr Res. 2014;157(1):112–119. doi:10.1016/j.schres.2014.05.040
3. Potkin SG, Saha AR, Kujawa MJ, et al. Aripiprazole, an antipsychotic with a novel mechanism of action, and risperidone vs placebo in patients with schizophrenia and schizoaffective disorder. Arch Gen Psychiatry. 2003;60(7):681–690. doi:10.1001/archpsyc.60.7.681
4. McEvoy JP, Daniel DG, Carson WH, McQuade RD, Marcus RN. A randomized, double-blind, placebo-controlled, study of the efficacy and safety of aripiprazole 10, 15 or 20mg/day for the treatment of patients with acute exacerbations of schizophrenia. J Psychiatr Res. 2007;41(11):895–905. doi:10.1016/j.jpsychires.2007.05.002
5. Chan HY, Lin WW, Lin SK, et al. Efficacy and safety of aripiprazole in the acute treatment of schizophrenia in Chinese patients with risperidone as an active control: a randomized trial. J Clin Psychiatry. 2007;68(1):29–36. doi:10.4088/JCP.v68n0104
6. Mailman RB, Murthy V. Third generation antipsychotic drugs: partial agonism or receptor functional selectivity? Curr Pharm Des. 2010;16(5):488. doi:10.2174/138161210790361461
7. Wood M, Reavill C. Aripiprazole acts as a selective dopamine D2 receptor partial agonist. Expert Opin Investig Drugs. 2007;16:771–775. doi:10.1517/13543784.16.6.771
8. Stip E, Tourjman V. Aripiprazole in schizophrenia and schizoaffective disorder: a review. Clin Ther. 2010;32:S3–S20. doi:10.1016/j.clinthera.2010.01.021
9. Bhattacharjee J, El‐Sayeh HG. Aripiprazole versus typical antipsychotic drugs for schizophrenia. Cochrane Lib. 2008;3:CD006617 .
10. Belgamwar RB, El-Sayeh H. Aripiprazole versus placebo for schizophrenia. Cochrane Database Syst Rev. 2007;8.
11. Khanna P, Suo T, Komossa K, et al. Aripiprazole versus other atypical antipsychotics for schizophrenia. Cochrane Lib. 2014.
12. Abbott NJ. Dynamics of CNS barriers: evolution, differentiation, and modulation. Cell Mol Neurobiol. 2005;25(1):5–23. doi:10.1007/s10571-004-1374-y
13. Abbott NJ, Patabendige AA, Dolman DE, Yusof SR, Begley DJ. Structure and function of the blood–brain barrier. Neurobiol Dis. 2010;37(1):13–25. doi:10.1016/j.nbd.2009.07.030
14. Kaur IP, Bhandari R, Bhandari S, Kakkar V. Potential of solid lipid nanoparticles in brain targeting. J Control Release. 2008;127(2):97–109. doi:10.1016/j.jconrel.2007.12.018
15. Patel M, Souto EB, Singh KK. Advances in brain drug targeting and delivery: limitations and challenges of solid lipid nanoparticles. Expert Opin Drug Deliv. 2013;10(7):889–905. doi:10.1517/17425247.2013.784742
16. Burgess A, Nhan T, Moffatt C, Klibanov A, Hynynen K. Analysis of focused ultrasound-induced blood–brain barrier permeability in a mouse model of Alzheimer’s disease using two-photon microscopy. J Control Release. 2014;192:243–248. doi:10.1016/j.jconrel.2014.07.051
17. Solans C, Izquierdo P, Nolla J, Azemar N, Garcia Celma M. Nano-emulsions. Curr Opin Colloid Interface Sci. 2005;10(3–4):102–110. doi:10.1016/j.cocis.2005.06.004
18. Lovelyn C, Attama AA. Current state of nanoemulsions in drug delivery. J Biomater Nanobiotechnol. 2011;2(05):626. doi:10.4236/jbnb.2011.225075
19. Bezerra MA, Santelli RE, Oliveira EP, Villar LS, Escaleira LA. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta. 2008;76(5):965–977. doi:10.1016/j.talanta.2008.05.019
20. Gilmour SG. Response surface designs for experiments in bioprocessing. Biometrics. 2006;62(2):323–331. doi:10.1111/j.1541-0420.2005.00444.x
21. Myers RH, Montgomery DC, Vining GG, Borror CM, Kowalski SM. Response surface methodology: a retrospective and literature survey. J Qual Technol. 2004;36(1):53–77.
