Back to Journals » Neuropsychiatric Disease and Treatment » Volume 15
Neurocognitive mechanisms of theory of mind impairment in neurodegeneration: a transdiagnostic approach
Authors Strikwerda-Brown C , Ramanan S , Irish M
Received 6 December 2018
Accepted for publication 18 January 2019
Published 20 February 2019 Volume 2019:15 Pages 557—573
DOI https://doi.org/10.2147/NDT.S158996
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yuping Ning
Cherie Strikwerda-Brown,1–3 Siddharth Ramanan,1–3 Muireann Irish1–3
1The University of Sydney, Brain and Mind Centre, Sydney, NSW, Australia; 2The University of Sydney, School of Psychology, Sydney, NSW, Australia; 3ARC Centre of Excellence in Cognition and its Disorders, Sydney, NSW, Australia
Abstract: Much of human interaction is predicated upon our innate capacity to infer the thoughts, beliefs, emotions, and perspectives of others, in short, to possess a “theory of mind” (ToM). While the term has evolved considerably since its inception, ToM encompasses our unique ability to apprehend the mental states of others, enabling us to anticipate and predict subsequent behavior. From a developmental perspective, ToM has been a topic of keen research interest, with numerous studies seeking to explicate the origins of this fundamental capacity and its disruption in developmental disorders such as autism. The study of ToM at the opposite end of the lifespan, however, is paradoxically new born, emerging as a topic of interest in its own right comparatively recently. Here, we consider the unique insights afforded by studying ToM capacity in neurodegenerative disorders. Arguing from a novel, transdiagnostic perspective, we consider how ToM vulnerability reflects the progressive degradation of neural circuits specialized for an array of higher-order cognitive processes. This mechanistic approach enables us to consider the common and unique neurocognitive mechanisms that underpin ToM dysfunction across neurodegenerative disorders and for the first time examine its relation to behavioral disturbances across social, intimate, legal, and criminal settings. As such, we aim to provide a comprehensive overview of ToM research in neurodegeneration, the resultant challenges for family members, clinicians, and the legal profession, and future directions worthy of exploration.
Keywords: prefrontal cortex, social cognition, mentalizing, executive function, dementia, empathy
Introduction
The term “theory of mind” (ToM) was first coined in 1978 to refer to an individual’s capacity to attribute mental states to oneself and others.1 Since then, over 1,450 studies have been published on this topic, exploring the cognitive and neural mechanisms that support this capacity and its disruption in clinical populations (Figure 1). The definition has since been updated to incorporate a broader range of abilities, including understanding and inferring the thoughts, feelings, beliefs, and intentions of ourselves (ie, first-person ToM) and others (ie, third-person ToM).2 Intuitively, the conceptualization of ToM overlaps with the related construct of empathy, which involves the basic recognition and understanding of another person’s affective state, in addition to the sharing of this emotional experience.3 The precise manner in which these two processes are related, however, remains a matter of debate.3,4 One argument holds that ToM is a “domain-specific” ability, distinct from other cognitive functions,5 providing the foundational mechanism upon which other complex social processes, such as empathy,6 are built. By contrast, the “domain-general” theory proposes that ToM involves the understanding of representations in general, invoking various component cognitive processes, some of which are shared with empathy3 and other social functions.7 Accordingly, affective ToM (ie, feelings and emotions) is separable from its cognitive counterpart (ie, thoughts, beliefs, and intentions).8
  | Figure 1 The exponential increase in number of published articles on theory of mind since its inception. |
As shall be demonstrated, this fractionation between cognitive and affective ToM has proven particularly useful in characterizing the nature of social cognitive dysfunction in neurodegeneration,9 as well as in differentiating between dementia syndromes.10 Nonetheless, parcellating dynamic and multifaceted social interactions into exclusive categories remains challenging,11 as cognitive and affective ToM are themselves multifaceted constructs, which are not always uniformly affected in neurodegeneration.12,13 Moreover, according to the domain-general account, multiple cognitive abilities support ToM capacity, including executive function,14,15 memory,16 language,17 and visuospatial skills,18 a pertinent issue to consider in the context of neurodegenerative disorders characterized by widespread cognitive impairment. Here, we provide an update on ToM research in neurodegeneration, considering how large-scale brain network dysfunction, and resultant cognitive impairment, impacts cognitive and affective expressions of ToM. In doing so, we aim to explicate the common and divergent neurocognitive mechanisms that subtend ToM dysfunction across neurodegenerative disorders and adjudicate between domain-specific vs domain-general accounts of ToM impairments in these syndromes.
Why study ToM in neurodegenerative disorders?
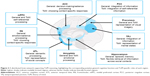
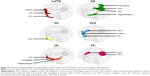
Functional neuroimaging studies in healthy individuals, complemented by lesion evidence from clinical populations, have accelerated our understanding of the neural circuitry supporting ToM performance. In the last decade, however, we have witnessed a shift from understanding the roles of localized brain regions to considering interactions between large-scale neural networks that support complex cognitive endeavors such as ToM (Figure 2). The study of neurodegenerative disorders offers compelling insights into the neurocognitive architecture of ToM, as these syndromes target large-scale brain networks implicated in ToM and many of its associated processes (Figure 3).19 Moreover, neurodegenerative disorders frequently present with co-occurring social cognitive, memory, and executive impairments, offering an opportunity to explore the intersection between ToM and cognitive function more broadly. In this review, we focus on the most common neurodegenerative syndromes that present with early impairments in social cognition, as well as emerging evidence of ToM dysfunction in syndromes not traditionally classified as disorders of social cognition.
Behavioral-variant of frontotemporal dementia (bvFTD)
BvFTD represents the prototypical disorder of social cognition. Characterized by insidious changes in personality and behavior, patients with bvFTD display flagrant violation of social norms, lack of social comportment, and apparent loss of empathy for others.20 The considerable parallels in social cognitive difficulties between bvFTD and developmental disorders, such as autism, provided an early clue that ToM might be altered in this dementia syndrome (John R Hodges, personal communication). From a neuroanatomical perspective, this observation prior to the advent of modern day neuroimaging techniques was particularly astute, given the now well-established vulnerability of large-scale networks specialized for socioemotional processing in bvFTD.21
The emergence of prominent ToM impairments as manifested in socially disruptive and inappropriate behaviors typically heralds the onset of bvFTD.22 Family members frequently report the affected individual to lack warmth, have an apparent disregard for others, and to display increasingly rigid and egocentric behavior. Collectively, these disturbances are posited to reflect a core, syndrome-specific difficulty in ToM,23–30 which in turn profoundly increases carer burden.31 The observation of significant cognitive and affective ToM disruption irrespective of modality of testing (ie, verbal or nonverbal) points to a primary ToM impairment in bvFTD. The evidence to date corroborates this position, with bvFTD patients presenting marked deficits across the broad spectrum of ToM tasks including first-order (ie, understanding others’ false beliefs about the world)32 and second-order false belief (ie, understanding others’ false beliefs about a third party’s mental state),22,33–35 detection of social faux pas,30,31 intention and emotion attribution on short video vignettes,36 and decoding emotion from visual cues (Reading the Mind in the Eyes Task; RMET).22
Mechanisms of ToM disruption in bvFTD
Arriving at a precise understanding of the cognitive origins of ToM disruption in bvFTD has proven challenging, in part due to the widespread nature of cognitive impairment in this syndrome. As such, ToM impairments may arise simply as a consequence of global cognitive and executive dysfunction, rather than a primary socioemotional impairment per se. By this view, bvFTD patients would be predicted to perform poorly on ToM tasks that demand greater executive resources. Paradoxically, however, bvFTD patients demonstrate deficits on even simple first-order false belief tasks32 as well as nonverbal ToM tasks with limited cognitive and executive loading,16 suggesting a fundamental, domain-specific impairment. Crucially, ToM impairments in bvFTD emerge statistically independent of overall cognitive and executive status when data-driven clustering and prediction models are employed,14,37 suggesting that social cognitive deficits are dissociable from general cognitive decline in this syndrome. The observation of significant difficulties on ToM, but not on related control tasks,38 reinforces the presence of a core ToM impairment, independent of co-occurring executive, memory, language, and other general cognitive deficits in this syndrome.14,25,37
Could ToM impairments arise simply as a function of widespread socioemotional dysfunction characteristic of the bvFTD syndrome? A recent meta-analysis suggests that ToM and emotion processing impairments are present to a commensurate degree.25 It is possible that the deterioration of affective ToM abilities co-occurs with a general decline in emotion processing in bvFTD,27,39 reflecting the shared neural structures implicated in these abilities. Importantly, however, the presence of early deficits on cognitive, as well as affective, ToM tasks in bvFTD suggests that emotion processing is unlikely to entirely account for disrupted ToM in this syndrome. Other mechanisms, such as an inability to integrate social and contextual cues, likely also contribute to ToM dysfunction in bvFTD.40 Furthermore, it remains unclear whether the ToM impairment in bvFTD reflects a general incapacity to inhibit one’s own mental perspective35 or adopt any perspective beyond the “here and now”,41,42 resonating with recent reports of egocentric, rigid behavior, and environmental dependency in this syndrome.43,44 As such, while ToM impairments in bvFTD appear to manifest independent of memory and executive functions, the precise contribution of socioemotional processes remains to be fully elucidated.
Neuroanatomy of ToM impairments in bvFTD
A common assumption is that ToM dysfunction in bvFTD reflects early atrophy to the medial prefrontal cortex (mPFC), one of the core regions of the ToM brain network (Figure 2).23,45 Indeed, the magnitude of visually rated mPFC atrophy in bvFTD patients directly correlates with objective impairments in false belief and faux-pas judgments.32 Reduced gray matter intensity of mPFC and neighboring prefrontal regions has been linked to performance impairments on the ToM component of the Frith–Happé animation task.16 Similarly, a reduction in coherent, resting state mPFC activity has been found to correlate with poorer performance on emotion attribution tasks employing short story vignettes.46 Finally, modulative neurostimulation via transcranial direct current stimulation over the mPFC has been demonstrated to improve accuracy on cognitive ToM (measured using intention attribution) tasks in bvFTD,36 suggesting a causal role for this region in ToM performance.
