Back to Journals » Journal of Hepatocellular Carcinoma » Volume 9
Neoadjuvant Therapy for Hepatocellular Carcinoma
Authors Yin Z, Chen D, Liang S, Li X
Received 9 January 2022
Accepted for publication 15 June 2022
Published 31 August 2022 Volume 2022:9 Pages 929—946
DOI https://doi.org/10.2147/JHC.S357313
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Imam Waked
Zongyi Yin,1– 3 Dongying Chen,3,4 Shuang Liang,1 Xiaowu Li1– 3
1Department of Hepatobiliary Surgery, Shenzhen University General Hospital, Shenzhen University, Shenzhen, 518055, People’s Republic of China; 2Guangdong Provincial Key Laboratory of Regional Immunity and Diseases & Carson International Cancer, Shenzhen University, Shenzhen, 518055, People’s Republic of China; 3Shenzhen University Clinical Medical Academy Center, Shenzhen University, Shenzhen, 518055, People’s Republic of China; 4Department of Anesthesiology, Shenzhen University General Hospital, Shenzhen University, Shenzhen, 518055, People’s Republic of China
Correspondence: Xiaowu Li, Department of Hepatobiliary Surgery, Shenzhen University General Hospital, Shenzhen University, Xueyuan AVE 1098, Nanshan District, Shenzhen, Guangdong, People’s Republic of China, Tel +86 755 2183 8184, Email [email protected]
Abstract: Hepatocellular carcinoma (HCC) is characterized by low resection and high postoperative recurrence rates, and conventional treatment strategies have failed to meet clinical needs. Neoadjuvant therapy (NAT) is widely employed in the routine management of several solid tumors because it increases resectability and reduces the rate of postoperative recurrence. However, a consensus has not been reached regarding the effects of NAT on HCC. As systemic therapy, particularly targeted therapy and immunotherapy, is given for HCC treatment, accumulating evidence shows that the “spring” of NAT for HCC is imminent. In the future, HCC researchers should focus on identifying biomarkers for treatment response, explore the mechanisms of resistance, and standardize the endpoints of NAT.
Keywords: neoadjuvant therapy, hepatocellular carcinoma, HCC, immunotherapy, systemic therapy, chemotherapy
Introduction
Neoadjuvant therapy (NAT) is the administration of therapeutic agents prior to definitive Surgery.1,2 It was first applied in cancer a half century ago. In 1955, Pisani et al first attempted to treat breast cancer with neoadjuvant radiotherapy (RT).3,4 Neoadjuvant chemotherapy (NAC) was then introduced in breast cancer treatment in 1985.5,6 At the same time as Pisani, neoadjuvant RT was introduced in colorectal cancer treatment by Stearns et al7,8 In 1986, Smith et al administered combined preoperative neoadjuvant RT and chemotherapy for anal and rectal cancer.9 Since the 1990s, an increasing number of patients with solid tumors, such as lung, ovarian, prostate, esophageal, and gastric cancers, have been treated with NAT, and they have achieved remarkable outcomes, including significant improvement in survival time and quality of life.10–18 This subverts the belief that surgery is the only curative treatment for tumors or that surgery should be the first consideration. Currently, it is unclear why these tumors benefit from NAT, but consensus has been established that NAT is an important part of the treatment of the aforementioned cancers19,20 (Figure 1).
In contrast to the aforementioned tumors, no convincing evidence of the effectiveness of NAT in hepatocellular carcinoma (HCC) has been reported for a long period of time.21–24 Among cancers, HCC consistently ranks high in terms of morbidity and mortality rates.25,26 More than 20% of patients who are initially eligible for liver transplantation (LT) drop out before transplantation due to tumor progression.27,28 Although considerable progress has been made in perioperative management and postoperative therapies, no significant improvement has been observed in postoperative 5-year overall survival (OS) rate and recurrence rate in patients with HCC.26,29–33 Thus, some preoperative interventions may be feasible methods to improve the prognosis of patients.34 This review provides an overview of the development of NATs and discusses how these various types of NATs can be used to downstage the tumor and increase the resectability of HCC. Articles were identified through a search of PubMed with the search terms “neoadjuvant radiotherapy”, “neoadjuvant chemotherapy”, “immunotherapy”, “neoadjuvant therapy”, “ablation therapy”, “systemic therapy”, “target therapy”, “hepatocellular carcinoma”, and “HCC” from 1950 until July 2021. Only papers published in English were reviewed. The final list of articles was generated on the basis of originality and relevance to the broad scope of this review.
Rationale of NAT for HCC
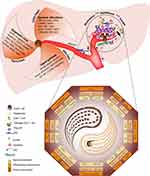
Unlike other organs, the liver has a complex anatomical structure. Various antigens from the digestive tract, such as gut nutrients, toxins, and metabolites, move into the bloodstream, and thus, the liver contains many immune cells, which confers the liver with high tolerance for immune function. Different compositions of immune cells have been observed in the immune microenvironment of primary and recurrent tumors.35 The relatively high tolerance threshold of the liver immune microenvironment facilitates immune escape during tumor development.36–41 Hence, the liver is widely considered as an immune organ.42 Additionally, compared with well-differentiated organs, the liver has renewable function. Even in adulthood, some pluripotent stem cells exist in various parts of the liver.43 Because of the presence of these cells, liver cancer exhibits strong temporal and spatial heterogeneity.44,45 Different clonal types are present in the same HCC lesion, and they progress over time according to different clonal patterns.46 The genomic and epigenomic expression profiles in the early and late disease stages are very different, even in the same tumor lesion.46–48 Many signaling pathways are involved in the occurrence and development of HCC. For example, vascular endothelial growth factor (VEGF) and VEGF receptor and platelet-derived growth factor receptor/fibroblast growth factor receptor (PDGFR/FGFR) participate in tumor angiogenesis and maintenance of mature blood vessels.49 EGFR/ IGF/ HGF/ e-MET, PI3K/ AKT/ mTOR, RAS/ RAF/ MEK/ ERK, and other signaling pathways are involved in tumor cell proliferation, motility, and apoptosis inhibition.50–52 According to recent molecular biology-related evidence, HCC can be categorized into many subtypes, eg, metabolism-driven, micro-environmental imbalance, and proliferation-driven subtypes.31,53 Different subtypes correspond to different therapeutic drugs or methods, which lead to significant differences in the prognosis of patients with HCC.53–56 The liver is the primary target organ for hepatitis viruses and harmful substances, such as alcohol. Long-term chronic interactions between liver cells and harmful agents lead to liver fibrosis and even cirrhotic nodules. These pathological structural changes will affect the physiological function of the liver; however, they will also affect drugs for liver cancer as they enter the target organ to kill cancer cells or activate the immune system to exert antitumor effects36,46,54,56,57 (Figure 2).
NAT for HCC
Similar to the role of NAT in other tumors, the application of NAT in HCC has many advantages and potential benefits. First, surgery may be allowed in patients with insufficient residual liver volume following NAT. Approximately 60% of patients with HCC have large tumors or tumor lesions that are distributed within multiple liver lobes at diagnosis.31,44 It is difficult for these patients to have sufficient residual liver volume (~30%) to support postoperative recovery (eg, extended right hemi-hepatectomy). Currently, the only method suitable for these patients is associating liver partition and portal vein ligation for staged hepatectomy (ALPPS), but the scope for application of this procedure is very limited.58–60 Second, NAT increases safety during the perioperative period. Large tumors, especially those at specific locations, often compress or invade important intrahepatic structures, such as the portal vein or bile ducts. This increases the difficulty of radical resection of liver tumors and the timing of surgery, and consequently, leads to an increased incidence of surgical complications and delays in the application of adjuvant therapy.61,62 These perioperative factors may undermine the potential benefits of the surgery. Third, NAT predicts anticancer drug sensitivity. After locoregional therapies, such as transarterial chemoembolization (TACE) and radiofrequency ablation (RFA), or surgery, the margins of liver tumors are significantly damaged, making it difficult to accurately evaluate the efficacy of anticancer drugs based on current detection methods (computed tomography or magnetic resonance imaging). However, NAT, especially neoadjuvant drug therapy, can be used to accurately evaluate the sensitivity of tumors to anticancer drugs in a relatively short time. This also provides some reference for adjuvant therapy in patients with HCC. Fourth, NAT might be an option to reduce the risk of early recurrences. HCC usually occurs as a multiclonal cancer. Preoperatively, tumor cells may have already migrated into the liver and the surrounding region.63 If only the macroscopic and visible tumor tissues are surgically removed, the patient will likely experience early postoperative recurrence. This somewhat explains why the early recurrence rate in patients with HCC is 70% postoperatively. This was also confirmed by differences in the early recurrence rate between patients with HCC who underwent partial hepatectomy and those who underwent LT.61,62,64 For microsatellite lesions, NAT achieves full contact between anticancer drugs and tumor cells, or alternatively, NAT changes the immune microenvironment of the lesions to maximize the tumor-killing effect, thereby reducing the risk of early postoperative recurrence. Briefly, NAT could be useful to downstage HCC to enable optimal surgical resection or to meet the criteria for liver resection (LR), which will lead to the improved prognosis of patients with HCC.
