Back to Journals » Vascular Health and Risk Management » Volume 11
Nonvitamin K antagonist oral anticoagulants (NOACs): the tide continues to come in
Authors Blann A
Received 2 June 2015
Accepted for publication 30 June 2015
Published 20 August 2015 Volume 2015:11 Pages 489—492
DOI https://doi.org/10.2147/VHRM.S89736
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Daniel Duprez
Andrew Blann
University of Birmingham Centre for Cardiovascular Sciences, City Hospital, Birmingham, UK
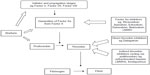
Thrombosis is the major common endpoint in most human diseases. In the coronary circulation, occlusive thrombi and/or the rupture of atherosclerotic plaque causes myocardial infarction, and in the cerebral circulation thrombosis, causes ischemic stroke. In the venous circulation, venous thromboembolism (VTE), manifesting clinically as pulmonary embolus and deep vein thrombosis (DVT), is a frequent complication among inpatients, and contributes to longer hospital stays with increased morbidity and mortality.1 Until perhaps 5 years ago, heparinoids (unfractionated heparin, low molecular weight heparin [LMWH], and fondaparinux) and vitamin K antagonists (VKAs: warfarin, acenocoumarol, phenocoumarol) were the only options for the prevention of thrombotic stroke in atrial fibrillation, and of VTE in general.2 Although effective, these traditional drugs have several practical, management, and clinical disadvantages, a fact that our colleagues in industry have not been slow to recognize and address by developing improved drugs, now collectively known as nonvitamin K antagonist oral anti coagulants (NOACs)2–4 (Table 1). These agents are steadily replacing the heparinoids and VKAs in both inpatient and outpatient prevention and treatment of thrombosis. Figure 1 illustrates the point of action of these drugs on the coagulation pathway.
  | Table 1 Disadvantages of traditional anticoagulants and NOAC reflection |
The first NOAC to be used in the clinic, dabigatran (a direct anti thrombin), was followed by three others that target coagulation Factor Xa (FXa): rivaroxaban, apixaban, and edoxaban.5–8 All have, or are expected to have, licenses for the treatment and/or prevention of VTE and stroke in a number of well-defined different clinical situations, such as in acute DVT or pulmonary embolus, after orthopedic surgery, and in atrial fibrillation. However, more NOACs are set to join the group,9 but many ask why do we need so many? The answer lies in consideration of the different pharmacokinetics and pharmacodynamics of each of the NOACs, and how these relate to the frequent comorbidities and other aspects of the patients. Although all of the current NOACs are preferable to the traditional drugs, each still has niggling problems that can be overcome with improved agents. Important factors influencing the choice of one particular NOAC over another include patient preference, once or twice daily dosing, drug–drug interactions, renal clearance, and hepatic metabolism. The latter is relevant as these drugs can influence and be influenced by the various cytochrome 450 enzymes and by the permeability glycoprotein (P-gp) pump. Furthermore, as the first NOACs to be trialed (ximelagratan) brought fears of long-term liver damage, all subsequent NOACs are now required to be assessed in this respect. Fortunately, all other NOACs have been shown to be hepato friendly and appear to be safer (in terms of increased liver function tests) than LMWH.10,11 An additional problem is of renal failure because of high degree of renal excretion of the four licensed NOACs; they cannot be used when the creatinine clearance/estimated glomerular filtration rate is very low, as this effectively means that the half-life of the NOAC is extended, leading to the increased risk of hemorrhage. Newcomers to this busy market, which include darexaban and betrixaban, both FXa inhibitors,11,12 must address these issues.
The new kids on the block
Preclinical studies of darexaban found it to be rapidly absorbed with or without a meal, with both blood and plasma concentrations peaking approximately 45 minutes after dosing, and with 51.9% excreted via feces and 46.4% via urine.12–14 It appears to have minimal interactions with digoxin and rifampicin, implying that the potential for drug–drug interactions between darexaban and CYP3A4 or P-gp-inducing agents is low.15,16 Although full-scale clinical development of this drug was stopped in 2011, darexaban has been trialed in the prevention of VTE after abdominal and orthopedic surgery.17–19 It has also been trialed in acute coronary syndromes,20 as has rixaroxaban (with successful end point outcomes),21 although guidelines from the European Society of Cardiology Working Group on Thrombosis recommends the use of newer antiplatelet agents over addition of NOACs in this setting.22 Thus, although there is a modest amount of promising literature on this drug, any advantage it can bring over its competitors remains to be seen and, should clinical development restart, robust additional data will be sought.