Neurodegenerative disorders, however, rarely target discrete regions in isolation,47 and progressive neural degradation in bvFTD reveals the importance of regions beyond the mPFC in modulating ToM performance. Emerging evidence suggests that in parallel with early atrophy of the mPFC, anterior cingulate cortices/frontoinsular (ACC/fIN) regions are particularly vulnerable to functional disruption and volumetric reduction in this syndrome.48 The importance of ACC/fIN regions in the context of ToM impairments in bvFTD deserves particular attention (Figure 3). A prominent hypothesis contends that accumulation of pathology around the ACC/fIN regions in bvFTD directly targets a specific population of neurons called von Economo neurons (VENs). VENs are an evolutionarily specialized set of cortical neurons suggested to play a critical role in ToM capacity, possibly through supporting the selection of quick and intuitive responses during uncertain social situations.49 Furthermore, the ACC/fIN cortices, along with the mPFC, are implicated in regulating and selecting context-specific emotional responses,50 and their degradation, in part, may contribute to co-occurring emotion dysregulation and ToM impairments in bvFTD. Finally, the ACC/fIN regions anchor the Salience Network of the brain, proposed to tag incoming salient information for further processing, thereby guiding appropriate behavioral and social responses.51 Degeneration of ACC/fIN regions reduces VEN counts,52 disrupts Salience Network functioning,53 and is directly implicated in ToM dysfunction in bvFTD.16 Atrophy of the ACC/fIN cortices in bvFTD is associated with compromised cognitive ToM on the Frith–Happé animations16 and affective ToM difficulties on emotion attribution tasks.54 Furthermore, lower emotion attribution scores on affective ToM tasks in bvFTD parametrically relate to reduced connectivity between ACC/fIN, PFC, and temporal cortices, as demonstrated using resting-state functional connectivity,46 suggesting that alterations in ACC/fIN circuitry disrupt ToM performance in bvFTD.
Looking beyond the prefrontal cortex
We next consider how damage to regions beyond the prefrontal cortex gives rise to ToM dysfunction in bvFTD. With disease progression, atrophy encroaches into the anterior and medial temporal lobes most noticeably on the right-hand side,55 including the hippocampus and amygdala,56 parietal cortices including temporoparietal junction (TPJ), precuneus,57 and posterior cingulate cortices (PCC)58 as well as subcortical regions including the thalamus and caudate nuclei.58 Importantly, atrophy to regions beyond the prefrontal cortex is associated with ToM dysfunction in bvFTD. For example, impairments on emotion attribution and faux-pas tasks in bvFTD have been shown to correlate with lateral temporal cortex integrity, including the right anterior temporal lobes (ATLs)59,60 and superior temporal cortices,31,61 as well as medial and lateral parietal regions such as the TPJ, precuneus,54 and PCC.61 Emergent evidence has further implicated cerebellar degeneration in modulating aspects of cognitive dysfunction62 as well as ToM difficulties on the Frith–Happé animations task.16 Longitudinal studies tracking how the evolution of ToM dysfunction in bvFTD relates to encroachment of pathology in temporoparietal cortices and the cerebellum are clearly warranted.
Alzheimer’s disease (AD)
At first glance, it seems somewhat counterintuitive to discuss AD in the context of ToM impairment, given its conception primarily as an amnestic disorder. Clinically, AD patients are noted to retain their warmth, affability, and social graces,63–65 with social cognitive impairments typically manifesting in later stages of the disease course.66 As a result, AD patients were historically included in ToM studies as disease control groups rather than syndromes of interest. With mounting evidence of episodic memory impairments in bvFTD,67,68 ToM performance was further suggested to hold high diagnostic utility in the discrimination of bvFTD from AD.69 The emerging picture, however, is less clear, with mixed reports of moderate impairments on social cognitive tasks in AD, modulated largely by the cognitive demands of the task itself.
Mechanisms of ToM disruption in AD
Two competing hypotheses have been advanced in relation to ToM disturbances in AD. One position holds that AD patients display a fundamental, domain-specific ToM impairment, in light of evidence for a degraded capacity to accurately infer and attribute beliefs, intentionality, and emotional states to characters on some studies of cognitive (eg, false belief tasks70,71) and affective (eg, RMET70) ToM. Other findings reveal significant impairments in AD in inferring mental exchanges of interlocutors from simple conversational exchanges in vignettes72 as well as in real life.73 The observation that impairment in attributing physical causality to characters in first-order false belief tasks is independent of comprehension abilities and executive performance70 has been interpreted as reflecting fundamental cognitive and affective ToM impairments in AD even on tasks with relatively low cognitive demand.
On the other hand, a more prominent view holds that ToM impairments in AD reflect a global decline in cognitive processes including episodic and working memory and executive function.24,74 In support of this position, general cognitive, executive, and memory performances in AD have been shown to predict subsequent ToM capacity.14,74 For example, Ramanan et al14 found that between 50% and 70% of cognitive ToM performance in AD (as measured by the faux-pas test) could be explained by patients’ overall attention and executive performance. Similarly, Synn et al16 found significant associations between ToM performance on the Frith–Happé animations and episodic memory retrieval impairments in AD. Moreover, AD performance on ToM tasks with low cognitive demands, such as first-order false belief, is relatively spared in the majority of studies,32,34,75,76 whereas its more cognitively demanding counterpart (second-order false belief) is impaired.72 Interestingly, AD patients are impaired even on control items of second-order false belief tasks75 that rely on working memory and comprehension abilities, further suggesting that ToM deficits in the syndrome arise due to the cognitive complexity of the tasks.
Although the aforementioned studies suggest that a decline in executive and memory functions influences ToM performance in AD, the precise nature of their contributions remains unclear. These processes may support the integration and maintenance of relevant information on ToM tasks, as well as facilitating the inference of mental states in the presence or absence of situational cues.77,78 With disease progression, increasing executive, visuospatial, and general cognitive dysfunction likely impact ToM performance, especially on tasks that tax visuospatial abilities or lack explicit situational cues.16,79 In this regard, the study by Synn et al16 is notable in demonstrating comparable difficulties on the ToM and “random movement” conditions of the Frith–Happé animations task, suggesting that ambiguity of stimuli and increasingly abstract task demands contribute to ToM impairments in AD.16 By contrast, with the provision of situational and social contexts, ToM deficits in AD are mitigated,79 presumably due to attenuation of the executive and mnemonic task demands. In summary, the bulk of evidence points to a domain-general impairment in ToM, attributable to a primary decline in memory, executive, and general cognitive abilities.
Neuroanatomy of ToM impairments in AD
Efforts to clarify the neuroanatomical signature of ToM disruption in AD have been limited as social cognitive impairments emerge relatively late in the disease course.66 Moreover, the diffuse gray and white matter neural damage at these later stages limits our capacity to attribute emergent impairments exclusively to the degeneration of the ToM network. Two recent studies, however, warrant discussion. First, Le Bouc et al35 found ToM performance impairments on a false belief task in a combined cohort of AD and bvFTD patients to correlate with hypometabolism of the left TPJ/inferior parietal cortex; however, the admixture of bvFTD patients in this analysis precludes any interpretation of these results as specific to AD. Second, Synn et al16 revealed significant associations between impaired overall performance on the Frith–Happé animations task and reduced gray matter integrity of the right hippocampus and bilateral cerebellum in AD. In addition to their key role in the ToM network, the TPJ, hippocampus, and cerebellum have also been implicated in general cognitive processes such as memory and executive function (Figure 2). As such, whether the involvement of these regions in ToM impairments in AD reflects a domain-specific or domain-general process remains unclear. For example, the TPJ and neighboring parietal regions play a well-established role in supporting the representation of mental states,80 but are also involved in the integration of information pertinent to successful semantic81 and episodic memory retrieval.82 Although typically discussed in relation to episodic memory dysfunction in AD,68 the hippocampus may facilitate ToM by allowing us to draw upon past experiences to anticipate social intentions and reactions83 and supporting cognitive flexibility by updating our knowledge of social structure through acquired information.84 Finally, cerebellar involvement has been reported during ToM tasks requiring high levels of abstraction, such as thinking about other’s traits,85 yet it is also implicated in executive control and working memory capacity.86 As such, further research delineating the precise role of these regions in ToM will enable us to adjudicate between domain-specific and domain-general accounts of ToM disruption in AD.
Amyotrophic lateral sclerosis (ALS)
ALS is a neurodegenerative disease involving the degradation of motor neurons in the brainstem, spinal cord, and motor cortex, manifesting in a progressive loss of motor function. Until recently, ALS was considered primarily a motor disease, although cognitive and behavioral changes are now accepted as common features of the condition. While a proportion of ALS patients will also be diagnosed with frontotemporal dementia (FTD), up to 50% of the individuals without dementia87 develop impairments in executive functioning, language, memory, and behavior, leading to the conceptualization of ALS and FTD as lying on a disease continuum.88 Accumulating evidence reveals ToM deficits in ALS, not unlike those found in bvFTD, leading to inclusion of deficits in “social cognition” in the revised diagnostic criteria for the ALS-FTD spectrum disorder.89 The neuroanatomical profiles of ALS and FTD, however, are not identical, with ALS spreading from its origin in the brain stem or spinal cord into motor cortex and subsequently frontotemporal, anterior cingulate, and basal ganglia regions.90 The cognitive and neurobiological processes underlying impaired ToM in ALS are thus unlikely to precisely mirror those implicated in FTD.
ToM impairments have been uncovered across the majority of tasks in ALS. This incorporates basic affective ToM tasks, involving the attribution of emotions to characters based on vignettes (emotional attribution tasks)91,92 or facial expressions (RMET).91,93 Performance on more complex measures of cognitive and affective ToM is also compromised, including false belief tasks,13,93 understanding thoughts and feelings of cartoon characters (Happé cartoons),94,95 detecting faux pas,96 and attributing intentions to characters in stories92,97 (but see Ref. 98). Findings are less consistent for making social inferences from dynamic videos (The Awareness of Social Inference Test; TASIT)94,99,100 and judging others’ preferences based on eye-gaze (Judgment of Preference task),12,101–103 with intact performance revealed by some, but not other, studies. On balance, though, ToM impairments appear relatively pervasive in ALS.
