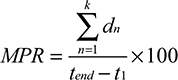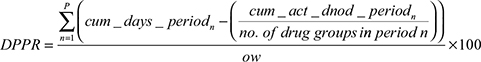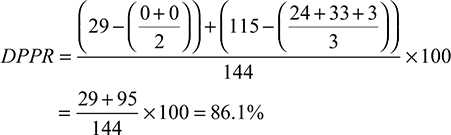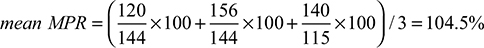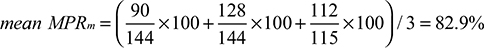Back to Journals » Clinical Epidemiology » Volume 10
Operationalization and validation of a novel method to calculate adherence to polypharmacy with refill data from the Australian pharmaceutical benefits scheme (PBS) database
Authors Arnet I , Greenland M , Knuiman MW, Rankin JM, Hung J , Nedkoff L , Briffa TG, Sanfilippo FM
Received 8 October 2017
Accepted for publication 29 April 2018
Published 6 September 2018 Volume 2018:10 Pages 1181—1194
DOI https://doi.org/10.2147/CLEP.S153496
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Vera Ehrenstein
Isabelle Arnet,1 Melanie Greenland,2 Matthew W Knuiman,2 Jamie M Rankin,3 Joe Hung,4 Lee Nedkoff,2 Tom G Briffa,2 Frank M Sanfilippo2
1Department of Pharmaceutical Sciences, Pharmaceutical Care Research Group, University of Basel, Basel, Switzerland; 2School of Population and Global Health, Faculty of Health and Medical Sciences, The University of Western Australia, Perth, WA, Australia; 3Cardiology Department, Fiona Stanley Hospital Murdoch, WA, Australia; 4School of Medicine, Sir Charles Gairdner Hospital Unit, The University of Western Australia, Perth, WA, Australia
Background: Electronic health care data contain rich information on medicine use from which adherence can be estimated. Various measures developed with medication claims data called for transparency of the equations used, predominantly because they may overestimate adherence, and even more when used with multiple medications. We aimed to operationalize a novel calculation of adherence with polypharmacy, the daily polypharmacy possession ratio (DPPR), and validate it against the common measure of adherence, the medication possession ratio (MPR) and a modified version (MPRm).
Methods: We used linked health data from the Australian Pharmaceutical Benefits Scheme and Western Australian hospital morbidity dataset and mortality register. We identified a strict study cohort from 16,185 patients aged ≥65 years hospitalized for myocardial infarction in 2003–2008 in Western Australia as an illustrative example. We applied iterative exclusion criteria to standardize the dispensing histories according to previous literature. A SAS program was developed to calculate the adherence measures accounting for various drug parameters.
Results: The study cohort was 348 incident patients (mean age 74.6±6.8 years; 69% male) with an admission for myocardial infarction who had cardiovascular medications over a median of 727 days (range 74 to 3,798 days) prior to readmission. There were statins (96.8%), angiotensin converting enzyme inhibitors (88.8%), beta-blockers (85.6%), and angiotensin receptor blockers (13.2%) dispensed. As expected, observed adherence values were higher with mean MPR (median 89.2%; Q1: 73.3%; Q3: 104.6%) than mean MPRm (median 82.8%; Q1: 68.5%; Q3: 95.9%). DPPR values were the most narrow (median 83.8%; Q1: 70.9%; Q3: 96.4%). Mean MPR and DPPR yielded very close possession values for 37.9% of the patients. Values were similar in patients with longer observation windows. When the traditional threshold of 80% was applied to mean MPR and DPPR values to signify the threshold for good adherence, 11.6% of patients were classified as good adherers with the mean MPR relative to the DPPR.
Conclusion: In the absence of transparent and standardized equations to calculate adherence to polypharmacy from refill databases, the novel DPPR algorithm represents a valid and robust method to estimate medication possession for multi-medication regimens.
Keywords: medication adherence, claims database, DPPR, medication possession ratio, algorithm, administrative data
Plain language summary
Adherence, a term for how much of the prescribed medications have been taken, can be estimated from medication claims data. Several measures of adherence to multiple medications (polypharmacy) have been reported in the literature as averaged metrics from single medications. Because they are historical and were not developed to estimate polypharmacy, the formulae represent a gross approximation of the actual medication intake and were therefore criticized. A call for transparency and standardization emerged, leading to the development of a novel calculation method. Due to an aging population and clinical guidelines recommending concurrent medication, there is a growing need for robust estimates of adherence to polypharmacy. A novel formula, the daily polypharmacy possession ratio (DPPR), was specifically developed to estimate daily availability for multiple medications. This is different to currently used measures of adherence that estimate multidrug adherence by averaging the measures of the individual medications that make up the regimen. In this article, DPPR is operationalized and validated from a rich database of population health records. The DPPR represents a robust and valid estimate of adherence to polypharmacy. Researchers, clinicians, and policy makers now have at their disposal a new method to discriminate non-adherence in big claims data.
