Back to Journals » Neuropsychiatric Disease and Treatment » Volume 11
Neurobehavioral burden of multiple sclerosis with nanotheranostics
Authors Sriramoju B, Kanwar R, Kanwar J
Received 12 February 2015
Accepted for publication 9 March 2015
Published 15 October 2015 Volume 2015:11 Pages 2675—2689
DOI https://doi.org/10.2147/NDT.S82768
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Roger Pinder
Bhasker Sriramoju, Rupinder K Kanwar, Jagat R Kanwar
Nanomedicine-Laboratory of Immunology and Molecular Biomedical Research (NLIMBR), School of Medicine, Molecular and Medical Research, Faculty of Health, Deakin University, VIC, Australia
Abstract: Multiple sclerosis (MS) is a chronic demyelinating neurological disorder affecting people worldwide; women are affected more than men. MS results in serious neurological deficits along with behavioral compromise, the mechanisms of which still remain unclear. Behavioral disturbances such as depression, anxiety, cognitive impairment, psychosis, euphoria, sleep disturbances, and fatigue affect the quality of life in MS patients. Among these, depression and psychosis are more common than any other neurological disorders. In addition, depression is associated with other comorbidities. Although anxiety is often misdiagnosed in MS patients, it can induce suicidal ideation if it coexists with depression. An interrelation between sleep abnormalities and fatigue is also reported among MS patients. In addition, therapeutics for MS is always a challenge because of the presence of the blood–brain barrier, adding to the lack of detailed understanding of the disease pathology. In this review, we tried to summarize various behavioral pathologies and their association with MS, followed by its conventional treatment and nanotheranostics.
Keywords: demyelination, behavioral disorders, behavioral tests
Introduction
Multiple sclerosis (MS) is a chronic neurological autoimmune disease characterized by erosions of myelin, the protective nerve sheath, with a partial preservation or complete loss of axonal activity and nerve transmission.1 However, remyelination is often possible once inflammation subsides and is mediated by oligodendrocytes, which secrete myelin. Remyelination process can be explained as the phenomenon of newer myelin sheath formation around the damaged axons. It is better explained as more the number of oligodendrocytes around the affected neurons, the faster the process of remyeliantion. Inadequate number of oligodendrocytes may lead to improper or no myelin formation, resulting in abnormal neuronal functioning.2 MS has affected more than 1 million people worldwide. The symptoms of MS are weakness, loss of senses, shuffling gait, loss of vision, and cognition.3,4 The main driving force involved in the pathology of MS is inflammation, which enhances autoreactivity, followed by demyelination and neuronal damage.5 Histopathological studies have also confirmed multicentered inflammatory lesions, which spread throughout the brain and spinal cord.6 Autoreactive T-cells are the key players involved in disease generation, breaking down the margin of autoreactivity and self-tolerance, and multiple etiological factors are responsible for this activation. The existence of autoreactive T-cells is evident in both normal and MS patients, but they turn active and seem to be devastating only in MS.7 Among all the T-cell clones, CD8 subtype is found to be more associated with the disease, both in number and infiltration when assessed in the brain and spinal cord. CD8 T-cells were also found to be persistent in blood and cerebrospinal fluid (CSF), implying the fact that they were activated consistently by an antigen driving the long-lasting autoimmune reactivity.8
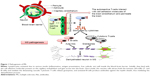
In brief, a group of activated T-cell subclones specific to the myelin protein will penetrate the blood–brain barrier (BBB), being potentiated by the existing inflammatory cytokines, while the resting T-cells have a restricted access to the BBB.9 The coupling of T-cell surface molecules – integrins, selectins, and cadherins – with the corresponding adhesion molecules present in brain capillary endothelial cells facilitates the entry of T-cells. Once the T-cells gain entry, they unleash the autoimmune reactions targeting the myelin antigen. The activation of antibody-producing B-cells further enhances this autoreactivity, thus driving the degeneration of neurons.10 Figure 1 shows MS pathogenesis. Symptom severity, disability levels, and rate of disease progression vary with each individual and even differs in the same patient with time.11 Significant sex differences were identified among MS patients, with a majority of females displaying the symptoms compared to males.
Also, MS was found to affect women at an early age (18–30 years), while it affected men at a later stage of life (30–40 years). These dissimilarities were possibly due to the protective effects of testosterone in men. Another interesting aspect observed in MS was the cessation of disease symptoms in pregnant women who were in the third trimester. The elevated levels of estriol were anticipated to be responsible for the protective effect, and the same was observed in mice models as well.12 The introduction of biomarkers has revolutionized the understanding of the disease pathology and its diagnostics. Some of the recent advancements are the discovery of elevated levels of astrocyte and axonal cytoskeletal proteins, namely, glial fibrillary acidic protein and neurofilament light protein in the CSF corresponding to MS progression.13
The elevated levels of the 14-3-3 protein in the CSF corresponded to the disease progression and disability, while a close correlation was established with the downregulation of cystatin-C, a protease inhibitor that neutralizes the actions of lysosomal cathepsins modulating lymphocytic activation.14 Some other recently found potential biomarkers associated with the disease are osteopontin and pentosidine.15,16 Based on the symptoms, MS is categorized into four subtypes. Patients with relapsing remitting form recover with no symptoms following disease onset. Patients with persistent disability symptoms between the cycles of relapses and recoveries are termed as “secondary-progressive” patients. “Primary-progressive” MS is characterized by symptoms of only progression, but no signs of remission. In “progressive-relapsing” MS, patients display severe symptoms and attacks during the period of remission.17
MS is not only associated with neurological symptoms but also with a variety of behavioral disturbances such as depression, anxiety, cognitive loss, psychosis, euphoria, sleep disturbances, and fatigue. The impact of these psychological disturbances is such that MS at times is misdiagnosed, supposing it to be a behavioral disorder. In patients with MS, depression is the most common behavioral disorder followed by psychosis.18,19 The disease profession is highly variable with a majority of the neurological compromise following the relapses. In particular, RRMS is a critical subtype that always follows a prolonged deterioration and poses a serious threat of progressive disability. The symptoms of depression and anxiety are very common during the early course of the disease, though the correlation is poor toward the lesional load.
Cognitive impairment is often seen as a forerunner of MS, presenting the symptoms of hampered levels of memory, attention, and execution. According to magnetic resonance imaging (MRI) studies, the severity of cognition loss is relative to the cerebral white matter lesions associated with MS progression. Correlations also exist between the white matter lesions and the onset of psychosis, which is seldom seen in MS.20 Hence, early identification of these behavioral abnormalities would certainly help in diagnosing the onset of MS and can be invaluable for the disease management. Behavioral disorders and their association with MS are described as follows:
Depression and MS
Depression is the most common behavioral disorder prevailing among MS patients. The frequency of occurrence and tendency to develop depressive symptoms were higher in patients with MS compared to patients with other chronic neurological disorders. However, depression often remains undiagnosed and untreated in MS patients. Several factors related to genetics, psychology, immunology, and inflammation have a role in mediating the depressive pathology in MS. The higher prevalence of depression in MS could possibly be explained as: both depression and MS are central nervous system disorders and share multiple etiological factors in their pathophysiology. Also, elevated levels of proinflammatory cytokines and imbalances in monoaminergic neurotransmitter metabolism observed in MS were also reflected in depression.21 Likewise, a study reported genetic influence,22 while a brain MRI study showed the evidence of inflammatory markers in the lesions and pleocytosis in the CSF.
