Back to Journals » Drug Design, Development and Therapy » Volume 10
Novel A20-gene-eluting stent inhibits carotid artery restenosis in a porcine model
Authors Zhou Z, Peng J, Meng Z, Chen L, Huang J, Huang H, Li L, Zeng W, Wei Y, Zhu C, Chen K
Received 23 August 2015
Accepted for publication 30 December 2015
Published 8 August 2016 Volume 2016:10 Pages 2341—2351
DOI https://doi.org/10.2147/DDDT.S94984
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Wei Duan
Zhen-hua Zhou,1 Jing Peng,1 Zhao-you Meng,1 Lin Chen,1 Jia-Lu Huang,1 He-qing Huang,1 Li Li,2 Wen Zeng,2 Yong Wei,2 Chu-Hong Zhu,2 Kang-Ning Chen1
1Department of Neurology, Cerebrovascular Disease Research Institute, Southwest Hospital, 2Department of Anatomy, Key Laboratory for Biomechanics of Chongqing, Third Military Medical University, Chongqing, People’s Republic of China
Background: Carotid artery stenosis is a major risk factor for ischemic stroke. Although carotid angioplasty and stenting using an embolic protection device has been introduced as a less invasive carotid revascularization approach, in-stent restenosis limits its long-term efficacy and safety. The objective of this study was to test the anti-restenosis effects of local stent-mediated delivery of the A20 gene in a porcine carotid artery model.
Materials and methods: The pCDNA3.1EHA20 was firmly attached onto stents that had been collagen coated and treated with N-succinimidyl-3-(2-pyridyldithiol)propionate solution and anti-DNA immunoglobulin fixation. Anti-restenosis effects of modified vs control (the bare-metal stent and pCDNA3.1 void vector) stents were assessed by Western blot and scanning electron microscopy, as well as by morphological and inflammatory reaction analyses.
Results: Stent-delivered A20 gene was locally expressed in porcine carotids in association with significantly greater extent of re-endothelialization at day 14 and of neointimal hyperplasia inhibition at 3 months than stenting without A20 gene expression.
Conclusion: The A20-gene-eluting stent inhibits neointimal hyperplasia while promoting re-endothelialization and therefore constitutes a novel potential alternative to prevent restenosis while minimizing complications.
Keywords: restenosis, A20, gene therapy, stent, endothelialization
Introduction
Ischemic stroke is a major cause of death and disability worldwide.1,2 Atherosclerotic stenosis of the carotid artery is a known major risk factor for ischemic stroke, and carotid angioplasty and stenting (CAS) has emerged as a potentially less invasive treatment alternative. Studies have suggested that CAS and carotid endarterectomy are associated with similar short- and long-term outcomes,3,4 and there is a growing evidence that the subgroup of patients aged <70 years may particularly benefit from CAS intervention.5,6
However, the occurrence of in-stent restenosis appears to limit the long-term efficacy and safety of CAS, and its occurrence is higher in intracranial atherosclerotic stenosis stenting.7,8
Unfortunately, fundamental research in the area of cerebrovascular ISR is very scarce presently.
Drug-eluting stents (DESs) have reduced the incidence of restenosis of the coronary artery;9,10 however, meta-analyses of long-term outcomes have raised questions about their safety,11,12 including late-stage poor stent adherence to the vascular wall, long-term damage to endothelial cells (ECs), and late stent thrombosis. All these untoward effects are believed to result from DES-mediated nonselective dual inhibition of smooth muscle cells (SMCs) and ECs, leading to incomplete endothelialization.
Compared with DES, gene-eluting stents can locally release biologically active agents to extend the agents’ half-life at vascular lesions.13–15 Therefore, it is an intriguing strategy to use a stent as a platform for localized and prolonged gene therapy on vascular wall lesions. This strategy is designed to treat restenosis by targeting the molecular mechanisms underlying the pathology.
If rapid endothelialization could be achieved after interventional surgery for stent placement, it could significantly reduce the risk of late-stent thrombosis. In addition, endothelial regeneration can decrease intimal hyperplasia by inhibiting the proliferation and migration of SMCs. Thus, protecting the function of ECs plays an important role in preventing restenosis.
