Back to Journals » OncoTargets and Therapy » Volume 13
Neuropilin-1 Interacts with Fibronectin-1 to Promote Epithelial–Mesenchymal Transition Progress in Gastric Cancer
Authors Wu C, Zeng M, Liao G , Qian K, Li H
Received 3 August 2020
Accepted for publication 28 September 2020
Published 20 October 2020 Volume 2020:13 Pages 10677—10687
DOI https://doi.org/10.2147/OTT.S275327
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sanjay Singh
Chao Wu, Meng-hua Zeng, Gang Liao, Kun Qian, Hui Li
Department of Gastrointestinal Surgery, The First Affiliated Hospital, Chongqing Medical University, Chongqing 400016, People’s Republic of China
Correspondence: Hui Li
Department of Gastrointestinal Surgery, The First Affiliated Hospital, Chongqing Medical University, Chongqing 400016, People’s Republic of China
Email [email protected]
Introduction: Neuropilin-1 (NRP1) binds to many ligands and co-receptors and affects cell survival and migration, which is essential for tumor progression. However, there are still largely unknowns about how NRP1 affects the epithelial–mesenchymal transition (EMT)-related malignant progression in gastric cancer.
Methods: We used TCGA to analyze the expression of NRP1 in gastric cancer and its impact on patient survival. In in vitro experiments, transwell, wound healing and colony formation assays were used to evaluate the effects of NRP1 and ginsenoside Rg3 on the invasion, migration and proliferation of gastric cancer cells. In in vivo experiments, we evaluated the overexpression and knockdown of NRP1 and the effect of ginsenoside Rg3 on tumor growth.
Results: We found that NRP1 is highly expressed in advanced gastric cancer and associated with poor prognosis. Knockdown of NRP1 expression can inhibit the proliferation and metastasis of gastric cancer cells. Mechanically. NRP1 interacts with fibronectin-1 (FN1) to promote the malignant progression of gastric cancer cells through ECM remodeling. In addition, we found that ginsenoside Rg3 can block the interaction of NRP1 and FN1 and inhibit the progression of gastric cancer.
Conclusion: Our study suggested that the interaction of NRP1 and FN1 is crucial for the malignant progression of gastric cancer. This may provide a new perspective and potential treatment methods for the treatment of gastric cancer.
Keywords: gastric cancer, neuropilin-1, fibronectin-1, epithelial–mesenchymal transition, ginsenoside Rg3
Introduction
Gastric cancer is one of the malignant tumors with the highest diagnosed and mortality in the world.1 It is characterized by a high recurrence and metastasis rate.2 Chemotherapy, surgery and targeted therapy are available for patients with early gastric cancer, among which surgical treatment is more effective.3 Meanwhile, the treatment of helicobacter pylori in recent years has indeed reduced the risk of gastric cancer. However, metastasis is still a major barrier to the successful treatment of gastric cancer that patients with primary advance gastric cancer or metastases occurrence are usually less effective in treatment.4
Epithelial–mesenchymal transition (EMT) is crucial for wound healing, embryogenesis and tumor progression.5 During EMT, loss of E-cadherin expression leads to decreased cell adhesion. While the expression of mesenchymal-related markers is up-regulated, notably N-cadherin, vimentin and fibronectin. Then cells acquire greater mobility and invasiveness.6,7 Extracellular matrix (ECM) remodeling is a necessary biological process during EMT to facilitate the dissemination of tumor cells. Fibronectin 1 (FN1) is distributed on the cell surface and ECM in the form of dimers or multimers, which is involved in cell adhesion and migration processes during tumor metastasis.8,9 Although the role of FN1 in tumor malignant progression and EMT is very clear, the molecular mechanism of how tumor cells participate in FN1-related extracellular matrix remodeling is still not very clear.
The transmembrane protein neuropilin-1 (NRP1) has been described to be involved in a wide range of pathological processes, including neuronal guidance, angiogenesis and tumor metastasis. NRP1 as a ligand can bind to extracellular components, such as VEGFA and SEMA3A. Here we found that NRP1 can bind to FN1 and promote EMT-related malignant progression of gastric cancer cells both in vitro and in vivo. In addition, we revealed that ginsenoside Rg3 can block the binding of NRP1 and FN1 to inhibit the malignant progression of gastric cancer cells.