Mechanisms of ToM disruption in ALS
Unsurprisingly, the cognitive mechanisms underlying ToM impairments in ALS vary according to the task in question. The so-called “pure” affective tasks (eg, RMET, emotion attribution) typically do not correlate with executive function in ALS,91,92,98 indicating that degraded affective ToM in ALS arises independently from executive dysfunction. Similarly, executive function is not associated with performance on more complex ToM tasks combining affective and cognitive components, such as faux pas96 and intention attribution,98 further suggesting a domain-specific impairment of ToM in ALS. By contrast, executive functions have been consistently implicated in performance on false belief13 and the Happé cartoons94,95 in ALS. This suggests that these latter tasks impose greater executive demands and as such may be less sensitive in detecting “pure” ToM impairment independent of other cognitive confounds. Studies examining the contribution of cognitive processes beyond executive function are limited; however, preliminary findings hint at a role for verbal memory and nonabstract reasoning in emotional attribution91 and semantic naming in Judgment of Preference.102
Neuroanatomy of ToM impairments in ALS
The neural substrates of ToM disruption in ALS appear to be task dependent. Emotion attribution performance is linked to gray matter intensity decrease in the ACC/fIN regions,98 as well as reduced resting state functional connectivity between PCC and mPFC and between left supramarginal gyrus and right frontoparietal regions.104 As such, impairments in affective ToM in ALS may result from disruption to a distributed frontotemporo–parietal network. A separate network, however, has been implicated in the more complex process of detecting false beliefs. Poor performance on this task in ALS is associated with reduced PET glucose consumption in bilateral dorsomedial and dorsolateral prefrontal cortex and supplementary motor area,13 potentially reflecting the executive demands of the test. In addition, in a task-based fMRI study, poor Judgment of Preference performance in ALS was related to reduced activation in the right precentral gyrus extending into the inferior frontal gyrus.103 Intriguingly, the same study revealed that reduced ToM in ALS was associated with increased brain activity in the right postcentral gyrus.103 Increased brain activity on PET imaging has also been documented in relation to the false belief task in ALS, with hypermetabolism in the left fusiform gyrus correlating with reduced performance.93 Together, these findings potentially reflect compensatory activity in the face of widespread degeneration of the ToM network.
Huntington’s disease (HD)
HD is a dominantly inherited neurodegenerative disease affecting motor, psychiatric, and cognitive functioning. Although formal diagnosis of HD is based on the presence of an extrapyramidal movement disorder, the advent of genetic testing has permitted assessment of symptoms in premanifest individuals harboring the CAG-repeat expansion in the Huntingtin gene, with mounting evidence to suggest that cognitive and psychiatric deficits can actually predate motor symptom onset.105 HD pathology primarily infiltrates subcortical regions including the striatum, putamen, and caudate, and related frontostriatal networks, though also affects amygdala, insula, and thalamus, with extension into wider cortical areas with disease progression.106 Given this neuroanatomical signature, it is unsurprising that deficits in ToM are a cardinal feature of the manifest disease.107
Mechanisms of ToM disruption in HD
In patients with manifest HD, cognitive and affective ToM impairments have been documented across the majority of studies, irrespective of task employed. This includes basic affective tasks of decoding emotions from visual cues (RMET),108,109 attributing emotions to story characters,108 as well as low demand cognitive tasks such as ascribing mental states to animated shapes (Frith–Happé animations)110 and spatial perspective taking.111 More complex measures encompassing both cognitive and affective elements are also impaired in HD, including faux pas detection,111,112 making social inferences (TASIT),108,113 understanding the thoughts and feelings of cartoon characters (Happé cartoons),29 and attributing intentions to others.114,115 Unlike other neurodegenerative disorders, ToM performance is closely linked to executive function in HD, with consistent correlations between ToM impairments and executive function emerging across all the aforementioned studies. More intriguingly, deficits in intention attribution were ameliorated when cognitive flexibility impairments were controlled for.115 Taken together, these findings suggest that the pervasive ToM impairments in HD are largely attributable to executive dysfunction, and as such, domain-general in nature. One anomaly is the Judgment of Preference task, which does not correlate with executive function in HD.29,116 Performance on this measure is not consistently impaired in HD,29 suggesting that it may capture slightly different aspects of ToM vs other tasks. Deficits in ToM have also been documented prior to motor symptom onset in premanifest HD (ie, individuals carrying the CAG-repeat expansion in the Huntingtin gene), specifically on tests of faux pas detection117 and mental state attribution (Frith–Happé task).117 Other tasks, including Judgment of Preference116 and RMET,118 while intact at the whole-group level in premanifest HD, are positively associated with time to symptom onset, suggesting that alterations in ToM ability may be an early feature of the disease.
Neuroanatomy of ToM impairments in HD
To date, no neuroimaging studies to our knowledge have directly related ToM performance in manifest HD to underlying brain changes. Based on the progression of pathology, ToM impairments in manifest HD are speculated to reflect widespread cortical atrophy of frontoparietal regions supporting higher-level cognition; a proposal that meshes well with the executive contribution to these deficits. Evidence from premanifest cases, however, suggests that alterations in functional brain networks likely play a mediating role. For example, in premanifest HD, reduced task-based functional connectivity between the left amygdala and the right fusiform face area during viewing of facial expressions was correlated with poorer attribution of emotions on a separate measure of RMET.118 By contrast, a task-based fMRI study of cognitive ToM, involving the attribution of intentions and beliefs to cartoon characters, failed to discriminate between premanifest and control cases on the behavioral level, with no significant differences in fMRI activation patterns emerging during task performance.119 As such, early impairments in affective ToM in HD may arise due to dysfunction to regions supporting emotional processing, such as the amygdala, with more extensive ToM deficits developing across the course of the disease, as a result of progressive neurodegeneration into frontoparietal cortices.107 The genetic nature of this condition offers a unique opportunity to test this hypothesis, by identifying individuals in advance of marked cognitive decline and tracking longitudinal structural and functional network changes that may give rise to ToM disruption.
Parkinson’s disease (PD)
PD is a progressive extrapyramidal movement disorder, involving the degeneration of dopaminergic neurons in substantia nigra, striatum, thalamus, and subthalamic nuclei, which secondarily affects frontostriatal loops. Although traditionally conceptualized as a motor disorder, nonmotor symptoms are common, with the majority (80%) of PD patients developing mild cognitive impairment followed by frank dementia.120 The characteristic cognitive complaints in PD include executive, visuospatial, memory, and processing speed impairments,121 with mounting evidence of significant social cognitive dysfunction.122
Mechanisms of ToM disruption in PD
Impairments in ToM are well established in PD, evident across the majority of tests employed including basic affective tasks of ascribing emotions to faces on the RMET (eg, reference 123–125 but see reference 18), determining the affective state of cartoon characters in stories (emotion attribution task),126 as well as low cognitive demand tasks such as Judgment of Preference,127–129 and more complex cognitive and affective measures of false belief130,131 (but see reference 125), faux pas detection,18,132,133 understanding of nonliteral utterances (Advanced Test of Theory of Mind),126,134 attribution of intentions to characters in comic strips,124 and other story-based tasks.135 It has been suggested that ToM impairments in PD may be at least partly attributable to executive dysfunction. Significant associations between executive function and ToM performance in PD have been demonstrated on complex measures combining cognitive and affective elements, such as faux pas detection,135,136 Advanced Test of Theory of Mind,126,134 intention attribution,124 and other story tasks.137 Mixed findings, however, have been reported on less cognitively demanding tasks, such as Judgment of Preference127,128 and RMET,123,124,128,138 with ToM correlating with executive function only at later disease stages.124,127,138 Collectively, this suggests the presence of an independent, domain-specific ToM deficit in early stages of PD, with executive dysfunction playing a modulating role in more complex tasks and with disease progression. The modality of testing also warrants consideration given prominent visuospatial impairments in PD. Although a recent meta-analysis found no evidence for a disproportionate impairment on visual vs verbal ToM tasks,122 impaired RMET performance has been associated with poor visuospatial function139 and reduced nonverbal reasoning140 in PD. Determining the precise role of visuospatial dysfunction in ToM impairment in PD is required, particularly given the heavy visual demands of many commonly used measures.
Neuroanatomy of ToM impairments in PD
The prefrontal cortex has long been implicated in ToM dysfunction in PD, given secondary disruption to frontostriatal loops as a consequence of dopaminergic degeneration.141 Indeed, recent studies point to a distributed frontoparietal network mediating ToM impairments in PD, suggesting that these deficits reflect damage beyond dopaminergic pathways.18 Specifically, understanding nonliteral utterances on the Advanced Test of Theory of Mind is associated with reduced gray matter volume in lateral frontal and parietal regions and reduced white matter connectivity in tracts adjacent to frontal and parietal regions (eg, superior longitudinal fasciculus).134 Interestingly, correlations with these gray and frontal white matter areas are negated when executive function performance is controlled for, with integrity of the superior longitudinal fasciculus adjacent to the parietal lobe remaining significant. These findings further imply a domain-specific ToM impairment in PD, associated with the disruption of frontoparietal tracts.
Other neurodegenerative disorders
Comparatively less attention has been paid to ToM impairments in the primary progressive aphasias (eg, semantic dementia [SD], progressive nonfluent aphasia [PNFA], and logopenic aphasia [LPA]), Parkinson-related syndromes (eg, dementia with Lewy Bodies [DLB], progressive supranuclear palsy [PSP], and corticobasal syndrome [CBS]), and vascular dementia (VD). Emerging evidence, however, suggests significant ToM deficits across the majority of these syndromes, reflecting disruption of key nodes of the canonical ToM network. While current consensus criteria emphasize aphasia as the most prominent clinical feature,142 mounting evidence of social cognitive dysfunction in primary progressive aphasia suggests that the cognitive profile extends beyond the domain of language. For example, ToM impairments have been characterized in SD on cartoon tasks designed to circumvent the semantic deficits of this syndrome.60,143 Importantly, a recent study revealed that impaired ToM capacity in SD persisted despite controlling for overall semantic comprehension, reflecting atrophy in right anterior temporal cortices, as well as the bilateral amygdala and temporal poles, left orbitofrontal, and insular cortices.60 Similarly, ToM impairments on RMET in PNFA have been shown to relate to atrophy in the insula, temporal pole, and amygdala,144 bilaterally. These studies add to a growing body of evidence implicating the right temporal lobe in the origin of social cognitive dysfunction in neurodegenerative disorders.145,146 While ToM has not been directly examined in LPA, early atrophy to the TPJ, along with reports of empathy loss,147 converges to suggest that ToM is likely to be affected.
