Back to Journals » International Journal of Nanomedicine » Volume 16
Nanomedicine for the SARS-CoV-2: State-of-the-Art and Future Prospects
Authors Varahachalam SP, Lahooti B, Chamaneh M, Bagchi S, Chhibber T, Morris K , Bolanos JF , Kim NY, Kaushik A
Received 24 September 2020
Accepted for publication 25 December 2020
Published 22 January 2021 Volume 2021:16 Pages 539—560
DOI https://doi.org/10.2147/IJN.S283686
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 6
Editor who approved publication: Dr Thomas Webster
Sree Pooja Varahachalam,1 Behnaz Lahooti,1 Masoumeh Chamaneh,1 Sounak Bagchi,1 Tanya Chhibber,1 Kevin Morris,2 Joe F Bolanos,3 Nam-Young Kim,4 Ajeet Kaushik5
1Department of Pharmaceutical Sciences, Jerry H. Hodge School of Pharmacy, Texas Tech University Health Sciences Center (TTUHSC), Amarillo, TX 79106, USA; 2Maharashtra University of Health Sciences (MUHS), Nashik, Maharashtra 422004, India; 3Facultad De Ciencias De La Salud “Dr.Luis Edmundo Vasquez” Santa Tecla, Universidad Dr. Jose Matias Delgado, Cd Merliot, El Salvador; 4RFIC Bio Center, Department of Electronics Engineering, Kwangwoon University, Seoul 01897, South Korea; 5NanoBioTech Laboratory, Department of Natural Sciences, Division of Sciences, Art, and Mathematics, Florida Polytechnic University, Lakeland, FL 3385, USA
Correspondence: Ajeet Kaushik
NanoBioTech Laboratory, Department of Natural Sciences, Division of Sciences, Art, and Mathematics, Florida Polytechnic University, Lakeland, FL 3385, USA
Email [email protected]
Abstract: The newly emerged ribonucleic acid (RNA) enveloped human beta-coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection caused the COVID-19 pandemic, severely affects the respiratory system, and may lead to death. Lacking effective diagnostics and therapies made this pandemic challenging to manage since the SARS-CoV-2 transmits via human-to-human, enters via ACE2 and TMPSSR2 receptors, and damages organs rich in host cells, spreads via symptomatic carriers and is prominent in an immune-compromised population. New SARS-CoV-2 informatics (structure, strains, like-cycles, functional sites) motivated bio-pharma experts to investigate novel therapeutic agents that act to recognize, inhibit, and knockdown combinations of drugs, vaccines, and antibodies, have been optimized to manage COVID-19. However, successful targeted delivery of these agents to avoid off-targeting and unnecessary drug ingestion is very challenging. To overcome these obstacles, this mini-review projects nanomedicine technology, a pharmacologically relevant cargo of size within 10 to 200 nm, for site-specific delivery of a therapeutic agent to recognize and eradicate the SARS-CoV-2, and improving the human immune system. Such combinational therapy based on compartmentalization controls the delivery and releases of a drug optimized based on patient genomic profile and medical history. Nanotechnology could help combat COVID-19 via various methods such as avoiding viral contamination and spraying by developing personal protective equipment (PPE) to increase the protection of healthcare workers and produce effective antiviral disinfectants surface coatings capable of inactivating and preventing the virus from spreading. To quickly recognize the infection or immunological response, design highly accurate and sensitive nano-based sensors. Development of new drugs with improved activity, reduced toxicity, and sustained release to the lungs, as well as tissue targets; and development of nano-based immunizations to improve humoral and cellular immune responses. The desired and controlled features of suggested personalized therapeutics, nanomedicine, is a potential therapy to manage COVID-19 successfully. The state-of-the-art nanomedicine, challenges, and prospects of nanomedicine are carefully and critically discussed in this report, which may serve as a key platform for scholars to investigate the role of nanomedicine for higher efficacy to manage the COVID-19 pandemic.
Keywords: COVID-19, SARS-CoV-2 viral infection, nanomedicine, personalized COVID-19 management, drug delivery
Introduction: Emergence of the COVID-19 Pandemic
The novel coronavirus disease 2019 (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), led to the current global pandemic, which started in Wuhan city, China, in December 2019 and led to a public health crisis. Initially classified as an outbreak of pneumonia of unknown etiology, it was subsequently termed as COVID-19 by the World Health Organization (WHO) and declared as a pandemic in March 2020.1 As witnessed, over time, various deadly viruses (Figure 1) emerged and transmitted to humans from multiple animal species. Notably, all these viruses are incredibly infectious, having transmissibility between humans, which has affected many lives and economies of the world.2 As of Jan 04, 2021, there have been 85,635,955 confirmed cases of COVID-19, including 1,853,813 deaths, reported to the World Health Organization (WHO).3 Coronaviruses (CoVs) classified into four genera, ie, α, β, γ, and δ, of which α-CoV and β-CoV are reported to infect humans.4 Six CoVs were previously identified as viruses to which humans are susceptible. Of these, four are regarded as low pathogenic CoVs.5 The other two- the SARS-CoV and the MERS-CoV are highly pathogenic and potentially fatal. The SARS-CoV-2 is the seventh discovered member of the family of CoVs. Based on the virus genome replication analysis and genetic research, bats are suspected of being the virus-host reservoir. Bats could transmit the SARS-CoV-2 through intermediate hosts, which have yet to be confirmed as having infected humans.
It was reported that SARS-CoV-2 virus transmission occurs through respiratory droplets. Besides this, it was also shown that the virus transmission could occur through direct personal contact and via aerial droplets.6,7 Recent findings have suggested that aerosol is a potential mode of the SARS-CoV-2 virus transmission. The report further explained that the SARS-CoV-2 virus would remain infectious on fomite surfaces for 5–8 hours and in aerosols for 5–7 days.8
Considering the effects of the current COVID-19 pandemic on the global economy and public care, along with medical and diagnostic infrastructure shortages, scientists need to research the fabrication, production, and standardization of the SARS-CoV-2 rapid diagnostic tests and treatment.9 COVID-19 infection is due to human-to-human transmission, which demands diagnostics at point-of-care (POC) without experienced labor and sophisticated laboratories.10 Besides exploring sensors for quick testing and intelligent healthcare, efforts are being made to investigate novel therapeutics to manage the COVID-19 pandemic. This is proving very challenging due to variations in the SARS-CoV-2 virus structure with reference region and genomic profiling. In an optimized combination, the available drugs can treat the severe symptoms of COVID-19, but this is proven to be far from complete control. Despite these accomplishments, there is a gap in managing the COVID-19 pandemic successfully due to the lack of effective therapeutic agents and an efficient delivery mechanism at the infection site.