The symptoms of depression were found to be associated with the lesions seen in the arcuate fasciculus23 and medial orbitofrontal regions. They were also associated with poor perfusion rate in the left cerebral hemisphere, while in the right cerebral hemisphere, the frontal lesion load and temporal brain volume were involved.24
Similarities between MS and depression
There are some similarities between the pathologies of MS and depression. The lesions in arcuate fasciculus were found to detach few regions of the cerebral cortex. The lesions were evident in the frontotemporal region, while inflammation had an influence on the hypothalamic endocrine functioning, corroborating the coexistence of depression in MS compared to other neurological disorders.25 The symptoms of depression are not closely related to disease severity and its progression, but they are more prevalent during the relapses and in increased neurological disability.20 Though much progress has been made in identifying the depressive pathological origins, their specificity still remains as a question. When observed in a group of RRMS patients with depression, the proinflammatory cytokine interferon (IFN)-γ was excessively produced and was associated with symptom severity. However, when treated with antidepressive medication, the levels of IFN-γ were lowered, explaining its possible role in depressive pathology.26 Similarly, a case-controlled study observed exacerbations of depression in MS patients undergoing treatment with IFN-β.27 A few studies reported contradictory results revealing the uncertainty of MS medication-induced depression.28–31 The major consideration of depression in MS is that it is associated with other behavioral disturbances such as disturbed cognition. Depression may aggravate cognitive dysfunction, which may induce depression, thus worsening the quality of life in all aspects.32
Observations by Alajbegovic et al33 revealed some interesting results. They found that depression was common among the MS population studied, with a majority of them being young, educated, unemployed, and single.33 A longitudinal study also confirmed that the depression associated with MS is chronic and is usually episodic.34 Finally, the most interesting concept to understand is depression and its association with MS relapses. Though there are no substantiating data to support this issue, it definitely seems to pose a fascinating question to be answered. In summary, depression stands as the major behavioral disorder observed in MS, with a potential of aggravating other behavioral abnormalities. Hence, diagnosis and treatment of depression in MS will significantly improve the quality of life in patients.
Cognition and MS
Cognitive disturbances have been reported in 40%–60% of people with MS. Common manifestations include defects in recent memory and impaired attention, analyzing, visuospatial perception, and executional activities.35 Impairment of cognition can be a potential indicator for the demyelination process36 and is more common and severe in the progressive form of the disease rather than the relapses.37 The results of the study conducted by Deloire et al38 also hypothesized the symptoms of cognitive impairment as a forerunner for MS disability, which follows several years later. The precise pathology of cognitive disturbances in MS has not been identified, but inflammatory lesions, regional degeneration, and repetitive black holes somehow relate them to MS pathology.38
It has been previously reported that inflammatory cytokines tend to cause cognitive disturbances, and the association of MS with inflammation indicates the role of cytokines in correlating MS pathology with that of cognitive impairment.39 Loss of gray matter is quiet common among MS patients, and this also leads to the loss of cognitive function. Also, disruptions of the cerebral cortex regions and the thalamus strongly associate memory impairment with that of MS pathology.
Causes of cognitive imbalance in MS
Old age, male sex, ApoE, the gene encoding for apolipoprotein E, and variations in brain-derived neurotrophic factor were evaluated as major risk factors associated with cognitive impairment among MS patients.40 Some studies reported disability of recognizing emotions in MS patients, which were elusive, and associated it with memory impairment,41 while some other studies correlated disability of emotion recognition and perception of behavioral aspects and quality of life.42 Functional MRI studies have also proved that noticeable differences exist between MS patients and controls in emotion recognition, highlighting the point of disturbed brain function and cognition.43–45 Other manifestations that tend to associate disturbed cognition with MS include fatigue and depression, which are more common in MS than in any other neurological diseases. These symptoms are correlated in MS, assuming that they share some commonness in pathology.46 Also, the levels of fatigue and depression were noticeably high in cognitively impaired MS patients and were evident from the measures of attention and memory, respectively.47 Clinically, an interesting question regarding the correlation between depression and cognition loss arises: would the treatment for depression in MS alleviate memory impairment?
Currently, no data are available, but in traumatic brain injury, patients have displayed improved cognition when treated for depression.48 Other factors that have a correlation toward MS pathology and impaired cognition include dysregulation of the hypothalamopitutary–adrenal axis49 and hypercortisolemia. Corticosteroids administered for MS treatment were found to worsen memory, particularly in the relapsing–remitting form of the disease.50
Biomarker measurement
Though modest in correlating cognitive impairment in MS with MRI markers, MRI has been invaluable in the diagnosis of cognitive impairment. The identified MRI markers are T2 and T1 lesional loads, with the former showing a limited correlation, while the latter better related brain atrophy with cognitive loss.20 Also, MS patients showed cholinergic imbalances; hence, measuring the levels of choline acetyltransferase and acetylcholinesterase will be ideal for managing cognitive impairment.51 In conclusion, cognitive loss is identified as an important symptom of MS that is found to significantly hamper daily activities, inclusive of social and emotional functionings. Though the exact pathology remains unclear, the inflammatory and degenerative processes observed in MS can be the major contributors of cognition impairment with a higher impact on executive functions.
Anxiety in MS
Though not much studied in MS patients, anxiety cannot be ignored as it can be an important contributor to disability in them. As per published data, many MS patients exhibit the symptoms of anxiety; in some cases, it is more frequent than depression.52,53
Comparatively, the symptoms of anxiety are more prevalent in MS patients than in controls, and risk factors include female sex, lack of caring, coexisting depression, and ignorance of anxiety symptoms.54 Animal studies have also found the association of inflammation, which is default in MS pathology, and anxiety underlying the probable occurrence of anxious symptoms in MS.21 Anxiety among MS patients needs strict medical attention as it was found to disrupt executive functioning. Intriguingly, cognition was severely affected than when compared to depression among the patient population studied.55 Depression and anxiety run together in a few MS patients and importance should be given to anxiety as it may enhance suicidal tendencies in these patients compared to patients exhibiting little or no anxiety.52 A cross-sectional study also observed symptoms of depression and anxiety occurring twice and thrice, respectively, among MS patients with no sex indifference for depression, while anxiety was reported more in males.56 Another interesting feature reported was that some patients presented the symptoms of anxiety well before the diagnosis of MS.57,58 Considering the predominance of anxious symptoms among MS patients, attempts had been made to identify the connection of exact brain regions involved in this pathology. But still it has not been fruitful as correlation between anxious symptoms and brain MRI scans lacked correlation.24 The impact of anxiety in different forms of the disease was studied and contradictory results were observed. Some studies had reported that relapses in RRMS were more often associated with anxiety and disclosed higher levels of anxious symptoms,59 whereas studies by Noy et al53 and Brown et al60 reported the association of anxiety and relapses to be rather conflicting.
The advent of self-administered immunomodulatory drugs for MS has led to the induction of anxiety named “self-injection anxiety” and was reported to affect almost half of the MS population following this practice.61 Clinical studies have also revealed an increased prevalence of anxiety among the MS patients adding an interesting finding towards the MS associated behavioural compromise. Patients who were included in the study did not show severe neurological compromise but were certainly distressed as they were unable to cope up with the disease and get relief. Other reasons for developing anxiety were anticipated to be strong fear of developing personal and professional lagging, as the patients were emotionally preoccupied and compromised for a complete cure.62 In summary, it is quiet natural that people are concerned with health and become anxious, particularly, if affected with a chronic disease or a disease with no cure at all. Thus, MS, being a chronic disorder with incomplete cure, poses the symptoms of anxiety. The question to be answered in this scenario is to identify the pathologies that are common to MS and anxiety and to target them specifically.