Introduction
Electronic health care data represent a precious source of information on medicine use and yield rich and objective data. The Australian Federal Government maintains the Pharmaceutical Benefits Scheme (PBS) database which contains detailed information related to claims and payments for medications dispensed from the community as well as private and some public hospital pharmacies.1
Calculations from dispensing data allow us to estimate how much of the prescribed medications have been taken (ie, adherence) and for how long (ie, persistence).2 Because metrics have been mostly developed to estimate the costs of health care,3 the literature includes a plethora of different measures, the most common being the medication possession ratio (MPR) and the proportion of days covered (PDC).4
The most significant differences between the MPR and PDC are that the latter measures medication availability over a given period of time (coverage), mostly a calendar year,5 and values are capped at 100%.6 Since the PDC is affected both by gaps and termination of treatment, its calculation is most appropriate to analyze discontinuation with a threshold indicating premature termination.7 On the other hand, the calculation of MPR is affected solely by gaps from refill to refill. Unfortunately, most of the formulae lack transparency and consistency,8 leading to controversial9–11 or even contradictory results12 and denote the poor understanding of the use and limitations of the metrics.3 As an example, calculations of PDC from automated US refill data demonstrated an artificial inflation of the adherence estimate depending on the variables used.13 Thus, recent effort has focused on a proposal to standardize new measures for adherence calculations for single medications from dispensing data,14 including their operationalization.15
Polypharmacy (the concurrent use of multiple medications) is common due to patients having multiple comorbid medical conditions,16 and aging,17 but is also due to multidrug combinations relied on by clinicians18 and recommended by evidence-based guidelines.19 Thus, assessment of adherence to polypharmacy is clinically relevant, not least because abnormal intake of one or more drugs in a polypharmacy regimen is common and may impact on clinical outcomes.
Few studies have attempted to calculate adherence to multiple concurrent medications,20,21 and have generally calculated an MPR or a PDC for each single medication and then taken an average to represent adherence for the multidrug regimen. This method has been shown to overestimate,22 but may also underestimate adherence to polypharmacy regimens. Briefly, the use of MPR for more than one medication distorts the adherence estimate, predominantly because it does not account for overlapping supply periods, or unexpected and same-day refills.22 Thus, an averaged estimate may not reflect the actual adherence that it was intended to measure. Thus, we previously developed a new method for calculating adherence to multiple medications from dispensing data, the daily polypharmacy possession ratio (DPPR).23 This method calculates the proportion of all medicines available on-hand each day. In brief, the DPPR is a score (%) weighted by the number of medication(s) to be taken each day, with a maximum value of 100% after adjusting for any oversupply.23 The aim of this study was to operationalize the new DPPR method, and validate it against the MPR, the most common measure of adherence, using linked Australian health data. The DPPR SAS program we developed is freely available in the Supplementary materials. We followed the RECORD statement24 for the reporting of studies conducted using observational routinely-collected health data.
Materials and methods
Ethics statement
Human Research Ethics Committee approvals were obtained from Departments of Health (Western Australian #2011/62 and Federal) and The University of Western Australia (RA/4/1/1130). Patient data were de-identified and a waiver of consent was received from the ethics committees.
Selection of patient cohort
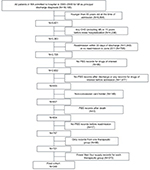
The PBS database contains detailed information related to pharmacy claims. Medications dispensed to eligible veterans, war widows/widowers, and their dependants are also included (repatriation benefits). In brief, patients pay a co-payment toward the cost of medications listed on the PBS, and the Australian Government pays the remainder.25,26 Linkage of the PBS data with core datasets of the Western Australian Data Linkage System (Hospital Morbidity data, Emergency Department data and Mortality Register) yields a unique and comprehensive patient-level database with growing use for population-wide research purposes.27 Details of the linked datasets have been published elsewhere.25 The myocardial infarction coding in the Western Australia Hospital Morbidity Data Collection has been validated previously.28 For this study, the investigators had access to the anonymized extract from the database population to create the study cohort. We identified a cohort of 16,185 people who were hospitalized in Western Australia between 2003 and 2008 with a principal discharge diagnosis of myocardial infarction (International Classification of Diseases [ICD]-10-AM I21) and for whom we had PBS data up to June 2011. Inclusion and exclusion criteria to identify the study dataset can be seen in Figure 1.
Inclusion criteria
- Has first hospital admission (index hospitalization) between January 1, 2003 and December 31, 2008 with a principal discharge diagnosis of acute myocardial infarction (ICD-10-AM I21);
- Is aged 65 years or older at time of admission;
- Has no admission for coronary heart disease (ICD-10-AM I20-I25) recorded in any diagnosis field within 15 years prior to the index hospitalization;
- Has dispensing records available in the PBS dataset after discharge and before a readmission (for any reason) for two or more (ie, polypharmacy) of the following four therapeutic groups: beta-blocker (ATC code C07Axxx), angiotensin converting enzyme (ACE) inhibitor (ATC code C09AAxx), angiotensin receptor blocker (ARB; ATC code C09CAxx), statin (ATC code C10AAxx). By doing this, we avoided changes to treatment during re-hospitalization;
- Has no dispensing records for the previously mentioned medications prior to the index hospitalization (lookback in PBS to June 2002);
- Has at least four dispensing records for each of the previously mentioned ATC groups. By doing this, we excluded primary non-adherence (ie, patients who obtained a first package but did not start the treatment) and enabled patients to build a medication taking habit, considering that 3 months is needed to adopt a new habit;
- Is a concession-card holder at the index hospitalization (to address a limitation of the PBS data where drugs are not recorded if the cost of the drug is less than the patient co-payment, so the patient pays in full). Of note, 82.4% of patients ≥65 years were concession-card holders in the PBS database.