In advancing COVID-19 treatment and vaccine production, nanotechnology tools will play a pivotal role. In the absence of a specific antiviral against SARS-CoV-2, the multifaceted molecular interactions involved in viral infections are targeted by current therapeutics. They consist primarily of repurposing existing antiviral molecules used for other RNA viruses. To make these repurposed therapeutics safer and more effective, it is equally important to look for a suitable nanocarrier delivery technology. Intrinsically, nanocarriers are also very helpful in delivering multiple drugs with distinct physicochemical properties that promise the full potential of combination therapies.11,12
In vitro and in vivo blood-brain barrier (BBB) transmigration with substantial anti-HIV activity in primary central nervous system (CNS) cells has been demonstrated by multidrug-loaded (antiretrovirals, delay reactivating agents, and substance addiction antagonist) pegylated-magneto-liposomal nanoformulations. To target SARS-CoV-2 virus that has migrated to the CNS, this multifunctional nanotherapeutic strategy can be implemented.13 Strategies for the production of COVID-19 nano carrier-based vaccines are similarly important and often overlap as compared to nano carrier-based therapeutics.14,15
One of the bigger challenges in COVID-19 vaccine research is discovering methods that activate the immunity of both T cells and B cells towards this virus. The need to accelerate the production of precise “next-generation” vaccine solutions that can also target particular demographic subgroups or persons with weakened immunity is another obstacle.16 The mRNA-based COVID-19 vaccine, using lipid nanoparticles (LNPs) as a medium, is now being clinically tested. Pure mRNAs are susceptible to extracellular RNase degradation, so it is essential to formulate their delivery vehicle.16 The LNPs are virus-sized particles (80–200 nm) synthesized by the self-assembly of an ionizable cationic lipid.17 As seen by many studies, they have the ability to deliver mRNA efficiently into the cytoplasm. It is possible to achieve sustained-release mRNA expression kinetics and thus protein translation by opting for intramuscular and intradermal pathways, supplying high antibody titers, and immune responses to both B cells and T cells.18 The spectrum of nanocarrier distribution becomes highly important because most of the COVID-19 vaccine candidates are sophisticated biological moieties (DNA, mRNA, recombinant proteins, engineered APCs, etc.).19
This focus of this review is to exploring the possibilities of developing novel nanomedicine for recognizing and eradicating SARS-CoV-2 virus. It becomes essential to study the structure, mechanism of infection, and pathogenesis of this virus.
Investigating SARS-CoV-2
The SARS-CoV-2 has a genome size of 24.4–31.7 kb and is the largest RNA virus genome.20 The genome for this coronavirus consists of approximately 30,000 nucleotides. This codes for four structural proteins and other non-structural proteins (nsp). The structural proteins included are the nucleocapsid (N) protein, the membrane (M) protein, the spike (S) protein, and the envelope (E) protein.21–23 The nuclear capsid, or N-protein, connects to the single positive-strand RNA virus, enabling the virus to capture human cells and convert them into virus factories. The N-protein covers the viral RNA genome and plays a crucial role in its replication and transcription. The M-protein is the most prevalent in the viral surface and is known to be the primary protein for the coronavirus unit’s assembly.21 The S-protein is integrated across the viral surface; it mediates the viral attachment to the host cell’s surface receptors and the fusion of the viral and host cell membranes to facilitate viral entry into the host cell.24 The E-protein is a tiny membrane protein that ranges between 76 and 109 amino acids and is a minor portion of the viral particle.25
The open-reading-frame (ORF1a or ORF1ab) viral polyproteins assemble a collection of non-structural proteins (nsp) generated as cleavage products to promote viral replication and transcription.26 The main part, the RNA-dependent RNA polymerase [(RdRp), also known as nsp12], catalyzes the viral RNA synthesis and plays a central role in the COVID-19 replication and transcription process, likely with the aid of nsp7 and nsp8 as cofactors.27 Therefore, nsp12 is considered to be a primary candidate for analog antiviral nucleotide inhibitors such as remdesivir that looked promising for the treatment of COVID-19 infection.28,29
The transmission of SARS-CoV-2 into the host cell depends on the host cell surface’s encounter with the angiotensin-converting enzyme 2 (ACE2), which acts as a receptor. Upon association with ACE2, the viral S-protein is cleaved and activated by the Transmembrane serine protease 2 (TMPRSS2) and furin to bind into the host’s cell membrane (Figure 2).30–33 Renin-angiotensin-aldosterone system (RAAS) is essential in controlling processes such as blood pressure, wound healing, and inflammation; angiotensin-converting enzyme (ACE2) plays a vital role in this biochemical pathway.34 ACE2 receptor is commonly found in humans in the airway epithelium, kidney cells, small intestine, lung parenchyma, vascular endothelium, and in the CNS.35 The TMPRSS2 is a protein on the cell surface mainly expressed in the respiratory and digestive tract by endothelial cells. As a serine protease, it is involved within the active site in the cleaving peptide bonds of proteins that have serine as the nucleophilic amino acid.36 The association of the SARS-CoV-2 with its ACE2 receptor is an appealing drug target, since receptor-binding domain (RBD) epitopes on the spike protein will serve as leads in the design of successful inhibitors for entry.37 The life cycle of the virus with the host is comprised of subsequent five steps: attachment, penetration, biosynthesis, maturation, and release.
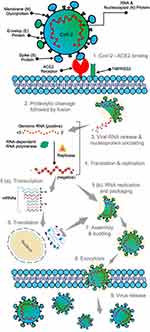 |
Figure 2 Mechanism of SARS-CoV-2 infects human host cells from an initial spike association with ACE2 and TMPRSS2 (Copyright ACS 2020).Notes: Reprinted with the permission from Chauhan G, Madou MJ, Kalra S, Chopra V, Ghosh D, Martinez-Chapa SO. Nanotechnology for COVID-19: therapeutics and Vaccine Research. ACS Nano. 2020;14(7):7760–7782. Copyright ACS under ACS COVID-19 Subset.19 |
The viruses bind to the host receptors (attachment), and then they penetrate the host cells via endocytosis or membrane fusion (penetration). When the viral components are released into the host cells, the viral RNA enters the nucleus for the replication process. The viral mRNA is utilized to produce viral proteins (biosynthesis). Finally, new viral particles are generated (maturation) and released.38 The coronavirus S-protein is then exposed to proteolytic cleavage through the host proteases (ie, furin and trypsin) in two sites situated at the boundary between the S1 and S2 subunits (S1/S2 site). Subsequently, to release the fusion peptide, the S2 domain (S2 site) cleavage takes place.21 The S glycoprotein comprises two subunits, mainly S1 and S2. S1 defines the host range and cellular tropism and enables viral attachment with the target cells. S2 is a component that facilitates the fusion of viral and cellular membranes, confirming viral entry through endocytosis. The affinity between the virus’s surface proteins and its receptors is a vital stage for viral entrance.39
One study conducted to explore the genomic variants of the SARS-CoV-2 involved a comprehensive investigation of 12,343 SARS-CoV-2 genome sequences isolated from patients by comparing them with reference to the SARS-CoV-2 sequence. It was identified that a total of 1234 mutations have occurred. In correlation analysis, it was found that variants of ORF1ab and S protein 614G, which are in strong association, displayed significant positive associations with fatality rates (r=0.41, P=0.029, and r=0.43, P=0.022, respectively). For the SARS-CoV-2 genome, it was found that there are several variants, and since December 2019, the D614G clade has become the most common variant.40,41
