Back to Journals » Drug Design, Development and Therapy » Volume 17
Network Pharmacology and Molecular Docking Reveal the Mechanism of Isodon ternifolius (D. Don) Kudo Against Liver Fibrosis
Authors Deng J , Qin L , Zhou Z
Received 14 April 2023
Accepted for publication 18 July 2023
Published 7 August 2023 Volume 2023:17 Pages 2335—2351
DOI https://doi.org/10.2147/DDDT.S412818
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Manfred Ogris
Jiasheng Deng,1,* Le Qin,2,* Zhipin Zhou3,*
1School of Pharmacy, Guangxi University of Chinese Medicine, Nanning, Guangxi, People’s Republic of China; 2Department of Pharmacy, Affiliated Hospital of Youjiang Medical University for Nationalities, Baise, Guangxi, People’s Republic of China; 3Department of Pharmacy, Liuzhou People’s Hospital, Liuzhou, Guangxi, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Zhipin Zhou, Department of Pharmacy, Liuzhou People’s Hospital, 8 Wenchang Road, Cheng Zhong District, Liuzhou, Guangxi, People’s Republic of China, Tel +86 − 18776921947, Email [email protected]
Aim: Many studies have demonstrated the hepatoprotective or anti-fibrotic effects of Isodon ternifolius, but its pharmacological basis and mechanism remain unclear. In this study, we used in vitro models to validate the predicted results and revealed the potential mechanism of action and active ingredients through network pharmacology methods and molecular docking.
Methods: The chemical components of Isodon ternifolius were identified by literatures. Potential targets of Isodon ternifolius were predicted by Swiss Target Prediction. The disease targets were collected through the databases of Gene Card. Common targets of Isodon ternifolius and liver fibrosis were obtained by online tool Venny 2.1. PPI protein interaction network was obtained using String database, and target protein interaction network was drawn using Cytoscape software. Signaling pathway enrichment analysis was performed on drug-disease targets with of DAVID database.
Results: Twenty-one potential active ingredients and 298 potential targets were predicted by Swiss Target Prediction platform. Ninety pathways related to liver fibrosis were obtained by KEGG enrichment. The TLR4, MAPK and PI3K-Akt signaling pathways are mostly associated with liver fibrosis. Molecular docking techniques were used to validate the core target proteins TNF, Akt1, MAPK1, EGFR and TLR4 binding to the ingredients of Isodon ternifolius, which showed that a multitude of ingredients of Isodon ternifolius were able to bind to the above target proteins, especially 2α-hydroxy oleanolic acid and (-)-Lambertic acid. Our experimental validation results showed that Isodon ternifolius inhibited the activation of PI3K-Akt and ERK1/2 signaling pathways.
Conclusion: Through a network pharmacology approach and in vitro cell assay, we predicted and validated the active compounds of Isodon ternifolius and its potential targets for LF treatment. The results suggest that the mechanism of Isodon ternifolius treating LF by inhibiting angiogenesis may be related to the ERK1/2 and PI3K/Akt signaling pathways.
Keywords: Chinese medicine, Isodon ternifolius (D. Don), network pharmacology, molecular docking, signaling pathway, liver fibrosis
Introduction
Liver fibrosis (LF) is a reversible damage repair response, which is characterized by the accumulation of extracellular matrix (ECM). If the injury was acute or self-limiting, these changes were transient, and the liver structure could return to its normal composition. However, if the insult is sustained, the liver parenchyma is replaced by fibrotic scar tissue, a process in which ECM and MMPs have recognized key functions.1 Chronic liver diseases (CLDs) are characterized by long-term chronic hepatic parenchymal cell injury, a persistent inflammation response and a sustained activation of LF and damage repair response, so LF is present in most chronic liver diseases.2 The prevalence and mortality of CLDs could be comparable to major publish healthy issue related to common chronic diseases, such as diabetes, tuberculosis, and cardiovascular diseases. Previous studies have shown that 844 million people worldwide suffer from CLDs, with 2 million deaths per year.3 Currently, curing LF remains a huge challenge globally, as drug treatment strategies can only slow down the process of LF but not cure it. Therefore, it is necessary to explore drugs for the treatment of LF.
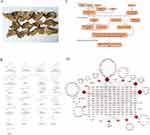
Traditional Chinese medicine (TCM) has a long history of preventing and treating diseases. TCM has curative effects in the treatment of liver diseases, especially in the field of anti-fibrosis, which has significant advantages and great development value. Isodon ternifolius is also known as the San Jie Mei in the Chinese folk (Figure 1A). The genus of Isodon ternifolius is Lamiaceae, which is sweet, slightly bitter, and cool. It has the effects of clearing heat and detoxification, promoting dampness and soothing the liver. It is used for treating enteritis, acute and chronic hepatitis and early cirrhosis of the liver.4 Chinese patent medicine containing Isodon ternifolius, “Fufang sanyexiangchacai Pian”, was used to treat acute and chronic hepatitis B.5,6 Previous studies have found that Isodon ternifolius has anti-LF effect, the mechanism may be through inhibiting the activation of TLR4/NF-κB signaling pathway,7,8 but its anti-fibrosis mechanism still needs further study. In this study, we used network pharmacology and molecular docking technology to reveal the mechanism and potential active ingredients of Isodon ternifolius against LF, and verified it through in vitro experiments. The workflow is shown in Figure 1C. The extract of Isodon ternifolius could inhibit the formation of blood vessels, possibly by inhibiting ERK1/ERK2 signaling pathway and the PI3K/Akt signaling pathway, inhibiting angiogenesis with inhibiting the progression of LF. Our results provide molecular insights into the mechanism of Chinese medicine Isodon ternifolius in the treatment of LF and will inform further development of anti-LF drugs from Isodon ternifolius.
Materials and Methods
The Source of Chemical Constituents and the Prediction of Drug Targets in Isodon ternifolius
The active compounds in Isodon ternifolius were collected from TCMSP (Traditional Chinese Medicine Database and Analysis Platform) (https://tcmsp-e.com/), ETCM (The Encyclopedia of Traditional Chinese Medicine) (http://www.tcmip.cn/ETCM/), and TCMID (Traditional Chinese Medicines Integrated Database) (http://47.100.169.139:8000/tcmid/). If there is no relevant data, we have collected the ingredients of Isodon ternifolius through literature search. Compound structures were drawn using ChemBioDraw Ultra 14.0, saved in the mol2 format and converted into 3D structures by ChemBio3D Ultra 14.0, and saved mol2 format file after minimizing MM2 energy. The corresponding targets of the putative compounds were searched in the TCMSP and the Swiss Target Prediction database (STP) (http://swisstargetprediction.ch/) get the drug target genes.
