Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 14
Nebulized Colistin And Continuous Cyclic Azithromycin In Severe COPD Patients With Chronic Bronchial Infection Due To Pseudomonas aeruginosa: A Retrospective Cohort Study
Authors Montón C, Prina E, Pomares X , Cugat JR , Casabella A, Oliva JC, Gallego M , Monsó E
Received 21 March 2019
Accepted for publication 9 August 2019
Published 17 October 2019 Volume 2019:14 Pages 2365—2373
DOI https://doi.org/10.2147/COPD.S209513
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Richard Russell
Concepción Montón,1,2 Elena Prina,1 Xavier Pomares,1,3 Jose R Cugat,4 Antonio Casabella,5 Joan Carles Oliva,6 Miguel Gallego,1,3 Eduard Monsó1,3
1Department of Respiratory Medicine, Hospital De Sabadell, Institut Universitari Parc Taulí-UAB, Sabadell, Spain; 2Health Services Research on Chronic Diseases Network-REDISSEC, Galdakao, Spain; 3CIBER de Enfermedades Respiratorias, CIBERES, Madrid, Spain; 4Department of Respiratory Medicine, Fundació Althaia, Manresa, Spain; 5Laboratory of Microbiology-UDIAT, Institut Universitari Parc Taulí-UAB, Sabadell, Spain; 6Epidemiology and Assessment Unit, Fundació Parc Taulí, Universitat Autònoma de Barcelona, Sabadell, Spain
Correspondence: Concepción Montón
Department of Respiratory Medicine, Hospital de Sabadell, Institut Universitari Parc Tauli-UAB, Parc Taulí 1, Sabadell, Barcelona 08208, Spain
Tel +34 93 7458262
Email [email protected]
Introduction: Long-term use of nebulized or oral antibiotics is common in the treatment of cystic fibrosis and non-cystic fibrosis bronchiectasis. To date, however, few studies have focused on the use of nebulized antibiotics in COPD patients. The aims of this study are: to establish whether a combination of nebulized colistin plus continuous cyclic azithromycin in severe COPD patients with chronic bronchial infection due to Pseudomonas aeruginosa reduces the frequency of exacerbations, and to assess the effect of this treatment on microbiological sputum isolates.
Material and methods: A retrospective cohort was created for the analysis of patients with severe COPD and chronic bronchial infection due to P. aeruginosa treated with nebulized colistin at the Respiratory Day Care Unit between 2005 and 2015. The number and characteristics of COPD exacerbations (ECOPD) before and up to two years after the introduction of nebulized colistin treatment were recorded.
Results: We analyzed 32 severe COPD patients who received nebulized colistin for at least three months (median 17 months [IQR 7–24]). All patients but one received combination therapy with continuous cyclic azithromycin (median 24 months [IQR 11–30]). A significant reduction in the number of ECOPD from baseline of 38.3% at two years of follow-up was observed, with a clear decrease in P. aeruginosa ECOPD (from 59.5% to 24.6%) and a P. aeruginosa eradication rate of 28% over the two-year follow-up.
Conclusion: In patients with severe COPD and chronic bronchial infection due to P. aeruginosa, combination therapy with nebulized colistin and continuous cyclic azithromycin significantly reduced the number of ECOPD, with a marked decrease in P. aeruginosa sputum isolates.
Keywords: COPD, exacerbation, azithromycin, colistin, Pseudomonas aeruginosa
Introduction
In patients with severe chronic obstructive pulmonary disease (COPD), infectious exacerbations (ECOPD) are associated with poor prognosis.1,2 Some interventions in this disease aim to prevent exacerbations, in order to improve the course of COPD.