Exclusion criteria
- Has a readmission (for any reason) within 30 days of discharge from the index hospitalization;
- Has no readmission after the index hospitalization up to June 2011 (by definition this will exclude patients who died and had no readmissions, and will standardize the study cohort);
- Has PBS records after the date of death (could be due to incorrect linkage when using probabilistic methods).
The exclusion criteria resulted in a strict cohort for validity because the aim was to test the performance of the DPPR in an administrative dataset of drug data, rather than assessing outcomes where precision is more important.
We have considered specific Australian conditions that may have interfered with the completeness of the PBS data. This included the availability of aspirin without prescription, which amounts to patients obtaining aspirin with no record in the PBS database,25 and an increase in the recommended duration of clopidogrel treatment from 6 months to 12 months during the study period.29 Therefore, we excluded both aspirin (ATC code B01AC06) and clopidogrel (ATC code B01AC04) from our analysis.
Definitions and assumptions
We define polypharmacy as the concurrent use of multiple medications, without restriction of the number, although the literature provides various definitions and numerical thresholds.30 For the operationalization of the DPPR, the following definitions and assumptions were made, in accordance with others,2,15,31 and integrated into the SAS programming:
- The observation window starts with the first dispensing (supply) date after the first hospitalization for myocardial infarction, and ends at the last refill date or the first readmission date, whichever came first.
- A gap may exist between two supplies when the current supply is depleted and before a refill supply is dispensed.
- Oversupply results from overlapping supply periods and leads to accumulated medications. Oversupply is permitted as patients may get a new supply before they have exhausted their current supply, which occurs in daily practice.
- Carry-over of oversupply occurs when accumulated oversupply is more than a subsequent gap. We assumed patients use their accumulated oversupply if there is a gap and carry-over any excess oversupply. A gap can only be compensated by a previous (not future) oversupply – with the rationale that this reflects real life, where patients exhaust a previous supply before starting the next. We assumed that medication is not consumed once there are no further refills and accumulated oversupply is exhausted.
- Oversupply beyond the observation window is not permitted (ie, extra doses beyond the end of the observation window are excluded from the calculation).
- Switching is defined as one drug being initially dispensed, then a different drug being dispensed at a later point within the observation window. We define brand switching as switching between drugs with identical ATC code at the fifth classification level which is switching between different brands of the drug (eg, C07AB02: Minax® 50 mg [Alphapharm] and Betaloc® 50 mg [AstraZeneca]). In this case, we consider the switch as additive use (because it is the same drug) and carry-over is granted under the previously mentioned conditions. We defined therapeutic switching as a switch between two different medications within the same therapeutic class. These either have different ATC codes at the fifth level (eg, C10AA01: simvastatin 40 mg and C10AA05: atorvastatin 40 mg; switching within chemical group) or at the fourth level (eg, A02BC: proton pump inhibitor and A02BA: H2-antagonist; switching between chemical subgroups within a pharmacological group). In this case, we consider switching as providing continuous use and no overlap is granted. That is, a possible oversupply of the previously used medication is ignored, with the rationale that a medical reason forced the physician to change medication (eg, lack of effectiveness, side effects or intolerance). Hence, we accounted for oversupply for brand switching but not for therapeutic switching.
- The prescribed daily dose (PDD) is seldom available in administrative health records and is often replaced by the World Health Organization (WHO) defined daily dose (DDD).32 The PBS dataset does not include PDD, and the DDD is a broad, and sometimes inaccurate, measure of actual dose. Hence, given that PBS quantities are intended for 1-month supplies, we defined by default a dose of one per day for all medications except for metoprolol tartrate and carvedilol (two times daily) according to Australian clinical recommendations.33
Operationalization of the measures
For each patient and for each therapeutic group, assuming there are a total of k dispensings during the observation window, we calculated the MPR and the modified MPR (MPRm) as follows (variable definitions in Table 1).34
|
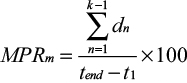 and calculated the mean value for each patient according to the number of therapeutic groups they were on throughout the observation window.
and calculated the mean value for each patient according to the number of therapeutic groups they were on throughout the observation window.