Target Genes of LF
GenCards integrates gene-centric data from 150 network sources, including genomics, transcriptomics, proteomics, genetics, clinical and functional information. Retrieve the disease “Liver Fibrosis” -related genes in the GenCard database (https://www.genecards.org/), download and save them.
Compound-Disease Target Network
The online tool Venny 2.1 (https://bioinfogp.cnb.csic.es/tools/venny/) was used to draw Venn diagrams to obtain target genes of compound-disease intersection. Finally, the visualization software Cytoscape 3.7.1 was used to construct the compound-disease target network.
Construction of Protein Protein Interaction (PPI) Network and Core Target Screening
To further identify the potential Isodon ternifolius therapeutic targets in LF. These targets were entered into the String Version database (https://string-db.org/) to obtain the protein–protein interaction (PPI) diagram. The search was limited to human species, the protein interaction confidence score threshold was set to 0.4, and disconnected nodes were hidden in the network representation. Finally, the PPI network is constructed and the results were exported and saved as files. Subsequently, the PPI network was visualized using Cytoscape Version 3.8.0 software to obtain the PPI diagram between the active components and LF-associated proteins.
GO (Gene Ontology) and KEGG (Kyoto Encyclopedia of Genes and Genomes) Enrichment Analysis and Construction of Core Target Pathways
The compound of Isodon ternifolius-LF common target genes were enriched by GO function and analyzed by KEGG pathway through the DAVID6.8 (https://david.ncifcrf.gov/) biological information annotation database. GO enrichment analysis includes Molecular Function (MF), Cellular Component (CC), and Biological Process (BC). P.Value and FDR have been used to assess the correlation and set the parameters P.Value <0.05 and FDR <0.05. The core target was imported into KEGG enrichment, and the relevant enrichment pathway was obtained through the website (https://www.genome.jp/kegg/tool/map_pathway1.html).
Molecular Docking
Autodock 4.2 is an automated program for predicting the interaction between ligands and biological macromolecular targets, which is used in drug research and development.9 We obtained the main enrichment pathways through enrichment analysis of the KEGG pathway and used Autodock 4.2 to verify the molecular docking of the core targets related to the middle value of the protein interaction network with the chemical components of Isodon ternifolius.
Acquisition and Processing of Target Protein
The 3D structure of candidate target is downloaded in the PDB protein database (https://www.rcsb.org/), and the result was saved in the pdb format, and then the visualization software PyMOL (TM) Molecular Graphics System, Version 2.4, was used to remove the water in the target protein molecule and separation of small molecules.
Docking Steps and Result Evaluation
(1) Ligand and receptor file preparation before docking: Small-molecule ligand compounds and core proteins are imported into AutoDockTools (v1.5.6) for hydrogenation, charge calculation, charge distribution and save as “pdbq”; (2) Grid file preparation: After opening the Grid option to import the receptor and ligand, select the Grid Box, adjust the center of the box to wrap the macromolecular protein, and save the output as “gpf”; (3) Parameter file preparation: Open Docking to import receptor and ligand files, set the parameters to default values, and operate as follows: Docking/Search Parameters/Genetic Algorithm Parameters, Docking/Docking Parameters, Output/Genetic Algorithm output is saved as “dpf”; (4) Put the prepared protein files, compound files and corresponding folders into the corresponding folders, and use AutoDock (v1.5.6) for molecular docking; (5) Evaluation of results: The docking possibility and stability are evaluated by the spatial position and binding energy after docking. It is generally believed that the lower the energy of the conformational stability of the ligand and the receptor, the greater the possibility of interaction. When the binding energy ≤ − 5.0 kcal/mol is taken as the screening criterion.10
Medicinal Materials Preparation
The dried roots of Isodon ternifolius were purchased from Guangxi Xianzhu Chinese Medicine Technology Company (20190501, Guangxi Province, China) in October 2019. About 6 kg of the rhizome of Isodon ternifolius, add 10 times the amount of water to soak for 30 minutes, then boiled for 60 minutes. Gauze was used to filter the herb residue, and the filtrate was collected. The above process was repeated three times. About 95% ethanol was used for extraction. After standing for 24 hours, the sediment was filtered off, and the filtrate was collected. The filtrate was concentrated into an extract and refrigerated at −80°C. The Isodon ternifolius extract was prepared with DMEM solution and used after filtration.
Cell Culture
Human umbilical vein endothelial cells (HUVECs) were Cell Bank of Type Culture Collection of Chinese Academy of Sciences (Shanghai, China). The HUVECs were cultured in DMEM complete medium (GIBCO, cat # 11965092) containing 10% fetal bovine serum FBS (GIBCO, cat # 10099–141) in 5% CO2, 37 °C, 100% saturated humidity CO2 incubator.
MTT Assay
The HUVECs were seeded in 96 well plates with 1×104 cells/well and cultured in DMEM medium containing different concentrations (2ug/mL, 5ug/mL, 10ug/mL, 40ug/mL, 75ug/mL) of Isodon ternifolius extract for 24 h. Each group was repeated three times. MTT cell proliferation and cytotoxicity kit (China, Solarbio, cat# M1020) was used to detect cell viability and proliferation inhibition rate. After 24 h of culture, 20 μL of MTT (5 mg/mL) was added to each well. After 4 h culture, the supernatant was centrifuged, and 150 μL of DMSO was added to each well to dissolve the crystalline particles. The absorbance was measured at 570nm.
Scratch Healing Assay
Scratch healing assay is used to assess changes in migration of HUVECs. Different groups of cells were seeded into 6-well plates. After the cells have grown to full, the central portion of the cells was removed by scoring the central area of cell growth with tip, and then the cells continue to be cultured with a culture medium containing different concentrations of Isodon ternifolius. After 24 hours, the cells were observed under microscope whether it migrated to the central scratch area and photographed for recording. Image J software was used to analyze migration and calculate migration rate.
Tube Formation Assay
To observe microvasculature, HUVECs treated by different concentration were seeded in 96-well plates with matrix adhesive with substrate gel (Solarbio, cat# 356234) and cultured at 37°C in a 5% CO2 incubator. After 6 hours of culture, the vascular network was imaged by an inverted microscope at 10×, and vascular network was quantified by Image J’s package with angiogenesis analyzer.