NAC
Similar to the role of NAT in other tumors, NAC was the first regimen investigated as a NAT for HCC. In 1989, Marvin et al treated three patients with unresectable HCC with the cytotoxic drug doxorubicin.65 Four years later, the same group conducted a pilot study comprising 20 patients.21 They concluded that neoadjuvant doxorubicin chemotherapy favorably alters the post-transplant survival of patients with HCC.21 Encouraged by their findings, more researchers treated patients with HCC with NAC.66–68 To reduce the systemic side effects of chemotherapy, TACE and hepatic arterial infusion chemotherapy (HAIC) were developed as alternatives to systemic chemotherapy.68–72 Chemotherapeutic drugs such as mitomycin, epirubicin, and cisplatin were delivered to the tumor site in high concentrations and were continuously administered through a tumor feeding artery. Most chemotherapeutic drugs are dose-dependent, and thus, locoregional administration enhances their killing effect on tumors.73–75 Tumors initially exceeding the Milan Criteria (MC) that were treated with neoadjuvant TACE achieved a post-transplant 5-year survival rate and HCC recurrence-free probability equal to those of patients with tumors within the MC.76 Among patients with advanced, Child–Pugh class A HCC, 11.7% were down staged to hepatectomy after neoadjuvant HAIC for 3 months.77 Neoadjuvant TACE was significantly associated with improved OS and recurrence-free survival after LR of large HCC tumors (≥10 cm).78,79 Additionally, TACE or HAIC combined with RT could serve as an alternative for a subset of patients with advanced HCC. Hepatectomy after downstaging by three-dimensional conformal RT combined with HAIC was safe and resulted in good long-term outcomes.80 For patients with portal vein tumor thrombosis (PVTT), a 26.5% resection rate and 100% response rate were recorded after downstaging with localized concurrent chemoradiotherapy (CCRT) followed by HAIC.81 The disease-specific survival rate differed significantly between patients who underwent resection after localized CCRT and those who underwent resection first (median, 62 months vs 15 months).81 For resectable HCCs, including small HCCs, a retrospective study that compared 1725 patients found that effective neoadjuvant TACE led to a 5-year disease-free survival rate of 56.8%, which was superior to that after surgical resection alone.82 However, some studies did not support the use of TACE/HAIC as NATs for HCC.78,83–87 Presently, clinicians have not reached a consensus. The establishment of an effective prognostic model and the accurate distinction of patients who are suitable for neoadjuvant TACE/HAIC are keys to promoting these strategies in clinical practice.85,88,89
Neoadjuvant RT
RT comprises external and internal RT and plays an increasingly important role in HCC treatment. The universal application of external RT includes stereotactic body RT (SBRT), 3-DCRT, and intensity-modulated RT.73 Compared with adjuvant RT, neoadjuvant RT is associated with improved long-term patient survival.90 A randomized, multicenter, controlled study conducted in patients with resectable HCC and PVTT reported that neoadjuvant 3-DCRT significantly reduced HCC-related mortality and HCC early recurrence rates compared with surgery alone.91 Histologically, the primary tumor and PVTT show complete necrosis.92 For HCCs with PVTT in the main trunk, the 5-year survival rate after neoadjuvant RT followed by hepatectomy is 20% higher than after hepatectomy alone (34.8% vs 13.1%).93 For patients in the waitlist for LT, SBRT helps to downsize or stabilize tumors before LT without notable side effects.94,95
Recently, selective internal RT, namely, transarterial radioembolization (TARE), has shown promise for patients with HCC. TARE is a microembolic procedure that delivers radioactive particles (eg, yttrium-90 microspheres [Y-90]) to the tumor site through the tumor feeding artery. Accumulating evidence has demonstrated the safety and efficacy of Y-90 radioembolization for HCC.96–105 A randomized, Phase 2 study of 179 patients with Barcelona Clinic Liver Cancer stage A or B demonstrated that Y-90 radioembolization led to a significantly longer time to progression (>26 months) than conventional TACE and reduced the drop-out rate from transplant waitlists.97 Moreover, 20% of patients with locally advanced unresectable HCC with intrahepatic portal vein thrombosis were successfully downstaged to surgery after neoadjuvant TARE.98 Patients who underwent liver surgery (LT or hepatectomy) after neoadjuvant TARE had a median OS of 47 months and a 5-year OS rate of 86%.99 Approximately 10% of the total functional remnant liver volume increased after 40 days of neoadjuvant TARE before LR.100–102 Hence, TARE might provide a survival benefit for patients with HCC with large tumors and major vessel invasion.104 Finally, TARE minimizes alterations to the hepatic arterial flow and is therefore suitable for most patients with HCC with or without good liver function.103 Few adverse events (AEs) were observed in patients with main PVTT and severe cirrhosis.96 Despite its advantages and potential therapeutic value, TARE does not appear as beneficial as targeted drugs, such as sorafenib.105,106 Hence, proposed prognostic stratification may help to better identify patients for whom TARE can be indicated and contraindicated.103,107 High tumor-absorbed radiation doses and low serum alpha-fetoprotein (AFP) levels were also found to be significantly associated with successful downstaging.98
Neoadjuvant Ablation Therapy
Tumor ablation is a form of locoregional-directed therapy in patients with HCC. Local ablation includes percutaneous ethanol injection therapy (PEIT), microwave ablation (MWA), nanoparticle-based photothermal ablation (PTA), and RFA. In 1987, Takanashi et al demonstrated that PEIT is a valuable treatment for HCC as 30% of the patients had completely necrotic tumors, as revealed by histopathologic analysis, and no damage to the noncancerous liver parenchyma distant to the injected sites was observed.108–110 In terms of high-risk patients with unresectable multifocal tumors, LR combined with intraoperative RFA could be safe and effective.111 Because LR has short operative times and is associated with minimal blood loss, RFA was considered a part of a multimodal therapeutic regimen.112–114 For LT candidates with unresectable HCC, RFA prolongs wait times for cadaveric livers,23,115 and 56% of patients with advanced HCC are successfully downstaged to LT criteria.116 Mazzaferro et al showed that RFA is more suitable for patients with small tumors (<3 cm), pretreatment AFP levels of >1000 ng/mL, and those who receive short-term therapy.117,118 However, because of new metastatic diseases that develop in approximately 30% of patients after undergoing RFA, multimodality treatment approaches that include RFA should be explored.24 At present, there are no surrogate markers to determine the optimal interval between locoregional therapies and definitive therapy (LT or LR).119
Similar to RFA, MWA also exerts effective antitumor effects in terms of local tumor control, treatment-related complications, and long-term survival.120 Recently, PTA has been intensively investigated. However, PTA has certain shortcomings, such as the poor biocompatibility of most PTA agents, potential long-term toxicity, and limitations to surface tumors because of insufficient penetration depth of near-infrared light for deep abdominal tumors. Hence, a minimally invasive laparoscopic-assisted PTA method may be effective for HCC.121 Finally, compared with RFA + TACE, the neoadjuvant MWA + TACE strategy provides more durable disease control in patients with early-stage HCC.122 MWA may be superior to PEIT for the local control of moderately or poorly differentiated small HCC.123
Neoadjuvant Systemic Therapy (NST)
NST is a broad concept and refers to a series of systemic drug delivery strategies. NST was originally developed for patients with advanced unresectable HCC as a palliative treatment and did not play a relevant role in the neoadjuvant setting. Recently, new evidence of NST for HCC has emerged, and some experts believe that a new era of the application of NST for HCC is on the horizon.27,73,124–126 Presently, the major NSTs for patients with HCC involve tyrosine kinase inhibitor (TKI) therapy, immunotherapy, and gene/cell therapy.
Neoadjuvant TKI Strategy
Since the approval of sorafenib in 2007, TKIs have played an extremely important role in HCC treatment.127–130 Most TKIs have multiple targets; for example, cabozantinib has nine targets (MET, VEGFR1, VEGFR2, VEGFR3, ROS1, RET, AXL, NTRK, and KIT). Thus, several signaling pathways involved in the occurrence and development of HCC could be targeted by TKIs. The safety and feasibility of TKIs as NATs for HCC were then explored.131–137 For patients with unresectable HCC, a subset analysis of a sorafenib study database showed a difference of approximately 10 months in the OS between patients who received and did not receive sorafenib before surgical resection.136,138 No serious adverse effect of preoperative sorafenib administration was observed during and immediately after LR.137 For patients with locally advanced HCC who were awaiting LT, the drop-out rate of patients receiving NAT was less than that of those treated with NAT for <6 months.131,134 Downstaging from beyond the MC using neoadjuvant sorafenib therapy was correlated with better post-LT tumor event-free survival and longer OS.139 However, compared with TACE alone, the combination of TACE and sorafenib before LT did not result in significant benefits.133 Thus, sorafenib administration before LT appears to be associated with an increase in acute graft rejection and biliary complications.132,135 Other TKIs, such as lenvatinib, regorafenib, and cabozantinib, have been administrated as NATs in multiple solid tumors, such as thyroid cancer, renal cell carcinoma, and rectal cancer, and promising results were observed, but few studies have been published on TKIs as NATs for HCC.140–144 Finally, to evaluate the survival variation of LR/LT after TKIs, a statistical model that allows personalized survival prediction using baseline clinical features is warranted.145,146 According to the high expression level of molecular markers such as BRAF-V600E and EGFR in tumor biopsy specimens or peripheral blood, a dual-targeted therapy strategy with multiple targets is also worthy of investigation.147–149
Neoadjuvant Immunotherapy (NIT) Strategy
Immune escape plays an important role in HCC development. Alterations in major histocompatibility complex class I and aberrant expression of tumor neoantigens induce the activation of tumor-killing T cells.36,150 The elimination of cancer-related immunosuppression, which is a crucial treatment strategy for HCC, is an important issue.57,150,151 Recently, immune checkpoint inhibitors (ICIs) were found to have significant antitumor effects in HCC through targeting either programmed cell death (PD) receptor/CTLA-4 on T cells or its ligands PDL-1 and PDL-2 on tumor cells.152–160 The current evidence for NIT in HCC is mainly focused on patients with macrovascular invasion, multifocal disease, or large tumors requiring vascular resections.161 For unresectable HCC, lenvatinib plus pembrolizumab has shown promising antitumor activity (median OS, 22 months; objective response rate [ORR], 46%).152,153 Dual PD-L1/VEGF blockade (atezolizumab–bevacizumab) resulted in better OS and progression-free survival outcomes than sorafenib or other targeted drugs.157,158,160,162 More than 20% of human HCCs that do not respond to single agents could benefit from this combination.163,164 The combination of nivolumab + ipilimumab (Check Mate 040) led to an ORR of 32% in patients who previously treated with sorafenib.165 The toxicities were manageable, with no unexpected safety signals. In studies on NITs for unresectable HCC, the rate of successful downstaging to surgical intervention ranged from 13% to 46%, and a near-complete pathologic response was observed.158,165–168 According to these evidences, a study named HIMALAYA was conducted to assess the efficacy and safety of durvalumab plus tremelimumab as first-line treatment in advanced HCC (NCT03298451).169 For cancers with distant metastases, NIT has significant therapeutic efficacy compared with the use of single adjuvant immunotherapy after primary tumor resection.170 Besides locoregional therapies, NIT is expected to lead to HCC downstaging from unmet MC.156 Comparable with patients who met the LT criteria, successful downstaging of HCC to within the LT criteria was associated with a low rate of HCC early recurrence and excellent post-transplant survival.171 Despite the many benefits of NIT, no consensus has been reached on its application in a transplant setting because activated T cells may cause allograft rejection.156,172,173 Some evidence appears to support that an optimal time interval exists between the termination of NIT and LT.119,156,172
Additionally, because surgical resection is associated with high early recurrence rates, the exploration of immunotherapy for resectable (early-stage) HCC may maximize patient benefits.30,53 Several clinical studies are currently focusing on this issue.174,175 An ongoing randomized Phase II pilot trial showed that treatment for 6 weeks with nivolumab + ipilimumab led to a pathologic complete response rate of 25% in patients with resectable HCC; the NIT regimen was safe, and surgical resection was not delayed.155,176 The OS and 2-year recurrence rates of the study are highly anticipated. Studies on NIT are currently underway to evaluate the safety of the combination of TKIs + ICIs or dual ICI therapy before hepatic resection in patients with locally resectable HCC. Furthermore, the efficacy and the development of immune-related AEs (irAEs) appear to be impacted by the time window between NIT and surgery.156,172 In an animal model, smaller doses of NIT scheduled close to the time of surgery may have led to optimal antitumor immunity and improved severe irAEs.170 Two cycles of NIT appear to be a tolerable neoadjuvant dosing schedule, in which less toxicity but equal effectiveness is observed.177 Overall, the use of NIT for the treatment of HCC in a preoperative setting is yet to be explored,178 and thus, more randomized controlled trials (RCTs) are warranted (Table 1).