For the calculation of DPPR, further parameters were defined (Table 1). As a patient may have new therapeutic groups dispensed at staggered time points, the calculations were applied for each patient for each therapeutic group by “period”. A period starts when a new therapeutic group commences. In our case the maximum is four periods. It was assumed that if a patient started a therapeutic group they were meant to continually take medication from the group until the end of the observation window. For the purpose of applying the DPPR to the study dataset under strict conditions and comparing with the traditional MPR, we allowed for staggered entry of drugs within the observation window (the end of the observation window signifies either the end of the study period or the last supply date beyond which there was no further supply). This rule also applied to the way we calculated the MPR and MPRm.
|
Estimates are calculated by period, summed, and then divided by the observation window. The underlying concept of the DPPR is to use a 1 or 0 to indicate the presence or absence of a medication on-hand for each day, respectively, and builds proportions for periods, taking into account all of the previously mentioned assumptions. Table 2 provides an illustrative example (SAS program can be seen in the Supplementary materials). The following calculations correspond to the example patient in Table 2.
|
|
|
Statistical methods
The data were obtained from administrative health datasets and contained no missing values. We combined a patient’s supply to a single supply if a patient received multiple supplies of the medication on the same day. Data cleaning is carried out by the data custodians of the individual datasets. We carried out standard data checks prior to analysis. We used SAS v9.4 for all computations, calculations, plots, and development of the program. For descriptive statistics, we reported mean values with SD or median values with range and interquartile range (IQR), where appropriate. Analysis was performed for all patients and the subset of non-switching patients, where appropriate. The MPR, MPRm, and DPPR were compared graphically by scatter plots and correlation coefficient, frequency distribution of the percentage change relative to the observation window, and histogram of the percentage change between mean MPR and DPPR. The histogram bar width was set at 2.5% on either side of the central value (ie, width of 5%) with increments of 5%.
Results
Patient characteristics
We identified 348 patients who met the inclusion criteria in whom to compare the different methods of adherence (Figure 1). The mean age was 74.6±6.8 years at hospital admission date (range 65–95 years) and the majority were males (240 patients; 69.0%). The number of concurrent therapeutic groups of interest was two (78 patients; 22.4%), three (246 patients; 70.7%) or four (24 patients; 6.9%) and included statins (337 patients; 96.8%), ACE inhibitors (309 patients; 88.8%), beta-blockers (298 patients; 85.6%), and ARBs (46 patients; 13.2%; Table 3). The majority of patients (67.8%) started all their drug groups at the same time, while initiation of medication occurred at two and three time points (or periods) for 29.9% and 2.3% of patients, respectively. The median length of the observation window was 727 days (IQR 363–1,345 days) with range from 74 to 3,798 days.
Switching from first dispensed drug to another drug in a therapeutic group occurred in 64 patients (18.4%) a total of 69 times, with multiple switching possible, and concerned mostly statins (33 patients; switch from initial atorvastatin for 20 patients), and beta-blockers (27 patients; switch from metoprolol tartrate for 23 patients) (Table 3). Switching from first dispensed ARB or ACE inhibitor occurred in five and four patients, respectively. Independently of switching, the most frequently dispensed medications for each therapeutic group were atorvastatin, ramipril, metoprolol, and irbesartan (Table 3).
Adherence estimates
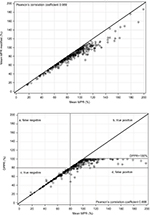
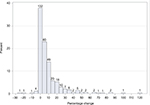
The mean values ranged from 80.4% (DPPR) to 89.2% (MPR), with a minimum value of 15.2% (MPRm) and a maximum value of 199.4% (MPR; Table 4) with each mean MPRm value being lower than the corresponding mean MPR value (Figure 2 top panel), and DPPR values not exceeding 100% (Figure 2 bottom panel), as expected. The DPPR method resulted in the smallest width of the frequency distribution of adherence estimates (Table 4). The percentage change between mean MPR and DPPR values ranged between –24% and +121% and measures the percent over/underestimation of DPPR when using the mean MPR (Figure 3). Very close MPR and DPPR values (ie, percentage changes between mean MPR and DPPR from –2.5% to +2.5%) were observed for 37.9% of patients, while over half (50.9%) of the DPPR values were overestimated up to +27.5%, and 12.1% of the patients had an overestimation of +22.5% or higher (Figure 3). An example of +25% overestimation is a mean MPR of 100% in comparison to a DPPR of 80%. When extrapolated to a scenario with a patient who should take two different medications once a day for 6 months (two different pills daily for 180 days), the 100% mean MPR suggests perfect intake but the 80% DPPR suggests the patient had a 36-day gap without either medication (144 days on two medications).
Since many researchers use a historical threshold of 80% to separate good adherence from poor adherence,35 we analyzed our data using this cutoff. When DPPR was considered the reference measure, the positive predictive value was 88.4%, implying the mean MPR method classified 11.6% of patients as good adherence (Figure 2, bottom panel) relative to the DPPR. Conversely, the negative predictive value was 100% implying the mean MPR method never misclassified poor adherence relative to the DPPR (Figure 2, bottom panel).
The DPPR and the mean MPR values were very similar in patients with longer observation windows, but there was more and larger overestimation of the DPPR in patients with observation window below 2 years (Figure 4). The mean percentage change was 9.28% for non-switching patients and 11.98% for switching patients (data not shown).
