Back to Journals » Infection and Drug Resistance » Volume 13
Non-Small Cell Lung Carcinoma with Concomitant Localized Pulmonary Melioidosis: A Rare Co-Existing Disease
Authors Laplertsakul G, Sutherasan Y , Suwatanapongched T, Incharoen P, Petnak T
Received 14 May 2020
Accepted for publication 27 July 2020
Published 24 August 2020 Volume 2020:13 Pages 2957—2961
DOI https://doi.org/10.2147/IDR.S262410
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Eric Nulens
Gunthiga Laplertsakul,1 Yuda Sutherasan,1 Thitiporn Suwatanapongched,2 Pimpin Incharoen,3 Tananchai Petnak1
1Division of Pulmonary and Pulmonary Critical Care Medicine, Department of Medicine, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand; 2Division of Diagnostic Radiology, Department of Diagnostic and Therapeutic Radiology, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand; 3Department of Pathology, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand
Correspondence: Tananchai Petnak
Division of Pulmonary and Pulmonary Critical Care Medicine, Department of Medicine, Faculty of Medicine Ramathibodi Hospital, Mahidol University, 270 Ramathibodi Hospital, Rama VI Road, Ratchathewi, Bangkok 10400, Thailand
Tel/Fax +66 2 201 1619
Email [email protected]
Abstract: Melioidosis is caused by Burkholderia pseudomallei, water-and-soil gram-negative bacteria predominantly found in Southeast Asia and Australia. Herein, we reported a 63-year-old Thai man presenting with prolonged fever, non-productive cough, and weight loss for 3 months. He underwent deceased donor kidney transplantation 4 years ago and was on many immunosuppressive agents after transplantation. At presentation, his chest radiograph showed a mass-like lesion in the left upper lobe. Histopathological examination of a transthoracic needle lung biopsy yielded adenocarcinoma, while tissue culture grew for B. pseudomallei. He was diagnosed with stage IIIA non-small cell lung cancer (T4N0M0) co-existing with localized pulmonary melioidosis. After intensive and eradication therapy for melioidosis, his well-being improved with the resolution of fever. He sequentially underwent left upper lobectomy, but the procedure was not accomplished due to severe adhesions surrounding the left lung and great vessels. After surgery, he received concurrent chemoradiation therapy for his lung cancer. Nevertheless, the disease progressed, and he finally passed away. Since fever is not a common manifestation of lung cancer, co-existing infection, such as tuberculosis, fungal infection, and melioidosis, should always be excluded in patients suspected of having lung cancer presenting with unexplained fever.
Keywords: non-small cell lung cancer, melioidosis, co-existing infection, kidney transplantation
Introduction
Co-existing infection simultaneously diagnosed with lung cancer is not a common manifestation. Therefore, the presence of a mass-like lesion and fever of unknown origin warrant further investigations to exclude primary infection mimicking malignancy or coinciding infection with lung cancer. Although concomitant infections secondary to post-obstructive pneumonia, the most common coinciding infection, occur approximately 20% of patients with lung cancer,1 a simultaneous presentation of lung cancer and melioidosis is rarely encountered in routine clinical practice. Herein, we reported a patient with an unusual simultaneous presentation of localized pulmonary Burkholderia pseudomallei infection and primary pulmonary adenocarcinoma in an old non-diabetic kidney transplant recipient who was a former smoker living in central Thailand, an endemic area of melioidosis.2
Case Report
A 63-year-old Thai male farmer residing in central Thailand presented with prolonged fever and non-productive cough for 3 months. He also complained of intermittent chest tightness at the left hemithorax and losing 4 kg of his weight. He was admitted to the local hospital and diagnosed with bacterial pneumonia at that time. However, his fever did not subside following 14 days of intravenous meropenem. He was, therefore, transferred to our hospital for further investigations. He was a former smoker of 25 pack-years quitted 2 years ago before the presentation. He had end-stage renal disease requiring deceased donor kidney transplantation 4 years ago. After transplantation, he was on immunosuppressive agents, including mycophenolate mofetil and cyclosporine. Acyclovir was also prescribed for herpes infection prophylaxis.