Psychosis and its association with MS
Psychosis and MS share a common pathophysiology and the symptoms are displayed due to regional demyelination during the disease progression. The association was also exacerbated due to the medications for MS. IFN-β administered to lower autoimmunity in MS produced psychotic symptoms, while a reduction in these symptoms was noticed on administration of antipsychotic medication.63 Brain MRI has also not shown any significant changes pre- and post-IFN treatment, strengthening the fact of medication-induced psychosis.64
In a group of comparative studies among MS patients with psychosis and those devoid of psychosis, lesions were found to be more prominent in the MS psychotic patients than in those without psychosis.65,66 These lesions were more specific to the periventricular white matter of the temporal horn regions, and many studies have also associated psychotic symptoms in MS with lesions, substantiating the role of regional demyelination in the pathology of MS psychosis.67,68 There are also studies focusing on the involvement of other brain regions such as temporal, frontotemporal, and temporoparietal lesions, associating psychic symptoms in MS patients69,70 with temporal and frontotemporal and temporoparietal abnormalities.71 There is a growing interest to explore the exact relation between psychosis and MS pathology, as several large-scale studies have also pointed out the coexistence of them. It poses a serious challenge in terms of both treatment and diagnosis when these pathologies run together.72,73 Clinicians have also highlighted the point that psychic patients who do not respond to standard antipsychotic regimen should be diagnosed for the underlying MS pathology. Hence, preferably, MS needs differential diagnosis both for effective therapeutic outcome and ruling out psychotic symptoms.74,75
Euphoria
Euphoria affects approximately 10% of the MS population and is best described as the phase of cheerfulness without displaying the symptoms of motor hyperactivity. It is related to the frontal lobe pathology, which is common in several other personality disorders, along with the involvement of brainstem, pontine, and periventricular lesions. Interesting correlation was drawn from a study where the association of euphoric symptoms in MS was related to severe cognitive disturbances rather than to disease exacerbations, indicating extensive brain pathology.
This study also concluded that euphoric symptoms are predisposed regardless of the sex and are mostly associated with the progressive form of MS and its severity.76
Sleep and fatigue
As MS is associated with behavioral symptoms, it strongly influences sleep patterns and the associated disturbances. Sleep disturbances in MS patients were related to the degenerated regions involving the basal ganglia, thalamus, and limbic system.77 It has been reported that a majority of the MS patients display symptoms of insomnia, narcolepsy, sleep-related breathing abnormalities, and rapid eye movement sleep behavior disorder, thus significantly influencing quality of life.78,79 Clinical studies also revealed sleep disturbances in terms of onset and duration of sleep among MS patients.80
Fatigue in MS
The other major manifestation of MS is the perception of fatigue, and multiple factors are involved in its origin, which include elevated body temperature and cytokine levels, hyperactive hypothalamic–pituitary–adrenal axis, and some medications for associated disorders like depression and spasticity.78,81,82 Though fatigue is quiet common among MS patients, it is incompletely understood. However, two subtypes of fatigue have been reported. These are motor fatigue, which develops due to excess muscular stress, and mental fatigue, which develops due to the intense level of tiresomeness. The etiology of fatigue overlaps with that of other associated disorders and hence can be considered as both primary fatigue, which is purely associated with MS, and secondary fatigue, which is related to the comorbidity.
The symptoms of fatigue mainly arise due to neuronal demyelination, which severely hampers neurotransmission. In the recent past, fatigue in MS had been related to parietotemporal and frontal region lesions.83 Neurotransmitter imbalances, excessive muscular stress, lesions in other brain regions, coexisting disturbances, and medications for MS are other possibilities of fatigue generation.84
Sleep–fatigue correlation
Interesting correlations also exist between symptoms of sleep disturbances and fatigue in MS patients. A comparative study of healthy controls and MS patients concluded that sleep abnormalities were common among MS patients and showed a strong correlation with fatigue. However, this correlation was not observed in the healthy controls.85 Altogether, a precise relationship has not been established between sleep and fatigue; the published data suggest an interrelation between them. Thus, sleep disturbances and fatigue either independently or collectively influence the quality of life in MS patients. The association of pathology of all the behavioral disorders with that of MS is shown in Table 1.
  | Table 1 Association of MS with behavioral disorders |
Behavioral screening studies for MS
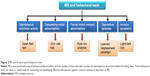
Although several animal models exist for studying behavioral changes in MS, the experimental autoimmune encephalomyelitis (EAE) induced by myelin oligodendrocyte glycoprotein is the most widely accepted mouse model that mimics MS in humans.86 As MS is associated with several behavioral abnormalities, animal models serve as extremely useful tools for assessing and developing novel therapeutic approaches. For example, analyzing the locomotor activities in these animal models provide useful information on the involvement of specific pathologies. A brief introduction of them is as follows:
Open field test
This test screens for spontaneous locomotor activity. In this test, animals are allowed to move and explore freely in an enclosed square area while being monitored for their locomotor activity.21
Grid walk test
This test provides valuable information on the formation of lesions in the dorsal column and descending motor control abnormalities by analyzing an animal’s ability to place its hindlimbs on bars. Animals are trained to walk on a runway consisting of bars placed at an elevation of 1 m above the ground. Comparatively, the number of falls were higher in the MS-induced animals than in the controls when they tried placing their hindlimbs firmly on the bars.87 Other tests for assessing locomotor activities include BBB open field locomotion score and narrow beam test.88
Rota rod analysis
This is the highly accepted test for assessing forced motor moments in rodents. Initially, animals are trained on a rotating rod at a speed of 4–40 rotations per minute and then the grip strength is analyzed by observing the animals’ latency fall from the rotating rod.86
Learned helplessness paradigm
This test mainly aims at studying the depressive episodes in animal models conducted in a customized shuttle box equipped with a shock generator and the software for controlling, processing, and recording. Initially, the animal is conditioned with a phase of inescapable foot shocks and then placed in a testing chamber, from where it can escape to a shock-free chamber.
After sufficient training, the animal is subjected to the same escapable foot shocks, but this time, a light stimulus strikes before the shock is applied. The ability of the animal to avoid the shock with each light stimulus and to escape into the shock-free chamber is tested. If the animal fails to do so, it is assumed as learned helplessness behavior.89
Light/dark box
The light/dark box model measures anxiousness. In this model, animals (mice) are placed in a light and dark compartment, and exploratory behavior is monitored. The analysis is done based on the length of time spent by the animals and number of crossings they make into the lighted compartment, assuming these activities to be reflective of anxious behavior.21 A summary of behavioral screening in MS is shown in Figure 2.
MS therapeutics and applications of nanotechnology
The strategic treatment for MS is mainly concerned to alleviate its symptoms and associated disease progression. The first-line drugs considered for the treatment of MS are IFNs, particularly type 1 and include IFN-β1a and IFN-β1b, along with glatiramer acetate. Their mechanism is mainly targeted against the reactive T-cell activity with a strong inhibition of T-cell proliferation, migration, and antigen presentation.90 Interesting outcomes were seen when the first-line drugs were combined with a natural metabolite, methyl thioadenosine, that possessed potent immunomodulatory activity preclinically.91 Corticosteroids with a mechanistic activity of immune suppression were also effective against MS and its progression. Dexamethasone, prednisone, prednisolone, and methylprednisolone are some of the corticosteroids used to treat MS. However, indiscriminate and long-term use of these drugs is strictly discouraged on account of their severe side effects.92
Chemotherapeutics like cyclophosphamide and mitoxantrone with a background history of immune suppression were found to be efficacious in treating MS. With respect to the mechanism of action, cyclophosphamide showed activity against T and B lymphocytes, while mitoxantrone was found to be cytotoxic.93,94
Monoclonal antibodies
Monoclonal antibodies (mAbs) have made significant contributions toward MS therapeutics. For instance, natalizumab, a humanized mAb, when tried in a patient population resulted in significant reduction in disease recurrence with an action against α-integrins (cell surface molecules essential for T-cell binding and entry). However, long-term use poses the risk of progressive multifocal leukoencephalopathy. mAbs under evaluation include alemtuzumab (Campath) and rituximab, which were observed to have a potential activity against the expression of CD52 and CD20 on leucocytes. When administered to a patient population, they resulted in a considerable reduction in the levels of inflammatory monocytes, lymphocytes, and dendritic cells.95,96 Daclizumab is a humanized mAb that acts against CD25 expression, thereby limiting the proliferation of reactive T-cell. This drug has recently finished its Phase II trial.97
Drug trials
The drug in trials with a prime motto of ameliorating disease progression and severity is the oral formulation BG00012. Its active principle is fumarate. It has the characteristic mechanisms of neuroprotection, relieving inflammatory cascade and oxidative stress.