The primary symptom of COVID-19 infection is fever, which may include other symptoms such as sore throat, difficulty breathing, dry cough, stomach ache, dizziness, chest pain, diarrhea, vomiting, and nausea.42 COVID-19 emerged in clusters and primarily impacted the pulmonary system, with some cases advancing quickly to acute respiratory distress syndrome (ARDS).43,44 The ARDS results from an intense endogenous inflammatory reaction triggered by direct or indirect injuries to the lungs. It was indicated that the COVID-19 related ARDS is primarily due to the injury to the alveolar epithelial cells. Several patients have an insufficient level of oxygenation index, which suggests severe respiratory failure.45 Cardiovascular problems such as myocarditis, pericarditis, heart failure, and vasculitis may also occur in patients with COVID-19 infection.46–48 Some patients are experiencing a significant ischemic stroke. Since the SARS-CoV-2 reduces the expression of ACE2, it can be hypothesized that patients infected with the SARS-CoV-2 are at greater risk of hemorrhagic stroke.49 Respiratory viruses can reach the CNS through the retrograde hematogenic or neuronal pathway. For the former, an infectious agent that utilizes the bloodstream invades the CNS. In the second, a virus infects the peripheral nerves and uses the axonal transmission mechanism to enter the CNS.50 The neuro-invasive potential of the SARS-CoV-2 was discussed in recent studies; also, a few infected subjects showed neurological symptoms. New multicenter research has reported recurrent gustatory and olfactory impairments (ie, more than 80% of the cohort) in the patients involved.51 Possible pathogenic pathways that could negatively impact the CNS of COVID-19 patients are aACE2, immune rampage or systemic inflammation, hypoxia lesion, the trajectory of blood supply, and neuronal pathway (Direct injury to infection).52–56 Cases of COVID-19 patients who possibly showed the effect of neurological manifestations have reported.57,58
Unlike its pathogenic ancestors, COVID-19 may often directly affect the function of the peripheral nervous system.56 Recently, it was observed that two groups of cells in the nose are the most probable initial points of contamination for viral entry into the peripheral nervous system (PNS). These two groups of cells are called goblet cells and ciliated cells. These two types of cells are linked because they both contain high ACE2 receptor concentrations, which enable the SARS-CoV-2 to enter the host cells.59 They also contain high concentrations of another protease named TMPRSS2. COVID-19 has a peripheral neural involvement that occurs when ACE2 receptors interact with the viral agent. These ACE2 receptors are located along with the tongue, throat, and nasal passages indicating the likelihood of the virus affecting the nervous system and having a considerable effect on certain senses.56
A few studies have demonstrated the SARS-CoV-2 association in other organs,27 including the liver and kidneys, impairing the digestive system and excretion of the drugs taken to treat the infection.60 The frequency of hepatic abnormalities altogether increments after disease with COVID-19 and amid the course of the infection, which may show the impact of the SARS-CoV-2 on the liver.61 Moreover, steatosis and liver damage within the liver biopsies of COVID-19 patients have been reported.62 Some studies have also reported an increased rate of intense renal damage following COVID-19. Higher mortality rates have been reported in patients with acute renal injury when compared to other patients.63,64
Initial progress for COVID-19 prevention and management was identified to be filtering out the potential of asymptomatic carriers giving rise to new infections. Asymptomatic COVID-19 carriers have been reported to be able to trigger their immediate associates.65 The viral load of an asymptomatic carrier is reportedly similar to that of symptomatic patients. Thus asymptomatic, or minimally symptomatic patients are likely to spread the virus.66 During the COVID-19 outbreak in Wuhan, one particular asymptomatic child (age 10) with a travel background from Shenzhen to Wuhan was identified in the COVID-19 family cluster study.43 He was quantitative Real-Time Polymerase Chain Reaction (qRT-PCR) positive for genes encoding this novel coronavirus, with confirmed Sanger sequencing. The child was confirmed virologically to have an asymptomatic infection.67 Besides, in one family, the father had signs of illness, reduced lymphocyte production, irregular Computed Tomography (CT) images of his chest, and a positive result on qRT-PCR for COVID-19. It is noteworthy that both the mother and the son were asymptomatic, with regular numbers of lymphocytes and chest CT scan images with positive COVID-19 infection for qRT-PCR.65 Asymptomatic carriers are more presumable participants than symptomatic patients. There is an assumption that the asymptomatic patients played a partial role in increasing the number of affected nations 10–20 days after the initial outbreak was reported. Face masking was recommended in crowded areas to stop the virus from transmitting to other uninfected individuals so that it might also deter asymptomatic carriers from spreading the virus (Figure 3).66
 |
Figure 3 Importance of facial covering for the control of on-going pandemic (Copyright ACS 2020).Notes: Reprinted with the permission from Kaushik AK, Dhau JS, Gohel H, et al. Electrochemical SARS-CoV-2 Sensing at Point-of-Care and Artificial Intelligence for Intelligent COVID-19 Management. ACS Applied Bio Materials. 2020;3(11):7306–7325. Copyright ACS under ACS COVID-19 Subset.10 |
There is also a shortage of formal guidance for how to treat asymptomatic carriers. Regional screening and tracking of asymptomatic carriers by simple PCR-based assays could control the carriers’ viral transmission.66,68 A drawback may be that oropharyngeal or nasopharyngeal swabs do not contribute to a confirmatory diagnosis of asymptomatic carriers, and further sampling of the respiratory tract could be needed.66
The Emergence of Nanotechnology to Manage COVID-19
The pulmonary drug distribution is relatively complicated since defense mechanisms have been developed by the respiratory tract to keep inhaled drug particles out of the lungs and extract or inactivate them once ingested. Most inhaled drug products deliver particles with a size range < 5 μm, but possible benefits can be achieved by using nanoparticle formulations, that is, particles < 1 μm. By obtaining a more reliable distribution of the drug in alveolar areas, nanoparticles may boost drug targeting. Several medications have low aqueous solubility and reducing the size of particles to the nanometer scale is a promising way to increase solubility. Which contributes to a higher surface area per drug unit mass, which may increase the amount of substance that can dissolve in the lung’s fluid lining.69 Often, nanoparticles (NPs) have been used to resolve mucociliary clearance and alveolar macrophages as drug carriers. Studies have shown that NPs can deposit and escape from mucociliary clearance and alveolar macrophages in the lining fluid. Polyethylene glycol (PEG) has recently been commonly used as a micro/nanocarrier coating for various drug distribution purposes. By forming a hydration layer over the NPs that sterically inhibits biofouling and phagocytosis, PEG offers stealth functionality.70 Since SARS-CoV-2 itself is a respiratory virus; nanoparticles can be used effectively in targeting this virus.