 |
Table 1 Clinical Trials Investigating Tyrosine Kinase Inhibitors and Immune Checkpoint Inhibitors as NATs for HCC |
Neoadjuvant Gene/Cell Therapy Strategy
With an in-depth understanding of the biological behavior of HCC and the development of cell/genetic engineering technologies, an increasing number of potential approaches are being explored as NITs in HCC. Most of these approaches are still at preclinical or early clinical stages. The mainstay approaches involve the use of chimeric antigen receptor (CAR) T cells, allogeneic natural killer (NK) cells, and oncolytic viruses.178–180
Because 70% of HCCs express glypican 3 (GPC3), GPC3-targeted CAR T cells could eliminate GPC3-positive HCC cells by inducing perforin- and granzyme-mediated apoptosis or by reducing Wnt signaling in tumor cells.181 Six registered Phase 1 studies are underway to evaluate the efficacy, tolerance, and safety of GPC3-targeted CAR T cells (NCT04121273, NCT04506983, NCT03198546, NCT02905188, NCT03884751, and NCT03980288). Other modified CAR T cell therapies are also being explored. For instance, a study of CD147-targeted CAR T cells administered by hepatic artery infusion for advanced HCC is underway (NCT03993743).182,183 Approximately 10 registered trials have been conducted to assess the safety and efficacy of allogeneic NK cells in patients with HCC (NCT04162158, NCT02008929, NCT04011033, NCT04106167, and NCT03203005). Apart from the sequence of NK therapy to LR, NK cell treatment was found to be well tolerated and resulted in no HCC early recurrence after LT (NCT01147380).184 NK cell-based anti-HCC therapeutic strategies alone or in combination with other therapies are highly promising for HCC treatment.185 Finally, vaccinia virus-based oncolytic immunotherapy, which is designed to preferentially replicate in and destroy tumor cells, does not improve OS as a second-line therapy after sorafenib failure, but it may be advantageous in the treatment of patients at earlier disease stages (NCT03071094).186 Although few trials have been conducted on neoadjuvant gene/cell therapy for HCC, theoretically, the approaches would have potential tumor-killing effects.
Combination Therapy
Although the function of NATs is increasingly obvious in HCC treatment, the rate of successful tumor downstaging or reduction of the risk of early recurrence are still not ideal. Several researchers have attempted to combine two or more treatments. NATs based on different mechanisms seem to have synergistic antitumor effects.187,188 The combination of ICIs and TKIs has been described in the section on systemic therapy.124 In clinical practice, complete tumor excision using unique locoregional therapy is difficult because local or remote microlesions are present in most patients with HCC.189–191 Hence, combination therapy, such as TACE + RFA, HAIC + RFA, or TACE + RFA + ICIs, is necessary.192 Locoregional therapy has been shown to induce a tumor immune response.187,192–196 For instance, RFA-derived tumor antigens led to the initiation of a systemic antitumor immune response that boosted the antitumor effect of ICIs.197 Tremelimumab (a CTLA-4 monoclonal antibody) in combination with tumor ablation led to the accumulation of intratumoral CD8+ T cells and resulted in a 6-month progression-free survival rate of 57.1% in patients with refractory HCC.198 Patients with recurrent HCC could achieve a longer survival time following combined treatment. TACE + sequential RFA within 30 days is more effective for recurrent HCCs. A certain time interval is required for the treatment of large or multiple HCCs but may not be necessary for the treatment of solitary medium-sized HCCs.199,200 Some trials of TACE + ICIs and/or TKIs are currently underway. Some examples are as follows: cabozantinib + ipilimumab/nivolumab and TACE in patients with HCC (NCT04472767), TACE + SBRT followed by dual immunotherapy for downstaging HCC (NCT04988945 and NCT03817736). For patients with potentially resectable HCC, TACE + HAIC vs HAIC, TACE + sintilimab injection, and TACE + durvalumab + bevacizumab therapy are being explored as preoperative strategies (NCT03591705, NCT04174781, and NCT03778957). However, not all combination therapies can reduce the tumor burden or improve prognosis; for example, HAIC + cisplatin before RFA did not significantly decrease the early recurrence rate in patients with early-stage HCC;192 hence, treatment decisions should be made strictly on the basis of clinical evidence-based results.
Selection of Appropriate NATs
As results about NATs applying in patients with HCC are not so strong at the moment, the identification of patients who will benefit and the selection of the appropriate NAT in advance are challenging.27 Previous models that were developed to predict the treatment effect and prognosis of patients are relatively imprecise and are mainly focused on the tumor burden, lesion number, tumor vascularity pattern, histological grade, hepatic reserve, and performance status.201,202 Multidimensional and multilevel evaluations are needed to predict the effect of NATs (eg, the combination of molecular/cell biology and imaging).
With a more in-depth understanding of the molecular biological behavior of HCC, numerous biomarkers have been established to predict the effect of NATs. In liquid biopsy, the serum AFP level was found to be correlated with tumor early recurrence after LT, and patients with AFP levels of >400 ng/mL responded well to ramucirumab (a VEGF-2 inhibitor).201,203–205 The reinvigoration of circulating exhausted-phenotype CD8+ T cells induces a strong immunological response to pembrolizumab.206 Lenvatinib-treated responders showed greater increases in FGF19 and FGF23 levels than nonresponders, but higher baseline VEGF, ANG2, and FGF21 levels were correlated with shorter OS after lenvatinib or sorafenib treatment.207 Circulating tumor cells, which escape from the tumor site, are the primary source of metastases or post-LT recurrence and have been shown to predict HCC early recurrence after LR and LT.208,209 Moreover, the integration of transcriptomic and genomic data provides a global tumor picture and describes the escape mechanisms.210 Analysis of circulating tumor DNA carrying a cancer-specific tumor framework, mutational load, immune composition, and antitumor immunity as well as immunosuppressive genetic and epigenetic aberrations enables monitoring of the effect of NAT in HCC in a noninvasive, dynamic manner.211 The expression levels of TP53, RET, FGFR3, and APC in the plasma were significantly higher in patients with multiple tumors or with metastasis than in patients with single tumors, which indicates that patients with high mutant allele frequency in these genes are more suitable for NAT.212 The predictive role of other biomarkers, such as des-γ-carboxy prothrombin, lens culinaris agglutinin-reactive AFP, and proteins induced by vitamin K absence/antagonist-II, are being explored.
In traumatic biopsy, the expression of PD-1 on subdominant T (CD8+) cells was found to be correlated with a favorable response to PD-1/PD-L1 blocking antibodies by enhancing CD8+ T cell survival and not by enhancing their proliferation.35,37,213 The low expression of Batf3 (+) DC in pretreatment tumor biopsies is related to relapse after neoadjuvant ipilimumab + nivolumab.214 Pre-existing T cell infiltration and/or PD-L1 expression in tumors may serve as indicators of the clinical response.206 The expansion and activation of CD103 (+) DC progenitors at the tumor site enhances tumor responses to therapeutic PD-L1.215 Increased IL-6 expression in pre-RT serum and tumor tissues was significantly associated with resistance to RT.91 Additionally, compared with the intermediate VEGF/FGF group, the VEGF/FGF-enriched groups demonstrated improved OS with lenvatinib administration.207 Recent studies found that lenvatinib reduced the tumor PD-L1 level and Treg differentiation to improve anti-PD-1 efficacy by blocking FGFR4. Furthermore, the levels of FGFR4 expression and Treg infiltration in the tumor could serve as biomarkers for screening patients with HCC for lenvatinib + anti-PD-1 combination therapy.163,216 For patients resistant to NAT within the treatment window, combinations of novel signal pathway inhibitors may revive the effect of TKIs or immunotherapy.147 Other biomarkers, such as TMB, MSI/DMMR, EBV infection, and POLE/ALK gene expression, also play an important role in predicting the effects of NAT.147,217–219
Overall, patients with a high tumor burden, high risk of recurrence, and potential resectable tumors are suitable for undergoing NAT. Although many biomarkers have been found to be associated with the effect of NAT, few have been confirmed by large RCTs. Thus, the comprehensive evaluation of these predictors is requisite in clinical practice.
Conclusion
A novel era for HCC treatment is imminent. NAT is the bridge leading to the development of personalized therapies and is constantly improving under the existing framework. In terms of treatment optimization, researchers should consider local and global aspects, short- and long-term effects, and toxicity. The main obstacle to adequate patients’ selection derives from the absence of predictive biomarkers. Future studies of NAT should focus on the identification of biomarkers of treatment response, explore the mechanisms of resistance, and standardize endpoints of NAT. Large RCTs are required to ensure the optimal components and sequence of multimodality therapy.
Abbreviations
HCC, hepatocellular carcinoma; NAT, neoadjuvant therapy; NAC, neoadjuvant chemotherapy; LT, liver transplantation; OS, overall survival; VEGF, vascular endothelial growth factor; PDGFR, platelet-derived growth factor receptor; FGFR, fibroblast growth factor receptor; ALPPS, associating liver partition and portal vein ligation for staged hepatectomy; TACE, transarterial chemoembolization; RFA, radiofrequency ablation; LR, liver resection; HAIC, hepatic arterial infusion chemotherapy; PVTT, portal vein tumor thrombosis; CCRT, concurrent chemoradiotherapy; SBRT, stereotactic body radiotherapy; TARE, transarterial radioembolization; AFP, alpha-fetoprotein; PEIT, percutaneous ethanol injection therapy; MWA, microwave ablation; PTA, photothermal ablation; NST, neoadjuvant systemic therapy; TKI, tyrosine kinase inhibitor; NIT, neoadjuvant immunotherapy; ICIs, immune checkpoint inhibitors; RCTs, randomized controlled trials.
Consent for Publication
All authors have agreed on the contents and publication of the manuscript.
Acknowledgments
We thank the editors and reviewers of the paper for their warm work earnestly.
Authors’ Information
Li Xiaowu M.D. PhD, Yin Zongyi M.D., Liang Shuang; Department of Hepatobiliary Surgery, Shenzhen University General Hospital, Shenzhen University, Shenzhen (518055), China; Guangdong Provincial Key Laboratory of Regional Immunity and Diseases & Carson International Cancer; Shenzhen University Clinical Medical Academy Center, Shenzhen University, Shenzhen (518055), China.
Chen Dongying M.D., Department of Anesthesiology, Shenzhen University General Hospital, Shenzhen University, Shenzhen (518055), China.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
This study was supported by grants from National Natural Science Foundation of China [grant numbers 81430063, 8210140740, 82104597], Guangdong Provincial Science and Technology Program [grant numbers 2019B030301009], Natural Science Foundation of Guangdong Province of China [grant numbers 2021A1515012161], Guangdong Province Regional Joint Fund-Key Projects [grant numbers 2020B1515120096], Guangdong Basic and Applied Basic Research Foundation, Sanming Project of Medicine in Shenzhen [grant numbers SZSM202003009], Shenzhen Key Laboratory Foundation [grant numbers ZDSYS20200811143757022] and Shenzhen International Cooperative Research Project [grant numbers GJHZ20200731095210030].
Disclosure
The authors declared that they have no conflicts of interest to this work.