With regard to Parkinson-related syndromes, deficits on false belief,148 faux pas,148,149 and RMET148,149 tasks have been uncovered in DLB. Interestingly, only RMET performance was correlated with executive function, supported on the neural level by an association with bilateral superior and middle frontal gyri atrophy in DLB.149 By contrast, poor performance on the faux-pas test in DLB correlated with atrophy to key nodes of the ToM network, including mPFC, TPJ, precuneus, and insula,149 suggesting that this test may capture core aspects of ToM impairment in this syndrome. In PSP, impairments in social inference-making have been documented on the TASIT, reflecting atrophy in mPFC.150 Finally, RMET impairments in a single case of CBS have been posited to reflect mPFC hypometabolism.151 The status of ToM capacity in VD remains unclear, with intact comprehension of insincere speech (TASIT),27 but impaired RMET, faux pas, and false belief performance in a single case study.148 These equivocal findings likely reflect the diffuse neuroanatomical predilection of this disease, which can target subcortical and/or cortical regions.
Research on ToM remains in its infancy in these disorders, yet the findings to date converge to suggest transdiagnostic alterations in ToM capacity in neurodegeneration. Future work uncovering the nature and extent of these impairments in the lesser researched syndromes is essential not only to validate this hypothesis but also to assist in identifying and managing these diagnostically challenging syndromes and providing feedback to clinicians and carers about the expected deficits. The careful study of ToM disruption in these syndromes will further refine our understanding of the neurocognitive architecture of ToM more broadly.
Summary: Toward a refined understanding of ToM impairment in neurodegeneration
Taken together, the evidence suggests that ToM impairments, and their respective neurocognitive mechanisms, vary in a syndrome-specific manner. Domain-specific impairments in ToM are present in bvFTD, ALS, and PD, reflecting pathological insult targeting, but not exclusive to, multiple nodes of the ToM network (Figure 3). These deficits, however, are exacerbated with encroachment of disease pathology into brain regions supporting ancillary cognitive processes implicated in ToM, such as executive function. By contrast, the bulk of the behavioral evidence in AD points to domain-general ToM impairments, attributable to disruption of brain regions supporting memory and executive processes. Finally, ToM impairments in manifest HD patients are strongly related to executive dysfunction, suggesting a domain-general impairment. Findings of amygdala involvement in ToM dysfunction in premanifest HD, however, raise the possibility of a domain-specific impairment of affective ToM in the earliest stages of the disease. Determining how this variability across syndromes manifests on the behavioral level is essential to arrive at a comprehensive understanding of ToM and its expression in everyday functioning. For example, it may be that neurodegenerative disorders with domain-specific ToM impairments (eg, bvFTD, ALS, PD, and HD) display significantly greater disruption to real-world social functioning than those with domain-general deficits (eg, AD), an issue we tackle in the next section.
Understanding the real-world impact of ToM disturbances in neurodegeneration
The literature reviewed thus far highlights ToM disruption as a potential transdiagnostic feature across neurodegenerative disorders, mediated by common and divergent neurocognitive mechanisms. The majority of these studies, however, have employed laboratory-based measures of ToM, which arguably lack many of the contextual and situational cues that inform social behavior in the real world. While more recently developed tasks involving dynamic videos of social interactions, such as the TASIT,11 offer improved ecological validity, the participant nevertheless remains an observer, rather than an active agent in the social milieu. As such, it is unclear to what extent ToM performance in the laboratory reflects real-world social functioning in its many permutations and settings. A more thorough picture of ToM deficits, and its resultant impact on interpersonal function, may be gleaned from carer observations and reports of patients’ behavior in daily life. This approach reveals syndrome-specific patterns of social functioning that do not always neatly map onto ToM performance as assessed formally on experimental tasks. For example, although significant impairments on ToM tasks have been documented in AD, such patients are widely reported to uphold appropriate social functioning, at least in the mild-to-moderate stages of the disease. In particular, social graces, interpersonal warmth, and the ability to form and maintain interpersonal relationships remain intact in AD,63–65 as does empathic concern and behavior.152 In fact, social–emotional sensitivity may be enhanced with AD onset, attributable to increased functional connectivity between the ACC/fIN and striatum.53 We suggest that this discrepancy between compromised task performance and intact real-world behavior in AD reflects the often ambiguous and contextually devoid nature of ToM tasks. For example, many of the tasks in which significant ToM impairments in AD have been documented rely upon interpretation of decontextualized stimuli, such as cartoons or photographs. Social function, however, hinges upon multifaceted interpersonal and contextual cues enabling us to interpret and understand other’s mental states (see also Ref. 79). Individuals with AD may find abstract tasks of ToM function overly cognitively demanding,16 yet if assessed within familiar and contextually rich social settings may display appropriate behavior. Our brief survey of the literature highlights the need for a new breed of ToM tasks to better capture this dimension of social cognitive functioning in AD.
Despite limited translation of laboratory-based ToM studies to daily functioning in AD, emerging evidence suggests that ToM impairments closely mirror real-world social dysfunction in other neurodegenerative disorders. This is exemplified by bvFTD, where carer reports of severe behavioral disruption in everyday life corroborate the stark ToM impairments displayed by patients on formal testing. For example, bvFTD patients are reported to display an array of inappropriate social behaviors such as loss of empathy,152 reduced tact, and failure to acknowledge the presence of others.153 Furthermore, ToM deficits on formal measures are strongly correlated with carer ratings of behavioral disturbance in bvFTD,32 suggesting that performance on laboratory measures accurately captures functionally relevant impairments in this syndrome. We note further parallels between ToM impairments and carer reports of behavioral dysfunction in SD. Although typically viewed as a language disorder, patients with SD can present with a similar profile of social behavioral disturbances to bvFTD,154 although to a milder degree. Such disturbances include reduced empathy145,155 and increased egocentric behavior manifesting in a failure to consider other’s viewpoints.156 In line with their ToM impairments, reductions in empathy are also reported by carers across the primary progressive aphasias, namely LPA and PNFA.147 Difficulties adopting or considering the mental state of others in daily life in neurodegenerative disorders result in profound carer stress and negatively impact the patient–carer relationship,157 underscoring the importance of studying real-world manifestations of ToM in these patients.
Emerging evidence also suggests an association between ToM deficits and real-world social functioning in PD, ALS, and HD. In PD, carer-reported reductions in empathy occur in parallel with impairments on ToM tasks.127 Furthermore, ToM impairments in PD are associated with disturbances in social behaviour127 and increased apathy.126 This relationship between ToM and apathy has also been uncovered in ALS,101,102 which may reflect reduced motivation for social engagement in the face of difficulties adopting other’s perspectives. The directionality of this relationship, however, is yet to be explored. In addition, carer reports reveal altered empathy and impaired social functioning in a subset of ALS patients,98,158 although whether this subset overlaps with those demonstrating ToM deficits remains to be determined. While the direct link between ToM and social functioning has not been explored in HD, alterations in social behavior such as empathy are characteristic of the disease,29 suggesting that impairments on ToM measures in HD extend to real-world dysfunction. By contrast, carers of PSP and CBS report intact empathy in these patients,145 and no study, to our knowledge, has explored empathic behavior in DLB or VD. Further examination of the potential relationship between ToM impairment and social functioning across each condition is critical to establish the ecological validity of formal ToM measures.
ToM and moral reasoning
Alterations in moral behavior, or transgressions from societal values about how its members should behave, have been documented in neurodegenerative disorders. Specifically, patients with bvFTD and PD have been shown to make abnormal responses to moral dilemmas, which involve a choice between two morally conflicting actions159,160 (but see Ref. 161). Such difficult moral decisions are proposed to require ToM, as the thoughts and feelings of other people in the scenario are often considered when choosing how to respond.7 The overlap between ToM and moral reasoning is supported at the neural level, with mPFC, ACC/fIN, and PCC implicated across both processes.162–164 Altered moral reasoning in bvFTD correlates with impaired performance on cognitive, but not affective, ToM tasks,165 and morality and ToM can also be simultaneously affected in these patients.166 No such relationship between ToM and moral reasoning, however, is apparent in PD.160,161 Furthermore, despite impaired ToM performance in AD, moral reasoning appears intact in this syndrome.159 As such, the evidence to date suggests that ToM dysfunction may contribute to altered moral behavior in some neurodegenerative disorders (ie, bvFTD), potentially attributable to atrophy in regions such as the mPFC; however, the extent to which this relationship is present across other syndromes requires further exploration.
ToM and criminality
Taken to the extreme, impairments in moral reasoning can translate into real-world transgressions, such as criminal behavior. Crimes such as theft, violence, hypersexuality, traffic violations, and even homicide have been reported in neurodegeneration.167 In fact, for some individuals, law-breaking represents the first-noticed symptom of the disease. Estimates of criminality range from 37% to 54% in bvFTD and 4.5% to 12% in AD, as well as 20%–40% in HD.167,168 Despite commonalities in the neural substrates of these processes (eg, mPFC, PCC, TPJ),169 no study, to our knowledge, has explored the relationship between ToM dysfunction and criminal behavior in neurodegeneration. The nature of crimes committed in each syndrome, however, suggests potential syndrome-specific associations between ToM and criminality. In bvFTD, for example, crimes are often of an antisocial disinhibited nature, such as harassing strangers, sexual advances, theft from other’s purses, and urinating in public.167 Similarly, in HD, antisocial behaviors of violence and reckless driving represent the most frequently committed crimes.170 The socially disruptive nature of criminal behavior in bvFTD and HD may reflect an inability to consider or comprehend how behavior affects others, mediated, in part, by impaired ToM. In AD, however, criminal behavior is less likely to be of a violent or aggressive nature (eg, traffic violations, theft) and potentially attributable to general cognitive dysfunction, such as forgetting to pay for items, or failure to notice a stop sign, rather than a genuine ToM impairment. Given the significant burden criminality places on patients, families, care facilities, and society more broadly,167 improved understanding of how ToM contributes to this behavior across specific syndromes is urgently required.