Nanotechnology has become an integral part of pharmaceutical research over the past few years. Nanomedicine is a term designating the application of nanotechnology for medical purposes, for monitoring, diagnosing, prevention, control, and treatment of diseases. Experts believe that the solution for reducing the outbreak of COVID-19 could lie in nanomedicine. Several attempts have already been made to combat COVID-19 infection using nanotechnology-based approaches, including the development of highly effective antiviral disinfectants engineered by nanotechnology for increasing the surface disinfection effect, and the development of rapid COVID-19 diagnostic kits. Several nanomaterials have been used in POC devices because they have sensor beneficial properties that include chemical stability, strong electrical conductivity, and the effect of Localized Surface Plasmon Resonance (LSPR) increased sensitivity and specificity of detection systems can be achieved by using nanomaterials in analytical devices.71 At various stages of COVID-19 pathogenesis, nanoparticles can play an important role, given their inhibition ability during viral entry and infected cell protein fusion during initial attachment and membrane fusion.72
In response to changes in environmental conditions such as temperature, pH, electric or magnetic field, light, ultrasound, salt concentration, among others, stimuli-responsive polymers alter their properties (solubility, form, surface characteristics, etc.). Stimulus-responsive polymers provide many possibilities to incorporate nanostructure functionalities and allow the development of various intelligent systems for biomedical applications such as imaging, disease detection, managed drug delivery, and bio separation due to their specific properties.73
Nanomaterials effectively adsorb biomolecules and form a so-called biomolecular corona, depending on surface properties. If the selected nanomaterial is relatively biocompatible, this passive, nontargeted adsorption could bind viruses.74 In particular, the use of copper salt nanoparticles or solutions (chloride, iodide, sulfide, etc.) proven to have an antiviral effect may help create PPE with improving shielding properties. Polyurethane/CuO nanocomposites have recently been developed, acting as an efficient air purification antimicrobial filter (Figure 4).75
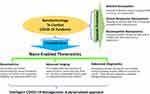 |
Figure 4 Projected nano-enabled technologies combating against COVID-19. |
It has been reported that before the acute respiratory distress syndrome and death resulting from COVID-19, there is a stage of viral-induced hyper inflammation due to a cytokine storm.76 Dormont et al proposed a novel formulation of prodrug-based nanoparticles (NPs) to mitigate uncontrolled inflammation, which was a multidrug nanoparticle, developed by conjugating squalene to adenosine before encapsulating it in α-tocopherol (an antioxidant). They claimed that, while using this technique, they could deliver the squalene encapsulated nano therapy in a targeted manner to the inflammation sites, leading to increased survival in an animal model of endotoxemia.77 To address the limitations of current antiviral therapy, nanomedicine has provided numerous approaches, making it a powerful tool to repurpose existing antiviral agents as possible therapeutic options for the treatment of COVID-19.19,78 Some basic challenges with conventional antiviral drug-delivery systems that can be overcome by nano-carrier-based systems, by modifying its pharmacokinetics/pharmacodynamics properties include low bioavailability, poor aqueous solubility, dose reduction, improved drug bioavailability, and reduced toxicity. Another major challenge is the need to maintain the presence of the antiviral drug at the reserve sites to reduce the chances of latency, as seen in the case of HIV patients despite suppressing the viral load in plasma.79 Using nanomedicine, targeted and controlled delivery to reserve sites can be achieved, which will help to lower the systemic toxic effects, attain the therapeutic concentration of a drug at these sites, and also mitigate the risk of viral rebound or poor patient compliance.19
Nanocarriers have been very useful for repurposing existing antiviral drugs, mimicking the cellular barriers, and being an effective tool for delivering biologicals like DNA, RNA, proteins, and neutralizing antibody (NAb), etc. Targeted nanocarriers with surface modifications can evade immune recognition and ensure the delivery of therapeutic compounds to the target cell, hence improving cellular uptake and efficacy. For the commercial success of an effective nanocarrier against the SARS-CoV-2, it is important to choose the right therapeutic candidate with the right nanocarrier and choose the right site to deliver the nanocarrier.19,80 In the future, nanotechnology-based approaches could help manage COVID-19 by developing an individual system by engineering nano theranostics to combine diagnostics and therapy or by providing personalized therapy for COVID-19 patients with chronic disease.81 Researchers propose that a reliable diagnostic or therapeutic strategy would be to use targeted drug delivery via nanostructured cargo directly to the infected cells. To use a similar approach, it becomes necessary to understand the cellular trail of the SARS-CoV-2 for combating this disease.82 Some companies have announced the intention to develop products such as DNA-Lipid nanoparticles and aerosolized mRNA nanoparticles.83 Exosomes (a type of extracellular vesicles) are biomolecular nanovesicles released from different cell types. Due to their unique biological properties and endogenous features, they have emerged as a promising candidate for use in theranostics application.84,85 Recently published literature suggests that exosomes can be used strategically to combat COVID-19 infection, especially mesenchymal stem cell-derived exosomes that may be an appealing candidate due to their various properties, including immunomodulatory, regenerative, and antimicrobial.86–88 Theranostic nanotechnologies can achieve the early diagnosis of thrombosis and thrombosis-inhibitor delivery to affected areas. NPs should be engineered to address certain biomarkers of thrombosis to be useful in detecting and delivering therapies. The latest approach for treating thrombosis by nanomedicine is that of delivering the nanoparticles, filled with antithrombotic agents, to the thrombosed site and targeting one or more proteins involved in coagulation. Thrombosed sites were detected using magnetic-particle imaging, with the use of iron oxide NP micelles with a fibrin-specific-binding peptide as well as bare NPs.89
Controlled or sustained-release drug nanocarriers are the safest remedy for alleviating the risk consequences of poor patient compliance and viral rebound during the treatment of viral infections. A nanomedicine approach is thus a potent tool for transmuting the antivirals, repurposing and enhancing COVID-19 therapeutic administration. Systemic toxicity, or immunotoxicity, is another significant concern, more particularly with protein and peptide-based drugs. Nanocarrier-based drug administration (comprised of biologic prodrugs) guarantees the enhanced half-life of biologicals by avoiding premature drug release and degradation and circumventing hepatic and renal clearance.19,90 Nanomedicine influences all medical disciplines and has been considered an essential tool for novel diagnostics, nanotherapeutics, vaccines, medical imaging, and the utilization of biomaterials for regenerative medicine. Soft materials on a nanometer scale, attained from polymeric NPs, surfactants (microemulsion, nano emulsions, liquid crystals), lipids (lipid-solid NPs, liposomes, nanostructured lipid carriers), and proteins NPs have been utilized in nanomedicine, specifically for drug delivery (Figure 5).72,91 The advantages offered by NPs for drug delivery systems include a high surface area; an organic or inorganic ionic surface charging range of biomaterial options; a targeted specificity via ligand binding (eg, tagging of antibodies), etc., as illustrated in (Figure 6). Transport to the CNS through the BBB is ideal with NPs of 100–150 nm in size.92 Biomedical uses of magnetic nanoparticles and surface engineering include Magnetic Resonance Imaging (MRI) and Magnetic Particle Imaging (MPI). Magnetic nanoparticles labeled with contrast agents will accomplish multimodal imaging with a combination of MRI or MPI and other imaging modalities such as X-ray, computerized tomography (CT) scan, positron emission tomography (PET), and photoacoustic (PA) imaging. Drug distribution, gene transfer, hyperthermia, and photodynamic therapy are the therapeutic applications that enable targeting particular diseases and cells so that that specific targeting can be obtained (Figure 7).93
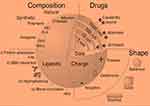 |