Betrixaban is further along the path to widespread acceptance, as fully described by the review from Chan et al in the current issue of the journal.23 This drug is important for several reasons. First, it has less than 1% metabolism via cytochrome P450, compared with 57%, <32%, and <25% for the other anti-FXa NOACs and <2% in the case of dabigatran. This means it is likely to have far fewer drug interactions and is more likely to be safe in those with liver disease. Second, renal excretion is in the region of 6%–13%, compared to >80%, 66%, 25%, and 35% for the other NOACs. The implication of this is that it is likely to be safe in those with severe renal failure, a feature unique to this drug, and so a highly sought-after characteristic. However, the fact that it has a slightly longer half-life (20 hours) compared to its competitors (9–15 hours) has both advantages (eg, less of a clinical issue if a dose is missed) and disadvantages (eg, longer time to wash out if hemorrhage). Betrixaban has been successfully trialed in the prevention of VTE in acutely ill medical patients, following orthopedic surgery and in stroke prevention in atrial fibrillation.
When things go wrong
A problem with all drugs is of overdose, and in the case of anticoagulants, this is hemorrhage. The short half-life of the NOACs (like that of heparins) means that simply stopping the drug should lead to a resumption of normal hemostasis. Nevertheless, antidotes have been called for and are in development.24 An antidote for dabigatran, idarucizumab (the antigen-binding site of a monoclonal antibody) is well into clinical development,25,26 as is a modified recombinant FXa (andexanet alpha), which lacks enzymatic activity, and which may inhibit all of the anti-FXa NOACs.27 Other antidotes in development include ciraparantag, a synthetic small molecule that reverses dabigatran, apixaban, and rivaroxaban, as well as subcutaneous fondaparinux and LMWH in vivo. Until such time as these become available (as possible afterward), severe hemorrhage is treated with blood components.28–32
A strength of the NOACs is that because their pharmacokinetics and pharmacodynamics are stable, routine blood monitoring is not required. However, there are several instances (as in overdose, or before emergency surgery) where the assessment of the actual anticoagulant status of the individual is needed. Regrettably, the most widely used laboratory tests, the prothrombin time and activated partial thromboplastin time, are unsuitable for the NOACs.32–36 Nevertheless, it is likely that the effectiveness of daraxaban (should it ever be needed) and of betrixaban can be determined, as with the other FXa inhibitors, by an anti-FXa assay such as the HepTest.
Conclusion
The NOACs are becoming an increasingly popular option for the most common causes and treatment of venous thrombosis and of thrombotic stroke in atrial fibrillation. The problem of renal excretion, and so, inadvisability in the face of low creatinine clearance, may be addressed by betrixaban, little of which is excreted via this organ. The problems of antidotes for these drugs are being addressed, and they may become available in 2016.
Disclosure
Dr Blann declares hospitality, speaker fees, and research funds from Pfizer, Bayer, Daiichi-Sankyo and Boehringer-Ingelheim. The University of Birmingham Centre for Cardiovascular Sciences at City Hospital, Birmingham, recruits into anticoagulant trials. The author reports no conflicts of interest in this work.
