Back to Journals » Drug Design, Development and Therapy » Volume 15
Optimal Empiric Polymyxin B Treatment of Patients Infected with Gram-Negative Organisms Detected Using a Blood Antimicrobial Surveillance Network in China
Authors Wu X, Huang C, Wang H, Ji J , Ying C , Xiao Y
Received 31 March 2021
Accepted for publication 2 June 2021
Published 17 June 2021 Volume 2021:15 Pages 2593—2603
DOI https://doi.org/10.2147/DDDT.S313714
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Manfred Ogris
Xingbing Wu,1 Chen Huang,2 Hui Wang,2 Jinru Ji,3 Chaoqun Ying,3 Yonghong Xiao3
1Department of Infectious Disease, Ningbo Medical Center Lihuili Hospital, Ningbo, People’s Republic of China; 2Department of Respiratory Medicine, Ningbo Medical Center Lihuili Hospital, Ningbo, People’s Republic of China; 3State Key Laboratory for Diagnosis and Treatment of Infectious Disease, Collaborative Innovation Center for Diagnosis and Treatment of Infectious Diseases, The First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, People’s Republic of China
Correspondence: Chen Huang
Department of Respiratory Medicine, Ningbo Medical Center Lihuili Hospital, Ningbo, 315000, People’s Republic of China
Email [email protected]
Yonghong Xiao
State Key Laboratory for Diagnosis and Treatment of Infectious Diseases, Collaborative Innovation Center for Diagnosis and Treatment of Infectious Diseases, The First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, 310003, People’s Republic of China
Tel +865 718 723 6421
Email [email protected]
Background: Few pharmacodynamics studies to date have evaluated the efficacy and safety of polymyxin B (PMB) in treating patients with bloodstream infections (BSIs) in China.
Methods: Patients with BSIs were identified using an antimicrobial surveillance network, and their pathogens were isolated. Patients were treated with a loading dose of PMB followed by either a weight-based or weight-independent maintenance dose. Monte Carlo simulation was utilized to calculate the probability of target attainment (PTA) and cumulative fraction of response (CFR) against Gram-negative organisms in patients with normal or decreased renal function.
Results: A total of 10,066 Gram-negative organisms, including 5500 Escherichia coli (Eco), 2519 Klebsiella pneumoniae (Kpn), 501 Acinetobacter baumannii (Aba), were isolated from patients with BSIs. Although these strains were highly resistant to carbapenem, they remained susceptible to PMB. Among patients with renal impairment (mean CrCL, 42 mL/min), a PMB 2.5 mg/kg loading dose followed by a maintenance dose of 60 mg q12h reached ≥ 90% PTA against isolates with an MIC of 2 mg/L, with a low risk of toxicity. Among patients with normal renal function (mean CrCL, 123 mL/min), all simulated regimens showed PTAs of 25– 80%. A weight-based loading dose followed by either a weight-based or weight-independent maintenance dose showed a promising CFR, especially in patients with renal impairment, with CFRs ≥ 90% against carbapenem-resistant Eco, Kpn, and Aba. Simulated regimens showed a disappointing CFR (< 80%) against carbapenem-resistant Pae in patients with normal renal function. Based on the optimal balance of efficacy and toxicity, a fixed maintenance dose of 60 mg q12h among patients with renal impairment yielded a CFR similar to regimens based on total body weight and was associated with a probability of toxicity (12.5%) significantly lower than that of simulations based on total body weight. Among patients with normal renal function, a weight-based maintenance dose of 1.25 mg/kg q12h achieved a higher CFR than a fixed maintenance dose, without significantly increasing toxicity.
Conclusion: A 2.5 mg/kg loading dose of PMB is optimal, regardless of renal function. A fixed maintenance dose of 60 mg q12h is recommended for empirical treatment of patients with renal impairment infected with Eco, Kpn, and Aba, whereas a weight-based maintenance dose of 1.25 mg/kg is recommended for patients with normal renal function.