He was referred to our hospital. Physical examination showed an asthenic patient with anicteric sclera. His vital signs revealed a body temperature of 38°c, blood pressure of 100/60 mm Hg, a pulse rate of 90/minute, and a respiratory rate of 17/minute. His oxygen saturation was 95% at ambient air. Respiratory examination revealed decreased breath sound at the left upper lung zone without crackles or egophony. Other physical examinations were unremarkable.
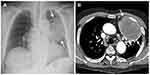
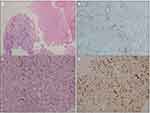
Complete blood count showed mild anemia with a hemoglobin level of 10.1 g/dL and leukocytosis with white blood cells count of 22 x 103/µL, 91% neutrophils. Blood chemistry tests remained within normal ranges. The chest radiograph (Figure 1A) and contrast-enhanced chest CT (Figure 1B) showed a large mass in the left upper lobe. A transthoracic core-needle biopsy of the left upper lobe mass was performed. The biopsy specimens were sent for pathological and microbiological studies. The histopathology revealed non-small cell carcinoma admixed with necrotic tissues. The immunohistochemical studies for cytokeratin (CK) AE1/AE3, CK7, and thyroid transcription factor-1 (TTF-1) were positive, compatible with primary lung adenocarcinoma. The Brown-Hopp stain revealed a single cluster of gram-negative bipolar rods with safety pin appearance characteristics of B. pseudomallei. (Figure 2A–D). B. pseudomallei subsequently grew on tissue culture. His serum antibody titer against B. pseudomallei was positive with a titer of 1:1280. His blood culture for aerobes and serum cryptococcal antigen were negative. The patient was finally diagnosed with stage IIIA non-small cell lung cancer (T4N0M0) co-existing with localized pulmonary melioidosis. The molecular analysis did not detect mutations of epidermal growth factor receptor (EGFR) genes and the rearrangement of anaplastic lymphoma kinase (ALK) gene.
After 3 weeks of intravenous ceftazidime, his fever subsided, and his well-being improved. He gradually gained his weight. Oral co-trimoxazole was sequentially prescribed for 20 weeks after the completion of intravenous ceftazidime treatment. He underwent surgery for left thoracotomy and left upper lobectomy. However, the procedure was not accomplished due to severe adhesion surrounding the entire left lung and great vessels. After surgery, he received concurrent chemoradiation therapy with a paclitaxel-based regimen for adenocarcinoma. Nevertheless, he developed progression of disease soon after. Three cycles of palliative chemotherapy using pemetrexed were delivered. Unfortunately, he finally passed away after 2 years after the initial presentation.
Discussion
Clinical presentations of lung cancer might be various. While common clinical symptoms include cough, chest tightness, and hemoptysis, patients with lung cancer incidentally discovered on radiological studies might be asymptomatic.3 Fever is considered as an atypical presentation of lung cancer and could be due to non-infectious causes related to cancer itself or related to psychogenic conditions, such as anxiety.4–6 However, infectious causes should always be excluded, particularly bacterial infection secondary to post-obstructive pneumonia, one of the most common causes of fever in patients with lung cancer. The differentiation between chronic infection, lung cancer, and a co-existing condition (akin to our case) is also challenging.