22. Tan SF, Masoumi HRF, Karjiban RA, et al. Ultrasonic emulsification of parenteral valproic acid-loaded nanoemulsion with response surface methodology and evaluation of its stability. Ultrason Sonochem. 2016;29:299–308. doi:10.1016/j.ultsonch.2015.09.015
23. Izadiyan Z, Basri M, Masoumi HRF, Karjiban RA, Salim N, Shameli K. Modeling and optimization of nanoemulsion containing Sorafenib for cancer treatment by response surface methodology. Chem Cent J. 2017;11(1):21. doi:10.1186/s13065-017-0248-6
24. Musa SH, Basri M, Masoumi HRF, et al. Formulation optimization of palm kernel oil esters nanoemulsion-loaded with chloramphenicol suitable for meningitis treatment. Colloids Surf B. 2013;112:113–119. doi:10.1016/j.colsurfb.2013.07.043
25. Masoumi HRF, Basri M, Samiun WS, Izadiyan Z, Lim CJ. Enhancement of encapsulation efficiency of nanoemulsion-containing aripiprazole for the treatment of schizophrenia using mixture experimental design. Int J Nanomedicine. 2015;10:6469. doi:10.2147/IJN
26. Samiun WS, Basri M, Masoumi HRF, Khairudin N. The prediction of the optimum compositions of a parenteral nanoemulsion system loaded with a low water solubility drug for the treatment of schizophrenia by artificial neural networks. RSC Adv. 2016;6(17):14068–14076. doi:10.1039/C5RA26243G
27. Geers H, Witt W. Direct calculation of the volume based particle size distribution from PCS or PCCS measurements. Proc Part Sys Anal. 2008.
28. Witt W, Geers H, Aberle L. Measurement of particle size and stability of nanoparticles in opaque suspensions and emulsions with Photon Cross Correlation Spectroscopy (PCCS). Part Sys Anal Harrogate UK. 2003.
29. Keng P, Basri M, Zakaria M, et al. Newly synthesized palm esters for cosmetics industry. Ind Crops Prod. 2009;29(1):37–44.
30. Krol SJJ. Challenges in drug delivery to the brain: nature is against us. J Controlled Release. 2012;164(2):145–155. doi:10.1016/j.jconrel.2012.04.044
31. Floyd AGJPS, Today T. Top ten considerations in the development of parenteral emulsions. Pharm Sci Technolo Today. 1999;2(4):134–143. doi:10.1016/S1461-5347(99)00141-8
32. Yilmaz E, Borchert -H-HJE. Design of a phytosphingosine-containing, positively-charged nanoemulsion as a colloidal carrier system for dermal application of ceramides. Biopharmaceutics. 2005;60(1):91–98.
33. Trotta M, Pattarino F, Ignoni TJE. Stability of drug-carrier emulsions containing phosphatidylcholine mixtures. Biopharmaceutics. 2002;53(2):203–208.
34. Juttulapa M, Piriyaprasarth S, Takeuchi H, Sriamornsak P. Effect of high-pressure homogenization on stability of emulsions containing zein and pectin. Asian J Pharm Sci. 2017;12(1):21–27. doi:10.1016/j.ajps.2016.09.004
35. Cui J, Wang L, Sun GG, Liu LN. Characterization of a Trichinella spiralis 31 kDa protein and its potential application for the serodiagnosis of trichinellosis. Acta Tropica. 2015;142:57–63. doi:10.1016/j.actatropica.2014.10.017
36. Song SH, Kim JH, Lee JH, Yun Y-M, Choi D-H, Kim HYJ. Elevated blood viscosity is associated with cerebral small vessel disease in patients with acute ischemic stroke. BMC Neurol. 2017;17(1):20.
37. Ott EO, Lechner H, Aranibar AJS. High blood viscosity syndrome in cerebral infarction. Stroke. 1974;5(3):330–333. doi:10.1161/01.str.5.3.330
38. Berteau C, Filipe-Santos O, Wang T, Rojas HE, Granger C, Schwarzenbach F. Evaluation of the impact of viscosity, injection volume, and injection flow rate on subcutaneous injection tolerance. Med Devices. 2015;8:473.
39. Roethlisberger D, Mahler H-C, Altenburger U, Pappenberger AJ. If euhydric and isotonic do not work, what are acceptable pH and osmolality for parenteral drug dosage forms? J Pharm Sci. 2017;106(2):446–456. doi:10.1016/j.xphs.2016.09.034
40. Fredrick E, Walstra P, Dewettinck KJA. Factors governing partial coalescence in oil-in-water emulsions. Adv Colloid Interface Sci. 2010;153(1–2):30–42. doi:10.1016/j.cis.2009.10.003
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