Recent studies have shown that the inflammatory response plays an important role in the genesis and development of restenosis.16,17 Histological and biochemical research has shown that platelets and white blood cells are activated following stent surgery, leading to a series of inflammatory responses. Therefore, the inflammatory response is also an important target for the prevention and treatment of restenosis.
The A20 gene belongs to the zinc-finger protein family. Its product can effectively enhance the ability of ECs to resist the damage caused by pathogenic agents and inhibit the pathological proliferation of SMCs. In addition, it downregulates the expression of some inflammatory mediators, thereby inhibiting the inflammatory response. We previously modified seed cells with A20 and effectively constructed tissue-engineered blood vessels with anti-atherosclerosis potential and demonstrated the therapeutic effects of these engineered blood vessels in animal experiments.18 The latter studies indicated that the A20 gene has the potential to inhibit atherosclerosis plaque formation and prevent restenosis.
In this study, we first locally delivered the A20 gene into the vascular wall via eluting stents and observed its effect on the prevention of restenosis of the carotid artery in a porcine model. The present study was aimed at exploring a new avenue for the prevention and treatment of carotid artery restenosis.
Materials and methods
Stents
Under sterile conditions, type I cowhide collagen (Kensey Nash Corporation, Exton, PA, USA) was added to the cross-linking agent ethyl-dimethyl-amino-propyl carbodiimide (Sigma-Aldrich Co., St Louis, MO, USA) (0.1 mg/mg collagen). A bare-metal stent (provided by Shanghai Weichuang Company, Ltd., Shanghai, People’s Republic of China) was dipped into the collagen solution and dried at 37°C. The latter steps were repeated several times until each stent was coated with ~1 mg of collagen. The stents were then examined under a scanning electron microscope to ensure proper collagen coating. The collagen-coated stents were placed in an N-succinimidyl-3-(2-pyridyldithiol) propionate (SPDP; Pierce, Holmdel, USA) solution (20 mmol/L) at room temperature for 2 hours. SPDP-treated stents were rinsed with phosphate-buffered saline (PBS) and then reacted with dithiothreitol (Beyotime, Shanghai, People’s Republic of China) at room temperature for 0.5 hours followed by five times rinsing with PBS. Stents were then placed at room temperature overnight in a solution containing a mouse monoclonal antibody to bovine DNA (US Biologicals, Swampscott, USA) that had been reacted with SPDP and purified (IgM-SPDP). The antibody-bound stents were fully rinsed with PBS and then placed into a pCDNA3.1EHA20 (with E-tag sequence: GGT GCG CCGGTG CCG TAT CCG GAC CCG CTG GAA CCG CGT) solution or a pCDNA3.1 void vector solution (100 μL PBS containing 100 μg plasmid DNA),18,19 incubated at 37°C for 1 hour, and then rinsed five times with PBS. Fifteen microliters of ESCORT liposome transfection reagent (Hoffman-La Roche Ltd., Basel, Switzerland) was added to the solution, and the treated stents were incubated at room temperature for 15 minutes before use in subsequent experiments. Bare-metal stents were used as controls.
In vitro transfection
To test whether the A20 gene carried by the stents could be transfected into ECs in vitro, the following procedures were carried out. Prior to the study, all patients were informed regarding the procedure and its possible complications, and agreed to participate. The Ethics Committee of the Third Military Medical University approved this study. Under sterile conditions, 15–20 cm of human umbilical cord was collected, washed with D-Hank’s solution, then perfused with 0.125% trypsin, and digested at 37°C for 20 minutes. The digestion was terminated with M199 culture medium containing 20% fetal calf serum (FCS). The digestion mixture was centrifuged at 1,000 rpm for 8 minutes, and the supernatant fluid was discarded. The recovered cells were resuspended in M199 medium containing 20% FCS, and inoculated at a concentration of 1×105/mL. The culture medium was M199 solution containing 20% FCS, 90 mg/mL sodium heparin, and 25 mg/L endothelial growth factor. The cells were cultured in an incubator with 5% CO2 at 37°C.