Pseudomonas aeruginosa is responsible for 5–10% of COPD exacerbations.3 Patients with severe and very severe COPD have a higher risk of suffering exacerbations due to this potentially pathogenic microorganism (PPM) than patients with mild or moderate COPD.4,5 In a recent 3-year follow-up study conducted in a cohort of severe COPD patients, P. aeruginosa was isolated in nearly 35%. Moreover, 12% of patients with P. aeruginosa isolates developed chronic bronchial colonization by this PPM, which also presented clonal persistence.6 Recovery of P. aeruginosa has also been associated with increased mortality.3,7
Inhaled antibiotics are approved by the US Food and Drug administration and the European Medicines Agency for the treatment of chronic bronchial infection by P. aeruginosa in cystic fibrosis (CF). In these patients, Saiman et al8 also demonstrated that continuous cyclic azithromycin significantly improved lung function and reduced exacerbations when added to standard therapy, which included a nebulized antibiotic in more than half of the patients. Since then, chronic antibiotic treatment with inhaled antibiotics and oral macrolides has been used to treat chronic bronchial infection by P. aeruginosa in CF, and the extension of these treatments to non CF-bronchiectasis has been recommended in recent guidelines.9–11 The last update of the GOLD guidelines for COPD recommends the use of oral azithromycin treatment in COPD patients with frequent ECOPD despite triple inhaled therapy, and the use of nebulized antibiotics in cases with associated bronchiectasis.12 However, these recommendations are mainly based on consensus; the scientific evidence is scarce and only a few small studies have focused on the use of nebulized antibiotics in COPD patients.13–15
The aim of our study is to evaluate the efficacy of combination therapy with nebulized colistin and continuous cyclic azithromycin on the frequency of exacerbations in severe COPD patients with chronic bronchial infection due to P. aeruginosa, when these antibiotics are added to the baseline treatment. We also aim to assess the eradicating effect of this antibiotic combination on the chronic presence of this PPM in the bronchial tree.
Materials And Methods
Design And Patients
A retrospective cohort was created including all patients with severe COPD and chronic bronchial infection due to P. aeruginosa attending the Respiratory Day Care Unit at Sabadell Hospital, Barcelona, who initiated a treatment with nebulized colistin between 2005 and 2015, in combination with long-term oral azithromycin. Patients with bronchiectasis as a main diagnosis were excluded. Chronic bronchial infection due to P. aeruginosa was defined as the presence of three or more positive sputum samples, collected at one-month intervals, over at least six months.9
The patients were enrolled in the cohort when starting nebulized colistin and were followed for a period of up to two years from their inclusion. Annual events occurring one year before and two years after enrolment were assessed, including the number of ECOPD and sputum cultures with isolates of PPMs, in order to evaluate the efficacy of the chronic antibiotic therapy. Only patients who had used inhaled colistin for a minimum of three consecutive months in the study period were included in the present analysis. All patients underwent the same scheduled clinical assessments at the Respiratory Day Care Unit conducted by the same team of pulmonologists before and after starting nebulized therapy.16 Data were collected at each 3-month control visit while the patient was stable, and when exacerbations appeared. The study was approved by the Ethics Committee of the ParcTaulí University Hospital (CEIC number 2017/555) and was conducted in accordance with the Declaration of Helsinki. All patients provided written informed consent.
Nebulized Colistin And Continuous Cyclic Azithromycin
All patients received nebulized colistin in two possible forms of administration: 1) Generic nebulized colistin with Jet nebulizer (PARI-LC-PLUS®, PARI): 1,000,000–2,000,000 UI/12 hrs; or 2) Colistin Promixin® with I-neb adaptive aerosol delivery device (Philips Respironics): 500,000–1,000,000 UI/12 hrs. The treatment regimen was administered according to current national guidelines on bronchiectasis.9,17
The first doses were administered at the hospital for tolerance assessment, and the treatment was discontinued in patients with bronchospasm on the first administration of nebulized colistin. A forced spirometry was performed before and after the first dose of nebulized antibiotic for closer monitoring of adverse events, and any FEV1 reduction of more than 200 mL and 12% was also considered as a contraindication for continuing the treatment. Continuous cyclic azithromycin (500 mg three times for week orally) was introduced at the same time as nebulized colistin, or during the previous months, in the patients enrolled.