Materials and Methods
Cell Lines
Gastric cancer cell lines MGC-803, MKN-45, MKN-28, Hs-746T, AGS, SNU-601, SNU-5 were obtained from the cell resource center of institute of basic medicine, Chinese Academy of Medical Sciences and cell resource center of shanghai institutes for biological sciences, Chinese Academy of Sciences. AGS, MKN-45 and MKN-28 cells were maintained in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS) at 37 °C with 5% CO2. SNU-5, Hs-746T, MGC-803 cells were maintained in DMEM medium supplemented with 10% fetal bovine serum (FBS) at 37 °C with 5% CO2. NRP1 overexpression or sh-NRP1 plasmids were transfected into cells using Lipofectamine 3000 reagent (Invitrogen, USA) according to the manufacturer’s instructions.
Clinical Specimens
Ten pairs of fresh tumor tissues and adjacent normal tissues were collected after surgery from patients in the First Affiliated Hospital, Chongqing Medical University. A written informed consent was obtained from each patient. Ethical approval from the First Affiliated Hospital, Chongqing Medical University was received before experiments and studies were performed in accordance with the Declaration of Helsinki.
Regents and Plasmids
Ginsenoside Rg3 was purchased from Aladdin china. NRP1 overexpression plasmid was obtained from Sino Biological (HG10011-NF, Sino Biological, China) with DNA sequencing confirmation. For NRP1 shRNA plasmid, the synthesized shRNA single-stranded nucleic acid (Genewiz, China) is annealed and cloned into pENTR/U6 vector. shRNA sequences were as followed: Top Strand,5ʹ-CACCGCTACGACCGGCTA GAAATCTCGAAAGATTTCTAGCCGGTCGTAGC −3ʹ, Bottom Strand, 5ʹ- AAAA GCTACGACCGGCTAGAAATCTTTCGAGATTTCTAGCCGGTCGTAGC −3ʹ
TCGA Analysis
The clinical stage and pathological grade information of gastric cancer obtained from the TCGA database were used to analyze the survival and expression of NRP1. Survival curves were calculated with the Kaplan–Meier method. The samples were divided into different clinical stages and pathological grades to analyze the expression and clinical value of NRP1.
Western Blot
Total proteins were isolated from cells and tissues using cell Extraction Reagent (Cat: P0013, Beyotime, China) with proteinase inhibitors. After quantified by Bicinchoninic Acid (BCA) method and separated in 10% SDS PAGE gel, proteins were transferred to polyvinylidene difluoride (PVDF) membrane. Then the membranes were blocked with 5% BSA for 2 h and subsequently incubated the following primary antibodies overnight at 4°C: NRP1 (1:500, DF7877, Affinity, China), E-Cadherin (1:1000, 14472, CST, USA), Vimentin, N-Cadherin (1:1000, 14215, CST, USA), and GAPDH (1:2000, 2118, CST, USA). After incubation with secondary antibody horseradish peroxidase-conjugated (HRP)-conjugated goat anti-rabbit antibody at room temperature for 2h. Bands were visualized using an enhanced chemiluminescence system (ECL, Millipore, USA) substrates according to the manufacturers’ instructions. Each experiment was performed in triplicate.
Colony Formation Assay
Cells were seeded into 6 well plate and transfected with NRP1 expression and shRNA plasmids. After 48 hours, the medium was removed, and the cells were digested into single cells. Then, the cells were seeded into a 6-well plate at a density of 1000 cells per well and cultured in an environment of 5% CO2 and 37°C for 2 weeks. Then the cells washed with PBS were fixed with absolute ethanol and stained with 1% crystal violet for 10 minutes. Finally, remove the background color and take a photo to record the number of clones. Each experiment was performed in triplicate.
Wound-Healing Assay
For cell migration analysis, transfected gastric cancer cells were seeded in 24-well plates to reach 90% confluence. Scrape the wound area with a sterile pipette tip, then wash with 1XPBS and continue to incubate in an environment of 5% CO2 and 37°C for 24 hours. The cells migration into the wound distance was imaged at 24 hours later. Each experiment was performed in triplicate.