The pCDNA3.1EHA20-bound stents were placed into a six-well plate, and 1 mL of ECs was added into each well at a concentration of 1 ×105/mL and incubated at 37°C for 2 hours. Stents were then rotated by 120° along the long axis, and the cell suspension was added to each well at the same concentration. These steps were repeated thrice. After 7 days of culture, stents were fixed with 4% paraformaldehyde for 30 minutes, and E-tag monoclonal antibody (Amersham Pharmacia Biotech Ltd., Shanghai, People’s Republic of China) was added and incubated at room temperature for 30 minutes. Phosphate buffer was used to wash away the excess monoclonal antibody, followed by addition of fluorescein isothiocyanate-labeled secondary antibody and staining of the nuclei with 4′,6-diamidino-2-phenylindole (DAPI). Stents were observed using a laser confocal microscope (Leica Microsystems, Wetzlar, Germany), with blue fluorescence indicating DAPI-labeled nuclei of ECs and green fluorescence indicating ECs transfected with A20.
Animals
The animal study was in conformity with national regulations and carried out after obtaining permits for the use of animals issued by the Ministry of Health. The animal use and experimental protocols were reviewed and approved by the Ethics Committee of Third Military Medical University. A total of 35 miniature pigs (12–14 weeks old), with a weight of 25–27 kg, were purchased from the Center of Laboratory Animals, Third Military Medical University (Chongqing, People’s Republic of China) and were fed in a standard manner. Pigs were randomly divided into three groups: pCDNA3.1EHA20 (n=12), pCDNA3.1 void vector (n=12), and bare-metal stent (n=11). From 3 days before surgery to the follow-up period, all animals were orally administered 300 mg of aspirin and 75 mg of clopidogrel every day. The animals were anesthetized with 25 mg/kg sodium pentobarbital, followed by tracheal intubation and mechanical ventilation. The animals were sacrificed 3 days, 2 weeks, and 3 months after surgery.
Porcine model of carotid artery stent
Three kinds of stents (pCDNA3.1EHA20, pCDNA3.1 void vector, and bare-metal stent) were randomly placed in the left and right carotid artery of each pig. Briefly, an 8Fr arterial sheath was inserted from the right femoral artery, and an 8Fr-guiding catheter was inserted into the carotid artery. A 0.014 in. guide wire was sent to the distal end of the carotid artery. The stents were advanced along the guide wire to the target vessel’s location and were placed at a balloon-to-vessel ratio of 1.2:1 by angiography. After the placement of the stents, angiography was performed to determine whether the lumen was unobstructed and whether filling defects, vascular wall dissection, embolization, stent migration, or remote vasospasm was present. Regular anti-infection treatment was employed after the surgery. The skin around the wound surface was disinfected routinely, washed with sterile normal saline, and dried. The animals were bred in a good environment. Subsequent follow-up angiography was carried out through catheterization of the contralateral femoral artery.
Analysis of A20 expression
Three days after stent placement, proteins were extracted from the pig carotid arteries. E-tag and A20 expression was confirmed by Western blot analysis, as described previously.20 Electrophoresis, membrane transfer, and signal development were performed. Tissue lysates were separated under reducing conditions by SDS-PAGE (Bio-Rad Laboratories Inc., Hercules, CA, USA), and transferred to polyvinylidenefluoride membranes (PerkinElmer Inc., Waltham, MA, USA) by semidry electroblotting. Membranes were probed with mouse anti-E-tag antibody (Amersham Pharmacia Biotech), mouse anti-human A20 antibody (1/1,000; Oncogene, NY, USA), and mouse antiglyceraldehyde3-phosphate dehydrogenase (EMD Millipore, Billerica, MA, USA). The blots were washed and exposed to horseradish peroxidase-conjugated antimouse IgG secondary antibody (Santa Cruz Biotechnology Inc., Dallas, TX, USA) and then developed using the enhanced chemiluminescence reagent (PerkinElmer Inc.). The results were normalized against glyceraldehyde-3-phosphate dehydrogenase gene expression.