ToM and gullibility
Patients with neurodegenerative disorders are particularly vulnerable to financial exploitation and abuse, often falling victim to scams or being easily misled.171,172 Avoiding this kind of deception requires complex social processes,173 including the advanced ToM function of correctly interpreting the speaker’s intention as insincere.174 The intersecting network of brain regions implicated across ToM, lie comprehension,175 and exploitation risk,176 namely mPFC, TPJ, ATL, and cerebellum, suggests a common neural substrate mediating ToM and the avoidance of deception. bvFTD patients are widely known to fall victim to scams, make irrational financial decisions, and engage in excessive gambling. Their concomitant difficulty in interpreting the sincerity of speech, related to their broader ToM deficits,27 combined with an inability to incorporate social contextual information to guide economic decisions,177 may explain the high levels of gullibility displayed by bvFTD patients in daily life.178,179 Misinterpretation of insincere speech is also found in AD180–183 (but see Ref. 27), along with an increasing tendency to believe things that are untrue.179 Financial exploitation in AD, however, likely reflects episodic memory dysfunction and overall cognitive decline.179 Collectively, these findings suggest that ToM underlies gullibility in some neurodegenerative syndromes, although further research directly testing this hypothesis is required.
Concluding remarks: beyond the clinic
Translating converging findings of ToM disruption from the cognitive neuroscience literature into socially relevant behaviors such as empathy, morality, criminality, and gullibility is essential to understand the real-world implications of these impairments and their underlying neurocognitive mechanisms to inform targeted interventions. In turn, this enhanced understanding will better equip carers, clinicians, and the legal system to cope with aberrant socioemotional behavior, particularly when viewed in light of underlying neuropathology. Moreover, the study of neurodegenerative syndromes in which ToM difficulties do not manifest in disruptive behaviors may offer compelling insights regarding potential protective factors for behavioral change. These findings have important implications for legal responsibility in neurodegeneration. In summary, our review highlights how extensive neural network degeneration can impair understanding of the effect of one’s behaviors on other people, as well as detection of other’s intentions, raising important questions regarding culpability for criminal activity and accountability for financial loss in neurodegenerative disorders. Moving forward, increased awareness and further rigorous investigation of these issues are required to ensure appropriate ethical treatment, care, and quality of life for individuals living with neurodegenerative disorders and their families.
Acknowledgments
This work was supported in part by an Australian Research Council (ARC) Centre of Excellence in Cognition and its Disorders (CE110001021). CS-B is supported by the National Health and Medical Research Council (NHMRC) Postgraduate Scholarship (APP1132764). SR is supported by the Faculty of Science PhD Research Scholarship from the University of Sydney. MI is supported by an ARC Future Fellowship (FT160100096) and an ARC Discovery Project (DP180101584). The authors would like to thank Ms Heidi Cartwright for assistance with preparation of Figures 2 and 3.
Disclosure
The authors report no conflicts of interest in this work.
References
Premack D, Woodruff G. Does the chimpanzee have a theory of mind? Behav Brain Sci. 1978;1(04):515–526. | ||
Baron-Cohen S. Mindblindness: An Essay On Autism and Theory of Mind. Cambridge: MIT Press; 1995. | ||
Decety J, Jackson PL. The functional architecture of human empathy. Behav Cogn Neurosci Rev. 2004;3(2):71–100. | ||
Happé F, Cook JL, Bird G. The Structure of Social Cognition: In(ter)dependence of Sociocognitive Processes. Annu Rev Psychol. 2017;68(1):243–267. | ||
Leslie AM, Thaiss L. Domain specificity in conceptual development: neuropsychological evidence from autism. Cognition. 1992;43(3):225–251. | ||
Dvash J, Shamay-Tsoory SG. Theory of mind and empathy as multidimensional constructs: neurological foundations. Top Lang Disord. 2014;34(4):282–295. | ||
Shany-Ur T, Rankin KP. Personality and social cognition in neurodegenerative disease. Curr Opin Neurol. 2011;24(6):550–555. | ||
Shamay-Tsoory SG, Harari H, Aharon-Peretz J, Levkovitz Y. The role of the orbitofrontal cortex in affective theory of mind deficits in criminal offenders with psychopathic tendencies. Cortex. 2010;46(5):668–677. | ||
Poletti M, Enrici I, Adenzato M. Cognitive and affective theory of mind in neurodegenerative diseases: neuropsychological, neuroanatomical and neurochemical levels. Neurosci Biobehav Rev. 2012;36(9):2147–2164. | ||
Dodich A, Cerami C, Crespi C, et al. Differential impairment of cognitive and affective mentalizing abilities in neurodegenerative dementias: evidence from behavioral variant of frontotemporal dementia, Alzheimer’s disease, and mild cognitive impairment. J Alzheimers Dis. 2016;50(4):1011–1022. | ||
Mcdonald S, Flanagan S, Rollins J, Kinch J. TASIT: a new clinical tool for assessing social perception after traumatic brain injury. J Head Trauma Rehabil. 2003;18(3):219–238. | ||
Burke T, Elamin M, Bede P, et al. Discordant performance on the “Reading the Mind in the Eyes” Test, based on disease onset in amyotrophic lateral sclerosis. Amyotroph Lateral Scler Frontotemporal Degener. 2016;17(7–8):467–472. | ||
Carluer L, Mondou A, Buhour MS, et al. Neural substrate of cognitive theory of mind impairment in amyotrophic lateral sclerosis. Cortex. 2015;65:19–30. | ||
Ramanan S, de Souza LC, Moreau N, et al. Determinants of theory of mind performance in Alzheimer’s disease: a data-mining study. Cortex. 2017;88:8–18. | ||
Friedman O, Leslie AM. Processing demands in belief-desire reasoning: inhibition or general difficulty? Dev Sci. 2005;8(3):218–225. | ||
Synn A, Mothakunnel A, Kumfor F, et al. Mental states in moving shapes: distinct cortical and subcortical contributions to theory of mind impairments in dementia. J Alzheimers Dis. 2018;61(2):521–535. | ||
Bora E, Walterfang M, Velakoulis D. Theory of mind in behavioural-variant frontotemporal dementia and Alzheimer’s disease: a meta-analysis. J Neurol Neurosurg Psychiatry. 2015;86(7):714–719. | ||
Péron J, Vicente S, Leray E, et al. Are dopaminergic pathways involved in theory of mind? A study in Parkinson’s disease. Neuropsychologia. 2009;47(2):406–414. | ||
Seeley WW, Crawford RK, Zhou J, Miller BL, Greicius MD. Neurodegenerative diseases target large-scale human brain networks. Neuron. 2009;62(1):42–52. | ||
Piguet O, Hornberger M, Mioshi E, Hodges JR. Behavioural-variant frontotemporal dementia: diagnosis, clinical staging, and management. Lancet Neurol. 2011;10(2):162–172. | ||
Seeley WW, Zhou J, Kim EJ. Frontotemporal dementia: what can the behavioral variant teach us about human brain organization? Neuroscientist. 2012;18(4):373–385. | ||
Torralva T, Gleichgerrcht E, Torres Ardila MJ, Roca M, Manes FF. Differential cognitive and affective theory of mind abilities at mild and moderate stages of behavioral variant frontotemporal dementia. Cogn Behav Neurol. 2015;28(2):63–70. | ||
Adenzato M, Cavallo M, Enrici I. Theory of mind ability in the behavioural variant of frontotemporal dementia: an analysis of the neural, cognitive, and social levels. Neuropsychologia. 2010;48(1):2–12. | ||
Bora E, Walterfang M, Velakoulis D. Theory of mind in behavioural-variant frontotemporal dementia and Alzheimer’s disease: a meta-analysis. J Neurol Neurosurg Psychiatry. 2015;86(7):714–719. | ||
Henry JD, Phillips LH, von Hippel C. A meta-analytic review of theory of mind difficulties in behavioural-variant frontotemporal dementia. Neuropsychologia. 2014;56:53–62. | ||
Kipps CM, Hodges JR. Theory of mind in frontotemporal dementia. Soc Neurosci. 2006;1(3–4):235–244. | ||
Shany-Ur T, Poorzand P, Grossman SN, et al. Comprehension of insincere communication in neurodegenerative disease: lies, sarcasm, and theory of mind. Cortex. 2012;48(10):1329–1341. | ||
Lough S, Hodges JR. Measuring and modifying abnormal social cognition in frontal variant frontotemporal dementia. J Psychosom Res. 2002;53(2):639–646. | ||
Snowden JS, Gibbons ZC, Blackshaw A, et al. Social cognition in frontotemporal dementia and Huntington’s disease. Neuropsychologia. 2003;41(6):688–701. | ||
Funkiewiez A, Bertoux M, de Souza LC, Lévy R, Dubois B. The sea (social cognition and emotional assessment): a clinical neuropsychological tool for early diagnosis of frontal variant of frontotemporal lobar degeneration. Neuropsychology. 2012;26(1):81–90. | ||
Brioschi Guevara A, Knutson KM, Wassermann EM, Pulaski S, Grafman J, Krueger F. Theory of mind impairment in patients with behavioural variant fronto-temporal dementia (bv-FTD) increases caregiver burden. Age Ageing. 2015;44(5):891–895. | ||