Definition And Treatment Of Exacerbations
ECOPD was defined as a sustained worsening of the patient’s condition from the stable state and beyond normal day-to-day variations, of acute onset, requiring a change in regular treatment (antibiotics with or without corticosteroids, with or without hospital admission) in a patient with underlying COPD.18 ECOPD were attended from Monday to Friday (8 am to 5 pm) at the Respiratory Day Care Unit, always by the same team of pulmonologists;16 outside these hours, exacerbations were attended at the emergency department. A chest radiograph was performed for each ECOPD to exclude acute pneumonia. ECOPD attended at primary care were also recorded. Antibiotics were prescribed during ECOPD according to Anthonisen’s criteria19 and were adjusted according to sputum culture antibiograms. Decisions regarding hospital admission were made in accordance with clinical practice guidelines.20 Nebulized colistin was stopped during hospital admissions.
Bacteriological Assessments
Sputum samples were collected from all patients for each ECOPD and during stable phases, in accordance with routine clinical practice, and were processed locally by Gram stain and bacteriological culture. All bacterial isolates were identified and antibiograms were performed following standardized microbiological protocols.21 Microorganisms isolated during ECOPD were classified into four groups: Group 1, common respiratory PPMs (Haemophilus influenzae, Streptococcus pneumoniae, Moraxella catarrhalis and methicillin-susceptible Staphylococcus aureus MSSA); Group 2, methicillin-resistant Staphylococcus aureus (MRSA); Group 3, Pseudomonas aeruginosa; and Group 4, other Gram-negative bacteria (Stenotrophomonas maltophilia, Achromobacter xylosoxidans, Serratia marcescens, Escherichia coli and Proteus mirabilis).
To evaluate the microbiological efficacy of the combination therapy of nebulized colistin and continuous cyclic azithromycin, eradication was defined as the absence of P. aeruginosa after the introduction of the treatment and during the first and second years of follow up.
Multidrug resistant P. aeruginosa was defined as resistance to at least one agent in three or more antibiotic groups, including beta-lactams (penicillins plus β-lactamase inhibitor, cephalosporins and carbapenems), quinolones, aminoglycosides, monobactam and polymyxins.22
Statistical Analysis
Means and standard deviations (SD) or medians and interquartile ranges (IQR) were used to summarize continuous variables, as appropriate. Absolute and relative frequencies were used for categorical variables. The number of exacerbations per year was adjusted according to the length of follow-up.
A random intercept linear mixed model for repeated measures with fixed effects of follow-up time was used to estimate the population mean of exacerbations, number of hospitalizations, and length of hospital stay at different time-points. These estimations allowed the calculation of the percentage changes over time from baseline. All statistical analyses were performed in R (The R Foundation for Statistical Computing Platform, version 3.5.0) and lme4 package for R.
Results
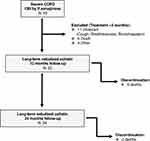
Fifty-three patients with severe COPD and chronic bronchial infection due to P. aeruginosa started treatment with nebulized colistin at Respiratory Care Day Unit at some point between 2005 and 2015. The main reason for discontinuation of nebulized colistin during the first three months was intolerance due to respiratory symptoms, mainly cough, breathlessness or bronchospasm, which was observed in 11 patients (20.8%). Of these patients with colistin intolerance, two presented bronchospasm at the first administration, while the remaining nine presented respiratory symptoms at home which obliged treatment withdrawal during the first three months. Ten additional patients who initiated nebulized colistin were excluded from the study due to death or other causes. Overall, 32 of the 53 patients who had initiated nebulized colistin had followed this treatment during at least three months, and were included in the study (Figure 1).
A good quality forced spirometry before and after the colistin nebulization trial (not showing significant changes) was obtained in 19 of these 32 participants (59.4%).