Keywords: polymyxin B, carbapenem-resistant gram-negative organisms, bloodstream infection, Monte Carlo, toxicity
Introduction
The widespread dissemination of bacterial strains resistant to antimicrobial treatment has posed a serious threat to public health.1 Disappointing clinical outcomes have been observed in patients with bloodstream infections (BSIs) with multidrug-resistant or carbapenem-resistant organisms.2,3 Data from the China Antimicrobial Surveillance Network (CHINET) have revealed significant increases in the rates of resistance of Klebsiella pneumoniae (Kpn) and Acinetobacter baumannii (Aba) strains to most classes of antimicrobial drugs, resulting in higher morbidity and mortality rates and higher medical costs in patients infected with such strains.4,5 Because of the limited effectiveness of antibiotics against life-threatening carbapenem-resistant isolates, the use of ceftazidime-avibactam, tigecycline, and polymyxins as last-resort options has increased. Ceftazidime-avibactam, which recently became clinical available in China, has exhibited excellent in vitro activity against carbapenem-resistant Enterobacteriaceae (mainly Kpn) isolated from CHINET 2018.6 However, ceftazidime-avibactam demonstrated limited activity (MIC50/90, 64/>64 mg/L) against Aba isolates, 79.2% of which were carbapenem-resistant. Polymyxins were approved for treatment in the 1950s, but they have a narrow therapeutic window and a potential to cause severe nephrotoxicity.7,8 Although polymyxins are regarded as alternative agents against carbapenem-resistant organisms in the “bad bugs, no drugs” era, their optimal dosing regimens remain poorly defined, especially that of polymyxin B (PMB). Because in vitro susceptibility is insufficient to select rational antibiotic dosing regimens, the integration of population pharmacokinetic (PPK) parameters and MIC data is needed to optimize PMB dosing regimens.
The pharmacokinetic/pharmacodynamic (PK/PD) relationships of the two clinically available polymyxins, colistimethate sodium (CMS) and PMB, are not fully understood.9,10 CMS is administrated parenterally as an inactive prodrug, with the in vivo conversion of CMS to colistin exhibiting a relatively large degree of interindividual variability. Thus, plasma concentrations of colistin increase slowly, leading to difficulties in optimal dose selection.11 Conversely, PMB appears to have remarkably low interpatient variability, making it easier to predict its PK.12 In addition, PMB has lower nephrotoxicity rates than CMS,13,14 suggesting that PMB has superior pharmacological characteristics in the treatment of infections. Because CMS is more widely available worldwide, clinical experience with PMB is lower than that with CMS.14,15
To date, no studies focusing on optimal PMB dosing regimens to treat patients in China with BSIs have been based on PK parameters and nationwide surveillance of MIC data. The present study was designed to (i) evaluate the distribution of PMB MCIs against Gram-negative organisms in patients with BSIs; (ii) re-evaluate the reasonable clinical breakpoints of PMB using Monte Carlo simulation (MCS) in Chinese populations; and (iii) determine the regimen involved in the prompt initiation of appropriate empirical antimicrobial therapy in patients with BSIs.
Materials and Methods
Bacterial Isolates
Gram-negative bacterial isolates, including Escherichia coli (Eco), Klebsiella pneumoniae (Kpn), Pseudomonas aeruginosa (Pae), Acinetobacter baumannii (Aba) and other strains, isolated from the Blood Bacterial Resistant Investigation Collaborative System (BRICS), were collected from patients in 60 tertiary and secondary hospitals in China between January 2018 and December 2019. Pathogens were isolated and identified by microbiological methods using matrix-assisted laser desorption ionization-time-of-flight (MALDI-TOF, Bruker) mass spectrometry (MS).16
Antimicrobial Susceptibility Testing
The MICs for imipenem and PMB were determined by the agar dilution and broth microdilution methods, respectively, as described by the Clinical and Laboratory Standards Institute (CLSI). For imipenem, MICs of ≤2 mg/L and ≥4 mg/L were defined as susceptible and resistant, respectively, whereas for PMB, MICs of ≤2 mg/L and ≥4 mg/L were defined as intermediate and resistant, respectively.17 E. coli strain 25,922 from the American Type Culture Collection (ATCC) was used for quality control.
Population PK Model and PK/PD Target
The PPK data for PMB were obtained from a recently published study on critically ill patients in China, with a two-compartment model used to describe the time-concentration relationship for PMB.18 In that study, patients were grouped into those with normal renal function (n=37) and renal insufficiency (n=33). The mean creatinine clearance (CrCL) in these two groups were 123.3 mL/min (range, 81.6–315.2 mL/min) and 42.0 mL/min (range, 15.6–77.6 mL/min), respectively. The detailed PPK parameters are listed in Table 1. Covariate modeling showed that PMB PK was not significantly associated with patient age, sex, CrCL or total body weight (WT). Patients receiving any form of renal replacement therapy were not enrolled in the PPK model.