Molecular Docking
NRP1 and FN1 protein crystal structures were downloaded from the PDB database (http://www.rcsb.org). Molecular docking was performed by using HEX software. The binding force was analyzed based on protein surface potential and surface structure. According to the docking results, we use protein interaction interface as an active pocket for high-throughput screening of the TCM database.
Invasion Assay
Transwell assay was performed to analyze cell invasion. A total of 105 cells were inoculated into the upper chamber coated with Matrigel and cultured in serum-free medium. Complete medium containing 10% FBS was added to the lower chamber and culture in an environment of 5% CO2 and 37°C. After 16 hours, cells on the bottom surface of the membrane were fixed with 4% formaldehyde and were stained with 1% crystal. After photographed, each group selects at least 6 fields of view to evaluate cell invasion ability. Each experiment was performed in triplicate.
Xenograft Model
In order to evaluate the role of NRP1 in tumor growth in vivo, we transfected NRP1 expression plasmid and shRNA plasmid into cells such as MKN-28 and MGC-803, respectively. Then use hygromycin or blasticidin to screen to obtain stable cell lines. Twenty-four 5-week-old Balb/c nude mice were randomly divided into 4 groups. These cells were inoculated subcutaneously at a concentration of 5X106 cells per mouse. To evaluate the effects of ginsenosides on tumor growth and metastasis, we inoculated MGC-803 subcutaneously or injected luciferase-labeled MGC-803 cells into the tail vein to make tumor growth or metastasis models. Ten days after inoculation, we evaluated the anti-tumor effect of ginsenoside Rg3 at concentrations of 5 mg/kg and 10 mg/kg. The tumor volume and animal survival status are monitored every three days. Thirty days after injection, animals were euthanized by intravenous injection of barbiturate at a final concentration of 100 mg/kg. Then, the solid tumors were harvested from the mice by surgery. Tumor volume was calculated as follows: V(volume) = (length × width2)/2. The ethical approval was obtained from The First Affiliated Hospital of Chongqing Medical University prior to the commencement of the study. All animal experiments were performed in accordance with the ethical standards of the Institutional Animal Care and Use Committee (IACUC) at The First Affiliated Hospital of Chongqing Medical University.
Immunohistochemistry
Formalin-fixed and paraffin-embedded tissue sections were blocked with 3% hydrogen peroxide for 10 min. Nonspecific binding was blocked by incubating the sections with 1% BSA in a humid chamber for 60 min. Incubation with the primary antibodies was subsequently performed overnight at 4°C using antibodies for E-cadherin (1:200, 14472, CST, USA) and vimentin (1:200, 14215, CST, USA), washed with phosphate-buffered saline (PBS) and then incubated with a biotin-conjugated secondary antibody for 25 min at 37 °C and visualized using diaminobenzidine (DAB). Staining intensity was scored as follows: 0 (no staining), 1(weak staining), 2 (moderate staining), 3 (strong staining). The extent of reactivity was scored as follows: 0 for negative cells, 1=1%-25%, 2=25%-50%, 3=50%-75%, 4=75%-100%. Each experiment was performed in triplicate.
Statistical Analysis
All statistical analyses were performed using SPSS 19.0 (SPSS, Chicago, IL, USA). Results of cell proliferation, migration and invasion were expressed as the mean ± standard deviation (SD). Significant differences among two groups were compared using Student’s t test. Comparisons among three or more groups were conducted using ANOVA with Dunnett’s post-test. P<0.05 was regarded as statistically significant and labeled with*.