Baseline characteristics of the 32 participating patients are shown in Table 1. Bronchiectasis was identified in 25 patients in the computed tomography scan (78%) and most patients were treated with inhaled triple therapy (78%).The median (IQR) duration of treatment with nebulized colistin was 17 (7–24) months. Only 9% of patients (3/32) presented bronchospasm as a late adverse event (after the first 3-month period of treatment). Continuous cyclic azithromycin was used during colistin therapy by all patients but one (due to pharmacological interactions). The median duration of azithromycin therapy was 24 (IQR 11–30) months. Azithromycin was started before nebulized colistin in 17 patients (55%) and simultaneously in the remaining 14 (45%).
 |
Table 1 Baseline Characteristics Of COPD Patients Chronically Colonized By P. Aeruginosa |
Respiratory Exacerbations
In the 12 months prior to the start of nebulized colistin, the 32 study participants presented a total of 126 ECOPD, with a median of four ECOPD per patient [IQR 3–5]. Eighty-five (67%) were severe enough to require hospitalization (median of three admissions per patient [IQR 1–4]), representing a total hospital stay of 1625 days (median 49 days per patient [IQR 14.5–70]). Data regarding respiratory exacerbations during the first and second years of follow-up are detailed in Table 2, which shows a clear reduction in ECOPD, hospitalizations and length of hospital stay. The mixed model applied showed that combination therapy with nebulized colistin and continuous cyclic azithromycin attained an estimated mean reduction in ECOPD of 38.3% with respect to baseline after 24 months (Figure 2), and parallel reductions in hospitalizations and hospital stays of up to 41.5% and 41.9% respectively (Figures 3 and 4).The data of the fixed effects of the model applied for ECOPD, hospitalizations and days of hospital stay are detailed in Tables 3–5; they reached statistical significance for ECOPD frequency, and borderline significance for hospitalizations due to respiratory disease.
 |
Table 2 Respiratory Exacerbations Before And After Combination Therapy With Nebulized Colistin And Continuous Cyclic Azithromycin. For Differences Between Periods, See Table 3–5 |
 |
Table 3 Random Intercept Linear Mixed Model For Repeated Measures With Fixed Effects Of Follow-Up Time Used To Estimate The Population Mean Of ECOPD |
 |
Table 4 Random Intercept Linear Mixed Model For Repeated Measures With Fixed Effects Of Follow-Up Time Used To Estimate The Population Mean Of Hospitalizations Due To Respiratory Disease |
 |
Table 5 Random Intercept Linear Mixed Model For Repeated Measures With Fixed Effects Of Follow-Up Time Used To Estimate The Population Mean Of Days Of Hospital Stay Due To Respiratory Disease |
Microbiology
After nebulized colistin plus azithromycin therapy, ECOPD caused by P. aeruginosa decreased markedly during the first and second year of follow-up. ECOPD caused by common PPMs showed only small variations, with a slight decrease during the first year of follow-up and a slight increase during the second year. ECOPD caused by MRSA and other Gram-negative bacteria also showed a slight increase during the two years of follow-up (Table 6, Figure 5).
 |
Table 6 Number Of Microbiological Isolates And Percentages Adjusted Per Total Number Of ECOPD Before And After Combination Therapy With Nebulized Colistin And Continuous Cyclic Azithromycin |
P. aeruginosa sputum isolates (related to the number of sputum samples obtained) clearly decreased during follow-up, not only in exacerbations but also in the regular stable phase sampling (Figure 6). Eradication of P. aeruginosa in the first year of follow-up was observed in 15 patients (47%), and in nine patients cultures remained negative for this PPM after two years (28%). Therefore, 6/15 (40%) patients presented a recurrence of P. aeruginosa isolation during the second year.
P. aeruginosa sputum isolates were multidrug resistant to antibiotics in 14 patients at baseline (43.7%), and this percentage did not increase throughout follow-up (11 patients, 34.3%). No resistance to colistin was observed during follow-up.