References
1. Available from: https://en.jinzhao.wiki/wiki/Neoadjuvant_therapy.
2. Available from: https://cancerstaging.org/.
3. Pisani G, Malaspina A. Results of preoperative x-ray therapy in 343 cases of breast cancer. Tumori. 1955;41(4):406–423. doi:10.1177/030089165504100404
4. Borgstrom S, Lindgren M. Preoperative roentgen therapy of cancer of the breast: preliminary communication. Acta radiol. 1962;58:9–16. doi:10.3109/00016926209169544
5. Ragaz J, Baird R, Rebbeck R, Goldie A, Coldman A, Spinelli J. Preoperative adjuvant chemotherapy (neoadjuvant) for carcinoma of the breast: rationale and safety report. Recent Results Cancer Res. 1985;98:99–105.
6. Jacquillat C, Baillet F, Auclerc G, et al. Neoadjuvant chemotherapy of breast cancer. Drugs Exp Clin Res. 1986;12(1–3):147–152.
7. Stearns MW, Deddish MR, Quan SH. Preoperative roentgen therapy for cancer of the rectum. Surg Gynecol Obstet. 1959;109(2):225–229.
8. Ruff CC, Dockerty MB, Fricke RE, et al. Preoperative radiation therapy for adenocarcinoma of the rectum and rectosigmoid. Surg Gynecol Obstet. 1961;112:715–723.
9. Smith DE, Muff NS, Shetabi H. Combined preoperative neoadjuvant radiotherapy and chemotherapy for anal and rectal cancer. Am J Surg. 1986;151(5):577–580. doi:10.1016/0002-9610(86)90552-0
10. Cox JD, Samson MK, Herskovic AM, et al. Cisplatin and etoposide before definitive radiation therapy for inoperable squamous carcinoma, adenocarcinoma, and large cell carcinoma of the lung: a Phase I-II study of the Radiation Therapy Oncology Group. Cancer Treat Rep. 1986;70(10):1219–1220.
11. Kelsen DP, Hilaris B, Martini N. Neoadjuvant chemotherapy and surgery of cancer of the esophagus. Semin Surg Oncol. 1986;2(3):170–176. doi:10.1002/ssu.2980020310
12. Gralla RJ, Kris MG. Chemotherapy in non-small cell lung cancer: results of recent trials. Semin Oncol. 1988;15(3 Suppl 4):2–5.
13. Schlag P, Herrmann R, Raeth U, Lehner B, Schwarz V, Herfarth C. Preoperative (neoadjuvant) chemotherapy in squamous cell cancer of the esophagus. Recent Results Cancer Res. 1988;110:14–20.
14. Lawton FG, Redman CW, Luesley DM, et al. Neoadjuvant (cytoreductive) chemotherapy combined with intervention debulking surgery in advanced, unresected epithelial ovarian cancer. Obstet Gynecol. 1989;73(1):61–65.
15. Vokes EE, Bitran JD, Hoffman PC, et al. Neoadjuvant vindesine, etoposide, and cisplatin for locally advanced non-small cell lung cancer. Final report of a phase 2 study. Chest. 1989;96(1):110–113. doi:10.1378/chest.96.1.110
16. Chambers JT, Chambers SK, Voynick IM, et al. Neoadjuvant chemotherapy in stage X ovarian carcinoma. Gynecol Oncol. 1990;37(3):327–331. doi:10.1016/0090-8258(90)90361-N
17. Kelsen D. Neoadjuvant therapy for gastrointestinal cancers. Oncology. 1993;7(9):
18. Sassine AM, Schulman CC. Neoadjuvant hormonal deprivation before radical prostatectomy. Eur Urol. 1993;24(Suppl 2):46–50. doi:10.1159/000474389
19. Korde LA, Somerfield MR, Carey LA, et al. Neoadjuvant chemotherapy, endocrine therapy, and targeted therapy for breast cancer: ASCO guideline. J Clin Oncol. 2021;39(13):1485–1505. doi:10.1200/JCO.20.03399
20. Available from: https://www.nccn.org/guidelines/.
21. Stone MJ, Klintmalm GBG, Polter D, et al. Neoadjuvant chemotherapy and liver transplantation for hepatocellular carcinoma: a pilot study in 20 patients. Gastroenterology. 1993;104(1):196–202. doi:10.1016/0016-5085(93)90852-4
22. Lygidakis NJ, Sgourakis G, Dedemadi G, et al. Preoperative main portal branch transection combined with liver locoregional transarterial neo and adjuvant immunochemotherapy for patients with hepatocellular carcinoma. Hepatogastroenterology. 2000;47(36):1546–1554.
23. Fontana RJ, Hamidullah H, Nghiem H, et al. Percutaneous radiofrequency thermal ablation of hepatocellular carcinoma: a safe and effective bridge to liver transplantation. Liver Transpl. 2002;8(12):1165–1174. doi:10.1053/jlts.2002.36394
24. Curley SA, Izzo F, Delrio P, et al. Radiofrequency ablation of unresectable primary and metastatic hepatic malignancies: results in 123 patients. Ann Surg. 1999;230(1):1–8. doi:10.1097/00000658-199907000-00001
25. Akinyemiju T, Abera S, Ahmed M. The burden of primary liver cancer and underlying etiologies from 1990 to 2015 at the global, regional, and national level: results from the global burden of disease study 2015. JAMA Oncol. 2017;3(12):1683–1691. doi:10.1001/jamaoncol.2017.3055
26. Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.21492
27. Liu K, McCaughan GW. How to select the appropriate “neoadjuvant therapy” for hepatocellular carcinoma. Expert Opin Pharmacother. 2018;19(11):1167–1170. doi:10.1080/14656566.2018.1498843
28. Kulik L, Heimbach JK, Zaiem F, et al. Therapies for patients with hepatocellular carcinoma awaiting liver transplantation: a systematic review and meta-analysis. Hepatology. 2018;67(1):381–400. doi:10.1002/hep.29485
29. Portolani N, Coniglio A, Ghidoni S, et al. Early and late recurrence after liver resection for hepatocellular carcinoma: prognostic and therapeutic implications. Ann Surg. 2006;243(2):229–235. doi:10.1097/01.sla.0000197706.21803.a1
30. Takeishi K, Maeda T, Tsujita E, et al. Predictors of intrahepatic multiple recurrences after curative hepatectomy for hepatocellular carcinoma. Anticancer Res. 2015;35(5):3061–3066.
31. Xie DY, Ren Z-G, Zhou J, et al. 2019 Chinese clinical guidelines for the management of hepatocellular carcinoma: updates and insights. Hepatobiliary Surg Nutr. 2020;9(4):452–463. doi:10.21037/hbsn-20-480
32. Zhu XD, Li KS, Sun HC. Adjuvant therapies after curative treatments for hepatocellular carcinoma: current status and prospects. Genes Dis. 2020;7(3):359–369. doi:10.1016/j.gendis.2020.02.002
33. Hirokawa F, Komeda K, Taniguchi K, et al. Is postoperative adjuvant transcatheter arterial infusion therapy effective for patients with hepatocellular carcinoma who underwent hepatectomy? A prospective randomized controlled trial. Ann Surg Oncol. 2020;27(11):4143–4152. doi:10.1245/s10434-020-08699-w
34. Lau WY. Future perspectives for hepatocellular carcinoma. HPB (Oxford). 2003;5(4):206–213. doi:10.1080/13651820310016779
35. Sun Y, Wu L, Zhong Y, et al. Single-cell landscape of the ecosystem in early-relapse hepatocellular carcinoma. Cell. 2021;184(2):404–421 e16. doi:10.1016/j.cell.2020.11.041
36. Zheng C, Zheng L, Yoo J-K, et al. Landscape of infiltrating T cells in liver cancer revealed by single-cell sequencing. Cell. 2017;169(7):1342–1356 e16. doi:10.1016/j.cell.2017.05.035
37. Zhang Q, He Y, Luo N, et al. Landscape and dynamics of single immune cells in hepatocellular carcinoma. Cell. 2019;179(4):829–845 e20. doi:10.1016/j.cell.2019.10.003
38. Su S, Liu Q, Chen J, et al. A positive feedback loop between mesenchymal-like cancer cells and macrophages is essential to breast cancer metastasis. Cancer Cell. 2014;25(5):605–620. doi:10.1016/j.ccr.2014.03.021
39. Zhou SL, Yin D, Hu Z-Q, et al. A positive feedback loop between cancer stem-like cells and tumor-associated neutrophils controls hepatocellular carcinoma progression. Hepatology. 2019;70(4):1214–1230. doi:10.1002/hep.30630
40. Dong LQ, Peng L-H, Ma L-J, et al. Heterogeneous immunogenomic features and distinct escape mechanisms in multifocal hepatocellular carcinoma. J Hepatol. 2020;72(5):896–908. doi:10.1016/j.jhep.2019.12.014
41. Liu LZ, Zhang Z, Zheng B-H, et al. CCL15 recruits suppressive monocytes to facilitate immune escape and disease progression in hepatocellular carcinoma. Hepatology. 2019;69(1):143–159. doi:10.1002/hep.30134
42. Ringelhan M, Pfister D, O’Connor T, et al. The immunology of hepatocellular carcinoma. Nat Immunol. 2018;19(3):222–232. doi:10.1038/s41590-018-0044-z
43. Zheng H, Pomyen Y, Hernandez MO, et al. Single-cell analysis reveals cancer stem cell heterogeneity in hepatocellular carcinoma. Hepatology. 2018;68(1):127–140. doi:10.1002/hep.29778
44. Dong LQ, Shi Y, Ma L-J, et al. Spatial and temporal clonal evolution of intrahepatic cholangiocarcinoma. J Hepatol. 2018;69(1):89–98. doi:10.1016/j.jhep.2018.02.029
45. Losic B, Craig AJ, Villacorta-Martin C, et al. Intratumoral heterogeneity and clonal evolution in liver cancer. Nat Commun. 2020;11(1):291. doi:10.1038/s41467-019-14050-z
46. Craig AJ, von Felden J, Garcia-Lezana T, et al. Tumour evolution in hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2020;17(3):139–152. doi:10.1038/s41575-019-0229-4
47. Ding X, He M, Chan AWH, et al. Genomic and epigenomic features of primary and recurrent hepatocellular carcinomas. Gastroenterology. 2019;157(6):1630–1645 e6. doi:10.1053/j.gastro.2019.09.005
48. Wang W, Mauleon R, Hu Z, et al. Genomic variation in 3010 diverse accessions of Asian cultivated rice. Nature. 2018;557(7703):43–49. doi:10.1038/s41586-018-0063-9
49. Haas NB, Manola J, Uzzo RG, et al. Adjuvant sunitinib or sorafenib for high-risk, non-metastatic renal-cell carcinoma (ECOG-ACRIN E2805): a double-blind, placebo-controlled, randomised, Phase 3 trial. Lancet. 2016;387(10032):2008–2016. doi:10.1016/S0140-6736(16)00559-6
50. Ikeda K, Kudo M, Kawazoe S, et al. Phase 2 study of lenvatinib in patients with advanced hepatocellular carcinoma. J Gastroenterol. 2017;52(4):512–519. doi:10.1007/s00535-016-1263-4
51. Zhai H, Zhong W, Yang X, et al. Neoadjuvant and adjuvant epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI) therapy for lung cancer. Transl Lung Cancer Res. 2015;4(1):82–93. doi:10.3978/j.issn.2218-6751.2014.11.08
52. Koulouris A, Tsagkaris C, Spyrou V, et al. Hepatocellular carcinoma: an overview of the changing landscape of treatment options. J Hepatocell Carcinoma. 2021;8:387–401. doi:10.2147/JHC.S300182
53. Jiang Y, Sun A, Zhao Y, et al. Proteomics identifies new therapeutic targets of early-stage hepatocellular carcinoma. Nature. 2019;567(7747):257–261. doi:10.1038/s41586-019-0987-8
54. Gao Q, Zhu H, Dong L, et al. Integrated proteogenomic characterization of HBV-related hepatocellular carcinoma. Cell. 2019;179(2):561–577 e22. doi:10.1016/j.cell.2019.08.052
55. Xue R, Chen L, Zhang C, et al. Genomic and transcriptomic profiling of combined hepatocellular and intrahepatic cholangiocarcinoma reveals distinct molecular subtypes. Cancer Cell. 2019;35(6):932–947 e8. doi:10.1016/j.ccell.2019.04.007
56. Duan M, Hao J, Cui S, et al. Diverse modes of clonal evolution in HBV-related hepatocellular carcinoma revealed by single-cell genome sequencing. Cell Res. 2018;28(3):359–373. doi:10.1038/cr.2018.11
57. Zhang Q, Lou Y, Yang J, et al. Integrated multiomic analysis reveals comprehensive tumour heterogeneity and novel immunophenotypic classification in hepatocellular carcinomas. Gut. 2019;68(11):2019–2031. doi:10.1136/gutjnl-2019-318912
58. Peng C, Li C, Liu C, et al. The outcome of the HCC patients underwent ALPPS: retrospective study. Medicine. 2019;98(38):e17182. doi:10.1097/MD.0000000000017182
59. Vennarecci G, Grazi GL, Sperduti I, et al. ALPPS for primary and secondary liver tumors. Int J Surg. 2016;30:38–44. doi:10.1016/j.ijsu.2016.04.031
60. Zhang J, Huang H, Bian J, et al. Safety, feasibility, and efficacy of associating liver partition and portal vein ligation for staged hepatectomy in treating hepatocellular carcinoma: a systematic review. Ann Transl Med. 2020;8(19):1246. doi:10.21037/atm-20-2214
61. Fitzmorris P, Shoreibah M, Anand BS, et al. Management of hepatocellular carcinoma. J Cancer Res Clin Oncol. 2015;141(5):861–876. doi:10.1007/s00432-014-1806-0
62. Llovet JM, Zucman-Rossi J, Pikarsky E, et al. Hepatocellular carcinoma. Nat Rev Dis Primers. 2016;2:16018. doi:10.1038/nrdp.2016.18
63. Fisher B. Biological and clinical considerations regarding the use of surgery and chemotherapy in the treatment of primary breast cancer. Cancer. 1977;40(1 Suppl):574–587.