Scanning electron microscopy
Stents were removed after either 14 days or 3 months in vivo, rinsed with physiological saline, fixed with 3% glutaraldehyde, and then cut open along the longitudinal axis. A scanning electron microscope (Model JSM 5510; JEOL, Tokyo, Japan) was used to examine the intimal layer. Photomicrographs of each specimen taken using scanning electron microscopy (SEM) were examined using a computer-assisted digital assessment system. The percent area of re-endothelialization compared with the total luminal surface area was calculated. For each specimen, five photomicrographs were taken at ×400 magnification using SEM, and the re-endothelialization area for each artery was represented by the sum of the data for the five photographed subareas.21
Stent endothelialization score was defined as the extent of the circumference of the arterial lumen covered by ECs and graded from 1 to 3 (1=25%, 2=25%–75%, and 3≥75%).22
Morphological measurement and analysis
Extracted blood vessels were embedded and sectioned as described earlier, and stained with hematoxylin and eosin. All sections were observed under an optical microscope (DFC300FX; Leica Microsystems) for intimal hyperplasia and vascular wall damage. Data analysis was carried out by use of an Image-Pro Plus computer-assisted digital system (Leica Qwin Plus V3.2.1; Leica Microsystems). Stent area and the lumen area (LA) were calculated, and the neointimal area (NA) was calculated using the formula (strut area − luminal area). Percentage stenosis area was calculated as (1 − luminal area/strut area) ×100%.
Evaluation of arterial injury and inflammation scores
The severity of arterial injury was scored as previously described by Schwartz et al23 (0= no injury; 1= break in the internal elastic membrane; 2= perforation of the media; and 3= perforation of the external elastic membrane to the adventitia). The inflammation score for each individual strut was graded according to the following criteria:22,24 0= no inflammatory cells surrounding the strut; 1= light non-circumferential lymphohistiocytic infiltrate surrounding the strut; 2= localized, moderate to dense cellular aggregate surrounding the strut non-circumferentially; and 3= circumferential, dense lymphohistiocytic cell infiltration of the strut. Arterial injury and inflammation scores for each cross-section were calculated by dividing the sum of the individual injury and inflammation scores by the total number of struts at the examined section, as previously described.
Assessment of inflammatory cytokines after stent implantation
Peripheral blood samples were drawn before stent implantation and 3 days after stent implantation in the carotid artery. To limit circadian variation in cytokine production, fasting blood samples were collected in the morning into pyrogen-free tubes. Within 30 minutes of blood collection, plasma was obtained after a centrifugation at 3,000 rpm at 4°C for 15 minutes and stored at −80°C until assay. Plasma cytokine concentrations were measured by commercial enzyme-linked immunosorbent assay for interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α) according to manufacturer’s instructions (R&D Systems, Inc., Minneapolis, MN, USA). Measurements were performed in duplicate, and the mean value of two measurements was used.
Statistical analysis
All values are expressed as mean ± standard deviation. Significance of variability among the means of the experimental groups was determined by one-way analysis of variance, using SPSS 11.0 software. P<0.05 was considered as statistically significant.
Results
Coated stents
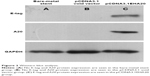
The stents were examined under a scanning electron microscope. The surface of bare-metal stents was smooth without any attachments (Figure 1A). The processed stents were evenly covered with a layer of collagen on their surface (Figure 1B).
Evaluation of in vitro transfection
Cultured ECs were seeded on the stents carrying A20. The transfection of these cells was evaluated by examination under a confocal laser scanning microscope. Figure 1C and D shows the general morphology of a stent in white balance mode. DAPI-stained nuclei appeared in blue (Figure 1E and F), indicating that cells were evenly distributed on the stent, covering it almost fully. Some of the cells emitted green fluorescence, suggesting that they were transfected with A20 (Figure 1G), while they were not transfected with A20 (Figure 1H).
Animals
Stents were successfully implanted in all animals. Angiography was performed 15 minutes later to confirm that the lumen was unobstructed, without filling defects, vascular wall dissection, embolization, stent migration, or distal vasospasm. No significant differences were observed in heart rate, blood pressure, and body weight between three groups in preoperative, intraoperative, and follow-up angiography. There were no obvious abnormalities in behavior, eating, and bowel movement after surgery.
Detection of A20 protein expression
Samples were collected from carotid arteries 3 days after they had been implanted with stents. Proteins were extracted from the collected blood vessel samples. Western blot analysis showed that there was no E-tag and A20 protein in the pCDNA3.1 void vector and bare-metal group (Figure 2A and B). There were E-tag and A20 protein expression in the pCDNA3.1EHA20 group (Figure 2C).