Results
NRP1 is High Expression in Gastric Cancer and is Associated with Poor Prognosis
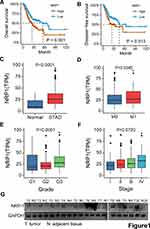
We obtained clinical information of gastric cancer samples from the TCGA database. All samples were divided into high and low groups based on the expression level of NRP1 for survival analysis. It was found that patients with high NRP1 expression often predicted a poor prognosis (Figure 1A and B). We also analyzed the expression of NRP1 in gastric cancer patients and control samples in TCGA database and found that NRP1 was highly expressed in gastric cancer patients (Figure 1C). Then we divided the samples according to different clinical stages, pathological grades and metastases to analyze the differences of NRP1 expression. The results showed that the expression of NRP1 was higher in gastric cancer patients with metastatic (Figure 1D), advanced high pathological grade (Figure 1E) and clinical stage (Figure 1F). In addition, we have detected the protein levels of NRP1 in 10 pairs of fresh tumor tissues and adjacent normal tissues by Western blot. Results showed that expression of NRP1 was higher in tumor (Figure 1G). Clinical information of the 10 patients was shown in Table S1. This shows that NRP1 is likely to promote the progression of gastric cancer.
NRP1 Expression is Associated with EMT-Related Proteins in Gastric Cancer
To further validate the function of NRP1in gastric cancer, we used GO and GSEA analysis to find that NRP1 contributes mainly to EMT-related ECM organization, tumor invasion and stemness (Figure 2A and B). These results suggest that NRP1 may be crucial for EMT-related gastric cancer progression. Analysis of the relationship between NRP1 and EMT-related factors in cervical cancer showed that NRP1 positively correlated with the expression of CA125, vimentin, MMPs. These results confirm the relationship between NRP1 and EMT and gastric cancer progression (Figure 2C-G).
NRP1 Overexpression Promotes Gastric Cancer Cell Proliferation and Invasion
In order to verify the role of NRP1 in gastric cancer, we tested the protein expression of NRP1 in 7 gastric cancer cells, and selected MGC-803 with the highest expression and MKN-28 with the lowest expression for subsequent experiments (Figure 3A). We knocked down or overexpressed NRP1 in MGC-803 and MKN-28 cells, respectively (Figure 3B). Then we used would healing, transwell and colony formation to verify the effect of NRP1 expression on cell migration, invasion and proliferation. The results showed that knocking down NRP1 could inhibit the migration (Figure 3C), invasion (Figure 3D) and proliferation (Figure 3E) of MGC-803, while overexpression of NRP1 in MKN-28 cells showed the opposite effect. GSEA, correlation analysis and cell function tests suggest that NRP1 is likely to be related to the evolution of EMT-related gastric cancer. Therefore, we detect the expression of EMT-related markers. The results showed that NRP1 can increase the expression of N-cadherin and Vimentin, but it inhibits the expression of E-cadherin (Figure 3F).
NRP1 Knockdown Inhibits the Growth in Xenograft Models
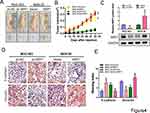
To confirm the effect of NRP1 on the growth of tumors in vivo, we established mouse xenograft models using MGC-803/NC, MGC-803/shNRP1, MKN-28/vector and MKN-28/NRP1 cells by subcutaneous injection. The results showed that NRP1 knockdown could significantly inhibit the growth of tumors, compared with the control group, while ectopic expression of NRP1 could promote tumor growth (Figure 4A and B). The expression of NRP1 in solid tumor was detected by Western blot (Figure 4C). The immunohistochemical staining results of the xenografted tumor tissues showed that NRP1 knockdown increased the expression of E-cadherin and decreased the expression of Vimentin (Figure 4D and E). Therefore, NRP1 knockdown could inhibit tumor growth of gastric cancer in vivo.
Ginsenoside Rg3 Blocks NRP1 and FN1 Interaction and Inhibits Gastric Cancer Cell Progression
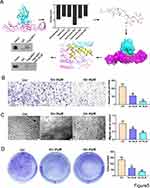
The above results show that the promotion of NRP1 on the progression of gastric cancer cells may be dependent on the EMT process. Since NRP1 is a membrane protein, we speculate that NRP1 may interact with extracellular matrix or cell motility-related proteins. By using Fpclass (http://dcv.uhnres.utoronto.ca/FPCLASS/) prediction, we obtained 80 proteins that potentially interacted with NRP1. Among these proteins, FN1 is an important component of extracellular matrix and is closely related to EMT. Next, we used molecular docking to simulate the interaction of NRP1 and FN1 and got the highest-scoring inhibitor, ginsenoside Rg3. The Co-IP test also confirmed the interaction of NRP1 and FN1 and the inhibitory effect of ginsenoside Rg3 on the interaction of the NRP1 (Figure 5A). Then we treated MGC-803 cells with 20uM and 40uM ginsenoside Rg3 to evaluate the effect of blocking NRP1 and FN1 interaction on the cells. The results showed that ginsenoside Rg3 can inhibit cell invasion (Figure 5B), metastasis (Figure 5C) and proliferation (Figure 5D). This revealed that blocking the interaction of NRP1 and FN1 can also achieve the inhibitory effect on the progression of gastric cancer cells caused by knocking down NRP1.