Fingolimod is one of the class of MS therapeutics with an activity of lymphocyte complexatoin. BG00012 is in Phase II clinical trials,98 and fingolimod has recently received approval for human use.99 Firategrast, an antagonist for the very late antigen-4 (a cell adhesion molecule expressed on brain capillary cells) has finished its Phase II clinical trial and acts by inhibiting the entry of T-lymphocytes into the BBB. Laquinimod and teriflunomide are the representatives for drugs in Phase III trials, and their mechanisms include the immune response shift toward Th2 type and inhibition of lymphocytic expansion.16,100 A proprietary recombinant human IFN-β1b (NU 100) and an extended-release polyethylene glycol coated IFN-β1a (BII-B017) are in Phase III trials for the treatment of RRMS.101
Applications of nanotechnology
Nanotechnology has always been fascinating as it deals with ultrasmall particles with a dimension range of 1–100 nm. Specific advantages attributed to these nanoparticles (NPs) considering their size and durability are that they offer huge surface area, improving bioavailability and absorption, and enhanced physicochemical stability of theranostics (therapeutic + diagnostics). A unique feature of this technology is that NPs can be tailor-made for specific targets, thus nullifying unwanted biodistribution and achieving the maximum efficacy at a reduced drug dose.91 The same application would also benefit drug permeation through the complex BBB, which is viewed as a major hurdle for conventional drugs to pass through. When applied, this technology would certainly benefit the nervous system, promoting the viability and integrity of neurons that have a limited regenerative capacity. Hence, nanotechnology holds a strong future potential for theranostic applications against a variety of neurological disorders and thus overcome the limitations of existing therapeutics. The following is a brief description of NP application in MS. It is evident that matrix metallopeptidase-9 (MMP-9) plays a pivotal role in disrupting the BBB, thereby attracting peripheral T-cells and thus initiating the neuroinflammation cascade. A study has shown that the quantum dot complexed with MMP-9–small interfering RNA (nanoplex) has significantly blocked the expression of MMP-9 in brain microvascular endothelial cells and leukocytes. In addition, upregulation of extracellular matrix proteins like collagen types I, IV, and V was observed, indicating the successful delivery and efficacy of small interfering RNA nanoplex in vitro.102 Tissue inhibitor of matrix metalloproteinase-1 is a potent inhibitor of MMP-9, which is one of the main culprits associated with neuroinflammation. A study reported encapsulation of tissue inhibitor of matrix metalloproteinase-1 in poly(lactic-co-glycolic) acid NPs and strong neuroprotective activity in vitro by inhibiting the gelatinase-mediated MMP-9 activity.103 Interesting results were reported showing the antioxidant activity of fullerene (C60), where C60 suspension on administration showed strong protective activity against carbon tetrachloride intoxication in rats.104 This could be a potential application if tested against disorders such as MS that are associated with free radical toxicity. In an EAE model induced by the proteolipid protein PLP139–151 administration, significant improved tolerance was observed in mice that were administered in prior with the PLP139–151-coupled poly(lactic-co-glycolic) acid NPs. Antigen-presenting cells internalized these NPs via scavenger receptors and induced both T-cell anergy and enhanced T-reg activity. The disease improvement was also confirmed with the vascular (AngioSense 750EX) and protease-activatable cathepsin B (Cat B 680 FAST) near-infrared fluorescence imaging agents, which showed a respective decrease in the BBB breakdown and inflammation.105
The T-cell regulatory activity was also improved in an autoimmune model, wherein the authors fabricated disease-relevant self-peptides and the class I peptide–major histocompatibility complex-coated iron oxide NPs.106 Thus, the antigen-specific T-reg cell expansion can be a potential approach if modified using the MS specific antigen. Similar results were also reported where gold NPs carrying 2-(1′H-indole-3′-carbonyl)-thiazole-4-carboxylic acid methyl ester and myelin oligodendrocyte glycoprotein showed significant disease remission. NP-treated dendritic cells showed a tolerogenic phenotype and induced the differentiation of T-reg cells, which significantly abrogated disease generation.107 The monolayer mannosylated liposomes packed with various immunodominant myelin basic protein (MBP) fragments showed significant reduction in the level of anti-MBP antibodies. This nanoformulation showed a synergetic therapeutic effect, wherein disease scores were reduced showing a first EAE wave inhibition followed by subsequent remission in vivo.108 Gold NPs encapsulated with potential antioxidant compounds epigallocatechin gallate, and α-lipoic acid were found to have strong wound healing and migration along with an increase in the antioxidant activity in vivo. This could be a future application for evaluating against MS pathology.109 Cerium oxide (CeO) NPs are regarded as potent antioxidants on account of their ability to either donate or receive electrons. It was also reported that CeO NPs showed an extremely small size of 2.9 nm with enhanced brain uptake and reduced reactive oxygen species in vivo. The authors claim that custom-made CeO NPs provide protection against autoimmune diseases associated with free radical toxicity.110 Further to this, a novel pH-sensitive IFN-β microparticle was synthesized using trimethyl-chitosan, poly(ethylene glycol)-D-methacrylate, and methacrylic acid. These microparticles showed effective drug release over 24 hours both in vitro and in vivo.111
Neuroprotection was also made possible with the implication of fullerenol (polyhydroxylated C60), which mitigated neuronal excitotoxicity antagonizing the glutamate receptors in amyotrophic lateral sclerosis model.112 This function of fullerenol also holds potential application for MS too. The implication of nanosurgery is underway and is at the threshold of revolutionizing neurological disorders. Earlier, nanosurgery was only a dream, but now the current nano tools have made the dream come true. The introduction of quantum dots for imaging, multipartite NPs for neuromodulations, and nano manipulations mediated by atomic force microscopy cantilever have all made it possible.113 A study reported the use of atomic force microscopy tip as nanoscissors for indenting the cell wall by only 1 μm, which otherwise is inevitably difficult employing conventional techniques.114 This kind of applications opens up new avenues for single-cell and chromosomal surgeries operating at submicron levels and would highly benefit the MS brain where damaged neurons can be successfully treated without affecting the healthy ones. NPs not only have a role in therapeutic application, but have also improvised the diagnostic strategy with enhanced sensitivity, enabling early detection of the disease. Likewise, the titanium dioxide nanocomposite film modified with alginate coat has been immobilized with MBP, and when tested for antigen detection, it showed an astounding sensitivity of 0.18 ng/mL. Thus, NPs have improved MS diagnosis, wherein a blood sample is sufficient instead of CSF samples.115 MS diagnosis was even improved with the application of very small superparamagnetic iron oxide particles, which showed a characteristic accumulation in the inflammatory lesions. The very small superparamagnetic iron oxide particles were also successful in discriminating inflammatory events between control and established EAE, with clear demarcation of the central nervous system alterations preceding immune cell infiltration.116
Similarly, detection sensitivity was enhanced using the streptavidin antibody-conjugated superparamagnetic iron oxide particles, as they are superior contrast agents because of their T2* relaxation properties. These target-specific NPs were able to visualize CD3+ T-cell infiltrates in a murine MS model, confirming the diagnostic potential of NPs.117 Some recent advancements were made with the introduction of gadolinium–diethylene triamine pentaacetic acid and ultrasmall superparamagnetic particles of iron oxide (USPIO). Gadolinium–diethylene triamine pentaacetic acid is a routinely employed inflammatory marker,118 while USPIO can detect cellular infiltration and inflammation progression in MS.119 The application of stem cell therapeutics has also garnered significant attention; it was found that patients administered with autologous hematopoietic stem cells showed much improved outcome in Phase II trials.120,121 Further, coupling nanotechnology platforms would improvise the stem cell-based theranostics. For instance, carbon nanotube substrates regulate growth and neurite regeneration, which can find potential application for neural regeneration.122 Intriguingly, USPIOs can label stem cells and help in tracking the fate of these cells using MRI.123 Table 2 summarizes the list of nanotechnology applications in MS.