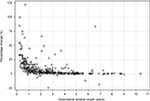  | Figure 4 Percentage change between mean MPR and DPPR relative to DPPR by observation window length. Abbreviations: MPR, medication possession ratio; DPPR, daily polypharmacy possession ratio. |
Discussion
Conventional and historical measures of adherence to polypharmacy are average metrics from single medications and have been recognized as a gross approximation of the actual medication intake. Thus, new methods are needed to provide more accurate estimates of medication use, especially using refill databases with longitudinal patient data. Comparison of the three possession measures MPR, MPRm, and DPPR, derived from a strictly defined patient population, shows the advantages of the DPPR estimates which indicate the proportion of all medicines available on-hand each day. As expected, of all adherence measures, the DPPR yielded the lower mean estimate and the narrowest distribution of values. In addition, a direct comparison between mean MPR and DPPR showed that percent over/underestimation of the DPPR when using the mean MPR ranged between –24% and +121%. The difference was in 62% of the values, and more than half of the patients were overestimated by up to +27.5% when using the mean MPR, especially for shorter observation windows below 2 years. Further, setting a threshold of 80% to signify good adherence resulted in 11.6% of patients being classified as good adherers with the mean MPR relative to the DPPR.
Our validation study has demonstrated that the disadvantages of the conventional MPR measures may be overcome with the DPPR. In particular, the DPPR estimates of adherence are more conservative mainly through strict accounting of oversupply. Hence, the DPPR accounts for oversupply solely to compensate a subsequent gap (time-forward-approach),2 and excludes oversupply beyond the end date which was shown to overestimate adherence measures36 by enlarging the value of the quantity dispensed. This is a disadvantage of the MPR methods because they count oversupplies and can lead to extreme outliers relative to the DPPR. However, because standardization of all variables is an integral part of the DPPR measure, it delivers a more realistic value of medication use and can be used in adherence research and for policy makers.
In our approach to operationalize the DPPR method for the first time, we deliberately applied strict iterative exclusion criteria to our original database as well as strict assumptions. By doing this, we standardized the dispensing histories and thus excluded noise in the analysis, enabling statistical comparison between the three possession metrics. We are aware that clinical practice might differ – possibly between countries – and have discussed, in the next section, each unique peculiarity of treatment history as well as its implication for the DPPR. This concerns possible stockpiling prior to the start date, therapeutic switching and non-persistence, dosage instructions, end date, and temporary gaps such as hospitalization, which can be summarized in elements related to the patient, the medication or the system.
Firstly, on a patient-level, we selected patients hospitalized with incident myocardial infarction who received the first dispensing of medication after discharge. By doing this, we assumed that the first intake occurred on the day of the first dispensing, and that patients had no stock at home. The influence of a pre-existing stockpile on the adherence metric and its implication on selecting the start date have been described previously37 and have intuitive appeal. Setting the start date of the observation window represents the first decision researchers have to make when defining their datasets. A look-back period can be included to identify if medication was dispensed previously.38 Depending on the purpose for which the adherence metric will be used, the start date may or may not take into account prior dispensing, with the inherent question of “For how long?”. Consequently, when running the DPPR SAS program with their datasets, researchers may need to adapt the raw data according to their needs, to adjust the original quantity values with the previous stockpile.
Secondly, on a medication level, we treated each initialized medication as if maintained until the end of the observation window. By doing this, we assumed that no treatment interruption or temporary discontinuation occurred during the observation window, thus excluding therapy cessation (so-called non-persistence). Also, we ignored switching between two different medications within the same therapeutic class, because we considered these as two separate drugs with no carry-over of supply from the switched drug. Depending on the local clinical guidelines, switching medications from different therapeutic groups may be recommended. For example, in Australia and in most countries during 2003–2008, an ACE inhibitor could be stopped and replaced by an ARB in case of intolerance39 while today, with new results and evolving guidelines,40 ACE inhibitors and ARB are considered interchangeable and treated as one drug, indicated as ACE/ARB in our study results. Consequently, when running the DPPR SAS program, researchers need to adapt it to their raw data according to local guidelines, and group the ATC codes for therapeutic equivalence together in one therapeutic group.
The challenge in defining non-persistence in refill databases is to determine the onset of cessation, that is, the last gap beyond which medication use ceases. Mathematically, it amounts to setting a specific period of time (or threshold) that needs to be exceeded after the supply from the previous refill ends. Therefore, an individual is classified as non-persistent if they did not refill a medication within the given time threshold. The value of this maximal gap can range from zero (no gaps allowed in medication history) to a specific number of days. Between those two extremes, almost every gap length from 7 to 180 days has been proposed in the literature.41 Setting a threshold is defining the sensitivity of the adherence measure, since the smaller the allowable gap, the higher the number of patients classified as having ceased treatment once the gap is exceeded. One method used with health records is the refill-sequence that determines if a patient has refilled a prescription within a predefined number of days.42 Similar analysis of persistence with cardiovascular medications from the Danish population registry showed that almost half of the patients re-initiated treatment after a gap of 90 days.43 Thus, a threshold of 90 days or more might be useful, but it will depend on the dosage of the individual drug. Consequently, when running the DPPR SAS program, researchers need to decide the magnitude of the gap according to the medications of interest, and to ensure that accumulated oversupply is accounted for prior to concluding that the defined gap has been exceeded.