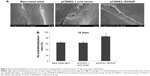
Detection of stent re-endothelialization
To determine the effect of three kinds of stents on re-endothelialization, stent coverage by ECs was evaluated by SEM. Two weeks after stent placement, there was a significantly higher extent of endothelial coverage above struts in the pCDNA3.1EHA20 group (85.10%±5.47%) compared with pCDNA3.1 void vector group (63.10%±3.93%) or bare-metal stent group (85.10%±5.47%) (P<0.05, Figure 3). There were no differences in the percentage of re-endothelialization between the pCDNA3.1 void vector group and bare-metal stent group (P>0.05, Figure 3).
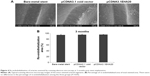
Three months after stent placement, the re-endothelialization areas for all stents were higher, there were no differences in the percentage of re-endothelialization (P>0.05), and the ECs completely covered the blood vessel in three groups (Figure 4).
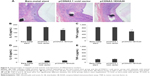
Histomorphometric analysis
There was no significant difference in arterial injury score and inflammation score among the three stent groups at 14 days and 3 months (P>0.05, Figure 5). Three months after stent implantation, morphological measurement results were as follows: 1) internal elastic lamina area (mm2): no significant difference among the three groups; 2) LA (mm2): the bare-metal stent group and pCDNA3.1 void vector group had significantly smaller LA than the pCDNA3.1EHA20 group (P<0.01); 3) NA (mm2): the bare-metal stent group and pCDNA3.1 void vector group had significantly larger NA than the pCDNA3.1EHA20 group (P<0.01); and 4) arterial stenosis rate (%): the bare-metal stent group and pCDNA3.1 void vector group had significantly larger stenosis rate than the pCDNA3.1EHA20 group (P<0.01). There was no significant difference in the above between the bare-metal stent group and pCDNA3.1 void vector group (P>0.05). (Refer to the specific values in Table 1, and typical pathological changes in Figure 6.)
Expression of inflammatory markers after stent implantation
We used enzyme-linked immunosorbent assay to determine the plasma levels of IL-1β and TNF-α before stent implantation and 3 days after stent implantation. TNF-α and IL-1β levels were not significantly different between the three groups before stent implantation. Their levels were significantly lower in the pCDNA3.1EHA20 group compared with the bare-metal stent group and the pCDNA3.1 void vector group (P<0.05, Figure 6). These results indicate that pCDNA3.1EHA20 coating on the stent downregulated inflammatory mediator expression and inhibited the inflammatory reaction.
Discussion
We have studied the possibility of using A20-modified intravascular stents to reduce neointimal formation and accelerate re-endothelialization as a treatment strategy. The objective of this study was to develop a treatment strategy that could reduce the incidence of ISR without increasing the risk of thrombosis. We used collagen to cover stents and then modified the stents with the A20 gene. Results showed that compared with stents in the control group, the A20-gene-modified stents significantly accelerated re-endothelialization in the blood vessels, which was apparent at day 14 and persisted at 3-month examinations. This effect was accompanied by a clear reduction of neointimal formation as detected by histomorphological and quantitative angiographic analysis. Therefore, for the first time, to our knowledge, we have proved that A20-gene-modified intravascular stents can reduce restenosis while promoting EC regeneration in a porcine carotid artery model.
Although the use of DES has significantly reduced restenosis, it has been found to be associated with long-term stent thrombosis. Recent reports indicate that after the discontinuation of clopidogrel administration, DES increases long-term mortality and Q-wave infarction rates, compared with bare-metal stents.11,25,26 Reducing restenosis while promoting re-endothelialization could be an alternative strategy to reduce stent thrombosis. Further benefits include avoiding the harmful effects of noncontinuous use of anti-platelet drugs. Compared with DES, gene-modified stents have the benefit of the combination of angioplasty and local gene delivery,27 for which the screening of appropriate functional genes is particularly important.