What is in the pipeline?
Agents possessing neuroprotective activity are worth considering as treatment for MS, and histone deacetylase inhibitors is one such class possessing strong anti-immune activity.124 Inflammatory insults play a major role in the onset and progression of MS, and hence therapeutics acting against it can be invaluable. Likewise, inhibitors of midkine (a cytokine that drives T-cell traffic followed by inflammatory cascade) are under development, as they have shown a remission preclinically in an MS model.125
Physiologically, carbon monoxide has some anti-inflammatory and cytoprotective activities. Interestingly, when carbon monoxide-releasing molecules were tested preclinically in the EAE model, they were found to alleviate the pathologic disease scores, driving the need for clinical evaluation.126 The potential immunomodulatory role of vitamin D is also being evaluated in a Phase II trial.127 Table 3 gives a summary of the drugs included.
  | Table 3 Current drugs in trials for MS treatment |
Future trends
As per the data published, there is commonness in the pathologies of MS and some behavioral disorders. Hence, selectively identifying and targeting the events of MS will also prove to be potential for the alleviation of comorbidities. Some of our own results are discussed here. We have adopted the principle of neuroprotection and observed anti-inflammatory effects of anti-mucosal addressin cell adhesion molecule-1 (MAdCAM-1) mAb, which protected oligodendrocytes. Also, simultaneous administration of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionate/kainate antagonist 2,3-dihydroxy-6-nitro-7-sulfamoylbenzo(f)quinoxaline and the neuroprotector glycine–proline–glutamic acid (N-terminal tripeptide of insulin-like growth factor) resulted in strong neuroprotector activity, confirmed with the significant reduction in impaired neurological scores in vivo.4 More rapid disease remittance was observed in mice when a combination of anti-MAdCAM-1, vascular cell adhesion molecule-1, intercellular cell adhesion molecule-1, and ligand for lymphocyte function-associated antigen integrin mAbs was administered.128 Also, the contribution of integrin (α4β7 and αEβ7)-mediated inflammation in MS was significantly remitted, administering antibodies against them.129
Thus, after the analysis of the results, we assume that the antibodies targeting the inflammatory ligands involved in MS may also impede the inflammatory component of the depressive episodes and cognitive impairment followed by their remission. Hence, it is postulated that identifying and targeting the pathological events specific to MS can definitely prove to be beneficial for relieving the comorbid behavioral symptoms as well. This is because of the findings of commonness in the pathologies of MS and behavioral disorders. Future also holds an interesting question to answer: shall the treatments initiated for behavioral symptoms improve MS too? Figure 3 shows strategic treatment options for MS.
Conclusion
Behavioral disturbances are found to be common among MS patients, irrespective of the severity and the course of disease progression. Depression seems to be the most common neuropsychiatric abnormality observed in MS compared to any other neurological disorders. It is also associated with cognitive impairment in few of the MS patients, raising the question of commonness in their pathology. It is worthwhile to identify the early symptoms of this memory loss, as it is considered to be a forerunner of MS in a group of patients. Psychosis, euphoric symptoms, sleep disturbances, and fatigue are other influential comorbidities associated with MS that will drastically affect the quality of a patient’s life. Hence, identifying disease-specific targets and coupling them with NPs will result in the effective diagnosis and treatment of the disease. This would also positively affect and help overcome the associated behavioral symptoms in MS. “The better the quality of life, the better the recovery”.
Acknowledgment
Jagat R Kanwar acknowledges the funding received from the Department of Innovation Industry Science, Research and Commonwealth Australia (BF030016/42), and National Health and Medical Research Council.
Disclosure
The authors report no conflicts of interest in this work.
References
Lassmann H, Bruck W, Lucchinetti CF. The immunopathology of multiple sclerosis: an overview. Brain Pathol. 2007;17(2):210–218. | ||
Reipert B. Multiple sclerosis: a short review of the disease and its differences between men and women. J Men’s Health Gend. 2004;1:334–340. | ||
Kanwar JR, Kanwar RK, Burrow H, Baratchi S. Recent advances on the roles of NO in cancer and chronic inflammatory disorders. Curr Med Chem. 2009;16(19):2373–2394. | ||
Kanwar JR, Kanwar RK, Krissansen GW. Simultaneous neuroprotection and blockade of inflammation reverses autoimmune encephalomyelitis. Brain. 2004;127(Pt 6):1313–1331. | ||
Hohlfeld R, Wekerle H. Autoimmune concepts of multiple sclerosis as a basis for selective immunotherapy: from pipe dreams to (therapeutic) pipelines. Proc Natl Acad Sci U S A. 2004;101(suppl 2):14599–14606. | ||
Frohman EM, Racke MK, Raine CS. Multiple sclerosis – the plaque and its pathogenesis. N Engl J Med. 2006;354(9):942–955. | ||
Viglietta V, Baecher-Allan C, Weiner HL, Hafler DA. Loss of functional suppression by CD4+CD25+ regulatory T cells in patients with multiple sclerosis. J Exp Med. 2004;199(7):971–979. | ||
Skulina C, Schmidt S, Dornmair K, et al. Multiple sclerosis: brain-infiltrating CD8+ T cells persist as clonal expansions in the cerebrospinal fluid and blood. Proc Natl Acad Sci U S A. 2004;101(8):2428–2433. | ||
Charo IF, Ransohoff RM. The many roles of chemokines and chemokine receptors in inflammation. N Engl J Med. 2006;354(6):610–621. | ||
Steinman L. A few autoreactive cells in an autoimmune infiltrate control a vast population of nonspecific cells: a tale of smart bombs and the infantry. Proc Natl Acad Sci U S A. 1996;93(6):2253–2256. | ||
Somerset M, Sharp D, Campbell R. Multiple sclerosis and quality of life: a qualitative investigation. J Health Ser Res Policy. 2002;7(3):151–159. | ||
Voskuhl RR. Gender issues and multiple sclerosis. Curr Neurol Neurosci Rep. 2002;2(3):277–286. | ||
Axelsson M, Malmestrom C, Nilsson S, Haghighi S, Rosengren L, Lycke J. Glial fibrillary acidic protein: a potential biomarker for progression in multiple sclerosis. J Neurol. 2011;258(5):882–888. | ||
Gajofatto A, Bongianni M, Zanusso G, Benedetti MD, Monaco S. Are cerebrospinal fluid biomarkers useful in predicting the prognosis of multiple sclerosis patients? Int J Mol Sci. 2011;12(11):7960–7970. | ||
Vogt MH, Lopatinskaya L, Smits M, Polman CH, Nagelkerken L. Elevated osteopontin levels in active relapsing-remitting multiple sclerosis. Ann Neurol. 2003;53(6):819–822. | ||