Quantitative Real-Time Polymerase Chain Reaction (PCR)
Total RNA was extracted by TriQuick Reagent Total RNA Extraction Kit (Solarbio, R1100) and stored at-80 °C. RNA was reverse transcribed to cDNA using PrimeScript™ RT reagent Kit with gDNA Eraser (Perfect Real Time) (Takara, RR036A) according to the instructions and stored at −80°C. The predicted target genes EGFR, VEGFR-1, ERK1, ERK2, Akt mRNA were amplified by VeritiTM 96-Well Thermal Cycler Applied Biosystems® in Real-Time PCR System (Agilent, AriaMx). PCR conditions: protein denaturation 95 °C for 30s, 40 cycles, each cycle 95 °C for 5s, 60 °C for 10s, 72 °C for 30s. β-ACTIN was used as the internal reference to correct the target Ct value, and 2−ΔΔ was used to calculate the relative expression of mRNA. All primers were synthesized by Sangon Biotech in Shanghai (Table 1).
 |
Table 1 List of Primer Sequences Used for qRT-PCR Analysis in This Study |
Statistics Analysis
The results were expressed as means ± S.E.M, and data from each group were compared by one-way ANOVA and Student-Newman-Keuls test. The results were considered statistically significant when the P value was less than 0.05.
Results
Compound of Isodon ternifolius
Ethyl acetate extraction and n-butanol extraction from the roots of Isodon ternifolius were isolated and identified 25 compounds,11 following 3-carboxy formate-6,7-dihydroxy-1-(3’,4’-dihydroxyphenyl)-naphthalene (1); 3-carboxy-6,7-dihydroxy-1-(3′,4′-dihydroxyphenyl)-naphthalene (2); 1-acetatexyl-2e,6e-dipiperonyl-3,7-dioxabcyclo[3,3,0]-octane (3); 2-(3,4,5-trimethoxyphenyl)-6-(4-hydroxy-3-methoxyphenyl)-3,7-dioxabicyclo[3.3.0]- octane (4); syringylglycerol-β-syringaresinol ether 4’,4- m-di-O-β-D-glucopyranoside (5); clemastanin A (6); 3-(3,4-dihydroxyphenyl) acrylic acid 1-(3,4-dihydroxy-phenyl)-2-methoxycarbonyl- ethyl ester (7); oxyneolignans A (8); 19-hydroxytotarol (9); (-)-lambertic acid (10); isodoternifolin B (11); longikaurin A (12); maoyecrystal F (13); 2α-hydroxy oleanolic acid (14); tormentic acid (15); myricetin 3-neohesperidoside (16); 3,4-dimethoxy-1-hydroxylphenyl-O-β-D–glucopyranoside (17); 2,6-dimethoxy-p-hydroquinone1-O-β-D-glucopyranoside (18); 3,4,α-trihydroxyl-methyl phenylpropionate (19); erythro-l-c-syringyl-glycerol (20); E-P-hydroxyl ethyl cinnamate (21); caffeic acid (22); 3, 4-dihydroxyphenacyl alco-hol (23); protocatechuic acid (24); Syringic acid (25) (Figure 1B). Additionally, the interaction network between compound of Isodon ternifolius and liver fibrosis target protein was discovered through network pharmacology analysis (Figure 1D).
Network Construction of Prediction Targets and Disease Targets of Isodon ternifolius
The SwissTargetPrediction platform database was used to predict the target of Isodon ternifolius. Compounds 13, 19, 20, and 23 were eliminated due to no prediction results, and the remaining 21 compounds were predicted to have 395 drug targets (Probability > 0, delete duplicate targets). Using “liver fibrosis” as the search term, GeneCards retrieved 6794 LF target genes. We intersected 395 drug targets with 6794 liver fibrosis-related targets to obtain 298 drug-disease common target genes. PPI network which contains 298 nodes and 626 edges was primitively constructed by STRING database, in which nodes represent proteins and edges stand for protein–protein interactions (Figure 2A).
Target Protein Interaction Network Construction and Core Target Screening
After the 298 predicted targets of Isodon ternifolius for the treatment of LF were imported into the STRING database (https://string-db.org/), the protein interaction network was constructed. In order to further visualize and analyze the protein interactions, the retrieved PPI data were presently imported into Cytoscape 3.6.2 to construct a new PPI network, which also includes 298 nodes and 626edges (Figure 2B). Subsequently, in the network in Figure 2B, the first 25 important target protein interaction networks were screened out (Figure 2C). Non-interacting targets were concealed, and other parameters are the default values. Finally, the results saved “.png” and “TSV” format (Figure 2).
Screening GO Function Enrichment and KEGG Signal Pathway Analysis
In order to explain the possible role and enriched pathways of the candidate targets of anti-LF of Isodon ternifolius, these targets were used for GO function enrichment and KEGG pathway analysis through the DAVID6.8 database.
The GO enrichment analysis yielded a total of 219 data results. Twenty-three results (Figure 3C) are used to describe the location of gene products in cellular components (CC), such as endoplasmic reticulum, mitochondria or nucleus. Thirty-three results (Figure 3B) were used to describe the functions of individual gene products in molecular functions (MF), such as binding activity and catalytic activity. One hundred and sixty-four results (Figure 3A) are used to describe an orderly biological process (BP) with multiple steps, such as cell growth, proliferation, apoptosis and signal transduction.
The KEGG pathways were applied to explore the functions and signaling pathways, the top 25 significant enrichment potential pathways with the most gene counts were presented in a bar plot diagram (Figure 3D), showing that Isodon ternifolius plays an important role in the treatment of LF through multiple targets and multiple pathways. Furthermore, the pathways with the top 5 highest gene counts were PI3K-Akt signaling pathway, MAPK signaling pathway, Viral carcinogenesis, TNF signaling pathway, and Toxoplasmosis, which would be the key pathways in the effect of Isodon ternifolius against LF. Among them, PI3K-Akt and MAPK signaling pathway, associated with the highest number of genes, may be the most important potential pathway (Figure 4B). In a word, we discovered that several target genes were involved in multiple pathways; meanwhile, these results demonstrated that Isodon ternifolius inpacts LF through multiple pathways, multiple targets, and total cooperation.