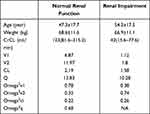 |
Table 1 Pharmacokinetic Parameters Used in the Monte Carlo Simulation |
PMB displays concentration-dependent antibacterial effects against bacterial isolates. AUC/MIC is the optimal PK/PD index, closely associated with clinical efficacy. Based on data from a murine thigh infection model, the PD target had to be over 50 mg∙h/L for promising outcomes.19 In addition, consensus guidelines indicated that fAUC/MIC was the PK/PD target that best correlated with bacterial killing.20 Because the molecular characteristics and in vitro antibacterial activities of colistin and PMB against pathogens are similar, a fAUC/MIC of approximately 20 for colistin was responsible for 2-log killing of Pae and Aba.21 The unbound fraction of PMB was defined as 0.42 derived from Sandri study,12 making the PK/PD targets of AUC/MIC and fAUC/MIC approximately identical. AUC/MIC was defined as the PK/PD target for further analysis. In addition, PMB has a narrow therapeutic window, and its nephrotoxicity is a dose‐limiting adverse effect. Based on a pharmacometric meta-analysis of PMB nephrotoxicity, an AUCss,24h of 100 mg∙h/L was regarded as a predictor of nephrotoxicity,18,22 and a target AUCss,24h >100 mg∙h/L was defined as a threshold associated with an increased risk of nephrotoxicity. All the simulated dosing regimens were tested to calculate the possibility of clinical cure and toxic effects.
Monte Carlo Simulation
To date, widely used PMB dosing regimens have been based on total body weight. International consensus guidelines recommend that PMB dosage not be adjusted in patients with renal impairment, a recommendation inconsistent with the FDA approved package inserts.23,24 Furthermore, little PK information is available for patients at the extremes of body weight.12 Because PMB has a narrow therapeutic window, optimal dosages that balance efficacy and toxicity have not yet been determined. To optimize the balance between efficacy and toxicity in different populations, fixed and weight-based PMB maintenance dose were simulated. These included a loading dose of 2.5 mg/kg followed by a maintenance dose of 60, 80, or 100 mg q12h; a loading dose of 2.2 mg/kg followed by a maintenance dose of 1 or 1.25 mg/kg q12h; a loading dose of 2.5 mg/kg followed by a maintenance dose of 1.25 or 1.5 mg/kg q12h; and a loading dose of 3 mg/kg followed by a maintenance dose of 1.5 mg/kg q12h. A 5000 patient MCS was utilized to calculate the probability of target attainment (PTA) and the cumulative fraction of response (CFR) of each dosage regimen against a bacterial MIC population using Crystal Ball software (version 11.1.2.4; Oracle). During simulation, the PK parameters obeyed a log-normal distribution, whereas MIC obeyed a discrete distribution. Moreover, the steady state Cmin and Cmax were calculated. Dosing regimens were considered optimal if they provided ≥90% CFR and a low risk of drug-related nephrotoxicity. Dosing regimens with a CFR between 80% and 90% were regarded as providing moderate probabilities of treatment success.25
Results
Microbiological Data
A total of 10,066 Gram-negative bacterial isolates (5500 Eco, 2519 Kpn, 546 Pae, and 501 Aba) were collected from BRICS (Table 2). Of the total, Eco, Kpn, Pae, and Aba Gram-negative isolates collected from patients with BSIs, 1169 (11.6%), 82 (1.5%), 506 (20.1%), 210 (38.5%), and 315 (62.9%), respectively, were carbapenem-resistant. Compared with most antibiotics, which show poor antibacterial activity against carbapenem-resistant organisms, PMB exhibited favorable in vitro activity against all the tested organisms, including the carbapenem-resistant strains. PMB had MIC50 and MIC90 values of 0.5 and 1.0 mg/mL, respectively, against carbapenem-resistant organisms, with PMB having both MIC50 and MIC90 values of 2 mg/mL against Gram-negative organisms. PMB had mildly reduced antibacterial activity against Pae than against the other Gram-negative species.