During their research on TNF response genes in ECs, Dixit et al28 revealed that the A20 gene has an open reading frame that encodes a novel zinc-finger protein. Currently, studies have shown that A20 binds to TRAF through its N-terminus, and binds to ABIN and IKK-γ via its C-terminus, thereby exerting its regulatory effects on the activity of nuclear factor κB and AP-1.29 On the one hand, A20 downregulates their activity levels, while on the other it also disrupts their synergistic regulatory effects on inflammatory response, exhibited as downregulation of the expression of inflammatory mediators such as TNF-α, IL-1, IL-6, and IL-8 and the inhibition of TNF-α-induced apoptosis.30 This will ultimately limit inflammation in the body and protect the cells from damage. Gene knockout experiments have shown that the A20 gene is an indispensable functional gene for the human body to resist inflammation, as well as an important gene for the physiological protection of ECs. A20 gene counteracts TNF, FAS, and natural killer cell-mediated cell death, and effectively blocks the NF-κB and the TOLL signaling pathways.31,32
Overexpression of the A20 gene prevents atherosclerosis in vascular grafts in allogenic kidney transplantation. In the ApoE−/− mouse model, A20 could effectively inhibit the occurrence of atherosclerosis.33 Furthermore, A20 protein overexpression prevented neointimal hyperplasia following rat carotid balloon injury and reverted pre-existing lesions.34,35
Previously, we effectively constructed tissue-engineered blood vessels with anti-atherosclerosis potential using A20-modified seed cells. Animal experiments demonstrated the therapeutic effects of these engineered blood vessels.18 Therefore, based on the dual function of A20 in inhibiting inflammatory response and protecting ECs, we used A20 to modify intravascular stents. The modified stents were implanted in vivo and investigated for their therapeutic effects and related mechanisms.
To accomplish this treatment strategy, it was necessary to identify an ideal stent/polymer/gene combination. Thus, we explored how to improve gene adherence to the stent. Because SPDP can effectively link two proteins without affecting their activities,36 it was used to link anti-DNA antibodies to collagen,37 thereby firmly attaching the A20 gene to the stents by taking advantage of antigen–antibody binding and allowing the A20 gene to withstand the impact of certain physiological conditions without falling off from the stents and to reach its maximum effectiveness at the target site. In addition, we chose liposome as the transfection reagent to minimize the adverse effects of other transfection reagents on cells. We first performed in vitro experiments, culturing the ECs on the stents carrying A20. Subsequent examination showed that A20 could be transfected into ECs. Further animal experiments showed that A20 protein expression could be detected 72 hours after stent implantation, indicating that this method can be used to successfully deliver genes to the vascular wall. Furthermore, we found that the A20 gene product downregulated the expression of inflammatory mediators such as TNF-α, IL-1 and inhibited the inflammatory reaction.
Our findings indicate that the effects of A20-modified stent could be seen on day 14. First, the degree of re-endothelialization was significantly higher in the experimental group than in the control group, indicating that A20-modified stent protected ECs. After 3 months, the results became even more evident. Although both the control group and the experimental group exhibited re-endothelialization, intimal hyperplasia was significantly reduced in the experimental group. Therefore, it is evident that A20 exerts its effects through protecting ECs and inhibiting proliferation of SMCs, which ultimately prevents the development of restenosis. Further studies are necessary to determine the mechanisms underlying these different processes.
Although vascular gene therapy is shown to be feasible in animal models, there is no definitive evidence on its benefits in humans. This may be due to many potential factors, one of which is the delivery method of the therapeutic genes to the blood vessel wall. Stent-based, liposome-mediated gene delivery has rarely been studied. However, the application of intravascular stents as carriers of therapeutic genes can extend the duration of the effect and achieve targeted delivery, which will generate ideal effects and have great application prospects. This study is among the very few reports on stent-based gene delivery in the vascular wall to reduce intimal hyperplasia and promote EC regeneration in a porcine model. It is also the first report to elucidate the effect of stent-based delivery of the A20 gene on the vascular wall.
Conclusion
Our studies have proved that stent-based overexpression of A20 inhibits ISR and promotes endothelial regeneration at the location of vascular lesions. This could be an ideal strategy to address the problem of ISR and provide an alternative to current DESs.
Acknowledgments
This work was supported by the National Natural Science Foundation of China for Distinguished Young Scholars (no 81000671), Natural Science Foundation of ChongQing (no 2010BB5190), and the National Natural Science Foundation of China (no 81471194). The authors also thank Ms Lu Ju for her assistance in SEM and confocal microscopic analysis.