The diagnosis of melioidosis is usually difficult due to various and nonspecific clinical presentation. Generally, melioidosis is considered a great mimicker because it can cause chronic cavitary lesions mimicking reactivation of tuberculosis, a mass-like lesion leading to superior vena cava obstruction mimicking lung cancer,7 and acute fatal pneumonia similar to other infections.8 According to the previous study in northeastern Thailand, melioidosis accounted for 20% of community-acquired sepsis, with mortality rate of 35–40%.8 The most common radiographic finding of pulmonary melioidosis is lobar or segmental consolidation, followed by pulmonary nodules, cavity, and pleural effusion.9,10 Occasionally, melioidosis manifests as a mass-like lesion indistinguishable from lung cancer.11 A simultaneous presentation of lung cancer and chronic infection, especially melioidosis, is rare. Mays et al previously described lung cancer developed after a long latency of chronic cavitary form of pulmonary melioidosis.12 Their observation was different from our case. In the present case, the diagnosis of lung cancer was simultaneously established with the initial presentation of pulmonary melioidosis. At present, there is no clear mechanism explaining why malignancy can be infected, or such finding is just a coincidence. Although our patient did not have common risk factors of melioidosis, such as diabetes mellitus and thalassemia diseases, we speculated that worsening of immune status from immunosuppressive therapy accompanied by living in the endemic area might be responsible for developing melioidosis.13
Antibiotics therapy for melioidosis is generally divided into 2 phases, the initial intensive phase and the eradication phase. The antibiotic of choice for the intensive phase is intravenous ceftazidime or meropenem in the setting of critical illness.2 The duration of treatment in the intensive phase is at least 14 days but can be prolonged depending on the severity of disease and clinical response. The eradication phase is initiated following the intensive therapy aiming to prevent later relapse of melioidosis. The antibiotic of choice for eradication therapy is oral co-trimoxazole.2 According to a recent multicenter randomized, double-blind trial for eradication therapy in Thailand, combination therapy using co-trimoxazole and doxycycline did not provide an additional benefit over co-trimoxazole alone.14 The treatment duration for the eradication phase is at least 12–20 weeks, according to a lower rate of relapse compared to 8 weeks of treatment.
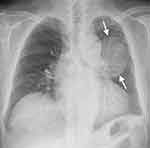
The median time of therapeutic response following the intravenous antibiotics was previously reported approximately 9 days, but delayed resolution of fever may occur up to 1 month.15 Therefore, an extended period of intensive therapy may be required in a slow-responsive patient. Our patient did not respond to a 14-day course of meropenem but responded well to a 21-day of intravenous ceftazidime. The follow-up chest radiograph obtained after intensive therapy and 8-weeks of co-trimoxazole monotherapy showing a slight decrease in size of the left upper lobe mass (Figure 3) suggesting regression of melioidosis within lung cancer.
Noteworthy, the most common pathogen coinciding with lung cancer is M. tuberculosis.16 In addition to melioidosis, other rare infections have been reported. The co-existence of endobronchial Talaromyces marneffei and adenocarcinoma has been reported in a 50-year-old healthy woman presenting with one month of chronic cough and prolonged fever.17 Entamoeba histolytica causing pulmonary infection in a patient with lung adenocarcinoma after receiving chemotherapy has also been reported.18
Conclusion
In conclusion, we have reported an uncommon simultaneous presentation of primary lung cancer and localized pulmonary melioidosis in an old man residing in the endemic area of B. pseudomallei. This case highlighted the importance to exclude an unusual cause of coinciding infection in patients with suspected lung cancer presenting with unexplained fever. Further histopathological and microbiological studies, including tissue culture for possible pathogens in endemic areas, are mandatory for a precise diagnosis and appropriate treatment.
Ethic and Consent Statement
This case report was approved by the Ethics Committee on Human Rights related to research involving human subjects at Faculty of Medicine Ramathibodi Hospital. The written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Disclosure
The abstract of this paper was presented at the CHEST Annual Meeting 2019 as a poster presentation with interim findings. The poster’s abstract was published in “Poster abstract” in the CHEST Journal Volume 156, Issue 4, Supplement: https://doi.org/10.1016/j.chest.2019.08.555. The authors report no conflicts of interest for this work.
References
1. Akinosoglou KS, Karkoulias K, Marangos M. Infectious complications in patients with lung cancer. Eur Rev Med Pharmacol Sci. 2013;17(1):8–18.