Gregory C, Lough S, Stone V, et al. Theory of mind in patients with frontal variant frontotemporal dementia and Alzheimer’s disease: theoretical and practical implications. Brain. 2002;125(4):752–764. | ||
Freedman M, Binns MA, Se B, Murphy C, Dt S, Black SE, Stuss DT. Theory of mind and recognition of facial emotion in dementia: challenge to current concepts. Alzheimer Dis Assoc Disord. 2013;27(1):56–61. | ||
Fernandez-Duque D, Baird JA, Black SE. False-belief understanding in frontotemporal dementia and Alzheimer’s disease. J Clin Exp Neuropsychol. 2009;31(4):489–497. | ||
Le Bouc R, Lenfant P, Delbeuck X, et al. My belief or yours? differential theory of mind deficits in frontotemporal dementia and Alzheimer’s disease. Brain. 2012;135(Pt 10):3026–3038. | ||
Cotelli M, Adenzato M, Cantoni V, et al. Enhancing theory of mind in behavioural variant frontotemporal dementia with transcranial direct current stimulation. Cogn Affect Behav Neurosci. 2018;18(6):1065–1075. | ||
Bertoux M, O’Callaghan C, Dubois B, Hornberger M. In two minds: Executive functioning versus theory of mind in behavioural variant frontotemporal dementia. J Neurol Neurosurg Psychiatry. 2016;87(3):231–234. | ||
Lough S, Gregory C, Hodges JR. Dissociation of social cognition and executive function in frontal variant frontotemporal dementia. Neurocase. 2001;7(2):123–130. | ||
Kipps CM, Nestor PJ, Acosta-Cabronero J, Arnold R, Hodges JR. Understanding social dysfunction in the behavioural variant of frontotemporal dementia: the role of emotion and sarcasm processing. Brain. 2009;132(Pt 3):592–603. | ||
Ibañez A, Manes F. Contextual social cognition and the behavioral variant of frontotemporal dementia. Neurology. 2012;78(17):1354–1362. | ||
Irish M, Piguet O, Hodges JR. Self-projection and the default network in frontotemporal dementia. Nat Rev Neurol. 2012;8(3):152–161. | ||
Irish M, Hodges JR, Piguet O. Episodic future thinking is impaired in the behavioural variant of frontotemporal dementia. Cortex. 2013;49(9):2377–2388. | ||
O’Callaghan C, Shine JM, Andrews-Hanna J, Irish M, Hodges JR. Hippocampal atrophy and intrinsic brain network alterations relate to impaired capacity for mind wandering in neurodegeneration. BioRxiv. 2018. | ||
Strikwerda-Brown C, Grilli MD, Andrews-Hanna J, Irish M. “All is not lost”: episodic and semantic contributions to continuity of self in dementia. 2019; [preprint]: 1–33. | ||
Amodio DM, Frith CD. Meeting of minds: The medial frontal cortex and social cognition. Nat Rev Neurosci. 2006;7(4):268–277. | ||
Caminiti SP, Canessa N, Cerami C, et al. Affective mentalizing and brain activity at rest in the behavioral variant of frontotemporal dementia. Neuroimage Clin. 2015;9:484–497. | ||
Ahmed RM, Devenney EM, Irish M, et al. Neuronal network disintegration: common pathways linking neurodegenerative diseases. J Neurol Neurosurg Psychiatry. 2016;87(11):1234–1241. | ||
Seeley WW, Crawford R, Rascovsky K, et al. Frontal paralimbic network atrophy in very mild behavioral variant frontotemporal dementia. Arch Neurol. 2008;65(2):249–255. | ||
Allman JM, Watson KK, Tetreault NA, Hakeem AY. Intuition and autism: a possible role for von Economo neurons. Trends Cogn Sci. 2005;9(8):367–373. | ||
Etkin A, Egner T, Kalisch R. Emotional processing in anterior cingulate and medial prefrontal cortex. Trends Cogn Sci. 2011;15(2):85–93. | ||
Menon V, Uddin LQ, Saliency ULQ. Saliency, switching, attention and control: a network model of insula function. Brain Struct Funct. 2010;214(5–6):655–667. | ||
Kim E-J, Sidhu M, Gaus SE, et al. Selective frontoinsular von Economo neuron and fork cell loss in early behavioral variant frontotemporal dementia. Cereb Cortex. 2012;22(2):251–259. | ||
Zhou J, Greicius MD, Gennatas ED, et al. Divergent network connectivity changes in behavioural variant frontotemporal dementia and Alzheimer’s disease. Brain. 2010;133(5):1352–1367. | ||
Cerami C, Dodich A, Canessa N, et al. Neural correlates of empathic impairment in the behavioral variant of frontotemporal dementia. Alzheimers Dement. 2014;10(6):827–834. | ||
Möller C, Hafkemeijer A, Pijnenburg YAL, et al. Different patterns of cortical gray matter loss over time in behavioral variant frontotemporal dementia and Alzheimer’s disease. Neurobiol Aging. 2016;38:21–31. | ||
Frings L, Yew B, Flanagan E, et al. Longitudinal grey and white matter changes in frontotemporal dementia and Alzheimer’s disease. PLoS One. 2014;9(3):e90814. | ||
Whitwell JL, Boeve BF, Weigand SD, et al. Brain atrophy over time in genetic and sporadic frontotemporal dementia: a study of 198 serial magnetic resonance images. Eur J Neurol. 2015;22(5):745–752. | ||
Landin-Romero R, Kumfor F, Leyton CE, Irish M, Hodges JR, Piguet O. Disease-specific patterns of cortical and subcortical degeneration in a longitudinal study of Alzheimer’s disease and behavioural-variant frontotemporal dementia. Neuroimage. 2017;151:72–80. | ||
Zahn R, Moll J, Iyengar V, et al. Social conceptual impairments in frontotemporal lobar degeneration with right anterior temporal hypometabolism. Brain. 2009;132(3):604–616. | ||
Irish M, Hodges JR, Piguet O. Right anterior temporal lobe dysfunction underlies theory of mind impairments in semantic dementia. Brain. 2014;137(4):1241–1253. | ||
Eslinger PJ, Moore P, Troiani V, et al. Oops! resolving social dilemmas in frontotemporal dementia. J Neurol Neurosurg Psychiatry. 2007;78(5):457–460. | ||
Chen Y, Kumfor F, Landin-Romero R, Irish M, Hodges JR, Piguet O. Cerebellar atrophy and its contribution to cognition in frontotemporal dementias. Ann Neurol. 2018;84(1):98–109. | ||
Rankin KP, Kramer JH, Mychack P, Miller BL. Double dissociation of social functioning in frontotemporal dementia. Neurology. 2003;60(2):266–271. | ||
Sabat SR, Gladstone CM. What intact social cognition and social behavior reveal about cognition in the moderate stage of Alzheimer’s disease. Dementia. 2010;9(1):61–78. | ||
Sabat SR, Lee JM. Relatedness among people diagnosed with dementia: social cognition and the possibility of friendship. Dementia. 2012;11(3):315–327. | ||
Kumfor F, Irish M, Leyton C, et al. Tracking the progression of social cognition in neurodegenerative disorders. J Neurol Neurosurg Psychiatry. 2014;85(10):1076–1083. | ||
Ramanan S, Bertoux M, Flanagan E, et al. Longitudinal executive function and episodic memory profiles in behavioral-variant frontotemporal dementia and Alzheimer’s disease. J Int Neuropsychol Soc. 2017;23(1):34–43. | ||
Irish M, Hornberger M, El Wahsh S, et al. Grey and white matter correlates of recent and remote autobiographical memory retrieval: insights from the dementias. PLoS One. 2014;9(11):e113081. | ||
Bertoux M, de Souza LC, O’Callaghan C, et al. Social cognition deficits: the key to discriminate behavioral variant frontotemporal dementia from Alzheimer’s disease regardless of amnesia? JAD. 2015;49(4):1065–1074. | ||
Laisney M, Bon L, Guiziou C, Daluzeau N, Eustache F, Desgranges B. Cognitive and affective theory of mind in mild to moderate Alzheimer’s disease. J Neuropsychol. 2013;7(1):107–120. | ||
Verdon CM, Fossati P, Verny M, Dieudonné B, Teillet L, Nadel J. Social cognition: an early impairment in dementia of the Alzheimer type. Alzheimer Dis Assoc Disord. 2007;21(1):25–30. | ||
Cuerva AG, Sabe L, Kuzis G, Tiberti C, Dorrego F, Starkstein SE. Theory of mind and pragmatic abilities in dementia. Neuropsychiatry Neuropsychol Behav Neurol. 2001;14(3):153–158. | ||
Moreau N, Rauzy S, Viallet F, Champagne-Lavau M. Theory of mind in Alzheimer disease: evidence of authentic impairment during social interaction. Neuropsychology. 2016;30(3):312–321. | ||
Fliss R, Le Gall D, Etcharry-Bouyx F, Chauviré V, Desgranges B, Allain P. Theory of Mind and social reserve: alternative hypothesis of progressive theory of mind decay during different stages of Alzheimer’s disease. Soc Neurosci. 2016;11(4):409–423. | ||
Zaitchik D, Koff E, Brownell H, Winner E, Albert M. Inference of beliefs and emotions in patients with Alzheimer’s disease. Neuropsychology. 2006;20(1):11–20. | ||
Zaitchik D, Koff E, Brownell H, Winner E, Albert M. Inference of mental states in patients with Alzheimer’s disease. Cogn Neuropsychiatry. 2004;9(4):301–313. | ||
Aboulafia-Brakha T, Christe B, Martory MD, Annoni JM. Theory of mind tasks and executive functions: a systematic review of group studies in neurology. J Neuropsychol. 2011;5(1):39–55. | ||
Choong CS, Doody GA. Can theory of mind deficits be measured reliably in people with mild and moderate Alzheimer’s dementia? BMC Psychol. 2013;1(1):28. | ||
Duclos H, Bejanin A, Eustache F, Desgranges B, Laisney M. Role of context in affective theory of mind in Alzheimer’s disease. Neuropsychologia. 2018;119:363–372. | ||
Saxe R, Kanwisher N. People thinking about thinking people. The role of the temporo-parietal junction in “theory of mind”. NeuroImage. 2003;19(4):1835–1842. | ||