Figure 6 Schematic presentation of a nanomedicine.Notes: Reprinted with the permission from Kaushik A, Jayant RD, Bhardwaj V, Nair M. Personalized nanomedicine for CNS diseases. Drug Discov Today. 2018;23 (5):1007–1015. Copyright © 2017 Elsevier Ltd.92 |
 |
Figure 7 Nano-enabled multi-model approaches to manage targeted diseases.Notes: Reprinted from the publication Tomitaka A, Kaushik A, Kevadiya BD, et al. Surface-engineered multimodal magnetic nanoparticles to manage CNS diseases. Drug Discov Today. 2019;24(3):873–882. Copyright 2019 with the permission from Elsevier.93 |
NPs might use the indirect or direct mechanisms, which mainly affect the virus’s entry into the host cell, and result in fighting the SARS-CoV-2 viral infection. Indirect interaction of nanomaterials does not suppress viruses of their own; instead, they enhance the activity of antiviral therapy and are used for delivery, stabilization, and increased bioavailability. Nanomaterials with direct action, on the other hand, function as the active compound when they inactivate viruses on their own, usually by changing the composition of the virus or its genetic material.94 The impediment of the viral surface proteins could lead to virus inactivation; therefore targeted NPs, particularly to virus expressed proteins, may decrease the viral internalization. Metal NPs have demonstrated the capability to hinder viral attachment to the cell surface, directing to the blocking of viral internalization, thereby weakening the viral replication during viral entry. NPs comprised of titanium (Ti), gold (Au), silver (Ag), and zinc (Zn) have previously demonstrated effects against the influenza virus, herpes simplex virus, HIV, respiratory syncytial virus, monkeypox virus, transmissible gastroenteritis virus, and zika virus. Actively targeted nanocarriers can cross the biological barriers and achieve therapeutic concentrations in protected viral reservoirs. Additionally, it is possible to target a specific organ and cellular, intercellular sites engaged in the pathophysiology of the SARS-CoV-2.19
Nanomedicine to Combat Against the SARS-CoV-2
Nanomedicine, along with its physicochemical attributes, might be a promising therapeutic approach to overcome the struggle between CoVs and host cells. Nanoparticles (NPs), which are covered with antibodies or viral antigens, may be used against the SARS-CoV-2 and any resurfacing CoV.95 Organic NPs have been utilized for delivering antivirals like acyclovir, zidovudine, dapivirine, and efavirenz, and for enhancing drug bioavailability and boosting efficient delivery of drugs and targeted antiviral activity. The significant drawback of antivirals is the absence of specific targeting, ensuring in cytotoxicity of the host cell, which can be tackled by organic NPs. Antimicrobial medications have been assessed in clinical trials for COVID-19; for instance, chloroquine, ritonavir, lopinavir, ribavirin, and remdesivir have exhibited positive outcomes against the SARS-CoV-2 viral infection. Nanoencapsulation of antimicrobial drugs might provide for the advancement of safer therapies for COVID-19 and other viral diseases.72
The usage of polymerics, ie, multilayer NPs, is one method that originated from recent multidisciplinary sciences. They have usually created in a layer-by-layer (LbL) method by manipulating almost every form of interaction in polymer networks (eg, electrostatic, hydrogen bonding, covalent bonding, and unique recognition).96–98 In fact, usage of the LbL method has many benefits in designing the distribution mechanism for biomolecules, such as proteins and nucleic acids, which have minimal solubility in non-aqueous solutions and are vulnerable to denaturation.99–101 In one analysis carried out, multilayered exenatide-loaded NPs were prepared using the LbL method and were described as a sustained delivery device to treat type 2 diabetes.102 In a study conducted, RBD specific antibody column for purification was formed, and the RBD using the Spy Tag/Spy Catcher method was shown on self-assembling protein nanoparticles (SApNPs). The heptad repeats 2 (HR2) stalks was identified as a significant cause of spike metastability, developed an HR2-deleted glycine-capped spike (S2GΔHR2), and showed S2GΔHR2 with high yield, purity, and antigenicity on three SApNPs. S2G’HR2 elicited two-fold higher Nab (neutralizing antibody) titers than the proline-capped spike (S2P), while S2G’HR2 SApNPs elicited up to 10-fold higher NAb titers from multilayered E2p and I3-01v9 60-mers. The S2G-HR2-presenting I3-01v9 SApNPs also induced critically needed T-cell immunity, providing a candidate for the next generation vaccine to combat the pandemic of COVID-19 infection.103 Another study has properly combined the inherent characteristics of insulin NPs with the LbL multilayer-assembly technique to establish a sustained insulin release for a transcutaneous delivery.104 To prevent binding to extracellular serum proteins, multivalent nanomedicines may be engineered to potentially contribute to the elimination of viruses. Multivalent associations, such as cell-cell coordination and adhesion, are responsible for conducting various biological functions in normal cells. The reversibility of ligand-receptor interactions during infections is dependent on multivalency. Multivalent NPs are presented as promising antiviral agents capable of blocking the virus that binds to the host cells.105 Multivalent conjugates of PAMAM have shown effective antiviral activity against infections with influenza. A study found that by multivalent binding to a hemagglutinin trimer, multivalent 6ʹ-sialyllactose-polyamidoamine (6SL-PAMAM) conjugates could act as anti-influenza inhibitors and demonstrated strong inhibition of H1N1 infection. It has long been known that silver NPs are potential antimicrobial agents. Silver ions released from silver NPs bind to the viral RNA genome and activate mitochondrial interferon signaling pathways, activating mitochondrial proteins that may have an antiviral effect.106
With rapid developments in nanomedicine, magnetic nanoparticles (MNPs) have appeared as an encouraging theranostics tool in biomedical applications involving drug delivery, diagnostic imaging, and novel therapeutics. Substantial pre-clinical and clinical studies have demonstrated their targeted delivery, controlled drug release, functionalization, and image-guided capabilities. MNPs may accumulate at the targeted site through active or passive targeting to enhance the therapeutic efficacy by functionalization with targeting ligands and drugs.107,108 Among numerous described methodologies, static-magnetic- field-based- magnetically guided brain delivery systems have been proven to be harmless and noninvasive. Successful delivery of drug-loaded nanoformulations (NFs) to the mouse brain has previously been achieved by applying this method. This technique appears to be appropriate for delivering magnetic, drug-loaded NFs into the CNS to offer maximum efficiency in tackling specific neurological diseases. It is expected that the delivered therapeutics will have very modest or no adverse side-effects on the patient in terms of motor coordination, motor function, and toxicity.109 To extract the SARS-CoV-2 viral RNA for detection by real-time polymerase chain reaction (RT-PCR), magnetic nanoparticles were used in research. By imparting magnetic nature to nanoparticles through encapsulation of superparamagnetic iron oxide nanoparticles (SPIONs), which helped to isolate the nanoparticle-bound virus pathogen using an external magnet. The virus extracted maintained its infectivity and could be identified using several diagnostic assays for viruses.110
Imaging plays a significant part in the pre-clinical assessment of nanomedicine-based drug delivery systems, and it has offered vital perceptions into their mechanism of action and therapeutic effect. From a progressive viewpoint, it allows the noninvasive assessment of the biodistribution and pharmacokinetics (PK) of these drug delivery approaches in animal models of disease, permitting researchers to choose the best candidates.111 The advancement of novel NPs comprising both therapeutic and diagnostic elements has improved over the past decade. Theranostic NPs have been created for an array of imaging modalities involving MRI, optical imaging, computed tomography (CT), ultrasound, and nuclear imaging encompassing both positron emission tomography (PET) and single-photon emission computed tomography (SPECT).112
Lately, nanoparticle-drug-delivery systems (NDDS), together with stimuli-responsive behavior, have been exhibited and earned encouraging attention by researchers.95 Researchers investigated abundant stimulus-responsive nanoplatforms with high disease specificity for targeted imaging and treatments. These stimuli-responsive nanosystems modify their physiochemical or biological performance and when subjected to subsequent stimuli, release agents for diagnosis and therapy.113