In the absence of dosage information in the PBS dataset, we allocated a daily dose of one or two for medications, with assumptions derived from Australian treatment guidelines. By doing this, we adopted the concept of the waiting-time-distribution method44 that estimates the prescription duration from data without dosage information, by using the 75th centile of the distribution of time to next supply date. In a recent study using the PBS database,45 the estimated duration was 35 days for most cardiovascular medications which corresponds to a 1-month supply and a once-daily intake for 30-day packages. This is consistent with the estimates of duration we determined from our data, and consistent with the PBS being a system of 1-month supplies for most drugs that are used in chronic diseases. Similarly, changes in medication strength and dose are not captured in most health care databases and need approximating from the quantity dispensed. Since the PBS was designed to deliver treatments of 30 days’ duration, approximating from quantity dispensed is possible. For other databases, calculations based on medication strength and the corresponding WHO DDD is an alternative way to approximate the dose. Consequently, when running the DPPR SAS program, researchers need to set daily doses according to the best method available to them. However, given that the MPR methods include oversupply and can overestimate the adherence, we expect the DPPR to be a more robust measure if the dose assumptions are incorrect because it accounts for oversupply.
Thirdly, on a system-level, we selected the next readmission date as the end of the observation window. Readmissions may not be possible to identify in some countries – eg, when hospital data are not linked to drug data – and can lead to longer gaps in refill sequences. If readmissions are included in the observation window, and the hospitalization period is known, it should be subtracted from the denominator of the metrics, assuming complete adherence to hospital medications during hospital stay. However, if the length of stay is short then adjusting for hospital admission(s) will probably have a negligible effect on the adherence calculation. Depending on the local hospital policies, patients might use their home medication in the hospital or medications might be dispensed at discharge. No adjustment of the calculation is needed in the former case, whilst medications dispensed at discharge should be taken into account in the latter case. Consequently, when running the DPPR SAS program, researchers need to adjust the end dates of some treatment episodes with hospitalizations, and include additional supply if medications are dispensed at discharge if these data are available.
We applied strict inclusion criteria, such as four or more dispensings of medications from two or more therapeutic groups, and eliminated outliers or abnormal patterns to obtain high quality datasets. We ended up with a robust algorithm that was able to be separated from the historical MPR measures. Thus, it is likely that larger effects will be observed when running our DPPR SAS program on a less restricted dataset that has more outliers and extreme dispensing patterns. To demonstrate whether the DPPR is an improved measure relative to the MPR or PDC, comparative data assessing adherence by a specific method to tangible outcomes (such as readmissions and death) is needed. Meanwhile, the DPPR can be considered as a better measure of adherence to polypharmacy than other estimates.
This study has several strengths. First, the data stem from a large national medications database and captured all PBS dispensing for concessional beneficiaries with heart disease in Western Australia. Thus, the data are population-based and adherence calculations yield real-world estimates. Second, we applied highly restricted criteria to select patients with first-time myocardial infarction. This approach enabled us to compare the different calculation methods under identical conditions and enhances the reliability of the results. Third, our approach allowed us to describe and discuss the unique peculiarities of treatment history such as hospitalization, and to propose ways to account for these in the analyses. By doing this, we hope to increase the awareness of other researchers when calculating medication adherence from administrative datasets. The DPPR method is applicable to all databases that contain information on dispensed medications (ie, prescribing, dispensing or claims databases), provided that the parameters “date of dispensing”, “quantity of medication dispensed”, and “duration of supply (days) at dispensing” are available or can be estimated.
We acknowledge some limitations. First, as with any analysis of adherence based on dispensing data, we assumed that medicines dispensed implied medicines were ingested. As a consequence, indices are surrogate measures of medication adherence and identify the highest possible level of medication possession. Second, several assumptions were made, the main one being that medication consumption commences on the day of dispensing. However, future standardization of the assumptions will lead to comparable estimates of adherence across different studies, for the same calculation method. Third, the PBS dataset does not include dosage, so we used a once-daily dose for each drug except for metoprolol tartrate and carvedilol (twice-daily doses used). This may have underestimated usage if a higher dose was prescribed or overestimated the dose if the patient was instructed to take half the dose in the case of tablets that can be cut in half. Fourth, claims databases may not capture the most non-adherent patients, ie, primary non-adherence. However, the DPPR is not designed to capture primary non-adherence, but to provide a measure of adherence in patients who have been supplied multidrug regimens. Lastly, our SAS program does not allow for the stopping of a drug group altogether. However, with the proposed periods of therapeutic group, one can calculate a DPPR for this time frame and then separately look at adherence after the removal of a drug group.
Conclusion
The new DPPR is an alternative method of estimating adherence to polypharmacy from administrative datasets that addresses usage from a combination of drugs rather than treating adherence as an individual drug measure where a regimen is prescribed. Our results support that the DPPR is a more robust method of measuring adherence than the traditional MPR methods, as it accounts for oversupply and thus leads to a more sound measure if the assumption for dose is incorrect. This study used strict conditions of record selection in administrative drug data to demonstrate the working application of the DPPR algorithm. The next step is to apply the algorithm under less restrictive study conditions to examine its impact in analyses of clinical outcomes.
Acknowledgments
The authors thank the Australian Department of Health for providing the cross-jurisdictional linked PBS dataset used in the study, and the Data Linkage Branch and data custodians of the WA Department of Health for providing the linked hospital morbidity and mortality data.