Ginsenoside Rg3 Inhibits Tumor Growth and Metastasis in vivo
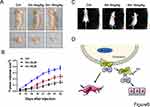
In order to confirm whether ginsenoside Rg3 inhibits the proliferation and metastasis of gastric cancer by blocking the interaction of NRP1 with FN1, we inoculated MGC-803 cells subcutaneously or luciferase labeled cells into Balb/c nude mice tail vein at a concentration of 5x106 cells per mice. Nine days after the injection, mice were treated with 5mg/kg and 10mg/kg ginsenoside Rg3. Thirty days after the injection, the mice were euthanized by carbon dioxide asphyxiation. The results show that ginsenoside Rg3 could indeed inhibit tumor growth (Figure 6A and B) and metastasis (Figure 6C). The above experiments showed that NRP1 could promote the malignant progression of EMT-dependent gastric cancer progression through interacting with FN1, and ginsenoside Rg3 can inhibit gastric cancer cells by blocking the interaction between NRP1 and FN1 (Figure 6D).
Discussion
Accumulating evidence indicates that EMT promotes cancer progression by conferring a mesenchymal phenotype associated with highly aggressive tumor cells. In the process of tumor malignant progression, tumor microenvironment and EMT-inducible transcription factors contribute to the occurrence of EMT. While, the expression and roles of different EMT inducers are diverse in different types of tumors. For example, Twist1 are essential for hepatocellular and breast cancer metastasis, but is dispensable in the process of pancreatic cancer metastasis.10−12 Similarly, ZEB2 has the effect of promoting the metastasis of glioma, gastric and colorectal cancer, but in melanoma, it activates melanocyte differentiation by regulating MITF levels to reduce the aggressiveness of melanoma.13–16 Although the EMT process in different tumors is triggered by different EMT-inducing factors, the similar is cell migration and invasion of carcinoma cells must pass through the basement membrane and extracellular matrix during EMT programs. Fibronectin is a ubiquitous ECM macromolecular protein involved in many physiological processes, including embryonic development, wound healing, and cell migration. During EMT progress, the interaction between FN and other ECM components participates in ECM remodeling, which is particularly important in the progression of tumors. For example, FN1 is very important for the polymerization collagen, which is a major component of ECM and is crucial for EMT-related tumor metastasis.17 In addition, the fusion of FN1 and FGF has an important biological effect on the promotion of mesenchymal tumors.18 In melanoma, FN1 inhibits cell apoptosis and promotes EMT process to promote tumor cell proliferation and metastasis.19 Study also showed that Cancer-associated fibroblasts mediated FN assemble is an important marker of prostate cancer and pancreatic cancer metastasis.20 In addition, FN1-mediated EMT process in tumors is also regulated by transcription factors and miRNAs.21,22
In the present study, we found that FN1 interacts with transmembrane proteins NRP1 and promotes the proliferation, migration and metastasis of gastric cancer cells mediated by EMT. And this combination can be blocked by ginsenoside Rg3, which was found to have anti-tumor effects both in vivo and in vitro experiments. It indicates that during the malignant progression of gastric cancer, transmembrane proteins are involved in ECM remodeling. Previous studies have shown that NRP1 is involved in the regulation of multiple tumor metastasis-related signal pathways. NRP1 was found to promote Met/β1-integrin internalization and activates the FAK/p130Cas pathway, thereby promoting colorectal cancer cell migration and metastasis.23 In lung cancer cells, NRP1-positive cells showed increased expression of OCT-4, Bmi-1 and NANOG, as well as higher cell migration and self-renewal capabilities.24 NRP1 was also found to promote radiation-induced lung adenocarcinoma cell EMT through TGF-β1/Smad signaling to enhance the invasion and migration of lung adenocarcinoma cells.25 However, in renal cell carcinoma, high expression of NRP1 often predicts improved survival.26 In gastric cancer, NRP1 is usually highly expressed and promotes tumor cell proliferation and metastasis.27–29 Similarly, here we also found high expression of NRP1 in gastric cancer and its association with poor prognosis. Interestingly, we found that the interaction of NRP1 and FN1 promotes EMT of gastric cancer cells. This may explain that NRP1’s promotion of tumor EMT process is based on the ECM remodeling. Results of knocking down NRP1 or using ginsenosides to block the interaction of NRP1 and FN1 further confirmed that the lack of NRP1 and the loss of interaction with FN1 can inhibit the proliferation and metastasis of gastric cancer cells.