Figure 4 Continued.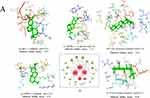
Molecular Docking Analysis
Due to the drug targets of Isodon ternifolius are mainly enriched in the MAPK signaling pathway and the PI3K-Akt signaling pathway (Figure 3D), Network pharmacological analysis revealed that five core proteins were highly expressed in these two pathways, so the core targets of this study are TNF (PDB ID:1EXT), Akt1 (PDB ID:4EKL), MAPK1 (PDB ID:6GLD), EGFR (PDB ID:5d41), TLR4 (PDB ID: 2z65), which were conducted molecular docking with two candidate bioactive compounds [(-)-lambertic acid, 2α-hydroxy oleanolic acid], whose results were visualized by LigPlus (3D) as shown in Figure 4A. The results revealed that Akt1 interacted with (-)-lambertic acid by forming hydrogen bonds with ILE-449 (length: 2.1Å), ILE-449 (2.5Å), ILE-180 (2.4Å), TYR-152 (2.6Å), GLU-151 (2.6Å), LYS-170 (2.4Å). There are also 4 hydrogen bonds, TYR-113 (2.5Å), PRO-152 (3.2Å), TYR-193 (2.5Å), and THR-190 (2.6Å) were predicted between MAPK1 and (-)-lambertic acid. Similarly, (-)-lambertic acid was predicted to dock into the binding pocket of EGFR via hydrogen bonds PRO-741 (2.7Å), PRO-749 (2.5Å), PRO-741 (2.1Å), VAL-738 (2.6Å), LYS-716 (1.9Å), LYS-728 (2.7Å), and LEU-792 (2.4Å). TLR4 bound to 2α-hydroxy oleanolic acid by forming hydrogen bonds with PHE-151 (2.6Å), VAL-135 (3.2Å), CYS-133 (2.5Å), LE-80 (2.8Å), PHE-121 (3.2Å), ILE-153 (2.7Å), ILE-52 (2.7Å) and ILE-32 (2.4Å). Lastly, 2α-hydroxy oleanolic acid was predicted to dock into the pocket of TNF via hydrogen bonds with ARG-104 (2.7Å), ILE-85 (2.9Å), ARG-99 (2.7Å), LYS-100 (2.6Å), GLN-102 (2.8Å), ASN-101 (2.6Å) and CYS-129 (2.3Å).
Therefore, there exists an interplay between these compounds and protein targets with hydrogen bonds. The binding energy was usually calculated to evaluate the affinity degree of ingredients with protein targets. It is generally accepted that binding energy which is less than −5.0 kcal/mol, fine and robust binding activity between the ligand and the receptor, respectively. The results of docking binding energy in Figure 4A revealed that these ingredients could bind well to the active sites of protein targets. Among them, the docking of Akt1 and (-)-lambertic acid had the lowest binding energy (−9.05 kcal/mol), the docking of TNF and 2α-hydroxy oleanolic acid had the highest binding energy (−6.06kcal/mol), and the average binding energy was −6.96 kcal/mol, mirroring a good or even strong binding activity in the molecular docking between candidate compounds and candidate protein targets. Taken together, these representative compounds of Isodon ternifolius could bind well with above core targets of UC (Akt1, TNF, MAPK1, TLR4, EGFR), all of which might play key roles in the treatment of LF.
Effects of Isodon ternifolius on Proliferation of HUVCs in vitro
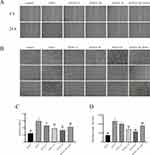
In aim of evaluating the effect of Isodon ternifolius extract on the proliferation viability of HUVECs, we experimented with a variety of concentrations of Isodon ternifolius extract (2μg/mL, 5μg/mL, 15μg/mL, 45μg/mL, 75μg/mL) to treat HUVECs. The MTT results showed that IC50 was 11.91μg/mL (Figure 5).
 |
Figure 5 Effect of different concentration of Isodon ternifolius on HUVECs activity. Note: SYXCC represents Isodon ternifolius. |
Isodon ternifolius Extract Inhibits Migration and Angiogenesis of HUVCs in vitro
Many evidences show that angiogenesis is involved in the development of liver fibrosis. Hypoxia and inflammation are the main triggers of angiogenesis. Fibroblasts were located at the intersection between angiogenesis and fibrosis and play a key role in angiogenesis.12,13 In the pharmacology network, we found that the mechanism of the anti-fibrotic of Isodon ternifolius may be related to angiogenesis. In vitro, we investigated whether Isodon ternifolius has an inhibitory effect on angiogenesis. As shown in Figure 6A–D, compared with the VEGF group, as the dose groups of SYXCC increase, the inhibitory effect on angiogenesis and migration becomes more pronounced. Therefore, the scratch healing and tube formation assays demonstrate that Isodon ternifolius extracts inhibited the migration of HUVECs in a dose-dependent manner and significantly inhibited the formation of blood vessels.
Isodon ternifolius Extract Inhibits Activation of PI3K/Akt and ERK1/2 Signaling Pathways in vitro
We have predicted that multiple ingredients of Isodon ternifolius were involved in the regulation of PI3K/Akt and ERK1/ERK2 signaling pathways via bioinformatics analysis. It has been demonstrated that ERK1/2 and PI3K/Akt were important kinase cascade signaling pathways in vivo, regulating HSCs of activation, proliferation, differentiation or intrahepatic vessels, resulting in the progression of liver fibrosis.14–19 Thus, to further investigate the underlying mechanism of how Isodon ternifolius exhibits its anti-LF activities, the mRNA levels of Akt1, ERK1, ERK2, EGFR and VEFGR-2 were examined. As shown in Figure 7A–E, the expression of the above mRNA decreased with the increase of Isodon ternifolius concentration. PCR suggested that Isodon ternifolius significantly inhibited the activation of PI3K/Akt and ERK1/2 signaling pathways and reduced ERFG, VEGFR-2 expression in vitro, which further verified the network pharmacology results of the PI3K/Akt signaling pathway and ERK1/2 signaling pathways in Isodon ternifolius for LF.
Discussion
Hepatic fibrosis is a pathological recovery process caused by various chronic liver damage factors. Its pathological characteristics are the imbalance of synthesis and degradation of extracellular matrix, which leads to excessive deposition of ECM in the liver, and eventually leads to abnormal hyperplasia of hepatic connective tissue and liver fibrosis. Angiogenesis has been found to play a crucial role in the progression of liver fibrosis, and hepatic angiogenesis occurs in chronic liver diseases (CLDs) characterised by inflammation and progressive fibrosis.20 Isodon ternifolius is a traditional Chinese medicine in southwest China, which was commonly used in early days to treat chronic liver diseases such as hepatitis B. Through research, we obtained 21 active ingredients of the Isodon ternifolius meeting the conditions at the same time, 395 potential targets, 298 targets overlapping with LF, involving 219 GO biological processes and 25 KEGG-related signal pathways. It revealed that the Isodon ternifolius treatment of LF has the characteristics of multiple components, multiple targets, and multiple pathways.