64. Meirelles Junior RF, Salvalaggio P, Rezende MBD, et al. Liver transplantation: history, outcomes and perspectives. Einstein. 2015;13(1):149–152. doi:10.1590/S1679-45082015RW3164
65. Stone MJ, Klintmalm G, Polter D, Husberg B, Egorin MJ. Neoadjuvant chemotherapy and orthotopic liver transplantation for hepatocellular carcinoma. Transplantation. 1989;48(2):344–347.
66. Rizzi PM, Ryder SD, Ramage JK, et al. Neoadjuvant chemotherapy after liver transplantation for hepatocellular carcinoma. Transplant Proc. 1994;26(6):3568–3569.
67. Holman M, Harrison D, Stewart A, et al. Neoadjuvant chemotherapy and orthotopic liver transplantation for hepatocellular carcinoma. N J Med. 1995;92(8):519–522.
68. Arcement CM, Towbin RB, Meza MP, et al. Intrahepatic chemoembolization in unresectable pediatric liver malignancies. Pediatr Radiol. 2000;30(11):779–785. doi:10.1007/s002470000296
69. Paye F, Jagot P, Vilgrain V, et al. Preoperative chemoembolization of hepatocellular carcinoma: a comparative study. Arch Surg. 1998;133(7):767–772. doi:10.1001/archsurg.133.7.767
70. Paye F, Farges O, Dahmane M, et al. Cytolysis following chemoembolization for hepatocellular carcinoma. Br J Surg. 1999;86(2):176–180. doi:10.1046/j.1365-2168.1999.01014.x
71. Yoshida T, Sakon M, Umeshita K, et al. Appraisal of transarterial immunoembolization for hepatocellular carcinoma: a clinicopathologic study. J Clin Gastroenterol. 2001;32(1):59–65. doi:10.1097/00004836-200101000-00014
72. Ramsey DE, Kernagis LY, Soulen MC, et al. Chemoembolization of hepatocellular carcinoma. J Vasc Interv Radiol. 2002;13(9 Pt 2):S211–21. doi:10.1016/S1051-0443(07)61789-8
73. Zhang T, Zhang L, Xu Y, et al.Neoadjuvant therapy and immunotherapy strategies for hepatocellular carcinoma. Am J Cancer Res. 2020;10:1658.
74. Galastri FL, Nasser F, Affonso BB, et al. Imaging response predictors following drug eluting beads chemoembolization in the neoadjuvant liver transplant treatment of hepatocellular carcinoma. World J Hepatol. 2020;12(1):21–33. doi:10.4254/wjh.v12.i1.21
75. Raoul JL, Forner A, Bolondi L, et al. Updated use of TACE for hepatocellular carcinoma treatment: how and when to use it based on clinical evidence. Cancer Treat Rev. 2019;72:28–36. doi:10.1016/j.ctrv.2018.11.002
76. Affonso BB, Galastri FL, Filho JM, et al. Long-term outcomes of hepatocellular carcinoma that underwent chemoembolization for bridging or downstaging. World J Gastroenterol. 2019;25(37):5687–5701. doi:10.3748/wjg.v25.i37.5687
77. Lee BH, Lee D-S, Cho CW, et al. Role and limitation of neoadjuvant hepatic arterial infusion chemotherapy in advanced hepatocelluar carcinoma patients with Child-Pugh class A. World J Surg Oncol. 2019;17(1):143. doi:10.1186/s12957-019-1685-6
78. Li C, Wang M-D, Lu L, et al. Preoperative transcatheter arterial chemoembolization for surgical resection of huge hepatocellular carcinoma (>/= 10 cm): a multicenter propensity matching analysis. Hepatol Int. 2019;13(6):736–747. doi:10.1007/s12072-019-09981-0
79. Chang Y, Jeong SW, Young Jang J, et al. Recent updates of transarterial chemoembolilzation in hepatocellular carcinoma. Int J Mol Sci. 2020;21(21). doi:10.3390/ijms21218165
80. Hamaoka M, Kobayashi T, Kuroda S, et al. Hepatectomy after down-staging of hepatocellular carcinoma with portal vein tumor thrombus using chemoradiotherapy: a retrospective cohort study. Int J Surg. 2017;44:223–228. doi:10.1016/j.ijsu.2017.06.082
81. Chong JU, Choi GH, Han DH, et al. Downstaging with localized concurrent chemoradiotherapy can identify optimal surgical candidates in hepatocellular carcinoma with portal vein tumor thrombus. Ann Surg Oncol. 2018;25(11):3308–3315. doi:10.1245/s10434-018-6653-9
82. Zhang Z, Liu Q, He J, et al. The effect of preoperative transcatheter hepatic arterial chemoembolization on disease-free survival after hepatectomy for hepatocellular carcinoma. Cancer. 2000;89(12):2606–2612. doi:10.1002/1097-0142(20001215)89:12<2606::AID-CNCR13>3.0.CO;2-T
83. Shi HY, Wang S-N, Wang SC, et al. Preoperative transarterial chemoembolization and resection for hepatocellular carcinoma: a nationwide Taiwan database analysis of long-term outcome predictors. J Surg Oncol. 2014;109(5):487–493. doi:10.1002/jso.23521
84. Lee KT, Lu Y-W, Wang S-N, et al. The effect of preoperative transarterial chemoembolization of resectable hepatocellular carcinoma on clinical and economic outcomes. J Surg Oncol. 2009;99(6):343–350. doi:10.1002/jso.21248
85. Yeh ML, Huang C-I, Huang C-F, et al. Neoadjuvant transcatheter arterial chemoembolization does not provide survival benefit compared to curative therapy alone in single hepatocellular carcinoma. Kaohsiung J Med Sci. 2015;31(2):77–82. doi:10.1016/j.kjms.2014.11.003
86. Chua TC, Liauw W, Saxena A, et al. Systematic review of neoadjuvant transarterial chemoembolization for resectable hepatocellular carcinoma. Liver Int. 2010;30(2):166–174. doi:10.1111/j.1478-3231.2009.02166.x
87. Zhou WP, Lai ECH, Li A-J, et al. A prospective, randomized, controlled trial of preoperative transarterial chemoembolization for resectable large hepatocellular carcinoma. Ann Surg. 2009;249(2):195–202. doi:10.1097/SLA.0b013e3181961c16
88. Han G, Berhane S, Toyoda H, et al. Prediction of survival among patients receiving transarterial chemoembolization for hepatocellular carcinoma: a response-based approach. Hepatology. 2020;72(1):198–212. doi:10.1002/hep.31022
89. Korita PV, Wakai T, Shirai Y, et al. Multidrug resistance-associated protein 2 determines the efficacy of cisplatin in patients with hepatocellular carcinoma. Oncol Rep. 2010;23(4):965–972. doi:10.3892/or_00000721
90. Lin H, Li X, Liu Y, et al. Neoadjuvant radiotherapy provided survival benefit compared to adjuvant radiotherapy for hepatocellular carcinoma. ANZ J Surg. 2018;88(10):E718–E724. doi:10.1111/ans.14387
91. Wei X, Jiang Y, Zhang X, et al. Neoadjuvant three-dimensional conformal radiotherapy for resectable hepatocellular carcinoma with portal vein tumor thrombus: a randomized, open-label, multicenter controlled study. J Clin Oncol. 2019;37(24):2141–2151. doi:10.1200/JCO.18.02184
92. Wada H, Sasaki Y, Yamada T, et al. Successful preoperative treatment for hepatocellular carcinoma with tumor thrombus in the major portal branch by three-dimensional conformal radiation therapy–two case reports. Hepatogastroenterology. 2005;52(62):343–347.