Weinstock-Guttman B, Ramanathan M. Multiple sclerosis in 2011: advances in therapy, imaging and risk factors in MS. Nat Rev Neurol. 2012;8(2):66–68. | ||
Multiple Sclerosis. Mayoclinic. Available from: www.mayoclinic.org/multiple-sclerosis. Accessed Nov 1, 2014. | ||
Chwastiak L, Ehde DM, Gibbons LE, Sullivan M, Bowen JD, Kraft GH. Depressive symptoms and severity of illness in multiple sclerosis: epidemiologic study of a large community sample. Am J Psychiatry. 2002;159(11):1862–1868. | ||
Harel Y, Barak Y, Achiron A. Dysregulation of affect in multiple sclerosis: new phenomenological approach. Psychiatry Clin Neurosci. 2007;61(1):94–98. | ||
McDonald WI, Ron MA. Multiple sclerosis: the disease and its manifestations. Philos Trans R Soc Lond B Biol Sci. 1999;354(1390):1615–1622. | ||
Peruga I, Hartwig S, Thone J, et al. Inflammation modulates anxiety in an animal model of multiple sclerosis. Behav Brain Res. 2011;220(1):20–29. | ||
Patten SB, Metz LM, Reimer MA. Biopsychosocial correlates of lifetime major depression in a multiple sclerosis population. Mult Scler. 2000;6(2):115–120. | ||
Pujol J, Bello J, Deus J, Marti-Vilalta JL, Capdevila A. Lesions in the left arcuate fasciculus region and depressive symptoms in multiple sclerosis. Neurology. 1997;49(4):1105–1110. | ||
Zorzon M, de Masi R, Nasuelli D, et al. Depression and anxiety in multiple sclerosis. A clinical and MRI study in 95 subjects. J Neurol. 2001;248(5):416–421. | ||
Andrade APTG. Prevalence of depression in multiple sclerosis. Arquivos de Neuro-Psiquiatria. 2011;69:415. | ||
Mohr DC, Goodkin DE, Islar J, Hauser SL, Genain CP. Treatment of depression is associated with suppression of nonspecific and antigen-specific T(H)1 responses in multiple sclerosis. Arch Neurol. 2001;58(7):1081–1086. | ||
Jacobs LD, Beck RW, Simon JH, et al. Intramuscular interferon beta-1a therapy initiated during a first demyelinating event in multiple sclerosis. CHAMPS Study Group. N Engl J Med. 2000;343(13):898–904. | ||
Feinstein A, O’Connor P, Feinstein K. Multiple sclerosis, interferon beta-1b and depression. A prospective investigation. J Neurol. 2002;249(7):815–820. | ||
Mohr DC, Likosky W, Dwyer P, Van Der Wende J, Boudewyn AC, Goodkin DE. Course of depression during the initiation of interferon beta-1a treatment for multiple sclerosis. Arch Neurol. 1999;56(10):1263–1265. | ||
Patten SB, Metz LM. Interferon beta-1a and depression in relapsing-remitting multiple sclerosis: an analysis of depression data from the PRISMS clinical trial. Mult Scler. 2001;7(4):243–248. | ||
Zephir H, De Seze J, Stojkovic T, et al. Multiple sclerosis and depression: influence of interferon beta therapy. Mult Scler. 2003;9(3):284–288. | ||
Feinstein A. Mood disorders in multiple sclerosis and the effects on cognition. J Neurol Sci. 2006;245(1–2):63–66. | ||
Alajbegovic A, Loga N, Tiro N, Alajbegovic S, Todorovic L, Jasminika D. Depression in multiple sclerosis patients. Med Arh. 2011;65(2):115–118. | ||
Koch MW, Patten S, Berzins S, et al. Depression in multiple sclerosis: a long-term longitudinal study. Mult Scler. 2015;21(1):76–82. | ||
Chiaravalloti ND, DeLuca J. Cognitive impairment in multiple sclerosis. Lancet Neurol. 2008;7(12):1139–1151. | ||
Achiron A, Barak Y. Cognitive changes in early MS: a call for a common framework. J Neurol Sci. 2006;245(1–2):47–51. | ||
Amato MP, Zipoli V, Portaccio E. Multiple sclerosis-related cognitive changes: a review of cross-sectional and longitudinal studies. J Neurol Sci. 2006;245(1–2):41–46. | ||
Deloire M, Ruet A, Hamel D, Bonnet M, Brochet B. Early cognitive impairment in multiple sclerosis predicts disability outcome several years later. Mult Scler. 2010;16(5):581–587. | ||
Kelley KW, Bluthe RM, Dantzer R, et al. Cytokine-induced sickness behavior. Brain Behav Immun. 2003;17(suppl 1):S112–S118. | ||
Benedict RH, Zivadinov R. Risk factors for and management of cognitive dysfunction in multiple sclerosis. Nat Rev Neurol. 2011;7:332–342. | ||
Henry JD, Phillips LH, Beatty WW, et al. Evidence for deficits in facial affect recognition and theory of mind in multiple sclerosis. J Int Neuropsychol Soc. 2009;15(2):277–285. | ||
Phillips LH, Henry JD, Scott C, Summers F, Whyte M, Cook M. Specific impairments of emotion perception in multiple sclerosis. Neuropsychology. 2011;25(1):131–136. | ||
Jehna M, Langkammer C, Wallner-Blazek M, et al. Cognitively preserved MS patients demonstrate functional differences in processing neutral and emotional faces. Brain Imaging Behav. 2011;5(4):241–251. | ||
Sandroff BM, Hillman CH, Benedict RH, Motl RW. Acute effects of walking, cycling, and yoga exercise on cognition in persons with relapsing-remitting multiple sclerosis without impaired cognitive processing speed. J Clin Exp Neuropsychol. 2015:1–11. | ||
Settle JR, Robinson SA, Kane R, Maloni HW, Wallin MT. Remote cognitive assessments for patients with multiple sclerosis: a feasibility study. Mult Scler. 2015;21(8):1072–1079. | ||
Krupp LB, Elkins LE. Fatigue and declines in cognitive functioning in multiple sclerosis. Neurology. 2000;55(7):934–939. | ||
Heesen C, Schulz KH, Fiehler J, et al. Correlates of cognitive dysfunction in multiple sclerosis. Brain Behav Immun. 2010;24(7):1148–1155. | ||
Fann JR, Uomoto JM, Katon WJ. Cognitive improvement with treatment of depression following mild traumatic brain injury. Psychosomatics. 2001;42(1):48–54. | ||
Heesen C, Gold SM, Raji A, Wiedemann K, Schulz KH. Cognitive impairment correlates with hypothalamo-pituitary-adrenal axis dysregulation in multiple sclerosis. Psychoneuroendocrinology. 2002;27(4):505–517. | ||
Brunner R, Schaefer D, Hess K, Parzer P, Resch F, Schwab S. Effect of corticosteroids on short-term and long-term memory. Neurology. 2005;64(2):335–337. | ||
Kooi EJ, Prins M, Bajic N, et al. Cholinergic imbalance in the multiple sclerosis hippocampus. Acta Neuropathol. 2011;122(3):313–322. | ||
Feinstein A, O’Connor P, Gray T, Feinstein K. The effects of anxiety on psychiatric morbidity in patients with multiple sclerosis. Mult Scler. 1999;5(5):323–326. | ||
Noy S, Achiron A, Gabbay U, et al. A new approach to affective symptoms in relapsing-remitting multiple sclerosis. Compr Psychiatry. 1995;36(5):390–395. | ||
Korostil M, Feinstein A. Anxiety disorders and their clinical correlates in multiple sclerosis patients. Mult Scler. 2007;13(1):67–72. | ||
Julian LJ, Arnett PA. Relationships among anxiety, depression, and executive functioning in multiple sclerosis. Clin Neuropsychol. 2009;23(5):794–804. | ||
Beiske AG, Svensson E, Sandanger I, et al. Depression and anxiety amongst multiple sclerosis patients. Eur J Neurol. 2008;15(3):239–245. | ||