Network pharmacology found that the Isodon ternifolius through the MAPK pathway,21 TNF pathway,22 PI3K-Akt pathway,23 TLR4/NF-κB pathway,24 VEGF pathway,25 HIF-1,26 which are involved in the regulation of viral infection, inflammatory response, angiogenesis, etc., and initiate the progress of fibrosis. Our previous studies have emphasized that Isodon ternifolius has the effects of protecting the liver and anti-LF, could down-regulate the activation of the TLR4 signaling pathway, and reduce interleukin-1β (IL-1β), interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α).7,8 GO and KEGG results indicated that ERK1/2 and PI3K/Akt pathways were most related to liver fibrosis. As two signaling pathways involved in cell survival, differentiation, apoptosis and receiving signals transmitted by tyrosine kinase receptors. They were important kinase cascade signaling pathways while regulating the activation, proliferation, and differentiation of HSCs and intrahepatic angiogenesis. These results reveal a new mechanism by which Isodon ternifolius can also intervene in liver fibrosis by regulating angiogenesis.14–19 HSC is the core of the pathogenesis of LF, and plays an important role among liver inflammation, fibrosis and angiogenesis. Quiescent HSCs could be stimulated by a variety of growth factors, pro-inflammatory mediators produced by damaged resident cell types, and hypoxia to promote the formation of new blood vessels, which in turn could become a bridge between acute and chronic inflammation.27 Evidence implies that under clinical and experimental conditions, liver angiogenesis and fibrosis were closely related. Hypoxia, inflammation and HSC were considered to be the main causes of angiogenesis. Pro-fibrosis cells were located at the intersection between inflammation, angiogenesis, and fibrosis, and also play a key role in angiogenesis.12,13
ERK1/2 and PI3K/Akt were regulated by various cell growth factors, including VEGF, PDGF, EGF, etc,28 and produce fibrosis through a variety of mechanisms, including promoting inflammation, activated endothelial cells releasing fibrosis enhancing molecules and activated HSC,29,30 up-regulate α-SMA, collagen, integrin, fibronectin, IFN-α, IL-6, IL-10.25 Once the liver was damaged, HSCs autocrine or paracrine cell growth factors such as VEGF, PDGF, EGF, etc., which growth factors bind to the tyrosine kinase receptor on the cell membrane to transmit the activation signal to GTPase Ras, and then Ras is recruited to the plasma membrane and binds to Raf. Ras was the common upstream molecule of ERK1/2 and PI3K/Akt signaling pathways.31 The activation signal was phosphorylated through the Ras/Raf/MEK/ERK cascade, and then also phosphorylates the phosphatidylinositol 3-kinase-Akt-p70 S6 kinase (PI3K-Akt-p70 S6k) cascade, which phosphorylates its substrate and expresses various transcription factors involved in cell proliferation and differentiation, secrete pro-fibrosis factors and angiogenesis in the progress of LF.15,32 During LF progression, the PI3K/Akt signaling pathway regulated the synthesis of MMP and the degradation of extracellular matrix through the activation of HSCs. Type I collagen was the main component of ECM and the main collagen secreted by activated HSC. PI3K regulated the transcriptional expression of collagen and plays an important regulatory role in the synthesis of collagen.33,34 Akt, also known as protein kinase B (PKB), was a serine/threonine protein kinase that plays an important role in the development of alcoholic hepatitis, including promoting cell proliferation, migration, and transcription, while impairing apoptosis. It was found that Akt1 and Akt2 in the Akt family are important in the pathological development of alcoholic hepatitis, and the mechanism is to regulate the occurrence of inflammatory response and fibrosis in the progression of alcoholic hepatitis.35 PI3K inhibitors could inhibit the activation of ERK and the expression of c-fos.36 PIK3R1 or Akt3 mice or HSC cells were knocked out, the expression of α-SMA and collagen I are reduced, and the degree of LF is reduced.19 As the main signal transduction pathway involved in the phenotypic response of liver myofibroblasts, the ERK signaling pathway plays an important role in the progression of LF.15 When Raf kinase inhibitory protein (RKIP) was blocked in rat liver tissue and primary HSCs, the ERK signaling pathway was activated to promote the production of α-SMA, MMP-1, MMP-2 and collagen, aggravate LF.37 In addition, It has been reported that when fed with choline deficiency and supplemented with ethionine diet, the degree of LF in ERK knockout mice was relieved, the expression of fibrosis maker α-SMA protein was reduced, and the percentage of proliferation and activated T cells was reduced.16 Epidermal growth factor receptor (EGFR) was a transmembrane receptor tyrosine kinase that was activated by variety of signaling pathways to mediate cell proliferation and differentiation.38 EGFR was important in the progression of LF,39,40 and EGFR was up-regulated in LF. When EGFR was blocked, it could alleviate the progress of LF by reducing lipid deposition, anti-oxidative stress, inhibiting HSCs activation and the release of inflammatory factors.39–41 VEGF was the most important pro-angiogenic cytokine and plays a central role in angiogenesis because it was related to all the steps of angiogenesis: increasing vascular permeability, inducing endothelial cell proliferation, and regulating the diameter of new blood vessel lumen.42 VEGF signaling pathway was blocked, it could effectively inhibit angiogenesis.43,44 Besides VEGF plays an important role in the progression of LF, promoting the activation, proliferation of HSC and collagen production.25 In the liver, VEGF and its receptor tyrosine kinases VEGFR1 (Flt-1) and VEGFR2 (Flt-2) were not only expressed in the endothelial cells and hepatocytes of the liver but also expressed in mesenchymal cells, especially in activation of HSC.45,46
In the drug-disease network, we found 3-carboxy formate-6,7-dihydroxy-1-(3’,4’-dihydroxyphenyl)-naphthalene (1), (-)-Lambertic acid (10), 2α-hydroxy oleanolic acid (14), tormentic acid (15), E-P-hydroxyl ethyl cinnamate (21), and they play an important regulatory role. Some compounds have been reported to have therapeutic effects on liver disease, such as ursolic acid and oleanolic acid. Oleanolic acid and ursolic acid were classified as pentacyclic triterpene acid compounds, which have been reported to treat acute and chronic liver diseases. Its mechanism includes anti-oxidative stress,47–50 inhibition of HSC activation and reduction of the production of pro-fibrotic factors,51,52 reduction of blood lipids, inhibition of liver lipid synthesis and promotion of lipid metabolism,53 and improvement of intestinal flora.54,55 Tormentic acid could prevent LF by blocking PI3K/Akt/mTOR and NF-κB signaling pathways to inhibit HSC activation.56 19-hydroxytotarol could antioxidant by inhibiting mitochondrial lipid peroxidation.57 3-carboxy formate-6,7-dihydroxy-1-(3’,4’-dihydroxyphenyl)-naphthalene (1) was first discovered as a component of Isodon ternifolius, which could relieve inflammation and reduce LPS-induced RAW264.7 cells secrete PGE2 and TNF-α.58 Network pharmacology is based on the theory of systems biology. It integrates the biological system network and the drug action network, and then analyzes the regulatory role of the drug in the disease network, emphasizing the overall regulatory role. The research method of network pharmacology is consistent with the multi-target and multi-path mechanism of TCM. Among them, 1-acetylacetonyl-2e-6e-dipiperonyl-3, 7-dioxabicyclo[3, 3, 0]-octane (3) has anti-inflammatory activity.11 Longikaurin A (12) activates the ROS/JNK/c-Jun signaling pathway by inhibiting the expression of Skp2, resulting in the induction of cycle arrest and apoptosis of liver cancer cells.59 It has been reported that (-)-lambertic acid exists in Lambertae plants such as Isodon macrophylla,60 Isodon amethystoides,61 which have anti-tumor and antioxidant effects.61,62 Particularly, (-)-lambertic acid was docked by multiple core target proteins, indicating that (-)-lambertic acid may be a potential active ingredient in Isodon ternifolius. The molecular docking results demonstrated that (-)-Lambertic acid (10), longikaurin A (12), 2α-hydroxy oleanolic acid (14), tormentic acid (15), may act on TLR4, Akt, MAPK, EGFR and other targets. These may play a role in the treatment of LF, but the specific role needs further study. Through network pharmacological analysis and in vitro experiments, it has been confirmed that the mechanism of Isodon ternifolius treating LF by inhibiting angiogenesis may be related to the ERK1/2 and PI3K/Akt signaling pathways.