93. Kamiyama T, Nakanishi K, Yokoo H, et al. Efficacy of preoperative radiotherapy to portal vein tumor thrombus in the main trunk or first branch in patients with hepatocellular carcinoma. Int J Clin Oncol. 2007;12(5):363–368. doi:10.1007/s10147-007-0701-y
94. Katz AW, Chawla S, Qu Z, et al. Stereotactic hypofractionated radiation therapy as a bridge to transplantation for hepatocellular carcinoma: clinical outcome and pathologic correlation. Int J Radiat Oncol Biol Phys. 2012;83(3):895–900. doi:10.1016/j.ijrobp.2011.08.032
95. Hasan S, Thai N, Uemura T, et al. Hepatocellular carcinoma with child Pugh-A Cirrhosis treated with stereotactic body radiotherapy. World J Gastrointest Surg. 2017;9(12):256–263. doi:10.4240/wjgs.v9.i12.256
96. Kulik LM, Carr BI, Mulcahy MF, et al. Safety and efficacy of 90Y radiotherapy for hepatocellular carcinoma with and without portal vein thrombosis. Hepatology. 2008;47(1):71–81. doi:10.1002/hep.21980
97. Salem R, Gordon AC, Mouli S, et al. Y90 radioembolization significantly prolongs time to progression compared with chemoembolization in patients with hepatocellular carcinoma. Gastroenterology. 2016;151(6):1155–1163 e2. doi:10.1053/j.gastro.2016.08.029
98. Tabone M, Calvo A, Russolillo N, et al. Downstaging unresectable hepatocellular carcinoma by radioembolization using 90-yttrium resin microspheres: a single center experience. J Gastrointest Oncol. 2020;11(1):84–90. doi:10.21037/jgo.2019.06.01
99. Labgaa I, Tabrizian P, Titano J, et al. Feasibility and safety of liver transplantation or resection after transarterial radioembolization with Yttrium-90 for unresectable hepatocellular carcinoma. HPB (Oxford). 2019;21(11):1497–1504. doi:10.1016/j.hpb.2019.03.360
100. Edeline J, Lenoir L, Boudjema K, et al. Volumetric changes after (90)y radioembolization for hepatocellular carcinoma in cirrhosis: an option to portal vein embolization in a preoperative setting? Ann Surg Oncol. 2013;20(8):2518–2525. doi:10.1245/s10434-013-2906-9
101. Lau WY, Sangro B, Chen P-J, et al. Treatment for hepatocellular carcinoma with portal vein tumor thrombosis: the emerging role for radioembolization using yttrium-90. Oncology. 2013;84(5):311–318. doi:10.1159/000348325
102. Lewandowski RJ, Donahue L, Chokechanachaisakul A, et al. 90 Y radiation lobectomy: outcomes following surgical resection in patients with hepatic tumors and small future liver remnant volumes. J Surg Oncol. 2016;114(1):99–105. doi:10.1002/jso.24269
103. Spreafico C, Sposito C, Vaiani M, et al. Development of a prognostic score to predict response to Yttrium-90 radioembolization for hepatocellular carcinoma with portal vein invasion. J Hepatol. 2018;68(4):724–732. doi:10.1016/j.jhep.2017.12.026
104. She WH, Cheung TT, Yau TCC, et al. Survival analysis of transarterial radioembolization with yttrium-90 for hepatocellular carcinoma patients with HBV infection. Hepatobiliary Surg Nutr. 2014;3(4):185–193. doi:10.3978/j.issn.2304-3881.2014.07.09
105. Vilgrain V, Pereira H, Assenat E, et al. Efficacy and safety of selective internal radiotherapy with yttrium-90 resin microspheres compared with sorafenib in locally advanced and inoperable hepatocellular carcinoma (SARAH): an open-label randomised controlled phase 3 trial. Lancet Oncol. 2017;18(12):1624–1636. doi:10.1016/S1470-2045(17)30683-6
106. Chow PKH, Gandhi M, Tan S-B, et al. SIRveNIB: selective internal radiation therapy versus sorafenib in asia-pacific patients with hepatocellular carcinoma. J Clin Oncol. 2018;36(19):1913–1921. doi:10.1200/JCO.2017.76.0892
107. Cheng S, Chen M, Cai J, et al. Chinese expert consensus on multidisciplinary diagnosis and treatment of hepatocellular carcinoma with portal vein tumor thrombus (2018 Edition). Liver Cancer. 2020;9(1):28–40. doi:10.1159/000503685
108. Shiina S, Tagawa K, Unuma T, et al. Percutaneous ethanol injection therapy for hepatocellular carcinoma. A histopathologic study. Cancer. 1991;68(7):1524–1530. doi:10.1002/1097-0142(19911001)68:7<1524::AID-CNCR2820680711>3.0.CO;2-O
109. Shiina S, Yasuda H, Muto H, et al. Percutaneous ethanol injection in the treatment of liver neoplasms. AJR Am J Roentgenol. 1987;149(5):949–952. doi:10.2214/ajr.149.5.949
110. Livraghi T, Festi D, Monti F, et al. US-guided percutaneous alcohol injection of small hepatic and abdominal tumors. Radiology. 1986;161(2):309–312. doi:10.1148/radiology.161.2.3020612
111. Lee SJ, Cho E-H, Kim R, et al. Hepatectomy, combined with intraoperative radiofrequency ablation in patients with multiple hepatocellular carcinomas. Korean J Hepatobiliary Pancreat Surg. 2015;19(3):98–102. doi:10.14701/kjhbps.2015.19.3.98
112. Amersi FF, McElrath-Garza A, Ahmad A, et al. Long-term survival after radiofrequency ablation of complex unresectable liver tumors. Arch Surg. 2006;141(6):
113. Pawlik TM, Izzo F, Cohen DS, et al. Combined resection and radiofrequency ablation for advanced hepatic malignancies: results in 172 patients. Ann Surg Oncol. 2003;10(9):1059–1069. doi:10.1245/ASO.2003.03.026
114. Xu LL, Zhang M, Yi P-S, et al. Hepatic resection combined with radiofrequency ablation versus hepatic resection alone for multifocal hepatocellular carcinomas: a meta-analysis. J Huazhong Univ Sci Technolog Med Sci. 2017;37(6):974–980. doi:10.1007/s11596-017-1836-3
115. Lu DS, Yu NC, Raman SS, et al. Percutaneous radiofrequency ablation of hepatocellular carcinoma as a bridge to liver transplantation. Hepatology. 2005;41(5):1130–1137. doi:10.1002/hep.20688
116. Barakat O, Wood RP, Ozaki CF, et al. Morphological features of advanced hepatocellular carcinoma as a predictor of downstaging and liver transplantation: an intention-to-treat analysis. Liver Transpl. 2010;16(3):289–299. doi:10.1002/lt.21994
117. Mazzaferro V, Battiston C, Perrone S, et al. Radiofrequency ablation of small hepatocellular carcinoma in cirrhotic patients awaiting liver transplantation: a prospective study. Ann Surg. 2004;240(5):900–909. doi:10.1097/01.sla.0000143301.56154.95
118. Yao FY, Kerlan RK, Hirose R, et al. Excellent outcome following down-staging of hepatocellular carcinoma prior to liver transplantation: an intention-to-treat analysis. Hepatology. 2008;48(3):819–827. doi:10.1002/hep.22412
119. Ersan V, Barut B, Yilmaz S. The timing of liver transplantation following downstaging: wait of not to wait? J Gastrointest Cancer. 2020;51(4):1152–1156. doi:10.1007/s12029-020-00491-z
120. Lu MD, Xu H-X, Xie X-Y, et al. Percutaneous microwave and radiofrequency ablation for hepatocellular carcinoma: a retrospective comparative study. J Gastroenterol. 2005;40(11):1054–1060. doi:10.1007/s00535-005-1671-3
121. Li Q, Chen K, Huang W, et al. Minimally invasive photothermal ablation assisted by laparoscopy as an effective preoperative neoadjuvant treatment for orthotopic hepatocellular carcinoma. Cancer Lett. 2021;496:169–178. doi:10.1016/j.canlet.2020.09.024
122. Thornton LM, Cabrera R, Kapp M, et al. Radiofrequency vs microwave ablation after neoadjuvant transarterial bland and drug-eluting microsphere chembolization for the treatment of hepatocellular carcinoma. Curr Probl Diagn Radiol. 2017;46(6):402–409. doi:10.1067/j.cpradiol.2017.02.006
123. Seki T, Wakabayashi M, Nakagawa T, et al. Percutaneous microwave coagulation therapy for patients with small hepatocellular carcinoma: comparison with percutaneous ethanol injection therapy. Cancer. 1999;85(8):1694–1702. doi:10.1002/(SICI)1097-0142(19990415)85:8<1694::AID-CNCR8>3.0.CO;2-3
124. Rizzo A. Tyrosine kinase inhibitors plus immune checkpoint inhibitors as neoadjuvant therapy for hepatocellular carcinoma: an emerging option? Expert Opin Investig Drugs. 2021;2021:1–3.
125. Pinato DJ, Fessas P, Sapisochin G, Marron TU. Perspectives on the neoadjuvant use of immunotherapy in hepatocellular carcinoma. Hepatology. 2020;74:483–490.
126. Dikilitas M. Why adjuvant and neoadjuvant therapy failed in HCC. Can the new immunotherapy be expected to be better? J Gastrointest Cancer. 2020;51(4):1193–1196. doi:10.1007/s12029-020-00497-7
127. Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359(4):378–390. doi:10.1056/NEJMoa0708857
128. Bruix J, Qin S, Merle P, et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;389(10064):56–66. doi:10.1016/S0140-6736(16)32453-9
129. Kudo M, Finn RS, Qin S, et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018;391(10126):1163–1173. doi:10.1016/S0140-6736(18)30207-1
130. Abou-Alfa GK, Meyer T, Cheng A-L, et al. Cabozantinib in patients with advanced and progressing hepatocellular carcinoma. N Engl J Med. 2018;379(1):54–63. doi:10.1056/NEJMoa1717002
131. Vitale A, Volk ML, Pastorelli D, et al. Use of sorafenib in patients with hepatocellular carcinoma before liver transplantation: a cost-benefit analysis while awaiting data on sorafenib safety. Hepatology. 2010;51(1):165–173. doi:10.1002/hep.23260
132. Truesdale AE, Caldwell SH, Shah NL, et al. Sorafenib therapy for hepatocellular carcinoma prior to liver transplant is associated with increased complications after transplant. Transpl Int. 2011;24(10):991–998. doi:10.1111/j.1432-2277.2011.01299.x
133. Hoffmann K, Ganten T, Gotthardtp D, et al. Impact of neo-adjuvant Sorafenib treatment on liver transplantation in HCC patients - a prospective, randomized, double-blind, Phase III trial. BMC Cancer. 2015;15:392. doi:10.1186/s12885-015-1373-z
134. Golse N, Radenne S, Rode A, et al. Liver transplantation after neoadjuvant sorafenib therapy: preliminary experience and literature review. Exp Clin Transplant. 2018;16(2):227–236. doi:10.6002/ect.2015.0299
135. Eilard MS, Andersson M, Naredi P, et al. A prospective clinical trial on sorafenib treatment of hepatocellular carcinoma before liver transplantation. BMC Cancer. 2019;19(1):568. doi:10.1186/s12885-019-5760-8
136. Ye SL, Chen X, Yang J, et al. Evaluation of sorafenib in Chinese unresectable hepatocellular carcinoma patients with prior surgery and portal vein tumor thrombosis: a subset analysis of GIDEON study data. Tumour Biol. 2017;39(3):1010428317695030. doi:10.1177/1010428317695030
137. Barbier L, Fuks D, Pessaux P, et al. Safety of liver resection for hepatocellular carcinoma after sorafenib therapy: a multicenter case-matched study. Ann Surg Oncol. 2013;20(11):3603–3609. doi:10.1245/s10434-013-3029-z
138. Bouattour M, Rosmorduc FL, Scatton O. BIOSHARE multicenter neoadjuvant phase 2 study: results of preoperative sorafenib in patients with resectable hepatocellular carcinoma (HCC)-From GERCOR IRC. J Clin Oncol. 2016;34:252–252. doi:10.1200/jco.2016.34.4_suppl.252
139. Mazzaferro V, Citterio D, Bhoori S, et al. Liver transplantation in hepatocellular carcinoma after tumour downstaging (XXL): a randomised, controlled, phase 2b/3 trial. Lancet Oncol. 2020;21(7):947–956. doi:10.1016/S1470-2045(20)30224-2