This work was funded by grants from the Heart Foundation of Australia (Vanguard grant ID 101455) and the National Health and Medical Research Council of Australia (project grant 1066242). LN is funded by an Early Career Fellowship from the National Health and Medical Research Council of Australia.
Disclosure
The authors declare there are no conflicts of interest.
References
Australian Government Department of Health. The Pharmaceutical Benefits Scheme (PBS); 2017. Available from: www.pbs.gov.au. Accessed June 7, 2017. | ||
Arnet I, Kooij MJ, Messerli M, Hersberger KE, Heerdink ER, Bouvy M. Proposal of standardization to assess adherence with medication records: methodology matters. Ann Pharmacother. 2016;50(5):360–368. | ||
Whalley Buono E, Vrijens B, Bosworth HB, Liu LZ, Zullig LL, Granger BB. Coming full circle in the measurement of medication adherence: opportunities and implications for health care. Patient Prefer Adherence. 2017;11:1009–1017. | ||
Sattler EL, Lee JS, Perri M, Medication PM. Medication (re)fill adherence measures derived from pharmacy claims data in older Americans: a review of the literature. Drugs Aging. 2013;30(6):383–399. | ||
Krueger K, Griese-Mammen N, Schubert I, et al. In search of a standard when analyzing medication adherence in patients with heart failure using claims data: a systematic review. Heart Fail Rev. 2018;23(1):63–71. | ||
Hess LM, Raebel MA, Conner DA, Malone DC. Measurement of adherence in pharmacy administrative databases: a proposal for standard definitions and preferred measures. Ann Pharmacother. 2006;40(7-8):1280–1288. | ||
Benner JS, Glynn RJ, Mogun H, Neumann PJ, Weinstein MC, Avorn J. Long-term persistence in use of statin therapy in elderly patients. JAMA. 2002;288(4):455–461. | ||
Wilke T, Groth A, Mueller S, et al. How to use pharmacy claims data to measure patient nonadherence? The example of oral diabetics in therapy of type 2 diabetes mellitus. Eur J Health Econ. 2013;14(3):551–568. | ||
Doró P, Benko R, Kosik E, Matuz M, Tóth K, Soós G. Utilization of oral antihyperglycemic drugs over a 7-year period (1998-2004) in a Hungarian population and adherence to drug therapy. Eur J Clin Pharmacol. 2005;61(12):893–897. | ||
Sajatovic M, Valenstein M, Blow FC, Ganoczy D, Ignacio RV. Treatment adherence with antipsychotic medications in bipolar disorder. Bipolar Disord. 2006;8(3):232–241. | ||
Salanitro AH, Kripalani S. Prescription refill management and its effect on adherence: comment on “the implications of therapeutic complexity on adherence to cardiovascular medications”. Arch Intern Med. 2011;171(9):822–823. | ||
Calip GS, Xing S, Jun DH, et al. Polypharmacy and adherence to adjuvant endocrine therapy for breast cancer. J Oncol Pract. 2017;13(5):e451–e462. | ||
Lester CA, Look KA, Chui MA. Is the Currently Used Prescription Adjudication Date a Good Proxy for Calculating Medication Refill Adherence? J Manag Care Spec Pharm. 2016;22(11):1311–1317. | ||
Raebel MA, Schmittdiel J, Karter AJ, Konieczny JL, Steiner JF. Standardizing terminology and definitions of medication adherence and persistence in research employing electronic databases. Med Care. 2013;51(8 Suppl 3):S11–S21. | ||
Dima AL, Dediu D. Computation of adherence to medication and visualization of medication histories in R with AdhereR: Towards transparent and reproducible use of electronic healthcare data. PLoS One. 2017;12(4):e0174426. | ||
Lien CT, Gillespie ND, Struthers AD, Mcmurdo ME. Heart failure in frail elderly patients: diagnostic difficulties, co-morbidities, polypharmacy and treatment dilemmas. Eur J Heart Fail. 2002;4(1):91–98. | ||
Fulton MM, Allen ER. Polypharmacy in the elderly: a literature review. J Am Acad Nurse Pract. 2005;17(4):123–132. | ||
Kingsbury SJ, Yi D, Simpson GM. Psychopharmacology: rational and irrational polypharmacy. Psychiatr Serv. 2001;52(8):1033–1036. | ||
Ibanez B, James S, Agewall S, et al. ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39(2):119-177. | ||
Choudhry NK, Setoguchi S, Levin R, Winkelmayer WC, Shrank WH. Trends in adherence to secondary prevention medications in elderly post-myocardial infarction patients. Pharmacoepidemiol Drug Saf. 2008;17(12):1189–1196. | ||
van Bruggen R, Gorter K, Stolk RP, Zuithoff P, Klungel OH, Rutten GE. Refill adherence and polypharmacy among patients with type 2 diabetes in general practice. Pharmacoepidemiol Drug Saf. 2009;18(11):983–991. | ||
Martin BC, Wiley-Exley EK, Richards S, Domino ME, Carey TS, Sleath BL. Contrasting measures of adherence with simple drug use, medication switching, and therapeutic duplication. Ann Pharmacother. 2009;43(1):36–44. | ||