Two methods can be adopted in devising stimuli-responsive drug delivery systems (DDS). The first approach is known as endogenous or internal stimuli, improving drug action specificity for diseased tissue. These include pH, redox, enzyme activity, heat, ionic strength, solvent polarity, and action of biomolecules. Physical or external stimuli are employed externally to targeted tissue after delivery of specific drug-laden nanocarriers in the second method. These exogenous stimuli consist of light, electric fields, magnetic fields, and ultrasound. Temperature can be recognized as either internal or external stimuli.80,114 Stimuli-responsive NPs can be utilized to deliver numerous bioactive components, such as vaccines, oligonucleotide peptides, antineoplastic agents, etc., which demonstrate excellent potential for clinical applications.115 By applying a stimuli-responsive DDS, the challenge of premature drug release can be solved. Consequently, a research trend has been diverted toward the direction of stimuli-responsive DDS. An amalgamation of two or more stimuli-responsive DDS has also been shown to enhance targeting efficiency.114
Integration of antigens in NPs can be attained by conjugation (covalent functionalization) or by encapsulation (physical entrapment). Investigations have revealed that NPs may shield antigens’ inherent structure from proteolytic degradation and enhance antigen delivery to antigen-presenting cells (APCs). Furthermore, NPs integrating antigens can exert a local depot outcome, confirming extended antigen presentation to immune cells. Remarkably, NPs have also shown inherent immunomodulatory activity.116 NPs used as unique immunotherapeutic platforms are desirable for numerous reasons. Firstly, these NPs can encapsulate a high density of bioactive components that can promote immunity against infection. Secondly, these delivery systems can be constructed from materials that can release drugs in a sustained manner. Finally, these delivery systems can be significantly modified to improve their bioactivity or targeting of the specific organs due to the flexibility of their synthesis and formulation.117
Our laboratory has demonstrated that the fabrication of nanomedicine formulations consists of a smart bio-compatible drug nanocarrier and an FDA approved, anti-HIV drug for site-specific eradication of HIV, even across the blood-brain barrier.118 Presently, chloroquine, an anti-malarial drug, is emerging as one of the potential drugs for managing COVID-19. Dr. Hu has explored the insights from nanomedicine into chloroquine efficacy against COVID-19.119 His research team stated that the SARS-CoV-2 protein might act like a nanoparticle due to its spherical shape and small size (2–600 nm) and could be uptaken by the host cell using a similar mechanism of endocytosis. Moreover, Dr. Kostas Kostarelos illustrated the peculiarity of virus particles as the most magnificent, smart, and efficient nanoparticles. Chloroquine is a weak base that interferes with acidification by being trapped in membrane-enclosed low pH organelles. In endoplasmic reticulum-Golgi intermediate compartment (ERGIC)-like structures, chloroquine can also inhibit Virion assembly. It has been demonstrated that chloroquine is an inhibitor of nanoparticle endocytosis by resident macrophages. Therefore, in response to clinically relevant doses of chloroquine, chloroquine reduces the accumulation of synthetic nanoparticles of different sizes (14–2,600 nm) and forms (spherical and discoidal) in cell lines, as well as in the mononuclear phagocyte system of mice.
Studies have shown that chloroquine decreases the expression of one of the three most abundant proteins in clathrin-coated pits, the phosphatidylinositol binding clathrin assembly protein (PICALM). The PICALM depletion has previously been shown to suppress endocytosis mediated by clathrin. One of the pathways responsible for chloroquine-mediated effects against SARS-CoV-2 can therefore be a general reduction in the capacity of cells to conduct clathrin-mediated endocytosis of nanosized structures due to suppression of PICALM.119
It is important to notice that many severe cases and deaths in patients with COVID-19 are due to an overreaction of the immune system, resulting in hyper-inflammation and macrophage activation syndrome (MAS). Hyper- inflammation and MAS result in an overproduction of pro-inflammatory cytokines, such as IL-1β, IL-6 and TNF-α (cytokine storm), as well as in coagulation abnormalities, which contribute to organ failure and fatalities.2,3 Dexamethasone is a steroid and anti-inflammatory. It has been stated that the administration of dexamethasone has lowered the number of COVID-19 associated deaths in patients by 35%.120 Based on the commonly accepted notion that NPs aggregate potently in macrophages upon intravenous administration and inhalation, the NFs of dexamethasone was proposed.121,122 Some long-lasting life-threatening SARS-CoV-2 symptoms include the development of cytokine storm, edema formation, and fibrosis development.121 Dexamethasone nanomedicines have been hypothesized to help in the day-to-day management of COVID-19 disease in three ways.4
First, the formulation of dexamethasone nanomedicine will help target inflammation-initiating and propagating phagocytic cells in the lung, blood, and myeloid and lymphoid tissues with the potent corticosteroid drug. This helps to better monitor the cytokine storm and MAS that have been involved in fatalities associated with COVID-19. As a result, it is expected that critically ill patients on ventilation or oxygen therapy can recover quicker and more effectively than on free-drug treatment.
Second, dexamethasone is an anti-edema drug that is particularly potent. By increasing drug availability and drug activity over time in hyper-activated immune cell populations in the lung’s inflamed sections, nano-formulating dexamethasone can further potentiate this effect.
Finally, dexamethasone is an anti-fibrotic drug that is highly active. In COVID-19, pulmonary fibrosis has recently emerged as a critical complication, especially in patients who have been ventilated for extended periods of time. Nanomedicine dexamethasone formulation will improve the anti-fibrotic action potential to meet an immediate medical requirement at this stage of COVID-19 management.
One research study was conducted on the rapid design and translation of microneedle arrays (MNAs) for SARS-CoV-2 vaccines capable of producing potent antigen-specific antibodies. MNA administration has proven to be more effective in triggering antibody neutralization for all vaccines. It was observed that the delivery of MERS-CoV-S1 vaccines by MNA induced greater humoral responses than traditional needles. In addition, transient mechanical stress occurring from the injection of microneedles will trigger a normal local innate immune response, which could act as a physical adjuvant to improve antigen-specific adaptive immunity. Stability studies are currently underway for MNA-based SARS-CoV-2 vaccines.123
To optimize the outcomes in patients with severe COVID-19 infections, macrophage-targeted nanomedicines may be used to attenuate cytokine storms and their related pathological effects.124 For macrophage targeting approaches, the nanoparticles used can differ considerably in their origin and structure, but macrophage uptake typically occurs by one of two distinct pathways: non-specific phagocytosis (passive targeting) controlled by physical properties of nanoparticles, or endocytosis (active targeting) mediated by receptors.125 These nanoparticles aggregate mainly at sites of infection or inflammation. Either natural or synthetic nanoparticles can be engineered through different methods to facilitate their active targeting of macrophages.124,126
One study has identified an effective medical countermeasure for COVID-19 by fabricating two types of cellular nanosponges. The nanosponges display the same type of receptors as those on which the viruses rely for cellular entry. Samples were tested for inhibitory effects on the human SARS-CoV-2 and displayed strong in-vitro antiviral efficacy. It was proposed that coronaviruses cannot invade their normal cellular targets while bound to nanosponges. The framework for nanosponges provides a distinct advantage over other therapies currently under development for COVID-19. To treat SARS-CoV-2 infection, the use of cellular nanosponges needs further research in suitable animal models.127
The latest findings reveal that 20%-30% of high-risk COVID-19 patients undergo blood clotting leading to stroke and sudden death. Rapid detection of proteins associated with blood clotting in COVID-19 patients’ plasma will save many lives. Many nanotechnology-based methods are being developed to diagnose patients at high risk of death due to complications from COVID-19 infections, including blood clots. Nanomedicine has great integration with proteomic strategies for disease identification and biomarker discovery applications.