Barker-Collo S, Cartwright C, Read J. Into the unknown: the experiences of individuals living with multiple sclerosis. J Neurosci Nurs. 2006;38(6):435–441, 46. | ||
Isaksson AK, Ahlstrom G. From symptom to diagnosis: illness experiences of multiple sclerosis patients. J Neurosci Nurs. 2006;38(4):229–237. | ||
Kalb R. The emotional and psychological impact of multiple sclerosis relapses. J Neurol Sci. 2007;256(suppl 1):S29–S33. | ||
Brown RF, Valpiani EM, Tennarnt CC, Dunn SM, Sharrock M, Hodgkinson S et al. Longitudinal assessment of anxiety, depression, and fatigue in people with multiple sclerosis. Psychology and Psychotherapy. 2009;82:41–56. | ||
Mohr DC, Cox D, Epstein L, Boudewyn A. Teaching patients to self-inject: pilot study of a treatment for injection anxiety and phobia in multiple sclerosis patients prescribed injectable medications. J Behav Ther Exp Psychiatry. 2002;33(1):39–47. | ||
Giordano A, Granella F, Lugaresi A, et al. Anxiety and depression in multiple sclerosis patients around diagnosis. J Neurol Sci. 2011;307(1–2):86–91. | ||
Manfredi G, Kotzalidis GD, Sani G, Koukopoulos AE, Savoja V, Lazanio S, et al. Persistent interferon-ss-1b-induced psychosis in a patient with multiple sclerosis. Psychiatry Clin Neurosci. 2010;64(5):584–586. | ||
Feinstein A, du Boulay G, Ron MA. Psychotic illness in multiple sclerosis. A clinical and magnetic resonance imaging study. Br J Psychiatry. 1992;161:680–685. | ||
Asghar-Ali AA, Taber KH, Hurley RA, Hayman LA. Pure neuropsychiatric presentation of multiple sclerosis. Am J Psychiatry. 2004;161(2):226–231. | ||
Honer WG, Hurwitz T, Li DK, Palmer M, Paty DW. Temporal lobe involvement in multiple sclerosis patients with psychiatric disorders. Arch Neurol. 1987;44(2):187–190. | ||
Chong SA, Ko SM. Clozapine treatment of psychosis associated with multiple sclerosis. Can J Psychiatry. 1997;42(1):90–91. | ||
Felgenhauer K. Psychiatric disorders in the encephalitic form of multiple sclerosis. J Neurol. 1990;237(1):11–18. | ||
Habek M, Brinar M, Brinar VV, Poser CM. Psychiatric manifestations of multiple sclerosis and acute disseminated encephalomyelitis. Clin Neurol Neurosurg. 2006;108(3):290–294. | ||
Mendez MF. Multiple sclerosis presenting as catatonia. Int J Psychiatry Med. 1999;29(4):435–441. | ||
Brinar V, Zadro I. Psychic manifestations in multiple sclerosis and other idiopathic demyelinating diseases of central nervous system. Neurol Croat. 2007;56:82–87. | ||
Dickstein LP, Viguera AC, Nowacki AS, et al. Thoughts of death and self-harm in patients with epilepsy or multiple sclerosis in a tertiary care center. Psychosomatics. 2015;56(1):44–51. | ||
Marrie RA, Reingold S, Cohen J, et al. The incidence and prevalence of psychiatric disorders in multiple sclerosis: a systematic review. Mult Scler. 2015:21:305–317. | ||
Diaz-Olavarrieta C, Cummings JL, Velazquez J, Garcia de la Cadena C. Neuropsychiatric manifestations of multiple sclerosis. J Neuropsychiatry Clin Neurosci. 1999;11(1):51–57. | ||
Ron MA, Logsdail SJ. Psychiatric morbidity in multiple sclerosis: a clinical and MRI study. Psychol Med. 1989;19(4):887–895. | ||
Kosmidis MH, Giannakou M, Messinis L, Papathanasopoulos P. Psychotic features associated with multiple sclerosis. Int Rev Psychiatry. 2010;22(1):55–66. | ||
Feinstein A, Feinstein K, Gray T, O’Connor P. Prevalence and neurobehavioral correlates of pathological laughing and crying in multiple sclerosis. Arch Neurol. 1997;54(9):1116–1121. | ||
Gottschalk M, Kumpfel T, Flachenecker P, et al. Fatigue and regulation of the hypothalamo-pituitary-adrenal axis in multiple sclerosis. Arch Neurol. 2005;62(2):277–280. | ||
Merlino G, Fratticci L, Lenchig C, et al. Prevalence of ‘poor sleep’ among patients with multiple sclerosis: an independent predictor of mental and physical status. Sleep Med. 2009;10(1):26–34. | ||
Kaynak H, Altintas A, Kaynak D, et al. Fatigue and sleep disturbance in multiple sclerosis. Eur J Neurol. 2006;13(12):1333–1339. | ||
Natelson BH, LaManca JJ, Denny TN, et al. Immunologic parameters in chronic fatigue syndrome, major depression, and multiple sclerosis. Am J Med. 1998;105(3A):43S–49S. | ||
Tachibana N, Howard RS, Hirsch NP, Miller DH, Moseley IF, Fish D. Sleep problems in multiple sclerosis. Eur Neurol. 1994;34(6):320–323. | ||
Krupp LB, Pollina DA. Mechanisms and management of fatigue in progressive neurological disorders. Curr Opin Neurol. 1996;9(6):456–460. | ||
Sepulcre J, Masdeu JC, Goni J, et al. Fatigue in multiple sclerosis is associated with the disruption of frontal and parietal pathways. Mult Scler. 2009;15(3):337–344. | ||
Paparrigopoulos T, Ferentinos P, Kouzoupis A, Koutsis G, Papadimitriou GN. The neuropsychiatry of multiple sclerosis: focus on disorders of mood, affect and behaviour. Int Rev Psychiatry. 2010;22(1):14–21. | ||
Attarian HP, Brown KM, Duntley SP, Carter JD, Cross AH. The relationship of sleep disturbances and fatigue in multiple sclerosis. Arch Neurol. 2004;61(4):525–528. | ||
Crawley JN. Behavioral phenotyping of transgenic and knockout mice: experimental design and evaluation of general health, sensory functions, motor abilities, and specific behavioral tests. Brain Res. 1999;835(1):18–26. | ||
Metz GA, Whishaw IQ. Cortical and subcortical lesions impair skilled walking in the ladder rung walking test: a new task to evaluate fore- and hindlimb stepping, placing, and co-ordination. J Neurosci Methods. 2002;115(2):169–179. | ||
Buddeberg BS, Kerschensteiner M, Merkler D, Stadelmann C, Schwab ME. Behavioral testing strategies in a localized animal model of multiple sclerosis. J Neuroimmunol. 2004;153(1–2):158–170. | ||
Chourbaji S, Zacher C, Sanchis-Segura C, Dormann C, Vollmayr B, Gass P. Learned helplessness: validity and reliability of depressive-like states in mice. Brain Res Brain Res Protoc. 2005;16(1–3):70–78. | ||
Kanwar JR SB, Kanwar RK. Recent Advances in the treatment of neurological autoimmune disorders. In: Huang F-P, editor. Autoimmune Disorders - Current Concepts and Advances from Bedside to Mechanistic Insights InTech; Intech Publishers. Croatia. 2011:289–322. | ||
Moreno B, Fernandez-Diez B, Di Penta A, Villoslada P. Preclinical studies of methylthioadenosine for the treatment of multiple sclerosis. Mult Scler. 2010;16(9):1102–1108. | ||
Chan A, Weilbach FX, Toyka KV, Gold R. Mitoxantrone induces cell death in peripheral blood leucocytes of multiple sclerosis patients. Clin Exp Immunol. 2005;139(1):152–158. | ||
Fauci AS, Wolff SM, Johnson JS. Effect of cyclophosphamide upon the immune response in Wegener’s granulomatosis. N Engl J Med. 1971;285(27):1493–1496. | ||
Buck D, Hemmer B. Treatment of multiple sclerosis: current concepts and future perspectives. J Neurol. 2011;258(10):1747–1762. | ||
Osborne R. Buzz around Campath proof-of-concept trial in MS. Nat Biotechnol. 2009;27(1):6–8. | ||