Discussion
To our knowledge, this is the first study to examine the long-term clinical and microbiological effectiveness of the introduction of nebulized colistin in combination with continuous cyclic azithromycin for the treatment of patients with severe COPD, chronic bronchial infection due to P. aeruginosa and frequent exacerbations. This combination therapy achieved a significant reduction in the number of ECOPD during the 2-year follow-up, with a marked decrease in P. aeruginosa sputum isolates not only in exacerbations but also in stable periods, and with good tolerance in most of the patients.
Inhaled antibiotics are approved by the US Food and Drug administration and the European Medicines Agency for treatment of chronic bronchial infection due to P. aeruginosa in CF. In these patients, Saiman et al have also demonstrated that continuous cyclic azithromycin significantly improves lung function and reduces exacerbations.8 Treatment with inhaled antibiotics and oral macrolides has been extended to chronic bronchial infection by P. aeruginosa in non CF-bronchiectasis, and has been included in different guidelines as a consensus recommendation,9–11 in spite of the limited evidence available in non-CF patients.
Althoughit is well known that P. aeruginosa frequently causes exacerbations in severe COPD, which evolves to chronic bronchial colonization in over 10% of patients,6 so far the experience with inhaled antibiotics in COPD patients is very limited. Steinfort et al15 included four severe COPD in a group of 18 patients with chronic respiratory disease (non-CF bronchiectasis) and chronically infected by Gram-negative bacteria, who were treated with nebulized colistin for at least six months. These authors reported a slower decline in lung function with this treatment and an improvement in quality of life. Later, Dal Negro et al13 used nebulized tobramycin for 14 days in 13 patients with severe COPD colonized by multiresistant P. aeruginosa, finding a short-term reduction in local inflammation and a long-term reduction in bacterial load and severe exacerbations during a 6-month follow-up. More recently, Bruguera-Avila et al14 reported a prospective study of 36 COPD patients with chronic or intermittent bronchial colonization by P. aeruginosa treated with long-term nebulized colistin (at least three months of treatment and 1-year follow-up), in which they found reductions in the number and length of admissions after treatment initiation. Similarly, our data confirmed a decrease in the number of hospitalizations due to ECOPD in severe COPD patients colonized by P. aeruginosa, and demonstrated an additional effect on moderate exacerbations, with a significant reduction in the total number of ECOPD over the two years of treatment. This reduction, previously suggested in studies with a shorter follow-up,13,14 highlights the long-term beneficial effect of inhaled colistin in this clinical setting, when added to oral macrolides.
The beneficial effects of macrolides in COPD patients are mediated by the inhibition of quorum sensing and biofilm formation, mechanisms which have been proven particularly useful in reducing the virulence of P. aeruginosa and in facilitating the activity of antipseudomonal antibiotics.23 In a previous study conducted by our group24 to evaluate the usefulness of long-term cyclic azithromycin therapy in reducing exacerbation frequency, a small subgroup of patients with chronic P. aeruginosa colonization also treated with nebulized colistin was included, and presented a statistically significant reduction in ECOPD of 43%. These preliminary results suggest that the combination of long-term cyclic azithromycin therapy and nebulized antibiotics may be useful in this group of patients. Our group has previously confirmed that continuous cyclic azithromycin therapy as a single antimicrobial therapy, for more than one year, reduces the number of ECOPD due to common PPMs, albeit associated with a relative increase in ECOPD due to P. aeruginosa.25 Therefore, considering that patients treated with long-term cyclic azithromycin are susceptible to developing chronic bronchial infection by P. aeruginosa, the addition of nebulized antibiotics active against this PPM seems justifiable.
The fall in exacerbations in COPD patients colonized by P. aeruginosa in our study is paralleled by a marked decrease in P. aeruginosa sputum isolates, not only in exacerbations but also in stable phase. This may be considered as the main explanation of the fall in the number of ECOPD, while the number of ECOPD due to other microorganisms did not change significantly. These microbiological effects have not been reported in previous studies conducted in severe COPD patients,14 but are particularly relevant for the identification of the mechanisms underlying the observed improvement in the evolution of severe COPD patients when using the combined therapy. On this issue, we can only say that our eradication rate of P. aeruginosa at 1-year follow-up is slightly higher than that observed by Bruguera-Avila et al14 (47% versus 39%), probably due to the addition of cyclic azithromycin to the treatment and its indirect antimicrobial effects.