Overall, after confirming that NRP1 is highly expressed in gastric cancer and is associated with gastric cancer metastasis and poor patient prognosis, we found that the promotion of NRP1 on the EMT process of gastric cancer cells depends on FN1 interaction. In addition, we found that ginsenosides can break the interaction between NRP1 and FN1 and thereby inhibit the proliferation and metastasis of gastric cancer cells. This may provide a new perspective on the mechanism of gastric cancer metastasis and potential therapeutic targets and drugs for the treatment of gastric cancer.
Acknowledgments
The results shown here are part based upon data generated by the TCGA Research Network: https://www.cancer.gov/tcga.
Disclosure
The authors declare that they have no conflict of interest.
References
1. Cao L, Chen Y, Zhang M, et al. Identification of hub genes and potential molecular mechanisms in gastric cancer by integrated bioinformatics analysis. Peer J. 2018;6:e5180. doi:10.7717/peerj.5180
2. Chen D, Liu G, Xu N, et al. Knockdown of ARK5 expression suppresses invasion and metastasis of gastric cancer. Cell Physiol Biochem. 2017;42:1025–1036. doi:10.1159/000478685
3. Shen Z, Kauttu T, Seppanen H, et al. Both macrophages and hypoxia play critical role in regulating invasion of gastric cancer in vitro. Acta Oncol. 2013;52(4):852–860. doi:10.3109/0284186X.2012.718444
4. Chen L, Shi Y, Zhu X, et al. IL10 secreted by cancer-associated macrophages regulates proliferation and invasion in gastric cancer cells via cMet/STAT3 signaling. Oncol Rep. 2019. doi:10.3892/or.2019.7206
5. Brabletz T, Kalluri R, Nieto MA, et al. EMT in cancer. Nat Rev Cancer. 2018;18:128–134.
6. Chaffer CL, San Juan BP, Lim E, et al. EMT, cell plasticity and metastasis. Cancer Metastasis Rev. 2016;35:645–654. doi:10.1007/s10555-016-9648-7
7. Derynck R, Weinberg RA. EMT and cancer: more than meets the eye. Dev Cell. 2019;49:313–316. doi:10.1016/j.devcel.2019.04.026
8. Peixoto P, Etcheverry A, Aubry M, et al. EMT is associated with an epigenetic signature of ECM remodeling genes. Cell Death Dis. 2019;10:205.
9. Li Y, Hong J, Jung BK, et al. Oncolytic Ad co-expressing decorin and Wnt decoy receptor overcomes chemoresistance of desmoplastic tumor through degradation of ECM and inhibition of EMT. Cancer Lett. 2019;459:15–29. doi:10.1016/j.canlet.2019.05.033
10. Tran HD, Luitel K, Kim M, et al. Transient SNAIL1 expression is necessary for metastatic competence in breast cancer. Cancer Res. 2014;74:6330–6340. doi:10.1158/0008-5472.CAN-14-0923
11. Meng J, Chen S, Han JX, et al. Twist1 regulates vimentin through Cul2 Circular RNA to promote EMT in hepatocellular carcinoma. Cancer Res. 2018;78:4150–4162.