Hauser SL, Waubant E, Arnold DL, et al. B-cell depletion with rituximab in relapsing-remitting multiple sclerosis. N Engl J Med. 2008;358(7):676–688. | ||
Saidha S, Eckstein C, Calabresi PA. New and emerging disease modifying therapies for multiple sclerosis. Ann N Y Acad Sci. 2012;1247:117–137. | ||
Lutterotti A. Advances in multiple sclerosis therapy: new oral disease-modifying agents. CML Mult Scler. 2010;2:1–10. | ||
Yang JS, Xu LY, Xiao BG, Hedlund G, Link H. Laquinimod (ABR-215062) suppresses the development of experimental autoimmune encephalomyelitis, modulates the Th1/Th2 balance and induces the Th3 cytokine TGF-beta in Lewis rats. J Neuroimmunol. 2004;156(1–2):3–9. | ||
Palmer AM. Pharmacotherapeutic options for the treatment of multiple sclerosis. Clin Med Insights Ther. 2012;4:145–168. | ||
Bonoiu A, Mahajan SD, Ye L, et al. MMP-9 gene silencing by a quantum dot-siRNA nanoplex delivery to maintain the integrity of the blood brain barrier. Brain Res. 2009;1282:142–155. | ||
Chaturvedi M, Figiel I, Sreedhar B, Kaczmarek L. Neuroprotection from tissue inhibitor of metalloproteinase-1 and its nanoparticles. Neurochem Int. 2012;61(7):1065–1071. | ||
Gharbi N, Pressac M, Hadchouel M, Szwarc H, Wilson SR, Moussa F. [60]fullerene is a powerful antioxidant in vivo with no acute or subacute toxicity. Nano Lett. 2005;5(12):2578–2585. | ||
Eaton VL, Vasquez KO, Goings GE, Hunter ZN, Peterson JD, Miller SD. Optical tomographic imaging of near infrared imaging agents quantifies disease severity and immunomodulation of experimental autoimmune encephalomyelitis in vivo. J Neuroinflammation. 2013;10:138. | ||
Tsai S, Shameli A, Yamanouchi J, et al. Reversal of autoimmunity by boosting memory-like autoregulatory T cells. Immunity. 2010;32(4):568–580. | ||
Yeste A, Nadeau M, Burns EJ, Weiner HL, Quintana FJ. Nanoparticle-mediated codelivery of myelin antigen and a tolerogenic small molecule suppresses experimental autoimmune encephalomyelitis. Proc Natl Acad Sci U S A. 2012;109(28):11270–11275. | ||
Stepanov AV, Belogurov AA, Mamedov AE, et al. Therapeutic effect of encapsulated into the nanocontainers MBP immunodominant peptides on EAE development in DA rats. Bioorg Khim. 2012;38(3):306–314. | ||
Leu JG, Chen SA, Chen HM, et al. The effects of gold nanoparticles in wound healing with antioxidant epigallocatechin gallate and alpha-lipoic acid. Nanomedicine. 2012;8(5):767–775. | ||
Heckman KL, DeCoteau W, Estevez A, et al. Custom cerium oxide nanoparticles protect against a free radical mediated autoimmune degenerative disease in the brain. ACS Nano. 2013;7(12):10582–10596. | ||
Kondiah PP, Tomar LK, Tyagi C, et al. A novel pH-sensitive interferon-beta (INF-beta) oral delivery system for application in multiple sclerosis. Int J Pharm. 2013;456(2):459–472. | ||
Dugan LL, Turetsky DM, Du C, et al. Carboxyfullerenes as neuroprotective agents. Proc Natl Acad Sci U S A. 1997;94(17):9434–9439. | ||
Jeffries GD, Edgar JS, Zhao Y, Shelby JP, Fong C, Chiu DT. Using polarization-shaped optical vortex traps for single-cell nanosurgery. Nano Lett. 2007;7(2):415–420. | ||
Obataya I, Nakamura C, Han S, Nakamura N, Miyake J. Nanoscale operation of a living cell using an atomic force microscope with a nanoneedle. Nano Lett. 2005;5(1):27–30. | ||
Derkus B, Emregul E, Emergul KC, Yucesan C. Alginate and alginate-titanium dioxide nanocomposite as electrode materials for anti-myelin basic protein immunosensing. Sens Actuators B Chem. 2014;192:294–302. | ||
Millward JM, Schnorr J, Taupitz M, Wagner S, Wuerfel JT, Infante-Duarte C. Iron oxide magnetic nanoparticles highlight early involvement of the choroid plexus in central nervous system inflammation. ASN Neuro. 2013;5(1):e00110. | ||
Luchetti A, Milani D, Ruffini F, et al. Monoclonal antibodies conjugated with superparamagnetic iron oxide particles allow magnetic resonance imaging detection of lymphocytes in the mouse brain. Mol Imaging. 2012;11(2):114–125. | ||
Heta Y, Kumaki K, Hifumi H, Citterio D, Tanimoto A, Suzuki K. Gadolinium containing photochromic micelles as potential magnetic resonance imaging traceable drug carriers. Photochem Photobiol. 2012;88(4):876–883. | ||
Richards JM, Shaw CA, Lang NN, et al. In vivo mononuclear cell tracking using superparamagnetic particles of iron oxide: feasibility and safety in humans. Circ Cardiovasc Imaging. 2012;5(4):509–517. | ||
Shevchenko JL, Kuznetsov AN, Ionova TI, et al. Long-term outcomes of autologous hematopoietic stem cell transplantation with reduced-intensity conditioning in multiple sclerosis: physician’s and patient’s perspectives. Ann Hematol. 2015;94(7):1149–1157. | ||
Mancardi GL, Sormani MP, Gualandi F, et al. Autologous hematopoietic stem cell transplantation in multiple sclerosis: a phase II trial. Neurology. 2015;84(10):981–988. | ||
Huang YJ, Wu HC, Tai NH, Wang TW. Carbon nanotube rope with electrical stimulation promotes the differentiation and maturity of neural stem cells. Small. 2012;8(18):2869–2877. | ||
Wu X, Hu J, Zhou L, et al. In vivo tracking of superparamagnetic iron oxide nanoparticle-labeled mesenchymal stem cell tropism to malignant gliomas using magnetic resonance imaging. Laboratory investigation. J Neurosurg. 2008;108(2):320–329. | ||
Faraco G, Cavone L, Chiarugi A. The therapeutic potential of HDAC inhibitors in the treatment of multiple sclerosis. Mol Med. 2011;17(5–6):442–447. | ||
Muramatsu T. Midkine: a promising molecule for drug development to treat diseases of the central nervous system. Curr Pharm Des. 2011;17(5):410–423. | ||
Fagone P, Mangano K, Coco M, et al. Therapeutic potential of carbon monoxide in multiple sclerosis. Clin Exp Immunol. 2012;167(2):179–187. | ||
Dorr J, Ohlraun S, Skarabis H, Paul F. Efficacy of vitamin D supplementation in multiple sclerosis (EVIDIMS Trial): study protocol for a randomized controlled trial. Trials. 2012;13:15. | ||
Kanwar JR, Kanwar RK, Wang D, Krissansen GW. Prevention of a chronic progressive form of experimental autoimmune encephalomyelitis by an antibody against mucosal addressin cell adhesion molecule-1, given early in the course of disease progression. Immunol Cell Biol. 2000;78(6):641–645. | ||
Kanwar JR, Harrison JE, Wang D, et al. Beta7 integrins contribute to demyelinating disease of the central nervous system. J Neuroimmunol. 2000;103(2):146–152. | ||
Pezeshki MZ, Zarrintan S, Zarrintan MH. Helicobacter pylori nanoparticles as a potential treatment of conventional multiple sclerosis. Med Hypotheses. 2008;70(6):1223. | ||
Warnke C, Meyer zu Horste G, Hartung HP, Stuve O, Kieseier BC. Review of teriflunomide and its potential in the treatment of multiple sclerosis. Neuropsychiatr Dis Treat. 2009;5:333–340. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.