 |
Table 2 The in vitro Activities of PMB Against Gram-Negative Organisms Isolated from BRICS |
Monte Carlo Simulation
A 5000 subject MCS based on PPK data for each dosing regimen was performed to calculate PTA and CFR. The relationships between MIC and PTA for various dosing regimens and CrCL are presented in Figure 1. Based on the PK/PD target of AUC/MIC ≥50, all the simulated regimens reached ≥90% PTA against isolates with MICs of ≤1 mg/L in patients weighing 65 kg, regardless of renal function. However, none of the simulated regimens achieved >90% PTA at the current CLSI breakpoint of 2 mg/L in patients with normal renal function, whereas most simulated regimens achieved promising PTA in subjects with renal impairment.
 |
Figure 1 Probability of target attainment (PTA) of 50 AUC/MIC for the simulated polymyxin B dosing regimens in patients with normal and decreased renal function. |
The possibility of nephrotoxicity of each simulated PMB dosing regimen was also determined (Figure 2). MICs of 1 mg/L for subjects with normal renal function and 2 mg/L for subjects with reduced renal function were used to calculate the PTA of different PMB dosing regimens reaching ≥50 AUC/MIC. Based on the combined efficacy for reaching the PTA target and the probability of developing drug-related toxicity, patients with normal renal function weighing 50, 65, and 80 kg should receive PMB loading doses of 2.5, 2.5, and 2 mg/kg, respectively, followed by maintenance doses of 1.25, 1.25, and 1 mg/kg, respectively, to treat organisms with a MIC of 1 mg/L. Total body weight-based dosing strategies, however, may not be optimal in balancing efficacy and toxicity among patients with renal impairment. These patients should receive a weight-based loading dose followed by a fixed maintenance dose. Compared with weight-independent dosing strategies, weight-based dosing resulted in increased toxicity in higher-weight patients. For example, the probability of toxicity was >50% in patients weighing 65 kg who received a PMB loading dose of 2 mg/kg loading dose followed by a maintenance dose of 1.25 mg/kg q12h. A higher risk was observed in patients weighing 80 kg, even in those receiving a maintenance dose of 1 mg/kg q12h. By contrast, fixed maintenance dose strategies provided a better balance between efficacy and toxicity in patients of different body weight strata with reduced renal function.
Calculations of simulated Cmin and Cmax showed that these values were highly heterogeneous in patients with reduced renal function (Table 3). Cmin and Cmax ranges were 0.94–3.01 mg/L and 4.55–14.56 mg/L, respectively, in patients with normal renal function, but were 0.07–0.23 mg/L and 12.94–41.41 mg/L, respectively, in patients with renal impairment.
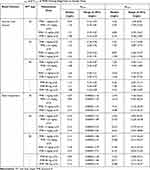 |
Table 3 Simulated Cmin and Cmax of PMB Dosing Regimens at Steady State |
The simulated CFR results for weight-based and weight-independent maintenance doses are shown in Figures 3 and 4, respectively. Among patients with normal renal function weighing 65 kg, a PMB loading dose of 3 mg/kg followed by a maintenance dose of 1.5 mg/kg q12h displayed the highest CFR, whereas a loading dose of 2.5 mg/kg followed by a maintenance 1.25 mg/kg q12h achieved similar CFR, with >90% probabilities against carbapenem-resistant Eco (CR-Eco), followed by 88.8% against CR-Kpn, 83.0% against CR-Aba and 70.0% against CR-Pae. The relatively low risk of toxicity and low medical cost of the latter regimen were beneficial to patients when compared with a maintenance dose of 1.5 mg/kg q12h.
Among patients with renal impairment, the CFR of a fixed maintenance dose was similar to that based on total body weight and was associated with a significantly lower probability of toxicity than that obtained in simulations based on total body weight. These findings indicate that a fixed maintenance dose was suitable for different body weight strata of patients with decreased renal function, especially at the extremes of body weight. Among underweight patients, a fixed maintenance dose of 60 mg q12h achieved higher CFRs than regimens based on total body weight. Meanwhile, the risk of toxicity remained relatively low (~12.5%). CFR could be further increased in overweight patients with renal impairment, as a weight-based loading dose was required, followed by a fixed maintenance dose. A fixed maintenance dose therefore resulted in a significantly lower risk of nephrotoxicity than a weight-based maintenance dose in patients with renal impairment.