Researchers have developed SARS-CoV-2-nucleocapsid-phosphoprotein-specific colorimetric assays that rely on gold nanoparticles coated with thiol-modified DNA antisense oligonucleotides, which could be used in the diagnosis of COVID-19 cases within 10 minutes.128 Such test kits could yield promising results, but the viral load quantity would also impact their performance. To overcome these limitations, researchers have come up with another favorable clinical diagnosis of COVID-19, consisting of a notable dual-functioning plasmonic biosensor that combines the plasmonic photothermal effect with the localized-surface-plasmon-resonance (LSPR) sensing transduction, and assisting in COVID-19 detection.
Surprisingly, this dual-functional LSPR biosensor showed high selectivity towards the sequences of the SARS-CoV-2 protein, with a very low detection limit of 0.22 pM.129 An ultra-sensitive field-effect-transistor (FET)-based biosensing system was developed by researchers in order to achieve rapid and accurate detection of the SARS-CoV-2 in clinical samples.130 The isolation and purification of selected molecules from biological fluids is another avenue where by the use of nanomaterials could lead to identification of the SARS-CoV-2 protein. Thus, nanomaterials with magnetic properties could be customized with unique virus receptors, resulting in virus molecules being attached to the NPs, that will facilitate their magnetic extraction using an external magnetic field. Consequently, nanomaterial-based detection can allow quicker and more precise identification of the virus, even at the initial stages of the infection, owing in large part to the versatility of the NPs surface alteration.131 Considering the multiple transmitting methods of the SARS-CoV-2, one strategy for tackling the virus has been to avoid its transmission by disinfecting the air, skin, or any other surfaces. Chemical disinfectants are also associated with disadvantages irrespective of their positive outcomes.132 Because of their extensive range of antiviral behavior and stability, and their potential to be successful at even lower dosages, metallic nanoparticles were suggested as alternatives. Also, the use of biodegradable nanomaterials could prevent the harmful effects of metallic nanomaterials on human health and the environment.75 Preliminary studies have indicated that a silver nanocluster/silica composite facial mask coating had viricidal effects on the SARS-CoV-2.133 Cellulose nanofibers are used by scientists to design a breathable and disposable filter cartridge, featuring the ability to filter off particles smaller than 100 nm. Nanomaterials can prevent viral transmission by inhibiting viral particle binding/penetration, either by facilitating surface oxidation, by releasing toxic ions, or by photothermal-based reactions.131
The nanomedicine hypothesis related to COVID-19 is studying the following approaches 1) exploring various drugs that can act as nano medications, and 2) exploring effective therapeutic agents that can further be formulated as nanomedicine and delivered at target tissues using an effective bio-compatible smart-drug nanocarrier.72 Investigating nano-therapy could also minimize the use of excessive anti-COVID-19 drug dosages, which exhibited adverse effects on the heart, retina, respiratory systems, etc., especially as associated with the elderly population. Recognizing the challenges of biotechnology involved with investigating future nano-therapies to manage COVID-19, we make the following recommendations:
- Investigate therapeutic agents that exhibit better efficacy without adverse effects.
- Optimize effective dosages and effective delivery at specific tissues using smart-drug nano carriers.
- Select drug nano carriers which are antiviral and anti-bacterial.
- Explore bio-functionalized nano-drug systems needed for high drug loading.
- Select stimuli-responsive nano-drug systems that can deliver to targeted tissues and control drug release.
- Design and develop nano-therapeutics that are specific to the liver, using immune-supportive formulations.
- Based on advancements, identify efficient therapeutic agents such as anti-malarial drugs, immuno-supportive agents, CRISPR-Cas9/gRNA, which can be delivered at required sites using nano-pharmacology approaches.
- Due to variations in selected therapeutic agents, design and develop nanomedicine that can eradicate the SARS-CoV-2.
- Customize nanomedicine formulations and performance according to the patient’s disease profile, and to research personalized nano-therapeutics.
Experts have suggested exploring nanomedicine as a potential therapy for managing COVID-19.134 Nanomedicine can be designed using a recommended therapeutic agent and an immuno-acceptable biomaterial to eradicate the SARS-CoV-2. The adjustable drug payload and the controlled drug release make nanomedicine suitable for personalized COVID-19 management. A multifunctional, efficient nano medication proposed to combat the SARS-CoV-2 is shown in Figure 8. To develop such a drug, federal support and public or private partnership is needed to fast-track research in this field and obtain timely FDA approval.
In the past decade, NP theranostics was described as a promising approach to deliver therapeutic substances effectively and selectively (ie, siRNA, drugs, peptide, vaccines) to target areas of infection. Furthermore, they permit supervising infectious sites and therapy responses employing noninvasive imaging modalities. Although intranasal delivery was proposed as the favored administration route for therapeutic moieties against viral pulmonary ailments, NP-based delivery approaches provide several advantages. This approach overcomes challenges linked with mucosal administration and guarantees a drug concentration that is many folds higher than anticipated in the selected areas of infection, while not presenting side effects on healthy cells.
Based on careful analysis of the advancements mentioned above, it is noticed that NPs might function as a crucial part of various phases of COVID-19 pathogenesis, taking into consideration their inhibitory role in the initial attachment and membrane fusion during viral entry and infected cell protein fusion. Irreversible damage by viruses and transcription, translation, and replication of viruses can be hindered by intracellular mechanisms, which can be initiated by nano-encapsulated drugs.72
Challenges
Physiological barriers such as mucus and alveolar fluid for intranasal and reticuloendothelial (RES) structures for systemic delivery are the main obstacles of nano-delivery. The nanoparticles need to be biodegradable and capable of supplying the chosen cell type with continuous drug delivery. The nano particle should be able to avoid the endocytic degradation machinery once within the cell.135 The NPs-established delivery methods have demonstrated considerable potential applications; however, investigations have revealed that these procedures might cause severe damage in respiratory sites and could even harm lung function. The four main pathobiological features, involving inflammation, genotoxicity, oxidative stress, and fibrosis, should be studied with the perspective of NPs and correlated methods.136 It is still not evident whether a nasal or oral nano-vaccine would attain a safer, prolonged mucosal immunity or extended protection against a virus. It is also still unclear whether adequate protection would arise when administered through various routes or to different age groups.137 Sekimukai et al reported that gold NP‐adjuvanted protein generated a powerful IgG reaction, but failed to enhance vaccine efficiency, or to diminish eosinophilic infiltration due to highly allergic inflammatory responses.138 From the perspective of nanomedicine, several nanomaterials have been established, varying from polymers to dendrimers, NPs, oligomers, liposomes, and small molecules. Nevertheless, fruitful clinical translation has been impeded by the point that, upon dilution, these nanomedicines lose their effectiveness as the virus compound-complex disassociates, causing the viruses to be set free, and to resume their replication cycle.74
The ROS are generated by nanoparticles causing oxidative stress due to disturbances in mitochondrial respiration or production of ROS in the cell. This can cause the initiation of harmful pathological processes within the cells, which leads to toxicity. Studies show that titanium dioxide (TiO2) nanoparticles accumulate in the lungs of mice after 90 days of the NP’s intra-tracheal uptake. These NPs result in ROS accumulation, increase lipid peroxidation level, inflammation, and decreased antioxidants. Cells treated with NPs show elevated levels of the AP-1 (Activator protein 1) transcription factor affecting transcriptional processes.139 Carbon nanotubes and other NPs interact with DNA structures. Hence, they can cause genotoxicity. Nanoparticles can accumulate in pulmonary sites and thus can cause inflammation. In an experiment with mice model/C57BL/6, pneumonia and fibrosis have been seen. It is important to address such factors before using nanoparticles for clinical use.136