Binder JR, Desai RH, Graves WW, Conant LL. Where is the semantic system? A critical review and meta-analysis of 120 functional neuroimaging studies. Cereb Cortex. 2009;19(12):2767–2796. | ||
Ramanan S, Piguet O, Irish M. Rethinking the role of the angular gyrus in remembering the past and imagining the future: the contextual integration model. Neuroscientist. 2018;24(4):342–352. | ||
Moreau N, Viallet F, Champagne-Lavau M. Using memories to understand others: the role of episodic memory in theory of mind impairment in Alzheimer disease. Ageing Res Rev. 2013;12(4):833–839. | ||
Montagrin A, Saiote C, Schiller D. The social hippocampus. Hippocampus. 2018;28(9):672–679. | ||
van Overwalle F, Baetens K, Mariën P, Vandekerckhove M. Social cognition and the cerebellum: a meta-analysis of over 350 fMRI studies. Neuroimage. 2014;86:554–572. | ||
Bellebaum C, Daum I. Cerebellar involvement in executive control. Cerebellum. 2007;6(3):184–192. | ||
Ringholz GM, Appel SH, Bradshaw M, Cooke NA, Mosnik DM, Schulz PE. Prevalence and patterns of cognitive impairment in sporadic ALS. Neurology. 2005;65(4):586–590. | ||
Neary D, Snowden JS, Mann DM, Northen B, Goulding PJ, Macdermott N. Frontal lobe dementia and motor neuron disease. J Neurol Neurosurg Psychiatry. 1990;53(1):23–32. | ||
Strong MJ, Abrahams S, Goldstein LH, et al. Amyotrophic lateral sclerosis – frontotemporal spectrum disorder (ALS-FTSD): revised diagnostic criteria. Amyotroph Lateral Scler Frontotemporal Degener. 2017;18(3–4):153–174. | ||
Shen D, Cui L, Fang J, Cui B, Li D, Tai H. Voxel-wise meta-analysis of gray matter changes in amyotrophic lateral sclerosis. Front Aging Neurosci. 2016;8(13):64. | ||
Trojsi F, Siciliano M, Russo A, et al. Theory of mind and its neuropsychological and quality of life correlates in the early stages of amyotrophic lateral sclerosis. Front Psychol. 1934;2016:7. | ||
Consonni M, Catricalà E, Dalla Bella E, Gessa VC, Lauria G, Cappa SF. Beyond the consensus criteria: multiple cognitive profiles in amyotrophic lateral sclerosis? Cortex. 2016;81:162–167. | ||
Buhour M-S, Doidy F, Mondou A, et al. Voxel-based mapping of grey matter volume and glucose metabolism profiles in amyotrophic lateral sclerosis. EJNMMI Res. 2017;7(1):21. | ||
Watermeyer TJ, Brown RG, Sidle KCL, et al. Executive dysfunction predicts social cognition impairment in amyotrophic lateral sclerosis. J Neurol. 2015;262(7):1681–1690. | ||
Gibbons ZC, Snowden JS, Thompson JC, Happé F, Richardson A, Neary D. Inferring thought and action in motor neurone disease. Neuropsychologia. 2007;45(6):1196–1207. | ||
Meier SL, Charleston AJ, Tippett LJ. Cognitive and behavioural deficits associated with the orbitomedial prefrontal cortex in amyotrophic lateral sclerosis. Brain. 2010;133(11):3444–3457. | ||
Cavallo M, Adenzato M, Macpherson SE, Karwig G, Enrici I, Abrahams S. Evidence of social understanding impairment in patients with amyotrophic lateral sclerosis. PLoS One. 2011;6(10):e25948. | ||
Cerami C, Dodich A, Canessa N, et al. Emotional empathy in amyotrophic lateral sclerosis: a behavioural and voxel-based morphometry study. Amyotroph Lateral Scler Frontotemporal Degener. 2014;15(1–2):21–29. | ||
Staios M, Fisher F, Lindell AK, Ong B, Howe J, Reardon K. Exploring sarcasm detection in amyotrophic lateral sclerosis using ecologically valid measures. Front Hum Neurosci. 2013;7:178. | ||
Savage SA, Lillo P, Kumfor F, Kiernan MC, Piguet O, Hodges JR. Emotion processing deficits distinguish pure amyotrophic lateral sclerosis from frontotemporal dementia. Amyotrop Lateral Scler Frontotemporal Degener. 2014;15(1–2):39–46. | ||
Girardi A, Macpherson SE, Abrahams S. Deficits in emotional and social cognition in amyotrophic lateral sclerosis. Neuropsychology. 2011;25(1):53–65. | ||
van der Hulst EJ, Bak TH, Abrahams S. Impaired affective and cognitive theory of mind and behavioural change in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry. 2015;86(11):1208–1215. | ||
Keller J, Böhm S, Aho-Özhan HEA, et al. Functional reorganization during cognitive function tasks in patients with amyotrophic lateral sclerosis. Brain Imaging Behav. 2018;12(3):771–784. | ||
Trojsi F, di Nardo F, Santangelo G, et al. Resting state fMRI correlates of theory of mind impairment in amyotrophic lateral sclerosis. Cortex. 2017;97:1–16. | ||
Vinther-Jensen T, Larsen IU, Hjermind LE, et al. A clinical classification acknowledging neuropsychiatric and cognitive impairment in Huntington’s disease. Orphanet J Rare Dis. 2014;9(1):114. | ||
Dogan I, Eickhoff SB, Schulz JB, et al. Consistent neurodegeneration and its association with clinical progression in Huntington’s disease: a coordinate-based meta-analysis. Neurodegener Dis. 2013;12(1):23–35. | ||
Bora E, Velakoulis D, Walterfang M. Social cognition in Huntington’s disease: a meta-analysis. Behav Brain Res. 2016;297:131–140. | ||
Larsen IU, Vinther-Jensen T, Gade A, Nielsen JE, Vogel A. Do I misconstrue? Sarcasm detection, emotion recognition, and theory of mind in Huntington disease. Neuropsychology. 2016;30(2):181–189. | ||
Lagravinese G, Avanzino L, Raffo de Ferrari A, et al. Theory of mind is impaired in mild to moderate Huntington’s disease independently from global cognitive functioning. Front Psychol. 2017;8(31):80. | ||
Eddy CM, Rickards HE. Interaction without intent: the shape of the social world in Huntington’s disease. Soc Cogn Affect Neurosci. 2015;10(9):1228–1235. | ||
Eddy CM, Sira Mahalingappa S, Rickards HE. Putting things into perspective: the nature and impact of theory of mind impairment in Huntington’s disease. Eur Arch Psychiatry Clin Neurosci. 2014;264(8):697–705. | ||
Eddy CM, Sira Mahalingappa S, Rickards HE. Is Huntington’s disease associated with deficits in theory of mind? Acta Neurol Scand. 2012;126(6):376–383. | ||
Philpott AL, Andrews SC, Staios M, Churchyard A, Fisher F. Emotion evaluation and social inference impairments in Huntington’s disease. JHD. 2016;5(2):175–183. | ||
Allain P, Havet-Thomassin V, Verny C, et al. Evidence for deficits on different components of theory of mind in Huntington’s disease. Neuropsychology. 2011;25(6):741–751. | ||
Brüne M, Blank K, Witthaus H, Saft C. “Theory of mind” is impaired in Huntington’s disease. Mov Disord. 2011;26(4):671–678. | ||
Adjeroud N, Besnard J, Massioui NE, et al. Theory of mind and empathy in preclinical and clinical Huntington’s disease. Soc Cogn Affect Neurosci. 2016;11(1):89–99. | ||
Eddy CM, Rickards HE. Theory of mind can be impaired prior to motor onset in Huntington’s disease. Neuropsychology. 2015;29(5):792–798. | ||
Mason SL, Zhang J, Begeti F, et al. The role of the amygdala during emotional processing in Huntington’s disease: from pre-manifest to late stage disease. Neuropsychologia. 2015;70:80–89. | ||
Saft C, Lissek S, Hoffmann R, et al. Mentalizing in preclinical Huntington’s disease: an fMRI study using Cartoon picture stories. Brain Imaging Behav. 2013;7(2):154–162. | ||
Aarsland D, Beyer MK, Kurz MW. Dementia in Parkinson’s disease. Curr Opin Neurol. 2008;21(6):676–682. | ||
Muslimović D, Post B, Speelman JD, Schmand B. Cognitive profile of patients with newly diagnosed Parkinson disease. Neurology. 2005;65(8):1239–1245. | ||
Bora E, Walterfang M, Velakoulis D. Theory of mind in Parkinson’s disease: a meta-analysis. Behav Brain Res. 2015;292:515–520. | ||
Xi C, Zhu Y, Mu Y, et al. Theory of mind and decision-making processes are impaired in Parkinson’s disease. Behav Brain Res. 2015;279:226–233. | ||
Enrici I, Mitkova A, Castelli L, Lanotte M, Lopiano L, Adenzato M. Deep brain stimulation of the subthalamic nucleus does not negatively affect social cognitive abilities of patients with Parkinson’s disease. Sci Rep. 2017;7(1):9413. | ||
Palermo S, Lopiano L, Zibetti M, Rosato R, Leotta D, Amanzio M. A novel framework for understanding reduced awareness of dyskinesias in Parkinson’s disease. Parkinsonism Relat Disord. 2017;39:58–63. | ||
Santangelo G, Vitale C, Trojano L, et al. Neuropsychological correlates of theory of mind in patients with early Parkinson’s disease. Mov Disord. 2012;27(1):98–105. | ||
Narme P, Mouras H, Roussel M, Duru C, Krystkowiak P, Godefroy O. Emotional and cognitive social processes are impaired in Parkinson’s disease and are related to behavioral disorders. Neuropsychology. 2013;27(2):182–192. | ||
Bodden ME, Mollenhauer B, Trenkwalder C, et al. Affective and cognitive theory of mind in patients with Parkinson’s disease. Parkinsonism Relat Disord. 2010;16(7):466–470. | ||
Bodden ME, Dodel R, Kalbe E. Theory of mind in Parkinson’s disease and related basal ganglia disorders: a systematic review. Mov Disord. 2010;25(1):13–27. | ||
Eddy CM, Beck SR, Mitchell IJ, Praamstra P, Pall HS. Theory of mind deficits in Parkinson’s disease: a product of executive dysfunction? Neuropsychology. 2013;27(1):37–47. | ||
Saltzman J, Strauss E, Hunter M, Archibald S. Theory of mind and executive functions in normal human aging and Parkinson’s disease. J Int Neuropsychol Soc. 2000;6(7):781–788. | ||