Although nebulized colistin was generally well tolerated in our study, adverse events such as bronchospasm were more frequent than in previous reports. Episodes of bronchospasm were not reported by Dal Negro et al13 and accounted for only 11% in the study conducted by Bruguera-Avila et al.14 However, the differences between these publications may be related to differences in study design, which means that comparison is difficult. The results of the present study point to the need for careful monitoring of COPD patients when initiating inhaled colistin, previous to their maintenance on long-term inhaled therapy.
This study has several limitations: its retrospective design, the limited sample size (with only men included), and the absence of a control group (either untreatred or treated only with azithromycin). However, among its strengths are the fact that it reflects the situation in real life and describes practices that are now recommended in clinical guidelines but are not backed as yet up by a large body of evidence.
In conclusion, this study shows that the use of nebulized colistin added to continuous cyclic azithromycin as a combination therapy in severe COPD patients with chronic bronchial infection by P. aeruginosa achieves a significant reduction in the number of ECOPD over a 2-year period, with a marked decrease in P. aeruginosa sputum isolates, not only in exacerbations but also in the stable phase, and relatively good tolerance. Further studies with prospective designs and with a larger number of patients are now needed to elucidate the role of each chronic antibiotic treatment (inhaled antibiotics and oral macrolides) and their interactions. These studies should also help to establish the most appropriate treatments for severe COPD patients with chronic bronchial infection by P. aeruginosa, and the timings of their introduction – a particularly important issue, given that this situation is often difficult to handle in clinical practice.
Acknowledgments
The authors thank Adela García, nurse at the Department of Respiratory Medicine, for her clinical assistance, and Michael Maudsley of the University of Barcelona’s Language Service for checking the English.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
None of the authors have any conflict of interests to declare regarding this manuscript.
References
1. Sethi S, Murphy TF. Infection in the pathogenesis and course of chronic obstructive pulmonary disease. N Engl J Med. 2008;359(22):2355–2365. doi:10.1056/NEJMoa0801936
2. Moreno A, Montón C, Belmonte Y, Gallego M, Pomares X, Real J. Causas de muerte en pacientes con EPOC grave. Factores pronósticos. Arch Bronconeumol. 2009;45(4):181–186. doi:10.1016/j.arbres.2008.09.004
3. Murphy TF. Pseudomonas aeruginosa in adults with chronic obstructive pulmonary disease. Curr Opin Pulm Med. 2009;15(2):138–142. doi:10.1097/MCP.0b013e328321861a
4. Eller J, Ede A, Schaberg T, Niederman MS, Mauch H, Lode H. Infective exacerbations of chronic bronchitis: relation between bacteriologic etiology and lung function. Chest. 1998;113(6):1542–1548. doi:10.1378/chest.113.6.1542
5. Miravitlles M, Espinosa C, Fernández-Laso E, Martos JA, Maldonado JA, Gallego M. Relationship between bacterial flora in sputum and functional impairment in patients with acute exacerbations of COPD. Study Group of Bacterial Infection in COPD. Chest. 1999;116(1):40–46. doi:10.1378/chest.116.1.40
6. Gallego M, Pomares X, Espasa M, et al. Pseudomonas aeruginosa isolates in severe chronic obstructive pulmonary disease: characterization and risk factors. BMC Pulm Med. 2014;14(1):103. doi:10.1186/1471-2466-14-103
7. Almagro P, Salvadó M, Garcia-Vidal C, et al. Pseudomonas aeruginosa and mortality after hospital admission for chronic obstructive pulmonary disease. Respiration. 2012;84(1):36–43. doi:10.1159/000331224
8. Saiman L, Marshall BC, Mayer-Hamblett N, et al. Azithromycin in patients with cystic fibrosis chronically infected with Pseudomonas aeruginosa: a randomized controlled trial. JAMA. 2003;290(13):1749–1756. doi:10.1001/jama.290.13.1749
9. Vendrell M, de Gracia J, Olveira C, et al. Diagnosis and treatment of bronchiectasis. Spanish Society of Pneumology and Thoracic Surgery. Arch Bronconeumol. 2008;44(11):629–640.