Discussion
The development of antibiotic resistance in Gram-negative bacterial isolates has emerged as a global public health concern. Inappropriate antibiotic therapy for patients with BSIs has been associated with poor outcomes.26,27 Unfortunately, our results revealed a high prevalence of CR-Kpn, Pae, and Aba in patients with BSIs, similar to the CHINET and Chinese Meropenem Surveillance Study (CMSS) results, indicating that highly resistant pathogens were widespread throughout China.5,28 Although PMB has shown promising in vitro antibacterial activity against carbapenem-resistant Gram-negative strains, few studies to date have compared the efficacy and safety of PMB and CMS dosing regimens in the treatment of BSIs. Most areas in China may have access only to CMS, not to PMB. In addition, large scale surveillance data of the MICs of bacterial strains toward PMB are urgently needed. Because PMB has superior PK characteristics, optimizing its clinical use is crucial in managing difficult-to-treat patients with BSIs.
In the present study, MCS was performed using different PMB dosage regimens against Gram-negative organisms isolated from BRICS. The number of pathogens included in the present study was 10,066, significantly larger than the numbers included in previous studies.29 Unlike previous studies, the focus of the present study was on the treatment of BSIs. Furthermore, the PPK data were derived from the Chinese population. The integration of domestic PPK parameters and population MIC data was useful in optimizing PMB dosage regimens in China.30 To our knowledge, the BRICS is the largest blood antimicrobial surveillance in China.
Although the efficacy of PMB may be enhanced by administration of higher loading doses, higher loading doses may increase the risk of toxicity, as PMB has a narrow therapeutic window. Our simulation results demonstrated that a loading dose of 2.5 mg/kg followed by a maintenance dose of 1.25 mg/kg could provide sufficient antimicrobial exposure with low risks of drug-related toxicity in the treatment carbapenem-resistant organisms (except for CR-Pae) among patients with normal renal function. Higher doses increased the risk of nephrotoxicity risk, with a maintenance dose of 1.5 mg/kg q12h resulting in a >50% possibility of toxicity in patients weighing 80 kg. This situation was significantly amplified in patients with renal impairment. Thus a fixed maintenance dose may be optimal in overcoming toxicity in renally impaired patients. Our findings suggest that a loading dose of 2.5 mg/kg is optimal, regardless of renal function, followed by a fixed maintenance dose in patients with impaired renal function. Consistent with our findings, a weight-based loading dose followed by a fixed maintenance dose of PMB has been reported to maximize efficacy while balancing toxicity concerns, whereas weight-based loading and maintenance doses did not.22 Furthermore, a PMB PPK study found that CL and Q values differed significantly in patients with normal and reduced renal function, whereas the variance and median AUCss,24h changes remained within a 2-fold range in patients treated with a weight-based maintenance dose, a finding commonly accepted as the threshold justification for dosing adjustment.18,31 Although dosing adjustment may not be necessary in patients with renal impairment, this strategy has been associated with increased toxicity rates in patients with decreased renal function, with over 50% of simulated AUCss,24h being >100 mg∙h/L.18 Moreover, weight-based maintenance doses have been associated with increased toxicity in higher-weight patients.32,33 In the present study, a fixed maintenance dose of 60 mg q12h provided sufficient antimicrobial coverage against carbapenem-resistant Gram-negative organisms in patients with renal impairment, along with a relative low risk of toxicity. Thus, a strategy that includes a weight independent maintenance dose may be optimal in patients with renal insufficiency, providing the optimal balance between CFR and the risk of toxicity. TDM was recommended if applicable and patients should be clinically monitored for adverse reactions to PMB, such as nephrotoxicity and neurotoxicity. An adaptive feedback control algorithm has been reported to optimize the personalization of PMB dosing regimens.34 This algorithm significantly increased the attainment of AUC0–24 values within the target window (>95% vs 71%).