140. Demir T, Lee SS, Kaseb AO. Systemic therapy of liver cancer. Adv Cancer Res. 2021;149:257–294.
141. El-Khoueiry AB, Hanna DL, Llovet J, et al. Cabozantinib: an evolving therapy for hepatocellular carcinoma. Cancer Treat Rev. 2021;98:102221. doi:10.1016/j.ctrv.2021.102221
142. Bregni G, Vandeputte C, Pretta A, et al. Rationale and design of REGINA, a phase II trial of neoadjuvant regorafenib, nivolumab, and short-course radiotherapy in stage II and III rectal cancer. Acta Oncol. 2021;60(4):549–553. doi:10.1080/0284186X.2020.1871067
143. Dang RP, McFarland D, Le VH, et al. Neoadjuvant therapy in differentiated thyroid cancer. Int J Surg Oncol. 2016;2016:3743420. doi:10.1155/2016/3743420
144. Kim M, Ahn J, Song DE, et al. Real-world experience of lenvatinib in patients with advanced anaplastic thyroid cancer. Endocrine. 2021;71(2):427–433. doi:10.1007/s12020-020-02425-y
145. Berhane S, Fox R, García-Fiñana M, et al. Using prognostic and predictive clinical features to make personalised survival prediction in advanced hepatocellular carcinoma patients undergoing sorafenib treatment. Br J Cancer. 2019;121(2):117–124. doi:10.1038/s41416-019-0488-4
146. Nault JC, Villanueva A. Intratumor molecular and phenotypic diversity in hepatocellular carcinoma. Clin Cancer Res. 2015;21(8):1786–1788. doi:10.1158/1078-0432.CCR-14-2602
147. Jin H, Shi Y, Lv Y, et al. EGFR activation limits the response of liver cancer to lenvatinib. Nature. 2021;595(7869):730–734. doi:10.1038/s41586-021-03741-7
148. Subbiah V, Lassen U, Élez E, et al. Dabrafenib plus trametinib in patients with BRAF(V600E)-mutated biliary tract cancer (ROAR): a phase 2, open-label, single-arm, multicentre basket trial. Lancet Oncol. 2020;21(9):1234–1243. doi:10.1016/S1470-2045(20)30321-1
149. Rimassa L, Danesi R, Pressiani T, et al. Management of adverse events associated with tyrosine kinase inhibitors: improving outcomes for patients with hepatocellular carcinoma. Cancer Treat Rev. 2019;77:20–28. doi:10.1016/j.ctrv.2019.05.004
150. Matsui M, Machida S, Itani-Yohda T, et al. Downregulation of the proteasome subunits, transporter, and antigen presentation in hepatocellular carcinoma, and their restoration by interferon-gamma. J Gastroenterol Hepatol. 2002;17(8):897–907. doi:10.1046/j.1440-1746.2002.02837.x
151. Kurebayashi Y, Ojima H, Tsujikawa H, et al. Landscape of immune microenvironment in hepatocellular carcinoma and its additional impact on histological and molecular classification. Hepatology. 2018;68(3):1025–1041. doi:10.1002/hep.29904
152. Zhu AX, Ikeda FR, Sung MW, et al. A phase Ib study of lenvatinib (LEN) plus pembrolizumab (PEMBRO) in unresectable hepatocellular carcinoma (uHCC). J Clin Oncol. 2020;20(38):v286–v287.
153. Finn RS, Ikeda M, Zhu AX, et al. Phase Ib study of lenvatinib plus pembrolizumab in patients with unresectable hepatocellular carcinoma. J Clin Oncol. 2020;38(26):2960–2970. doi:10.1200/JCO.20.00808
154. Rusch VW, Johnson CJ, Wistuba II, et al. Neoadjuvant atezolizumab in resectable non-small cell lung cancer (NSCLC): initial results from a multicenter study (LCMC3). J Clin Oncol. 2018;20(36):8541.
155. Kaseb AO, Tran Cao DD, Abugabal YI, et al. Randomized, open-label, perioperative phase II study evaluating nivolumab alone or nivolumab plus ipilimumab in patients with resectable HCC. J Clin Oncol. 2020;1(38):185.
156. Schwacha-Eipper B, Minciuna I, Banz V, et al. Immunotherapy as a downstaging therapy for liver transplantation. Hepatology. 2020;72(4):1488–1490. doi:10.1002/hep.31234
157. Casak SJ, Donoghue M, Fashoyin-Aje L, et al. FDA approval summary: atezolizumab plus bevacizumab for the treatment of patients with advanced unresectable or metastatic hepatocellular carcinoma. Clin Cancer Res. 2021;27(7):1836–1841. doi:10.1158/1078-0432.CCR-20-3407
158. Finn RS, Qin S, Ikeda M, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382(20):1894–1905. doi:10.1056/NEJMoa1915745
159. Hack SP, Spahn J, Chen M, et al. IMbrave 050: a Phase III trial of atezolizumab pParklus bevacizumab in high-risk hepatocellular carcinoma after curative resection or ablation. Future Oncol. 2020;16(15):975–989. doi:10.2217/fon-2020-0162
160. Lee MS, Ryoo B-Y, Hsu C-H, et al. Atezolizumab with or without bevacizumab in unresectable hepatocellular carcinoma (GO30140): an open-label, multicentre, phase 1b study. Lancet Oncol. 2020;21(6):808–820. doi:10.1016/S1470-2045(20)30156-X
161. Pinato DJ, Fessas P, Sapisochin G, et al. Perspectives on the neoadjuvant use of immunotherapy in hepatocellular carcinoma. Hepatology. 2021;74(1):483–490. doi:10.1002/hep.31697
162. Iwamoto H, Shimose S, Noda Y, et al. Initial experience of atezolizumab plus bevacizumab for unresectable hepatocellular carcinoma in real-world clinical practice. Cancers. 2021;13(11):2786. doi:10.3390/cancers13112786
163. Torrens L, Montironi C, Puigvehí M, et al. Immunomodulatory effects of lenvatinib plus anti-PD1 in mice and rationale for patient enrichment in hepatocellular carcinoma. Hepatology. 2021;74:2652–2669. doi:10.1002/hep.32023
164. Yau T, Park JW, Finn RS, et al. Nivolumab versus sorafenib in advanced hepatocellular carcinoma (CheckMate 459): a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2022;23(1):77–90. doi:10.1016/S1470-2045(21)00604-5
165. Yau T, Kang Y-K, Kim T-Y, et al. Efficacy and safety of nivolumab plus ipilimumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib: the checkmate 040 randomized clinical trial. JAMA Oncol. 2020;6(11):e204564. doi:10.1001/jamaoncol.2020.4564
166. Ho WJ, Sharma G, Zhu Q, et al. Integrated immunological analysis of a successful conversion of locally advanced hepatocellular carcinoma to resectability with neoadjuvant therapy. J Immunother Cancer. 2020;8(2):e000932. doi:10.1136/jitc-2020-000932
167. Yarchoan M, Durham ZQ, Durham JN, et al. Feasibility and efficacy of neoadjuvant cabozantinib and nivolumab in patients with borderline resectable or locally advanced hepatocellular carcinoma (HCC). J Clin Oncol. 2021;39(suppl 3):335–335. doi:10.1200/JCO.2021.39.3_suppl.335
168. Zhu AX. A phase Ib study of lenvatinib (LEN) plus pembrolizumab (PEMBRO) in unresectable hepatocellular carcinoma (uHCC). ASCO, 2020(Abstract 4519). 2020.