10. Martínez-García MÁ, Máiz L, Olveira C, et al. Normativa sobre el tratamiento de las bronquiectasias en el adulto. Arch Bronconeumol. 2018;54(2):88–98.
11. Polverino E, Goeminne PC, McDonnell MJ, et al. European respiratory society guidelines for the management of adult bronchiectasis. Eur Respir J. 2017;50(3):1700629. doi:10.1183/13993003.00711-2017
12. Global Initiative for Chronic Obstructive Lung Disease; 2019. Available from: www.goldcopd.org. Accessed March 1, 2019.
13. Dal Negro R, Micheletto C, Tognella S, Visconti M, Turati C. Tobramycin Nebulizer Solution in severe COPD patients colonized with Pseudomonas aeruginosa: effects on bronchial inflammation. Adv Ther. 2008;25(10):1019–1030. doi:10.1007/s12325-008-0105-2
14. Bruguera-Avila N, Marín A, Garcia-Olive I, et al. Effectiveness of treatment with nebulized colistin in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2017;12:2909–2915. doi:10.2147/COPD.S150887
15. Steinfort DP, Steinfort C. Effect of long-term nebulized colistin on lung function and quality of life in patients with chronic bronchial sepsis. Intern Med J. 2007;37(7):495–498. doi:10.1111/j.1445-5994.2007.01404.x
16. Pomares Amigó X, Montón Soler C. Respiratory day hospital: what have we learned? Med Clin (Barc). 2011;136(10):454–455. doi:10.1016/j.medcli.2009.07.028
17. Miravitlles M, Soler-Cataluña JJ, Calle M, et al. Spanish COPD guidelines (GesEPOC): pharmacological treatment of stable COPD. Aten Primaria. 2012;44(7):425–437. doi:10.1016/j.aprim.2012.04.005
18. Rodriguez-Roisin R. Toward a consensus definition for COPD exacerbations. Chest. 2000;117(5Suppl 2):398S–401S. doi:10.1378/chest.117.5_suppl_2.398S
19. Anthonisen NR, Manfreda J, Warren CP, Hershfield ES, Harding GK, Nelson NA. Antibiotic therapy in exacerbations of chronic obstructive pulmonary disease. Ann Intern Med. 1987;106(2):196–204. doi:10.7326/0003-4819-106-2-196
20. Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease 2017 report. GOLD executive summary. Am J Respir Crit Care Med. 2017;195(5):557–582. doi:10.1164/rccm.201701-0150WS
21. Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Twentieth Informational Supplement. Wayne (PA): Clinical and Laboratory Standards Institute; 2010.
22. Magiorakos A-P, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268–281. doi:10.1111/j.1469-0691.2011.03570.x
23. Aliberti S, Chalmers JD, Pletz MW, editors. Anti-Infectives and the Lung. Vol. 75. European Respiratory Society;2017.
24. Pomares X, Montón C, Espasa M, Casabon J, Monsó E, Gallego M. Long-term azithromycin therapy in patients with severe COPD and repeated exacerbations. Int J Chron Obstruct Pulmon Dis. 2011;6:449–456. doi:10.2147/COPD.S23655
25. Pomares X, Montón C, Bullich M, et al. Clinical and safety outcomes of long-term azithromycin therapy in severe COPD beyond the first year of treatment. Chest. 2018;153(5):1125–1133. doi:10.1016/j.chest.2018.01.044
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.