A relatively disappointing CFR was observed in patients infected with Pae, regardless of renal function. Suboptimal PMB monotherapy dosing regimens have yielded relatively low clinical cure rates (57–75%).35,36 Moreover, PMB monotherapy often resulted to the amplification of colistin-resistant subpopulations in heteroresistant strains, especially in Kpn.37 Acquisition of resistance has been reported after PMB therapy.38,39 Although PMB showed promising CFR against carbapenem-resistant Eco, Kpn, and Aba, studies have reported that PMB combination therapy may be required. PMB monotherapy showed excellent simulated results in treating carbapenem-resistant organisms, the combination of PMB with other agents could further prevent the emergence of pan-resistant organisms and improve patient outcomes. A previous meta-analysis indicated that mortality was significantly higher in patients treated with PMB monotherapy than in patients treated with combinations of PMB with tigecycline, Fosfomycin, and aminoglycosides, especially those infected with Kpn BSIs. These findings may be useful in empirically treating patients infected with carbapenem-resistant Gram-negative organisms.
This study had several limitations. First, the PPK model was developed from 462 plasma samples obtained from 37 normal patients and 33 with reduced renal function.18 The rich PK properties of PMB may be not fully evaluated, resulting in a bias in simulating PMB concentration. Second, the MIC of PMB against organisms from China may not be representative of the MIC distributions in other regions of the world. Third, combination regimens are often empirically prescribed for patients infected with carbapenem-resistant or pandrug-resistant organisms. Unfortunately, MCS was inapplicable in these situations.
Conclusions
To our knowledge, this is the largest study to date evaluate the efficacy and safety of PMB dosing regimens in Chinese patients infected with carbapenem-resistant microorganisms. Patients should be administered a loading dose of 2.5 mg/kg, regardless of renal function. This should be followed by a fixed maintenance dose of 60 mg q12h in the empirical treatment of CR-Eco, Kpn, and Aba infections in patients with impaired renal function. By contrast, patients with normal renal function should be administered a weight-based maintenance dose of 1.25 mg/kg PMB. Additional studies are needed to formulate the exact treatment strategies for patients with renal impairment.
Data Sharing Statement
The datasets used and analyzed during the current study are available from the corresponding author of Yonghong Xiao on reasonable request.
Acknowledgments
We thank all BRICS participants for their contribution to the programme.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
This work was supported by the Natural Science Foundation of NingBo (grant number. 2019A610232).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Ferri M, Ranucci E, Romagnoli P, Giaccone V. Antimicrobial resistance: a global emerging threat to public health systems. Crit Rev Food Sci Nutr. 2017;57(13):2857–2876. doi:10.1080/10408398.2015.1077192
2. Xu L, Sun X, Ma X. Systematic review and meta-analysis of mortality of patients infected with carbapenem-resistant Klebsiella pneumoniae. Ann Clin Microbiol Antimicrob. 2017;16(1):18. doi:10.1186/s12941-017-0191-3
3. Giannella M, Trecarichi EM, De Rosa FG, et al. Risk factors for carbapenem-resistant Klebsiella pneumoniae bloodstream infection among rectal carriers: a prospective observational multicentre study. Clin Microbiol Infect. 2014;20(12):1357–1362. doi:10.1111/1469-0691.12747
4. Hu FP, Guo Y, Zhu DM, et al. Resistance trends among clinical isolates in China reported from CHINET surveillance of bacterial resistance, 2005–2014. Chin J Infect Chemother. 2017;17(1):93–99.
5. Hu F, Guo Y, Zhu D. CHINET surveillance of bacterial resistance across tertiary hospitals in 2019. Chin J Infect Chemother. 2020;20(03):6–16.
6. Yang Y, Guo Y, Yin D, et al. In vitro activity of cefepime-zidebactam, ceftazidime-avibactam, and other comparators against clinical isolates of Enterobacterales, Pseudomonas aeruginosa, and Acinetobacter baumannii: results from China Antimicrobial Surveillance Network (CHINET) in 2018. Antimicrob Agents Chemother. 2020;65:1. doi:10.1128/AAC.01726-20
7. Koch-Weser J, Sidel VW, Federman EB, Kanarek P, Finer DC, Eaton AE. Adverse effects of sodium colistimethate. Manifestations and specific reaction rates during 317 courses of therapy. Ann Intern Med. 1970;72(6):857–868. doi:10.7326/0003-4819-72-6-857
8. Storm DR, Rosenthal KS, Swanson PE. Polymyxin and related peptide antibiotics. Annu Rev Biochem. 1977;46:723–763. doi:10.1146/annurev.bi.46.070177.003451
9. Nation RL, Velkov T, Li J. Colistin and polymyxin B: peas in a pod, or chalk and cheese? Clin Infect Dis. 2014;59(1):88–94. doi:10.1093/cid/ciu213