2. Wiersinga WJ, Virk HS, Torres AG, et al. Melioidosis. Nat Rev Dis Primers. 2018;4(1):17107. doi:10.1038/nrdp.2017.107
3. Buccheri G, Ferrigno D. Lung cancer: clinical presentation and specialist referral time. Eur Respir J. 2004;24(6):898–904. doi:10.1183/09031936.04.00113603
4. Kim MS, Koh JS, Baek HJ, Ryoo BY, Kim CH, Lee JC. Neoplastic fever caused by lung cancer. J Thorac Oncol. 2007;2(2):158–159. doi:10.1016/S1556-0864(15)30045-9
5. Zee YK, Soo RA. Non-small cell lung cancer presenting with neoplastic fever at diagnosis and relapse. Int J Infect Dis. 2010;14(6):e518–e521. doi:10.1016/j.ijid.2009.06.009
6. Xu M, Zhang X, Xu Z, et al. Psychogenic fever in a patient with small cell lung cancer: a case report. BMC Cancer. 2015;15(1):445. doi:10.1186/s12885-015-1462-z
7. Wilson M, Smith S, Brown J, Hanson J. Melioidosis mimicking primary lung malignancy with superior vena cava obstruction. IDCases. 2016;6:58–59. doi:10.1016/j.idcr.2016.09.014
8. Hinjoy S, Hantrakun V, Kongyu S, et al. Melioidosis in Thailand: present and future. Trop Med Infect Dis. 2018;3(2):38. doi:10.3390/tropicalmed3020038
9. Maneechotesuwan K. An exotic pulmonary infection in Thailand: melioidosis. Respirology. 1999;4(4):419–422. doi:10.1046/j.1440-1843.1999.00216.x
10. Burivong W, Wu X, Saenkote W, Stern EJ. Thoracic radiologic manifestations of melioidosis. Curr Probl Diagn Radiol. 2012;41(6):199–209. doi:10.1067/j.cpradiol.2012.01.001
11. Zaw KK, Wasgewatta SL, Kwong KK, Fielding D, Heraganahally SS, Currie BJ. Chronic pulmonary melioidosis masquerading as lung malignancy diagnosed by EBUS guided sheath technique. Respir Med Case Rep. 2019;28:100894.
12. Mays EE, Ricketts EA. Melioidosis: recrudescence associated with bronchogenic carcinoma twenty-six years following initial geographic exposure. Chest. 1975;68(2):261–263. doi:10.1378/chest.68.2.261
13. Suputtamongkol Y, Chaowagul W, Chetchotisakd P, et al. Risk factors for melioidosis and bacteremic melioidosis. Clin Infect Dis. 1999;29(2):408–413. doi:10.1086/520223
14. Chaowagul W, Suputtamongkol Y, Dance DA, Rajchanuvong A, Pattara-arechachai J, White NJ. Relapse in melioidosis: incidence and risk factors. J Infect Dis. 1993;168(5):1181–1185.
15. Simpson AJ, Suputtamongkol Y, Smith MD, et al. Comparison of imipenem and ceftazidime as therapy for severe melioidosis. Clin Infect Dis. 1999;29(2):381–387. doi:10.1086/520219
16. Morales-Garcia C, Parra-Ruiz J, Sanchez-Martinez JA, Delgado-Martin AE, Amzouz-Amzouz A, Hernandez-Quero J. Concomitant tuberculosis and lung cancer diagnosed by bronchoscopy. Int J Tuberc Lung Dis. 2015;19(9):1027–1032. doi:10.5588/ijtld.14.0578
17. Lin F, Qiu Y, Zeng W, Liang Y, Zhang J. Talaromyces marneffei infection in a lung cancer patient: a rare case report. BMC Infect Dis. 2019;19(1):336. doi:10.1186/s12879-019-3968-5
18. Liu YY, Ying Y, Chen C, et al. Primary pulmonary amebic abscess in a patient with pulmonary adenocarcinoma: a case report. Infect Dis Poverty. 2018;7(1):34. doi:10.1186/s40249-018-0419-2
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.