Arnet I, Abraham I, Messerli M, Hersberger KE. A method for calculating adherence to polypharmacy from dispensing data records. Int J Clin Pharm. 2014;36(1):192–201. | ||
Benchimol EI, Smeeth L, Guttmann A, et al. The REporting of studies Conducted using Observational Routinely-collected health Data (RECORD) statement. PLoS Med. 2015;12(10):e1001885. | ||
Gunnell AS, Knuiman MW, Geelhoed E, et al. Long-term use and cost-effectiveness of secondary prevention drugs for heart disease in Western Australian seniors (WAMACH): a study protocol. BMJ Open. 2014;4(9):e006258. | ||
Page E, Kemp-Casey A, Korda R, Banks E. Using Australian Pharmaceutical Benefits Scheme data for pharmacoepidemiological research: challenges and approaches. Public Health Res Pract. 2015;25(4):e2541546. | ||
Holman CD, Bass AJ, Rouse IL, Hobbs MS. Population-based linkage of health records in Western Australia: development of a health services research linked database. Aust N Z J Public Health. 1999;23(5):453–459. | ||
Sanfilippo FM, Hobbs MS, Knuiman MW, et al. Can we monitor heart attack in the troponin era? Evidence from a population-based cohort study. BMC Cardiovasc Disord. 2011;11:35. | ||
Acute Coronary Syndrome Guidelines Working Group. Guidelines for the management of acute coronary syndromes 2006. Med J Aust. 2006;184(8 Suppl):S9–29. | ||
Gnjidic D, Tinetti M, Allore HG. Assessing medication burden and polypharmacy: finding the perfect measure. Expert Rev Clin Pharmacol. 2017;10(4):345–347. | ||
Gardarsdottir H, Souverein PC, Egberts TC, Heerdink ER. Construction of drug treatment episodes from drug-dispensing histories is influenced by the gap length. J Clin Epidemiol. 2010;63(4):422–427. | ||
who.int [homepage on the Internet] WHO Collaborating Centre for Drug Statistics Methodology. DDD, Definitions and general considerations. Available from: www.whocc.no/atc_ddd_index. Accessed June 7, 2017. | ||
Australian Government Department of Health TGA. Product Information (PI) of metoprolol tartrate. Carvedilol. Available from: www.tga.gov.au/product-information-pi. Accessed June 8, 2017. | ||
Karve S, Cleves MA, Helm M, Hudson TJ, West DS, Martin BC. An empirical basis for standardizing adherence measures derived from administrative claims data among diabetic patients. Med Care. 2008;46(11):1125–1133. | ||
Hansen RA, Kim MM, Song L, Tu W, Wu J, Murray MD. Comparison of methods to assess medication adherence and classify nonadherence. Ann Pharmacother. 2009;43(3):413–422. | ||
Hess LM, Raebel MA, Conner DA, Malone DC. Measurement of adherence in pharmacy administrative databases: a proposal for standard definitions and preferred measures. Ann Pharmacother. 2006;40(7–8):1280–1288. | ||
Bryson CL, Au DH, Young B, Mcdonell MB, Fihn SD. A refill adherence algorithm for multiple short intervals to estimate refill compliance (ReComp). Med Care. 2007;45(6):497–504. | ||
Messerli M, Blozik E, Vriends N, Hersberger KE. Impact of a community pharmacist-led medication review on medicines use in patients on polypharmacy--a prospective randomised controlled trial. BMC Health Serv Res. 2016;16:145. | ||
Swedberg K, Cleland J, Dargie H, et al. Guidelines for the diagnosis and treatment of chronic heart failure: executive summary (update 2005): The Task Force for the Diagnosis and Treatment of Chronic Heart Failure of the European Society of Cardiology. Eur Heart J. 2005;26(11):1115–1140. | ||
Adams KF, Giblin EM, Pearce N, Patterson JH. Integrating new pharmacologic agents into heart failure care: role of heart failure practice guidelines in meeting this challenge. Pharmacotherapy. 2017;37(6):645–656. | ||
Andrade SE, Kahler KH, Frech F, Chan KA. Methods for evaluation of medication adherence and persistence using automated databases. Pharmacoepidemiol Drug Saf. 2006;15(8):565–574. | ||
Caetano PA, Lam JM, Morgan SG. Toward a standard definition and measurement of persistence with drug therapy: Examples from research on statin and antihypertensive utilization. Clin Ther. 2006;28(9):1411–1424. | ||
Gislason GH, Rasmussen JN, Abildstrøm SZ, et al. Long-term compliance with beta-blockers, angiotensin-converting enzyme inhibitors, and statins after acute myocardial infarction. Eur Heart J. 2006;27(10):1153–1158. | ||
Pottegård A, Hallas J. Assigning exposure duration to single prescriptions by use of the waiting time distribution. Pharmacoepidemiol Drug Saf. 2013;22(8):803–809. | ||
Gadzhanova S, Roughead EE, Bartlett LE. Long-term persistence to mono and combination therapies with angiotensin converting enzymes and angiotensin II receptor blockers in Australia. Eur J Clin Pharmacol. 2016;72(6):765–771. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.