References
ISTH Stpering Committee for World Thrombosis Day. Thrombosis: a major contributor to the global disease burden. J Thromb Haemost. 2014;12:1580–1590. | |
Blann AD, Khoo CW. The prevention and treatment of venous thromboembolism with LMWHs and new anticoagulants. Vasc Health Risk Manag. 2009;5:693–704. | |
Riva N, Ageno W. Pros and cons of vitamin K antagonists and non-vitamin K antagonist oral anticoagulants. Semin Thromb Hemost. 2015;41:178–187. | |
Husted S, De Caterina R, Andreotti F, et al. ESC Working Group on Thrombosis Task Force on anticoagulants in heart disease. Non-vitamin K antagonist oral anticoagulants (NOACs): No longer new or novel. Thromb Haemost. 2014;111:781–782. | |
Stangier J, Rathgen K, Stahle H, Gansser D, Roth W. The pharmacokinetics, pharmacodynamics and tolerability of dabigatran etexilate, a new oral direct thrombin inhibitor, in healthy male subjects. Br J Clin Pharmacol. 2007;64:292–303. | |
Kubitza D, Becka M, Voith B, Zuehlsdorf M, Wensing G. Safety, pharmacodynamics, and pharmacokinetics of single doses of BAY59-7939, an oral, direct factor Xa inhibitor. Clin Pharmacol Ther. 2005;78:412–421. | |
Wong PC, Crain EJ, Xin B, et al. Apixaban, an oral, direct and highly selective factor Xa inhibitor: in vitro, antithrombotic and antihemostatic studies. J Thromb Haemost. 2008;6:820–829. | |
Ogata K, Mendell-Harary J, Tachibana M, et al. Clinical safety, tolerability, pharmacokinetics, and pharmacodynamics of the novel factor Xa inhibitor edoxaban in healthy volunteers. J Clin Pharmacol. 2010;50:743–753. | |
Ahrens I, Peter K, Lip GY, Bode C. Development and clinical applications of novel oral anticoagulants. Part II. Drugs under clinical investigation. Discov Med. 2012;13:445–450. | |
Caldeira D, Barra M, Santos AT, et al. Risk of drug-induced liver injury with the new oral anticoagulants: systematic review and meta-analysis. Heart. 2014;100:550–556. | |
Palladino M, Merli G, Thomson L. Evaluation of the oral direct factor Xa inhibitor – betrixaban. Expert Opin Investig Drugs. 2013;22:1465–1472. | |
Iwatsuki Y, Sato T, Moritani Y, et al. Biochemical and pharmacological profile of darexaban, an oral direct factor Xa inhibitor. Eur J Pharmacol. 2011;673:49–55. | |
Hashimoto T, Suzuki K, Kihara Y, et al. Absorption, metabolism and excretion of darexaban (YM150), a new direct factor Xa inhibitor in humans. Xenobiotica. 2013;43(6):534–547. | |
Kaku S, Suzuki M, Saitoh M, et al. Darexaban: anticoagulant effects in mice and human plasma in vitro, antithrombotic effects in thrombosis and bleeding models in mice and effects of anti-inhibitor coagulant complex and recombinant factor VIIa. Thromb Res. 2013;131:450–456. | |
Kadokura T, Groenendaal D, Heeringa M, et al. Darexaban (YM150), an oral direct factor Xa inhibitor, has no effect on the pharmacokinetics of digoxin. Eur J Drug Metab Pharmacokinet. 2014;39:1–9. | |
Groenendaal D, Strabach G, Garcia-Hernandez A, et al. The pharmacokinetics of darexaban are not affected to a clinically relevant degree by rifampicin, a strong inducer of P-glycoprotein and CYP3A4. Br J Clin Pharmacol. 2013;75(2):440–449. | |
Sakon M, Nakamura M. Darexaban (YM150) prevents venous thromboembolism in Japanese patients undergoing major abdominal surgery: phase III randomized, mechanical prophylaxis-controlled, open-label study. Thromb Res. 2012;130:e52–e59. | |
Fuji T, Nakamura M, Takeuchi M. Darexaban for the prevention of venous thromboembolism in Asian patients undergoing orthopedic surgery: results from 2 randomized, placebo-controlled, double-blind studies. Clin Appl Thromb Hemost. 2014;20:199–211. | |