12. Zheng X, Carstens JL, Kim J, et al. Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature. 2015;527:525–530. doi:10.1038/nature16064
13. Nagaishi M, Fujii Y, Sugiura Y, et al. Increased Twist and ZEB2 expression in a cutaneous metastasis of high-grade glioma. Neuropathology. 2020;40:196–201. doi:10.1111/neup.12621
14. Li MZ, Wang JJ, Yang SB, et al. ZEB2 promotes tumor metastasis and correlates with poor prognosis of human colorectal cancer. Am J Transl Res. 2017;9:2838–2851.
15. Dai YH, Tang YP, Zhu HY, et al. ZEB2 promotes the metastasis of gastric cancer and modulates epithelial mesenchymal transition of gastric cancer cells. Dig Dis Sci. 2012;57:1253–1260. doi:10.1007/s10620-012-2042-6
16. Caramel J, Papadogeorgakis E, Hill L, et al. A switch in the expression of embryonic EMT-inducers drives the development of malignant melanoma. Cancer Cell. 2013;24:466–480. doi:10.1016/j.ccr.2013.08.018
17. Velling T, Risteli J, Wennerberg K, et al. Polymerization of type I and III collagens is dependent on fibronectin and enhanced by integrins alpha 11beta 1 and alpha 2beta 1. J Biol Chem. 2002;277:37377–37381. doi:10.1074/jbc.M206286200
18. Lee JC, Su SY, Changou CA, et al. Characterization of FN1-FGFR1 and novel FN1-FGF1 fusion genes in a large series of phosphaturic mesenchymal tumors. Mod Pathol. 2016;29:1335–1346. doi:10.1038/modpathol.2016.137
19. Li B, Shen W, Peng H, et al. Fibronectin 1 promotes melanoma proliferation and metastasis by inhibiting apoptosis and regulating EMT. Onco Targets Ther. 2019;12:3207–3221.
20. Erdogan B, Ao M, White LM, et al. Cancer-associated fibroblasts promote directional cancer cell migration by aligning fibronectin. J Cell Biol. 2017;216:3799–3816. doi:10.1083/jcb.201704053
21. Wu J, Wang Y, Xu X, et al. Transcriptional activation of FN1 and IL11 by HMGA2 promotes the malignant behavior of colorectal cancer. Carcinogenesis. 2016;37:511–521. doi:10.1093/carcin/bgw029
22. Liang H, Yu M, Yang R, et al. A PTAL-miR-101-FN1 axis promotes EMT and invasion-metastasis in serous ovarian cancer. Mol Ther Oncolytics. 2020;16:53–62.
23. Huang X, Ye Q, Chen M, et al. N-glycosylation-defective splice variants of neuropilin-1 promote metastasis by activating endosomal signals. Nat Commun. 2019;10(1):3708. doi:10.1038/s41467-019-11580-4
24. Jimenez-Hernandez LE, Vazquez-Santillan K, Castro-Oropeza R, et al. NRP1-positive lung cancer cells possess tumor-initiating properties. Oncol Rep. 2018;39:349–357.
25. Chen Z, Gao H, Dong Z, et al. NRP1 regulates radiation-induced EMT via TGF-beta/Smad signaling in lung adenocarcinoma cells. Int J Radiat Biol. 2020;1-15.
26. Morin E, Lindskog C, Johansson M, et al. Perivascular Neuropilin-1 expression is an independent marker of improved survival in renal cell carcinoma. J Pathol. 2020;250(4):387–396. doi:10.1002/path.5380
27. Li L, Jiang X, Zhang Q, et al. Neuropilin-1 is associated with clinicopathology of gastric cancer and contributes to cell proliferation and migration as multifunctional co-receptors. J Exp Clin Cancer Res. 2016;35(1):16. doi:10.1186/s13046-016-0291-5
28. Akagi M, Kawaguchi M, Liu W, et al. Induction of neuropilin-1 and vascular endothelial growth factor by epidermal growth factor in human gastric cancer cells. Br J Cancer. 2003;88(5):796–802. doi:10.1038/sj.bjc.6600811
29. Van Cutsem E, de Haas S, Kang YK, et al. Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a biomarker evaluation from the AVAGAST randomized Phase III trial. J Clin Oncol. 2012;30(17):2119–2127. doi:10.1200/JCO.2011.39.9824
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.