Disclosure
The authors report no conflicts of interest in this work.
References
Demaerschalk BM, Hwang HM, Leung G. US cost burden of ischemic stroke: a systematic literature review. Am J Manag Care. 2010;16(7):525–533. | ||
Rothwell PM, Algra A, Amarenco P. Medical treatment in acute and long-term secondary prevention after transient ischaemic attack and ischaemic stroke. Lancet. 2011;377(9778):1681–1692. | ||
Bonati LH, Dobson J, Featherstone RL, et al. Long-term outcomes after stenting versus endarterectomy for treatment of symptomatic carotid stenosis: the International Carotid Stenting Study (ICSS) randomised trial. Lancet. 2015;385(9967):529–538. | ||
Mantese VA, Timaran CH, Chiu D, Begg RJ, Brott TG; CREST Investigators. The carotid revascularization endarterectomy versus stenting trial (CREST): stenting versus carotid endarterectomy for carotid disease. Stroke. 2010;41(10):S31–S34. | ||
Brott TG, Hobson RW, Howard G, et al. Stenting versus endarterectomy for treatment of carotid-artery stenosis. N Engl J Med. 2010;363(1):11–23. | ||
Bonati LH, Dobson J, Algra A, et al. Short-term outcome after stenting versus endarterectomy for symptomatic carotid stenosis: a preplanned meta-analysis of individual patient data. Lancet. 2010;376(9746):1062–1073. | ||
Shin YS, Kim BM, Suh SH, et al. Wingspan stenting for intracranial atherosclerotic stenosis: clinical outcomes and risk factors for in-stent restenosis. Neurosurgery. 2013;72(4):596–604. | ||
Wasser K, Schnaudigel S, Wohlfahrt J, et al. Clinical impact and predictors of carotid artery in-stent restenosis. J Neurol. 2012;259(9):1896–1902. | ||
Morice MC, Serruys PW, Sousa JE, et al. A randomized comparison of a sirolimus-eluting stent with a standard stent for coronary revascularization. N Engl J Med. 2002;346(23):1773–1780. | ||
Kedhi E, Joesoef KS, McFadden E, et al. Second-generation everolimus-eluting and paclitaxel-eluting stents in real-life practice (COMPARE): a randomised trial. Lancet. 2010;375(9710):201–209. | ||
Shuchman M. Debating the risks of drug-eluting stents. N Engl J Med. 2007;356(4):325–328. | ||
Stone GW, Moses JW, Ellis SG. Safety and efficacy of sirolimus and paclitaxel eluting coronary stents. N Engl J Med. 2007;356(10):998–1008. | ||
Hharif F, Hynes SO, Cooney R, et al. Gene-eluting stents: adenovirus-mediated delivery of eNOS to the blood vessel wall accelerates re-endothelialization and inhibits restenosis. Mol Ther. 2008;16(10):1674–1680. | ||
Liu J, Xu Y, Yang Q, et al. Reduction biodegradable brushed PDMAEMA derivatives synthesized by atom transfer radical polymerization and click chemistry for gene delivery. Acta Biomater. 2013;9(8):7758–7766. | ||
Sharif F, Hynes SO, McMahon J, et al. Gene-eluting stents: comparison of adenoviral and adeno-associated viral gene delivery to the blood vessel wall in vivo. Hum Gene Ther. 2006;17(7):741–750. | ||
Bonta PI, Pols TW, van Tiel CM, et al. Nuclear receptor Nurr1 is expressed in and is associated with human restenosis and inhibits vascular lesion formation in mice involving inhibition of smooth muscle cell proliferation and inflammation. Circulation. 2010;121(18):2023–2032. | ||
Takahashi M. Genetic susceptibility to restenosis: role of bone marrow cells and inflammatory response. Arterioscler Thromb Vasc Biol. 2009;29(10):1407–1408. | ||
Zhu CH, Ying DJ, Mi JH, et al. Development of anti-atherosclerosis tissue engineering blood vessel using zinc finger protein A20 regulated endothelial progenitor cells. Biomaterials. 2008;29(17):2628–2636. | ||