Conclusion
Network pharmacology and molecular docking technology explored the mechanism of anti-hepatic fibrosis and potential active ingredients of Isodon ternifolius. The experiment verified that the anti-LF mechanism of the Isodon ternifolius might be the inhibition of intrahepatic blood vessels. We concluded that (1) Isodon ternifolius multi-target regulation PI3K/Akt, ERK1/ERK2 signaling pathways, inhibits the synthesis and release of inflammatory cytokines, fibrosis-related factors and other cytokines, and inhibits the formation of blood vessels to achieve the effect of treating LF. (2) The most potential activities of Isodon ternifolius include 19-hydroxytotarol, (-)-lambertic acid, longikaurin A, 2α-hydroxy oleanolic acid, tormentic acid, which are easily combined with TLR4, TNF, MAPK, EGFR and other core targets were docked.
Ethics Approval and Informed Consent
All research involving human data in this study has been approved by the local Institutional Review Board (IRB). This use of the database does not require the consent of the owner. The databases on humans used in this paper are publicly available and allow for unrestricted re-use through an open licence. This study was approved by the Ethics Committee of Liuzhou People’s Hospital, Guangxi Province, China. Permit numbers: KY2023-022-01
Acknowledgments
This work was supported by grants of the National Natural Scientific Foundation of China (No.81760751), Guangxi Provincial Natural Scientific Foundation (No. 2021GXNSFAA075020), and Science and Technology Program of Liuzhou province (2020NBAB0815).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Hernandez-Gea V, Friedman SL. Pathogenesis of liver fibrosis. Annu Rev Pathol. 2011;6(1):425–456. doi:10.1146/annurev-pathol-011110-130246
2. Parola M, Pinzani M. Liver fibrosis: pathophysiology, pathogenetic targets and clinical issues. Mol Aspects Med. 2019;65:37–55. doi:10.1016/j.mam.2018.09.002
3. Marcellin P, Kutala BK. Liver diseases: a major, neglected global public health problem requiring urgent actions and large-scale screening. Liver Int. 2018;38(Suppl 1):2–6. doi:10.1111/liv.13682
4. Clemants S, Ma J. A history and overview of the Flora Reipublicae Popularis Sinicae (FRPS, Flora of China, Chinese edition, 1959–2004). Taxon. 2006;55(2). doi:10.2307/25065592
5. Liu M, Wang WG, Sun HD, Pu JX. Diterpenoids from Isodon species: an update. Nat Prod Rep. 2017;34(9):1090–1140. doi:10.1039/c7np00027h
6. Rui LI, Liansan Z, Huici X. Observation on curative effect of treatment of 98 cases of chronic hepatitis B with compound rabdosia ternifolia tablets. Sichuan Med J. 2008;29(4):397–398. doi:10.16252/j.cnki.issn1004-0501-2008.04.004
7. Zhipin Z, Xiquan X, Qin W, Honghan Q, Xiaoxue YE. Effect of Herba Isodonis Ternifoliae on toll -like receptor 4 pathway in rats with hepatic fibrosis induced by carbon tetrachloride. Trad Chin Drug Res Clin Pharmacol. 2017;28(2):183–186+262. doi:10.19378/j.issn.1003-9783.2017.02.008
8. Zhipin Z, Xiquan X, Qin W, Honghang Q, Xiaoxue Y. The Isodonis ternifoliae protects rat against hepatic fibrosis induced by CCl4 via down-regulation of NF-κB/IL-6 pathway. Drug Des Devel Ther. 2016;32(6):150–153. doi:10.13412/j.cnki.zyyl.2016.06.040
9. Harris R, Olson AJ, Goodsell DS. Automated prediction of ligand-binding sites in proteins. Proteins. 2008;70(4):1506–1517. doi:10.1002/prot.21645
10. Du A, Zheng R, Disoma C, et al. Epigallocatechin-3-gallate, an active ingredient of Traditional Chinese Medicines, inhibits the 3CLpro activity of SARS-CoV-2. Int J Biol Macromol. 2021;176:1–12. doi:10.1016/j.ijbiomac.2021.02.012
11. Ke W. The Constituents and anti-inflammatory activity of the roots of Isodon ternifolia (D.Don) Kudo; 2018.
12. Li H. Angiogenesis in the progression from liver fibrosis to cirrhosis and hepatocelluar carcinoma. Expert Rev Gastroenterol Hepatol. 2021;15(3):217–233. doi:10.1080/17474124.2021.1842732
13. Lefere S, Devisscher L, Geerts A. Angiogenesis in the progression of non-alcoholic fatty liver disease. Acta Gastroenterol Belg. 2020;83(2):301–307.