10. Huang Chen, Xiao Yonghong. Polymyxins: review on clinical use and dilemma. Her Med. 2020;39(1):10–16.
11. Garonzik SM, Li J, Thamlikitkul V, et al. Population pharmacokinetics of colistin methanesulfonate and formed colistin in critically Ill patients from a Multicenter Study provide dosing suggestions for various categories of patients. Antimicrob Agents Chemother. 2011;55(7):3284–3294. doi:10.1128/AAC.01733-10
12. Sandri AM, Landersdorfer CB, Jacob J, et al. Population pharmacokinetics of intravenous polymyxin B in critically ill patients: implications for selection of dosage regimens. Clin Infect Dis. 2013;57(4):524–531. doi:10.1093/cid/cit334
13. Kassamali Z, Danziger L. To B or not to B, that is the question: is it time to replace colistin with polymyxin B? Pharmacotherapy. 2015;35(1):17–21. doi:10.1002/phar.1510
14. Phe K, Lee Y, McDaneld PM, et al. In vitro assessment and multicenter cohort study of comparative nephrotoxicity rates associated with colistimethate versus polymyxin B therapy. Antimicrob Agents Chemother. 2014;58(5):2740–2746. doi:10.1128/AAC.02476-13
15. Akajagbor DS, Wilson SL, Shere-Wolfe KD, Dakum P, Charurat ME, Gilliam BL. Higher incidence of acute kidney injury with intravenous colistimethate sodium compared with polymyxin B in critically ill patients at a tertiary care medical center. Clin Infect Dis. 2013;57(9):1300–1303. doi:10.1093/cid/cit453
16. Wattal C, Oberoi JK, Goel N, Raveendran R, Khanna S. Matrix-assisted laser desorption ionization time of flight mass spectrometry (MALDI-TOF MS) for rapid identification of micro-organisms in the routine clinical microbiology laboratory. Eur J Clin Microbiol. 2017;36(5):807–812. doi:10.1007/s10096-016-2864-9
17. Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing.
18. Wang P, Zhang Q, Zhu Z, et al. Comparing population pharmacokinetics and acute kidney injury of polymyxin B in Chinese patients with and without renal insufficiency. Antimicrob Agents Chemother. 2021;65(2):e01900–20.
19. Cheah SE, Wang J, Nguyen VT, Turnidge JD, Li J, Nation RL. New pharmacokinetic/pharmacodynamic studies of systemically administered colistin against Pseudomonas aeruginosa and Acinetobacter baumannii in mouse thigh and lung infection models: smaller response in lung infection. J Antimicrob Chemother. 2015;70(12):3291–3297. doi:10.1093/jac/dkv267
20. Xellia Pharmaceuticals APS (2016) Polymyxin B sulfate injection [package Insert]. 16 February 2016. Xellia Pharmaceuticals APS, Denmark.
21. Dudhani RV, Turnidge JD, Nation RL, Li J. fAUC/MIC is the most predictive pharmacokinetic/pharmacodynamic index of colistin against Acinetobacter baumannii in murine thigh and lung infection models. J Antimicrob Chemother. 2010;65(9):1984–1990. doi:10.1093/jac/dkq226
22. Miglis C, Rhodes NJ, Avedissian SN, et al. Population pharmacokinetics of polymyxin B in acutely ill adult patients. Antimicrob Agents Chemother. 2018;62(3):3. doi:10.1128/AAC.01475-17
23. Li J, Coulthard K, Nation RL. Labelling conventions and product package insert of parenteral polymyxins: factors causing potential medication errors and impeding optimal clinical use. Adv Exp Med Biol. 2019;1145:133–141.