Roca M, Torralva T, Gleichgerrcht E, et al. Impairments in social cognition in early medicated and unmedicated Parkinson disease. Cogn Behav Neurol. 2010;23(3):152–158. | ||
Nobis L, Schindlbeck K, Ehlen F, et al. Theory of mind performance in Parkinson’s disease is associated with motor and cognitive functions, but not with symptom lateralization. J Neural Transm. 2017;124(9):1067–1072. | ||
Díez-Cirarda M, Ojeda N, Peña J, et al. Neuroanatomical correlates of theory of mind deficit in parkinson’s disease: a multimodal imaging study. PLoS One. 2015;10(11):e0142234. | ||
Yu RL, Rm W, Chiu MJ, Tai CH, Lin CH, Hua MS. Advanced theory of mind in patients at early stage of Parkinson’s disease. Parkinsonism Relat Disord. 2012;18(1):21–24. | ||
Costa A, Bak T, Caffarra P, et al. The need for harmonisation and innovation of neuropsychological assessment in neurodegenerative dementias in Europe: consensus document of the joint program for neurodegenerative diseases Working Group. Alzheimers Res Ther. 2017;9(1):27. | ||
Monetta L, Grindrod CM, Pell MD. Irony comprehension and theory of mind deficits in patients with Parkinson’s disease. Cortex. 2009;45(8):972–981. | ||
Poletti M, Vergallo A, Ulivi M, Sonnoli A, Bonuccelli U. Affective theory of mind in patients with Parkinson’s disease. Psychiatry Clin Neurosci. 2013;67(4):273–276. | ||
Mckinlay A, Albicini M, Kavanagh P. The effect of cognitive status and visuospatial performance on affective theory of mind in Parkinson’s disease. NDT. 2013;9:1071–1076. | ||
Roca M, Manes F, Chade A, et al. The relationship between executive functions and fluid intelligence in Parkinson’s disease. Psychol Med. 2012;42(11):2445–2452. | ||
Mengelberg A, Siegert RJ. Is theory-of-mind impaired in Parkinson’s disease? Cogn Neuropsychiatry. 2003;8(3):191–209. | ||
Gorno-Tempini ML, Hillis AE, Weintraub S, et al. Classification of primary progressive aphasia and its variants. Neurology. 2011;76(11):1006–1014. | ||
Duval C, Bejanin A, Piolino P, et al. Theory of mind impairments in patients with semantic dementia. Brain. 2012;135(1):228–241. | ||
Couto B, Manes F, Montañés P, et al. Structural neuroimaging of social cognition in progressive non-fluent aphasia and behavioral variant of fronttemporal dementia. Front Hum Neurosci. 2013;7:467. | ||
Rankin KP, Gorno-Tempini ML, Allison SC, et al. Structural anatomy of empathy in neurodegenerative disease. Brain. 2006;129(11):2945–2956. | ||
Irish M, Kumfor F, Hodges JR, Piguet O. A tale of two hemispheres: contrasting socioemotional dysfunction in right- versus left-lateralised semantic dementia. Dement Neuropsychol. 2013;7(1):88–95. | ||
Hazelton JL, Irish M, Hodges JR, Piguet O, Kumfor F. Cognitive and affective empathy disruption in non-fluent primary progressive aphasia syndromes. Brain Impairment. 2017;18(01):117–129. | ||
Modinos G, Obiols JE, Pousa E, Vicens J. Theory of mind in different dementia profiles. J Neuropsychiatry Clin Neurosci. 2009;21(1):100–101. | ||
Heitz C, Noblet V, Phillipps C, et al. Cognitive and affective theory of mind in dementia with Lewy bodies and Alzheimer’s disease. Alzheimers Res Ther. 2016;8(1):10. | ||
Ghosh BCP, Calder AJ, Peers PV, et al. Social cognitive deficits and their neural correlates in progressive supranuclear palsy. Brain. 2012;135(7):2089–2102. | ||
Poletti M, Bonuccelli U. Impairment of affective theory of mind in corticobasal degeneration. J Neuropsychiatry Clin Neurosci. 2012;24(1):E7. | ||
Dermody N, Wong S, Ahmed R, Piguet O, Hodges JR, Irish M. Uncovering the neural bases of cognitive and affective empathy deficits in Alzheimer’s disease and the behavioral-variant of frontotemporal dementia. J Alzheimers Dis. 2016;53(3):801–816. | ||
Mendez MF, Fong SS, Shapira JS, et al. Observation of social behavior in frontotemporal dementia. Am J Alzheimers Dis Other Demen. 2014;29(3):215–221. | ||
Hodges JR, Patterson K. Semantic dementia: a unique clinicopathological syndrome. Lancet Neurol. 2007;6(11):1004–1014. | ||
Rankin KP, Kramer JH, Miller BL. Patterns of cognitive and emotional empathy in frontotemporal lobar degeneration. Cogn Behav Neurol. 2005;18(1):28–36. | ||
Bon L, Belliard S, Eustache F, Desgranges B. Behavioural egocentrism in semantic dementia: effect of deficits in theory of mind or cognitive egocentrism? Rev Neuropsychol. 2009;1(2):133–149. | ||
Hsieh S, Irish M, Daveson N, Hodges JR, Piguet O. When one loses empathy: its effect on carers of patients with dementia. J Geriatr Psychiatry Neurol. 2013;26(3):174–184. | ||
Abrahams S, Newton J, Niven E, Foley J, Bak TH. Screening for cognition and behaviour changes in ALS. Amyotroph Lateral Scler Frontotemporal Degener. 2014;15(1–2):9–14. | ||
Mendez MF, Shapira JS. Altered emotional morality in frontotemporal dementia. Cogn Neuropsychiatry. 2009;14(3):165–179. | ||
Rosen JB, Rott E, Ebersbach G, Kalbe E. Altered moral decision-making in patients with idiopathic Parkinson’s disease. Parkinsonism Relat Disord. 2015;21(10):1191–1199. | ||
Rosen JB, Brand M, Polzer C, Ebersbach G, Kalbe E. Moral decision-making and theory of mind in patients with idiopathic Parkinson’s disease. Neuropsychology. 2013;27(5):562–572. | ||
Chiong W, Wilson SM, D’Esposito M, et al. The salience network causally influences default mode network activity during moral reasoning. Brain. 2013;136(6):1929–1941. | ||
Andrews-Hanna JR, Smallwood J, Spreng RN. The default network and self-generated thought: component processes, dynamic control, and clinical relevance. Ann NY Acad Sci. 2014;1316(1):29–52. | ||
van den Stock J, Stam D, de Winter FL, et al. Moral processing deficit in behavioral variant frontotemporal dementia is associated with facial emotion recognition and brain changes in default mode and salience network areas. Brain Behav. 2017;7(12):e00843. | ||
Gleichgerrcht E, Torralva T, Roca M, Pose M, Manes F. The role of social cognition in moral judgment in frontotemporal dementia. Soc Neurosci. 2011;6(2):113–122. | ||
Lough S, Kipps CM, Treise C, et al. Emotion and empathy in frontotemporal dementia. Neuropsychologia. 2006;44(6):950–958. | ||
Liljegren M, Naasan G, Temlett J, et al. Criminal behavior in frontotemporal dementia and Alzheimer’s disease. JAMA Neurol. 2015;72(3):295–300. | ||
Dewhurst K, Oliver JE, Mcknight AL. Socio-psychiatric consequences of Huntington’s disease. Br J Psychiatry. 1970;116(532):255–258. | ||
Darby RR, Horn A, Cushman F, Fox MD. Lesion network localization of criminal behavior. Proc Natl Acad Sci U S A. 2018;115(3):601–606. | ||
Jensen P, Fenger K, Bolwig TG, Sørensen SA. Crime in Huntington’s disease: a study of registered offences among patients, relatives, and controls. J Neurol Neurosurg Psychiatry. 1998;65(4):467–471. | ||
Lichtenberg PA. Financial exploitation, financial capacity, and Alzheimer’s disease. Am Psychol. 2016;71(4):312–320. | ||
Tronetti P. Evaluating abuse in the patient with dementia. Clin Geriatr Med. 2014;30(4):825–838. | ||
Spreng RN, Karlawish J, Marson DC, Cognitive MDC. Cognitive, social, and neural determinants of diminished decision-making and financial exploitation risk in aging and dementia: a review and new model. J Elder Abuse Negl. 2016;28(4–5):320–344. | ||
Winner E, Brownell H, Happé F, Blum A, Pincus D. Distinguishing lies from jokes: theory of mind deficits and discourse interpretation in right hemisphere brain-damaged patients. Brain Lang. 1998;62(1):89–106. | ||
Harada T, Itakura S, Xu F, et al. Neural correlates of the judgment of lying: a functional magnetic resonance imaging study. Neurosci Res. 2009;63(1):24–34. | ||
Asp E, Manzel K, Koestner B, Cole CA, Denburg NL, Tranel D. A neuropsychological test of belief and doubt: damage to ventromedial prefrontal cortex increases credulity for misleading advertising. Front Neurosci. 2012;6(100):100. | ||
O’Callaghan C, Bertoux M, Irish M, et al. Fair play: social norm compliance failures in behavioural variant frontotemporal dementia. Brain. 2016;139(1):204–216. | ||
Pressman PS, Miller BL. Diagnosis and management of behavioral variant frontotemporal dementia. Biol Psychiatry. 2014;75(7):574–581. | ||
Wong S, Irish M, O’Callaghan C, et al. Should I trust you? learning and memory of social interactions in dementia. Neuropsychologia. 2017;104:157–167. | ||
Ag C, Sabe L, Kuzis G, et al. Theory of mind and pragmatic abilities in dementia. Cogn Behav Neurol. 2001;14(3):153–158. | ||
Maki Y, Yamaguchi T, Koeda T, Yamaguchi H. Communicative competence in Alzheimer’s disease: metaphor and sarcasm comprehension. Am J Alzheimers Dis Other Demen. 2013;28(1):69–74. | ||
Yamaguchi T, Maki Y, Takatama M, Yamaguchi H. Gullibility may be a warning sign of Alzheimer’s disease dementia. Int Psychogeriatr. 2018;1:1–8. | ||
Yamaguchi T, Maki Y, Yamaguchi H. Pitfall intention explanation task with clue questions (pitfall task): assessment of comprehending other people’s behavioral intentions in Alzheimer’s disease. Int Psychogeriatr. 2012;24(12):1919–1926. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.