169. Abou-Alfa GK, Chan SL, Kudo M, et al.Phase 3 randomized, open-label, multicenter study of tremelimumab (T) and durvalumab (D) as first-line therapy in patients (pts) with unresectable hepatocellular carcinoma (uHCC): HIMALAYA. Am Soc Clin Oncol. 2022. doi:10.1200/JCO.2022.40.4_suppl.379
170. Liu J, Blake SJ, Yong MCR, et al. Improved efficacy of neoadjuvant compared to adjuvant immunotherapy to eradicate metastatic disease. Cancer Discov. 2016;6(12):1382–1399. doi:10.1158/2159-8290.CD-16-0577
171. Yao FY, Mehta N, Flemming J, et al. Downstaging of hepatocellular cancer before liver transplant: long-term outcome compared to tumors within Milan criteria. Hepatology. 2015;61(6):1968–1977. doi:10.1002/hep.27752
172. Nordness MF, Hamel S, Godfrey CM, et al. Fatal hepatic necrosis after nivolumab as a bridge to liver transplant for HCC: are checkpoint inhibitors safe for the pretransplant patient? Am J Transplant. 2020;20(3):879–883. doi:10.1111/ajt.15617
173. Gassmann D, Weiler S, Mertens JC, et al. Liver allograft failure after nivolumab treatment-a case report with systematic literature research. Transplant Direct. 2018;4(8):e376. doi:10.1097/TXD.0000000000000814
174. Pinato DJ, Cortellini A, Sukumaran A, et al. PRIME-HCC: phase Ib study of neoadjuvant ipilimumab and nivolumab prior to liver resection for hepatocellular carcinoma. BMC Cancer. 2021;21(1):301. doi:10.1186/s12885-021-08033-x
175. Marron TU, Fiel MI, Hamon P, et al. Neoadjuvant cemiplimab for resectable hepatocellular carcinoma: a single-arm, open-label, phase 2 trial. Lancet Gastroenterol Hepatol. 2022;7(3):219–229. doi:10.1016/S2468-1253(21)00385-X
176. Kaseb AO, Vence L, Blando J, et al. Immunologic correlates of pathologic complete response to preoperative immunotherapy in hepatocellular carcinoma. Cancer Immunol Res. 2019;7(9):1390–1395. doi:10.1158/2326-6066.CIR-18-0605
177. Rozeman EA, Menzies AM, van Akkooi ACJ, et al. Identification of the optimal combination dosing schedule of neoadjuvant ipilimumab plus nivolumab in macroscopic stage III melanoma (OpACIN-neo): a multicentre, phase 2, randomised, controlled trial. Lancet Oncol. 2019;20(7):948–960. doi:10.1016/S1470-2045(19)30151-2
178. Foerster F, Galle PR. The current landscape of clinical trials for systemic treatment of HCC. Cancers. 2021;13(8):1962. doi:10.3390/cancers13081962
179. Tagliamonte M, Mauriello A, Cavalluzzo B, et al. Tackling hepatocellular carcinoma with individual or combinatorial immunotherapy approaches. Cancer Lett. 2020;473:25–32. doi:10.1016/j.canlet.2019.12.029
180. Kim JM, Cho SY, Rhu J, et al. Adjuvant therapy using ex vivo -expanded allogenic natural killer cells in hepatectomy patients with hepatitis B virus related solitary hepatocellular carcinoma: MG4101 study. Ann Hepatobiliary Pancreat Surg. 2021;25(2):206–214. doi:10.14701/ahbps.2021.25.2.206
181. Correction. Gastroenterology. 2021;160(4):1433. doi:10.1053/j.gastro.2021.02.025
182. Tseng HC, Xiong W, Badeti S, et al. Efficacy of anti-CD147 chimeric antigen receptors targeting hepatocellular carcinoma. Nat Commun. 2020;11(1):4810. doi:10.1038/s41467-020-18444-2
183. Sun B, Yang D, Dai H, et al. Eradication of hepatocellular carcinoma by NKG2D-based CAR-T cells. Cancer Immunol Res. 2019;7(11):1813–1823. doi:10.1158/2326-6066.CIR-19-0026
184. Ohira M, Hotta R, Tanaka Y, et al. Pilot study to determine the safety and feasibility of deceased donor liver natural killer cell infusion to liver transplant recipients with hepatocellular carcinoma. Cancer Immunol Immunother. 2021;71:589–599. doi:10.1007/s00262-021-03005-3
185. Sun C, Sun H-Y, Xiao W-H, et al. Natural killer cell dysfunction in hepatocellular carcinoma and NK cell-based immunotherapy. Acta Pharmacol Sin. 2015;36(10):1191–1199. doi:10.1038/aps.2015.41
186. Moehler M, Heo J, Lee HC, et al. Vaccinia-based oncolytic immunotherapy Pexastimogene Devacirepvec in patients with advanced hepatocellular carcinoma after sorafenib failure: a randomized multicenter Phase IIb trial (TRAVERSE). Oncoimmunology. 2019;8(8):1615817. doi:10.1080/2162402X.2019.1615817
187. Slovak R, Ludwig JM, Gettinger SN, et al. Immuno-thermal ablations - boosting the anticancer immune response. J Immunother Cancer. 2017;5(1):78. doi:10.1186/s40425-017-0284-8
188. Chu KF, Dupuy DE. Thermal ablation of tumours: biological mechanisms and advances in therapy. Nat Rev Cancer. 2014;14(3):199–208. doi:10.1038/nrc3672
189. Yoshida S, Kornek M, Ikenaga N, et al. Sublethal heat treatment promotes epithelial-mesenchymal transition and enhances the malignant potential of hepatocellular carcinoma. Hepatology. 2013;58(5):1667–1680. doi:10.1002/hep.26526
190. Obara K, Matsumoto N, Okamoto M, et al. Insufficient radiofrequency ablation therapy may induce further malignant transformation of hepatocellular carcinoma. Hepatol Int. 2008;2(1):116–123. doi:10.1007/s12072-007-9040-3
191. Marin HL, Furth EE, Olthoff K, et al. Histopathologic outcome of neoadjuvant image-guided therapy of hepatocellular carcinoma. J Gastrointestin Liver Dis. 2009;18(2):169–176.
192. Oyama A, Nouso K, Yoshimura K, et al. Randomized controlled study to examine the efficacy of hepatic arterial infusion chemotherapy with cisplatin before radiofrequency ablation for hepatocellular carcinoma. Hepatol Res. 2021;51(6):694–701. doi:10.1111/hepr.13633
193. Mehta A, Oklu R, Sheth RA. Thermal ablative therapies and immune checkpoint modulation: can locoregional approaches effect a systemic response? Gastroenterol Res Pract. 2016;2016:9251375. doi:10.1155/2016/9251375
194. den Brok MH, Sutmuller RPM, Nierkens S, et al. Synergy between in situ cryoablation and TLR9 stimulation results in a highly effective in vivo dendritic cell vaccine. Cancer Res. 2006;66(14):7285–7292. doi:10.1158/0008-5472.CAN-06-0206
195. Zhang Y, Zhang M-W, Fan -X-X, et al. Drug-eluting beads transarterial chemoembolization sequentially combined with radiofrequency ablation in the treatment of untreated and recurrent hepatocellular carcinoma. World J Gastrointest Surg. 2020;12(8):355–368. doi:10.4240/wjgs.v12.i8.355
196. Lemaire M, Lucidi V, Bouazza F, et al. Selective internal radiation therapy (SIRT) before partial hepatectomy or radiofrequency destruction for treatment of hepatocellular carcinoma in cirrhotic patients: a feasibility and safety pilot study. HPB (Oxford). 2018;20(7):641–648. doi:10.1016/j.hpb.2018.01.006
197. van den Bijgaart RJ, Eikelenboom DC, Hoogenboom M, et al. Thermal and mechanical high-intensity focused ultrasound: perspectives on tumor ablation, immune effects and combination strategies. Cancer Immunol Immunother. 2017;66(2):247–258. doi:10.1007/s00262-016-1891-9
198. Duffy AG, Ulahannan SV, Makorova-Rusher O, et al. Tremelimumab in combination with ablation in patients with advanced hepatocellular carcinoma. J Hepatol. 2017;66(3):545–551. doi:10.1016/j.jhep.2016.10.029
199. Liu D, Liu M, Su L, et al. Transarterial chemoembolization followed by radiofrequency ablation for hepatocellular carcinoma: impact of the time interval between the two treatments on outcome. J Vasc Interv Radiol. 2019;30(12):1879–1886. doi:10.1016/j.jvir.2019.07.029
200. Choe WH, Kim YJ, Park HS, Park SW, Kim JH, Kwon SY. Short-term interval combined chemoembolization and radiofrequency ablation for hepatocellular carcinoma. World J Gastroenterol. 2014;20(35):12588–12594. doi:10.3748/wjg.v20.i35.12588
201. Fujiki M, Aucejo F, Kim R. General overview of neo-adjuvant therapy for hepatocellular carcinoma before liver transplantation: necessity or option? Liver Int. 2011;31(8):1081–1089. doi:10.1111/j.1478-3231.2011.02473.x
202. Andreana L, Burroughs AK. Treatment of early hepatocellular carcinoma: how to predict and prevent recurrence. Dig Liver Dis. 2010;42(Suppl 3):S249–57. doi:10.1016/S1590-8658(10)60513-0
203. Zhu AX, Kang Y-K, Yen C-J, et al. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased alpha-fetoprotein concentrations (REACH-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(2):282–296. doi:10.1016/S1470-2045(18)30937-9
204. Zhu AX, Park JO, Ryoo B-Y, et al. Ramucirumab versus placebo as second-line treatment in patients with advanced hepatocellular carcinoma following first-line therapy with sorafenib (REACH): a randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol. 2015;16(7):859–870. doi:10.1016/S1470-2045(15)00050-9
205. Zhu AX, Baron AD, Malfertheiner P, et al. Ramucirumab as second-line treatment in patients with advanced hepatocellular carcinoma: analysis of REACH trial results by child-pugh score. JAMA Oncol. 2017;3(2):235–243. doi:10.1001/jamaoncol.2016.4115
206. Huang AC, Postow MA, Orlowski RJ, et al. T-cell invigoration to tumour burden ratio associated with anti-PD-1 response. Nature. 2017;545(7652):60–65. doi:10.1038/nature22079
207. Finn RS, Kudo M, Cheng A-L, et al. Pharmacodynamic biomarkers predictive of survival benefit with lenvatinib in unresectable hepatocellular carcinoma: from the phase III REFLECT study. Clin Cancer Res. 2021;27:4848–4858. doi:10.1158/1078-0432.CCR-20-4219
208. Xue F, Shi S, Zhang Z, et al. Application of a novel liquid biopsy in patients with hepatocellular carcinoma undergoing liver transplantation. Oncol Lett. 2018;15(4):5481–5488. doi:10.3892/ol.2018.8019
209. Sun YF, Xu Y, Yang X-R, et al. Circulating stem cell-like epithelial cell adhesion molecule-positive tumor cells indicate poor prognosis of hepatocellular carcinoma after curative resection. Hepatology. 2013;57(4):1458–1468. doi:10.1002/hep.26151
210. Bagaev A, Kotlov N, Nomie K, et al. Conserved pan-cancer microenvironment subtypes predict response to immunotherapy. Cancer Cell. 2021;39(6):845–865 e7. doi:10.1016/j.ccell.2021.04.014
211. Xu RH, Wei W, Krawczyk M, et al. Circulating tumour DNA methylation markers for diagnosis and prognosis of hepatocellular carcinoma. Nat Mater. 2017;16(11):1155–1161. doi:10.1038/nmat4997
212. He G, Chen Y, Zhu C, et al. Application of plasma circulating cell-free DNA detection to the molecular diagnosis of hepatocellular carcinoma. Am J Transl Res. 2019;11(3):1428–1445.
213. Memarnejadian A, Meilleur CE, Shaler CR, et al. PD-1 blockade promotes epitope spreading in anticancer CD8 + T cell responses by preventing fratricidal death of subdominant clones to relieve immunodomination. J Immunol. 2017;199(9):3348–3359. doi:10.4049/jimmunol.1700643
214. Liu J, Rozeman EA, O’Donnell JS, et al. Batf3 + DCs and type I IFN are critical for the efficacy of neoadjuvant cancer immunotherapy. Oncoimmunology. 2019;8(2):e1546068. doi:10.1080/2162402X.2018.1546068
215. Salmon H, Idoyaga J, Rahman A, et al. Expansion and activation of CD103(+) dendritic cell progenitors at the tumor site enhances tumor responses to therapeutic PD-L1 and BRAF inhibition. Immunity. 2016;44(4):924–938. doi:10.1016/j.immuni.2016.03.012
216. Yi C, Chen L, Lin Z, et al. Lenvatinib targets FGFR4 to enhance antitumor immune response of anti-PD-1 in hepatocellular carcinoma. Hepatology. 2021;74:2544–2560. doi:10.1002/hep.31921
217. Liu L, Bai X, Wang J, et al. Combination of TMB and CNA stratifies prognostic and predictive responses to immunotherapy across metastatic cancer. Clin Cancer Res. 2019;25(24):7413–7423. doi:10.1158/1078-0432.CCR-19-0558
218. Samstein RM, Lee C-H, Shoushtari AN, et al. Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat Genet. 2019;51(2):202–206. doi:10.1038/s41588-018-0312-8
219. Cristescu R, Mogg R, Ayers M, et al. Pan-tumor genomic biomarkers for PD-1 checkpoint blockade-based immunotherapy. Science. 2018;362(6411). doi:10.1126/science.aar3593
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.