24. Tsuji BT, Pogue JM, Zavascki AP, et al. International consensus guidelines for the optimal use of the polymyxins: endorsed by the American College of Clinical Pharmacy (ACCP), European Society of Clinical Microbiology and Infectious Diseases (ESCMID), Infectious Diseases Society of America (IDSA), International Society for Anti-infective Pharmacology (ISAP), Society of Critical Care Medicine (SCCM), and Society of Infectious Diseases Pharmacists (SIDP). Pharmacotherapy. 2019;39(1):10–39. doi:10.1002/phar.2209
25. Bradley JS, Dudley MN, Drusano GL. Predicting efficacy of antiinfectives with pharmacodynamics and Monte Carlo simulation. Pediatr Infect Dis J. 2003;22(11):982–992. doi:10.1097/01.inf.0000094940.81959.14
26. De Rosa FG, Pagani N, Fossati L, et al. The effect of inappropriate therapy on bacteremia by ESBL-producing bacteria. Infection. 2011;39(6):555–561. doi:10.1007/s15010-011-0201-x
27. Micek S, Johnson MT, Reichley R, Kollef MH. An institutional perspective on the impact of recent antibiotic exposure on length of stay and hospital costs for patients with gram-negative sepsis. BMC Infect Dis. 2012;12(1):56. doi:10.1186/1471-2334-12-56
28. Wang Q, Wang Z, Zhang F, et al. Long-term continuous antimicrobial resistance surveillance among nosocomial gram-negative bacilli in China from 2010 to 2018 (CMSS). Infect Drug Resist. 2020;13:2617–2629. doi:10.2147/IDR.S253104
29. Wang C, Hao W, Jin Y, Shen C, Wang B. Pharmacokinetic/pharmacodynamic modeling of seven antimicrobials for empiric treatment of adult bloodstream infections with gram-negative bacteria in China. Microb Drug Resist. 2020;26(12):1559–1567. doi:10.1089/mdr.2019.0152
30. Yu X-B, Jiao Z, Zhang C-H, et al. Population pharmacokinetic and optimization of polymyxin B dosing in adult patients with various renal functions. Br J Clin Pharmacol. 2021;87(4):1869–1877. doi:10.1111/bcp.14576
31. Manchandani P, Thamlikitkul V, Dubrovskaya Y, et al. Population pharmacokinetics of polymyxin B. Clin Pharmacol Ther. 2018;104(3):534–538. doi:10.1002/cpt.981
32. Kubin CJ, Ellman TM, Phadke V, Haynes LJ, Calfee DP, Yin MT. Incidence and predictors of acute kidney injury associated with intravenous polymyxin B therapy. J Infect. 2012;65(1):80–87. doi:10.1016/j.jinf.2012.01.015
33. Rigatto MH, Behle TF, Falci DR, et al. Risk factors for acute kidney injury (AKI) in patients treated with polymyxin B and influence of AKI on mortality: a multicentre prospective cohort study. J Antimicrob Chemother. 2015;70(5):1552–1557. doi:10.1093/jac/dku561
34. Lakota EA, Landersdorfer CB, Nation RL, et al. Personalizing polymyxin B dosing using an adaptive feedback control algorithm. Antimicrob Agents Chemother. 2018;62(7). doi:10.1128/AAC.00483-18
35. Garnacho-Montero J, Ortiz-Leyba C, Jimenez-Jimenez FJ, et al. Treatment of multidrug-resistant Acinetobacter baumannii ventilator-associated pneumonia (VAP) with intravenous colistin: a comparison with imipenem-susceptible VAP. Clin Infect Dis. 2003;36(9):1111–1118. doi:10.1086/374337
36. Kallel H, Hergafi L, Bahloul M, et al. Safety and efficacy of colistin compared with imipenem in the treatment of ventilator-associated pneumonia: a matched case-control study. Intensive Care Med. 2007;33(7):1162–1167. doi:10.1007/s00134-007-0675-2
37. Poudyal A, Howden BP, Bell JM, et al. In vitro pharmacodynamics of colistin against multidrug-resistant Klebsiella pneumoniae. J Antimicrob Chemother. 2008;62(6):1311–1318. doi:10.1093/jac/dkn425
38. Lopez-Rojas R, McConnell MJ, Jimenez-Mejias ME, Dominguez-Herrera J, Fernandez-Cuenca F, Pachon J. Colistin resistance in a clinical Acinetobacter baumannii strain appearing after colistin treatment: effect on virulence and bacterial fitness. Antimicrob Agents Chemother. 2013;57(9):4587–4589. doi:10.1128/AAC.00543-13
39. Beyrouthy R, Robin F, Lessene A, et al. MCR-1 and OXA-48 in vivo acquisition in KPC-producing Escherichia coli after colistin treatment. Antimicrob Agents Chemother. 2017;61(8). doi:10.1128/AAC.02540-16
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