14. Lavoie H, Gagnon J, Therrien M. ERK signalling: a master regulator of cell behaviour, life and fate. Nat Rev Mol Cell Biol. 2020;21(10):607–632. doi:10.1038/s41580-020-0255-7
15. Foglia B, Cannito S, Bocca C, Parola M, Novo E. ERK pathway in activated, myofibroblast-like, hepatic stellate cells: a critical signaling crossroad sustaining liver fibrosis. Int J Mol Sci. 2019;20(11). doi:10.3390/ijms20112700
16. Jeng KS, Lu SJ, Wang CH, Chang CF. Liver fibrosis and inflammation under the control of ERK2. Int J Mol Sci. 2020;21(11). doi:10.3390/ijms21113796
17. Mannella P, Brinton RD. Estrogen receptor protein interaction with phosphatidylinositol 3-kinase leads to activation of phosphorylated Akt and extracellular signal-regulated kinase 1/2 in the same population of cortical neurons: a unified mechanism of estrogen action. J Neurosci. 2006;26(37):9439–9447. doi:10.1523/JNEUROSCI.1443-06.2006
18. Ge X, Guo R, Qiao Y, et al. The G protein-coupled receptor GPR30 mediates the nontranscriptional effect of estrogen on the activation of PI3K/Akt pathway in endometrial cancer cells. Int J Gynecol Cancer. 2013;23(1):52–59. doi:10.1097/IGC.0b013e31827912b8
19. Wang J, Chu ES, Chen HY, et al. microRNA-29b prevents liver fibrosis by attenuating hepatic stellate cell activation and inducing apoptosis through targeting PI3K/AKT pathway. Oncotarget. 2015;6(9):7325–7338. doi:10.18632/oncotarget.2621
20. Xu M, Xu HH, Lin Y, et al. LECT2, a ligand for tie1, plays a crucial role in liver fibrogenesis. Cell. 2019;178(6):1478–1492.e20. doi:10.1016/j.cell.2019.07.021
21. Shen X, Guo H, Xu J, Wang J. Inhibition of lncRNA HULC improves hepatic fibrosis and hepatocyte apoptosis by inhibiting the MAPK signaling pathway in rats with nonalcoholic fatty liver disease. J Cell Physiol. 2019;234(10):18169–18179. doi:10.1002/jcp.28450
22. Kalliolias GD, Ivashkiv LB. TNF biology, pathogenic mechanisms and emerging therapeutic strategies. Nat Rev Rheumatol. 2016;12(1):49–62. doi:10.1038/nrrheum.2015.169
23. Gong Z, Lin J, Zheng J, et al. Dahuang Zhechong pill attenuates CCl4-induced rat liver fibrosis via the PI3K-Akt signaling pathway. J Cell Biochem. 2020;121(2):1431–1440. doi:10.1002/jcb.29378
24. Lai L, Chen Y, Tian X, et al. Artesunate alleviates hepatic fibrosis induced by multiple pathogenic factors and inflammation through the inhibition of LPS/TLR4/NF-κB signaling pathway in rats. Eur J Pharmacol. 2015;765:234–241. doi:10.1016/j.ejphar.2015.08.040
25. Luo J, Liang Y, Kong F, et al. Vascular endothelial growth factor promotes the activation of hepatic stellate cells in chronic schistosomiasis. Immunol Cell Biol. 2017;95(4):399–407. doi:10.1038/icb.2016.109
26. Ju C, Colgan SP, Eltzschig HK. Hypoxia-inducible factors as molecular targets for liver diseases. J Mol Med. 2016;94(6):613–627. doi:10.1007/s00109-016-1408-1
27. Zadorozhna M, Di Gioia S, Conese M, Mangieri D. Neovascularization is a key feature of liver fibrosis progression: anti-angiogenesis as an innovative way of liver fibrosis treatment. Mol Biol Rep. 2020;47(3):2279–2288. doi:10.1007/s11033-020-05290-0
28. Karaman S, Leppänen VM, Alitalo K. Vascular endothelial growth factor signaling in development and disease. Development. 2018;145(14). doi:10.1242/dev.151019
29. Yoshiji H, Kuriyama S, Yoshii J, et al. Vascular endothelial growth factor and receptor interaction is a prerequisite for murine hepatic fibrogenesis. Gut. 2003;52(9):1347–1354. doi:10.1136/gut.52.9.1347
30. Sahin H, Borkham-Kamphorst E, Kuppe C, et al. Chemokine Cxcl9 attenuates liver fibrosis-associated angiogenesis in mice. Hepatology. 2012;55(5):1610–1619. doi:10.1002/hep.25545
31. Castellano E, Downward J. Role of RAS in the regulation of PI 3-kinase. Curr Top Microbiol Immunol. 2010;346:143–169. doi:10.1007/82_2010_56
32. Parsons CJ, Takashima M, Rippe RA. Molecular mechanisms of hepatic fibrogenesis. J Gastroenterol Hepatol. 2007;22(Suppl 1):S79–84. doi:10.1111/j.1440-1746.2006.04659.x
33. Son G, Hines IN, Lindquist J, Schrum LW, Rippe RA. Inhibition of phosphatidylinositol 3-kinase signaling in hepatic stellate cells blocks the progression of hepatic fibrosis. Hepatology. 2009;50(5):1512–1523. doi:10.1002/hep.23186
34. Reif S, Lang A, Lindquist JN, et al. The role of focal adhesion kinase-phosphatidylinositol 3-kinase-akt signaling in hepatic stellate cell proliferation and type I collagen expression. J Biol Chem. 2003;278(10):8083–8090. doi:10.1074/jbc.M212927200
35. Reyes-Gordillo K, Shah R, Arellanes-Robledo J, Cheng Y, Ibrahim J, Tuma PL. Akt1 and Akt2 isoforms play distinct roles in regulating the development of inflammation and fibrosis associated with alcoholic liver disease. Cells. 2019;8(11). doi:10.3390/cells8111337
36. Marra F, Pinzani M, DeFranco R, Laffi G, Gentilini P. Involvement of phosphatidylinositol 3-kinase in the activation of extracellular signal-regulated kinase by PDGF in hepatic stellate cells. FEBS Lett. 1995;376(3):141–145. doi:10.1016/0014-5793(95)01261-0
37. Huang Q, Liang C, Wei L, et al. Raf kinase inhibitory protein down-expression exacerbates hepatic fibrosis in vivo and in vitro. Cell Physiol Biochem. 2016;40(1–2):49–61. doi:10.1159/000452524
38. Komposch K, Sibilia M. EGFR Signaling in Liver Diseases. Int J Mol Sci. 2015;17(1). doi:10.3390/ijms17010030