Hou CL, Zhang W, Wei Y, et al. Zinc finger protein A20 overexpression inhibits monocyte homing and protects endothelial cells from injury induced by high glucose. Genet Mol Res. 2011;10(2):1050–1059. | ||
Zhu CH, Ying DJ, Mi JH, et al. The zinc finger protein A20 gene protects endothelial cell from burns serum injury. Burns. 2004;30(2):127–133. | ||
Ma X, Hibbert B, Dhaliwal B, et al. Delayed re-endothelialization with rapamycin-coated stents is rescued by the addition of a glycogen synthase kinase-3beta inhibitor. Cardiovasc Res. 2010;86(2):338–345. | ||
Carter AJ, Aggarwal M, Kopia GA, et al. Long-term effects of polymer-based, slow-release, sirolimus-eluting stents in a porcine coronary model. Cardiovasc Res. 2004;63(4):617–624. | ||
Schwartz RS, Huber KC, Murphy JG, et al. Restenosis and the proportional neointimal response to coronary artery injury: results in a porcine model. J Am Coll Cardiol. 1992;19(2):267–274. | ||
Steigerwald K, Merl S, Kastrati A, et al. The pre-clinical assessment of rapamycin-eluting, durable polymer-free stent coating concepts. Biomaterials. 2009;30(4):632–637. | ||
Lassen JF. Long-term safety and efficacy of drug-eluting stents. Lancet. 2011;377(9773):1213–1214. | ||
Cook S, Ladich E, Nakazawa G, et al. Correlation of intravascular ultrasound findings with histopathological analysis of thrombus aspirates in patients with very late drug-eluting stent thrombosis. Circulation. 2009;120(5):391–399. | ||
Fishbein I, Alferiev I, Bakay M, et al. Local delivery of gene vectors from bare-metal stents by use of a biodegradable synthetic complex inhibits in-stent restenosis in rat carotid arteries. Circulation. 2008;117(16):2096–2103. | ||
Dixit VM, Green S, Sarma V, et al. Tumor necrosis factor-alpha induction of novel gene products in human endothelial cells including a macrophage-specific chemotaxin. J Biol Chem. 1990;265(5):2973–2978. | ||
Heyninck K, Beyaert R. A20 inhibits NF-kappaB activation by dual ubiquitin-editing functions. Trends Biochem Sci. 2005;30(1):1–4. | ||
Lee EG, Boone DL, Chai S, et al. Failure to regulate TNF-induced NF-kappaB and cell death responses in A20-deficient mice. Science. 2000;289(5488):2350–2354. | ||
Shembade N, Harhaj NS, Parvatiyar K, et al. The E3 ligase Itch negatively regulates inflammatory signaling pathways by controlling the function of the ubiquitin-editing enzyme A20. Nat Immunol. 2008;9(3):254–262. | ||
Wertz IE, O’Rourke KM, Zhou H, et al. De-ubiquitination and ubiquitin ligase domains of A20 downregulate NF-kappaB signalling. Nature. 2004;430(7000):694–699. | ||
Wolfrum S, Teupser D, Tan M, Chen KY, Breslow JL. The protective effect of A20 on atherosclerosis in apolipoprotein E-deficient mice is associated with reduced expression of NF-kappaB target genes. Proc Natl Acad Sci U S A. 2007;104(47):18601–18606. | ||
Patel VI, Daniel S, Longo CR, et al. A20, a modulator of smooth muscle cell proliferation and apoptosis, prevents and induces regression of neointimal hyperplasia. FASEB J. 2006;20(9):1418–1430. | ||
Damrauer SM, Fisher MD, Wada H, et al. A20 inhibits post-angioplasty restenosis by blocking macrophage trafficking and decreasing adventitial neovascularization. Atherosclerosis. 2010;211(2):404–408. | ||
Strehblow C, Schuster M, Moritz T, Kirch HC, Opalka B, Petri JB. Monoclonal antibody-polyethyleneimine conjugates targeting Her-2/neu or CD90 allow cell type-specific nonviral gene delivery. J Control Release. 2005;102(3):737–747. | ||
Jin X, Mei L, Song C, et al. Immobilization of plasmid DNA on an anti-DNA antibody modified coronary stent for intravascular site-specific gene therapy. J Gene Med. 2008;10(4):421–429. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.