Eriksson BI, Agnelli G, Gallus AS, et al. Darexaban (YM150) versus enoxaparin for the prevention of venous thromboembolism after total hip arthroplasty: a randomised phase IIb dose confirmation study (ONYX-3). Thromb Haemost. 2014;111:213–225. | |
Steg PG, Mehta SR, Jukema JW, et al. RUBY-1 investigators. RUBY-1: a randomized, double-blind, placebo-controlled trial of the safety and tolerability of the novel oral factor Xa inhibitor darexaban (YM150) following acute coronary syndrome. Eur Heart J. 2011;32:2541–2554. | |
Mega JL, Braunwald E, Wiviott SD, et al; ATLAS ACS 2–TIMI 51 Investigators. Rivaroxaban in patients with a recent acute coronary syndrome. N Engl J Med. 2012;366:9–19. | |
Ganetsky VS, Hadley DE, Thomas TF. Role of novel and emerging oral anticoagulants for secondary prevention of acute coronary syndromes. Pharmacotherapy. 2014;34:590–604. | |
Chan NC, Bhagirath V, Eikelboom JW. Profile of betrixaban and its potential in the prevention and treatment of venous thromboembolism. Vasc Health Risk Manag. 2015;11:343–351. | |
Greinacher A, Thiele T, Selleng K. Reversal of anticoagulants: an overview of current developments. Thromb Haemost. 2015;113:931–942. | |
Glund S, Moschetti V, Norris S, et al. A randomised study in healthy volunteers to investigate the safety, tolerability and pharmacokinetics of idarucizumab, a specific antidote to dabigatran. Thromb Haemost. 2015;113:943–951. | |
Pollack CV Jr, Reilly PA, Bernstein R, et al. Design and rationale for RE-VERSE AD: a phase 3 study of idarucizumab, a specific reversal agent for dabigatran. Thromb Haemost. 2015;114(1):198–205. | |
Lu G, DeGuzman FR, Hollenbach SJ, et al. A specific antidote for reversal of anticoagulation by direct and indirect inhibitors of coagulation factor Xa. Nat Med. 2013;19:446–451. | |
Fawole A, Daw HA, Crowther MA. Practical management of bleeding due to the anticoagulants dabigatran, rivaroxaban, and apixaban. Cleve Clin J Med. 2013;80:443–451. | |
Holster IL, Hunfield NGM, Kuipers EJ, Kruip MJHA, Tjwa ETTL. On the treatment of new oral anticoagulant-associated gastrointestinal haemorrhage. J Gastrointestin Liver Dis. 2013;22:229–231. | |
Majeed A, Schulman S. Bleeding and antidotes in new oral anticoagulants. Best Pract Res Clin Haematol. 2013;26:191–202. | |
Steiner T, Bohm M, Dichgans M, et al. Recommendations for the emergency management of complications associated with the new direct oral anticoagulants (DOACs) apixaban, dabigatran and rivaroxaban. Clin Res Cardiol. 2013;102:399–412. | |
Cushman M, Lim W, Zakai NA. Clinical Practice Guide on Antithrombotic Drug Dosing and Management of Anti-Thrombotic Drug-Associated Bleeding Complications in Adults. 2014. Available from: http://www.hematology.org/Clinicians/Guidelines-Quality/Quick-Ref/2869.aspx. February 2014 Edition. Accessed August 3, 2015. | |
Dinkelaar J, Patiwael S, Harenberg J, Leyte A, Brinkman HJ. Global coagulation tests: their applicability for measuring direct factor Xa- and thrombin inhibition and reversal of anticoagulation by prothrombin complex concentrate. Clin Chem Lab Med. 2014;52:1615–1623. | |
Blann AD. Non-vitamin K antagonist oral anticoagulants (NOACs): a view from the laboratory. Br J Biomed Sci. 2014;71:158–167. | |
Baglin T, Hillarp A, Tripodi A, Elalamy I, Buller H, Ageno W. Measuring oral direct inhibitors (ODIs) of thrombin and factor Xa: a recommendation from the Subcommittee on Control of Anticoagulation of the Scientific and Standardisation Committee of the ISTH. J Thromb Haemost. 2013;11:756–760. | |
Kitchen S, Gray E, Mackie I, Baglin T, Makris M. Measurement of non-coumarin anticoagulants and their effects on tests of haemostasis: guidance from the British Committee for standards in haematology. Br J Haematol. 2104;166:830–841. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.