39. Fuchs BC, Hoshida Y, Fujii T, et al. Epidermal growth factor receptor inhibition attenuates liver fibrosis and development of hepatocellular carcinoma. Hepatology. 2014;59(4):1577–1590. doi:10.1002/hep.26898
40. Liang D, Chen H, Zhao L, et al. Inhibition of EGFR attenuates fibrosis and stellate cell activation in diet-induced model of nonalcoholic fatty liver disease. Biochim Biophys Acta Mol Basis Dis. 2018;1864(1):133–142. doi:10.1016/j.bbadis.2017.10.016
41. Qian Y, Han J, Zhou L, et al. Inhibition of Epidermal Growth Factor Receptor (EGFR) reduces Lipopolysaccharide (LPS)-induced activation and inflammatory cytokines in hepatic stellate cells in vitro. Med Sci Monit. 2018;24:5533–5541. doi:10.12659/MSM.909901
42. Carmeliet P, Jain RK. Molecular mechanisms and clinical applications of angiogenesis. Nature. 2011;473(7347):298–307. doi:10.1038/nature10144
43. Ding Q, Tian XG, Li Y, Wang QZ, Zhang CQ. Carvedilol may attenuate liver cirrhosis by inhibiting angiogenesis through the VEGF-Src-ERK signaling pathway. World J Gastroenterol. 2015;21(32):9566–9576. doi:10.3748/wjg.v21.i32.9566
44. Park S, Kim JW, Kim JH, Lim CW, Kim B. Differential roles of angiogenesis in the induction of fibrogenesis and the resolution of fibrosis in liver. Biol Pharm Bull. 2015;38(7):980–985. doi:10.1248/bpb.b15-00325
45. Ankoma-Sey V, Matli M, Chang KB, et al. Coordinated induction of VEGF receptors in mesenchymal cell types during rat hepatic wound healing. Oncogene. 1998;17(1):115–121. doi:10.1038/sj.onc.1201912
46. Ishikawa K, Mochida S, Mashiba S, et al. Expressions of vascular endothelial growth factor in nonparenchymal as well as parenchymal cells in rat liver after necrosis. Biochem Biophys Res Commun. 1999;254(3):587–593. doi:10.1006/bbrc.1998.9984
47. He W, Shi F, Zhou ZW, et al. A bioinformatic and mechanistic study elicits the antifibrotic effect of ursolic acid through the attenuation of oxidative stress with the involvement of ERK, PI3K/Akt, and p38 MAPK signaling pathways in human hepatic stellate cells and rat liver. Drug Des Devel Ther. 2015;9:3989–4104. doi:10.2147/DDDT.S85426
48. Ma JQ, Ding J, Zhang L, Liu CM. Protective effects of ursolic acid in an experimental model of liver fibrosis through Nrf2/ARE pathway. Clin Res Hepatol Gastroenterol. 2015;39(2):188–197. doi:10.1016/j.clinre.2014.09.007
49. Yang Y, Zhao Z, Liu Y, Kang X, Zhang H, Meng M. Suppression of oxidative stress and improvement of liver functions in mice by ursolic acid via LKB1-AMP-activated protein kinase signaling. J Gastroenterol Hepatol. 2015;30(3):609–618. doi:10.1111/jgh.12723
50. Gan D, Zhang W, Huang C, et al. Ursolic acid ameliorates CCl4-induced liver fibrosis through the NOXs/ROS pathway. J Cell Physiol. 2018;233(10):6799–6813. doi:10.1002/jcp.26541
51. Xiang H, Han Y, Zhang Y, et al. A new oleanolic acid derivative against CCl₄-induced hepatic fibrosis in rats. Int J Mol Sci. 2017;18(3). doi:10.3390/ijms18030553
52. Yu SS, Chen B, Huang CK, et al. Ursolic acid suppresses TGF-β1-induced quiescent HSC activation and transformation by inhibiting NADPH oxidase expression and Hedgehog signaling. Exp Ther Med. 2017;14(4):3577–3582. doi:10.3892/etm.2017.5001
53. Kwon EY, Shin SK, Choi MS. Ursolic acid attenuates hepatic steatosis, fibrosis, and insulin resistance by modulating the circadian rhythm pathway in diet-induced obese mice. Nutrients. 2018;10(11). doi:10.3390/nu10111719
54. Wan S, Huang C, Wang A, Zhu X. Ursolic acid improves the bacterial community mapping of the intestinal tract in liver fibrosis mice. PeerJ. 2020;8:e9050. doi:10.7717/peerj.9050
55. Wan S, Luo F, Huang C, Liu C, Luo Q, Zhu X. Ursolic acid reverses liver fibrosis by inhibiting interactive NOX4/ROS and RhoA/ROCK1 signalling pathways. Aging. 2020;12(11):10614–10632. doi:10.18632/aging.103282
56. Lin X, Li Y, Zhang X, et al. Tormentic acid inhibits hepatic stellate cells activation via blocking PI3K/Akt/mTOR and NF-κB signalling pathways. Cell Biochem Funct. 2021;39(1):77–87. doi:10.1002/cbf.3564
57. Haraguchi H, Ishikawa H, Kubo I. Antioxidative action of diterpenoids from Podocarpus nagi. Planta Med. 1997;63(3):213–215. doi:10.1055/s-2006-957655
58. Zhang Y, Wang K, Chen H, et al. Anti-inflammatory lignans and phenylethanoid glycosides from the root of Isodon ternifolius (D.Don) Kudô. Phytochemistry. 2018;153:36–47. doi:10.1016/j.phytochem.2018.05.017
59. Liao YJ, Bai HY, Li ZH, et al. Longikaurin A, a natural ent-kaurane, induces G2/M phase arrest via downregulation of Skp2 and apoptosis induction through ROS/JNK/c-Jun pathway in hepatocellular carcinoma cells. Cell Death Dis. 2014;5(3):e1137. doi:10.1038/cddis.2014.66
60. Qin S, Chen SH, Guo YW, Gu YC. Diterpenoids of Isodon macrophylla. Helv Chim Acta. 2007;90:2041–2046. doi:10.1002/hlca.200790212
61. Duan H, Wang GC, Khan GJ, et al. Identification and characterization of potential antioxidant components in Isodon amethystoides (Benth.) Hara tea leaves by UPLC-LTQ-Orbitrap-MS. Food Chem Toxicol. 2021;148:111961. doi:10.1016/j.fct.2020.111961
62. Thao T, Lieu NT, Ninh PT, et al. Study on the chemical constituents of Dacrydium elatum and their cytotoxic activity. Zeitschrift fur Naturforschung B. 2019:74. doi:10.1515/znb-2018-0214
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.