While nanotechnology has gained tremendous prominence in the research industry because of its enhanced effectiveness relative to conventional equivalents, it is important to explain toxicological concerns and their regulations. Behavior of the nanoparticles post human body exposure and its deposition up to certain extent raises a big concern. For instance, platinum (Pt)-containing drugs (cisplatin, carboplatin and oxaliplatin) are among the most widely used and most potent anti-cancer chemotherapeutic drugs for treatment of lung, colorectal, ovarian, breast, head and neck, bladder and testicular cancers.140,141 Side effects caused by off-target delivery to normal tissue and organs, notably nephrotoxicity in the kidneys, limit the use of Pt-based drugs.142 Nanotechnology-based chemotherapeutics, has the potential to improve drug delivery and overcome this limitation. While on the other hand a major challenge for translating nanodrugs to clinical settings is their rapid clearance by the RES, hence reducing efficacy and increasing toxicities on off-target organs like spleen, liver, and kidney in this regards.143–145 Administering Intralipid via intravenous route one hour before injection of Pt-containing drugs, decreases Pt accumulation in off-target organs and increases the bioavailability of active drug by tumor cells.146 The rational for this hypothesis is that the infusion of Intralipid has been reported to inhibit RES function by possibly inhibiting peritoneal clearance and impairing the phagocytic activity of Kupffer cells.147 Kupffer cells in the liver play an important role in the uptake and metabolism of Intralipid.148
Iron oxide particles have also been found to exhibit harmful characteristics both in vitro and in vivo, mainly due to generation of ROS.149–151 A polymer coating on the surface of iron oxide nanoparticles dramatically improved cell viability. Surface modifications another approach to reduce harmful effect of nanoparticles. As an illustration, the addition of hydroxyl groups to gadolinium fullerene particles prevented the generation of ROS, thereby reducing toxicity.152
Circulating levels of pro-inflammatory cytokines are generally associated with the seriousness of numerous infections. Severe infections can cause uncontrolled immune reactions complemented by excessive release of pro-inflammatory cytokines, commonly described as a “cytokine storm”, which can account for a considerable amount of the pathology and mortality correlated with severe infections.153 For instance, excessive cytokine release acts as a common characteristic of COVID-19, particularly in severe cases, where it can generate ARDS to exacerbate lung pathology, and sometimes leads to multiple organ dysfunction and failure.154,155
Macrophages and associated monocyte-lineage immune cells play vital functions in controlling cytokine-facilitated inflammatory responses as well as innate and adaptive immune responses.156 Exaggerated pro-inflammatory reactions to SARS-CoV-2 infection are the foremost factor of the disease severity, and patients with severe COVID-19 cases demonstrate cytokine profiles comparable to those discovered in macrophage activation like syndrome (MALS), which is associated with macrophage dysfunction.157
Immunosuppression has been suggested as a method for combating cytokine storms in severe COVID-19 cases. Immunosuppressive drugs (eg, hydroxychloroquine, methylprednisolone, chloroquine, and leflunomide) and inhibitors of pro-inflammatory cytokines (eg, IFN-γ, IL-6, IL-17A, IL-1β, M-CSF, and TNF-α) were chosen as candidates for clinical trials. Modulators of factors that control adaptive and innate immune responses (CD47, C5, M-CSF, GM-CSF, and sphingosine-1-phosphate) in COVID-19 patients are also currently underway. Nevertheless, the immunomodulatory effect of targeting one or two cytokines might not be adequate to mitigate cytokine storms and their ensuing pathology. As an alternative for treating severe COVID-19 cases, it is more beneficial to utilize therapeutic approaches that target the essential immune cells that facilitate cytokine storm advancement.124
Prior findings have shown that liposome-mediated macrophage depletion has a remedial function of reducing cytokine production in infectious and autoimmune diseases.158,159 Possible harmful effects of systemic macrophage depletion (eg, enhanced risk of infection and weakening of homeostatic macrophage functions in healthy tissues) are not addressed by enhancing target specificity, either. To diminish pro-inflammatory cytokine production during excessive immune responses, reprogramming dysfunctional macrophages with stem-cell derived EVs (extracellular vesicles) or synthetic nanoparticles can act as a beneficial tactic as compared with total macrophage depletion.124
Functionalizing NPs-based therapeutics with peptides or ligands that have a high affinity for extracellular macrophage membrane factors can improve their specific uptake in order to mitigate dysfunctional macrophage reactions.126 Though these synthetic NPs may perhaps be more likely than unmodified EVs and liposomes to initiate adverse immune reactions, their biosafety needs to be thoroughly assessed in clinical trials.124
The impact of virus-like particles (VLPs) on macrophage-facilitated immune reactions is not well elucidated; however, VLP therapy has been demonstrated to have remedial effects in animal models of infection or chronic inflammation.160,161 VLPs might be a good platform for prophylactic therapy approaches, although investigations are needed to exhibit their capacity to operate as therapeutic agents that can restrict macrophage dysfunction. Research is required to determine the stability and intrinsic immunogenicity of these particles.124
Conclusion
This review is an attempt to propose nanomedicine technology as a potential COVID-19 therapy, due to the recent bioinformatics research into the potential pharmacological target sites of the SARS-CoV-2 protein along with new therapeutic agents (eg, vaccines, antibodies, natural products) to eradicate the SARS-CoV-2. Therefore, investigation of nano-enabled therapeutic cargos emerged very usefully for achieving therapeutics delivery at the desired target tissue sites and for managing drug release per patient viral load and other symptoms. Such nanomedicine drugs can be formulated by adopting aspects of pharmacology and advanced surface-active functional nanosystem approaches. This pharmacologically relevant nano cargo, along with specially designed stimuli-responsive systems, exhibited customizable properties that set a command to design and develop therapies for managing COVID-19 in a personalized manner. Nanomedicine has an impact on all medical fields and has been considered a significant method for new diagnostics, medical imaging, nanotherapeutics, vaccines and the development of biomaterials for regenerative medicine. Drug-based nanoparticles have been developed for decades, and many clinical trials are being conducted for cancer, neurodegenerative, autoimmune, cardiovascular, and infectious disorders, but few are certified for human use. While it is well known that DDS focused on nanotechnology strengthen current therapeutics in medicine, their use in viral diseases is underexplored and underused, as seen in the SARS-CoV-2 pandemic. At various stages of COVID-19 pathogenesis, nanoparticles can play an important role, given their inhibition ability during viral entry and infected cell protein fusion during initial attachment and membrane fusion.72,162
The outcomes of this review help scholars of pharmacology, biomedical engineering, and nano-systems engineering who are engaged in the field of exploring efficient manipulative nanomedicine as future COVID-19 infection therapy, where the key is to control drug delivery and release without adverse effects.
Acknowledgment
The authors acknowledge their respective departments and institutions for providing support and the use of facilities. AK acknowledges a research grant, namely GR-2000024, funded by Florida Polytechnic University, Lakeland, FL, USA.
Author Contributions
All authors made a significant contribution to the work reported, whether that was in: the conception, study design, execution, acquisition of data, analysis, and interpretation, or all of these; taking part in drafting, revising or critically reviewing the article; giving final approval of the version to be published; agreeing on the journal to which the article would be submitted; and/or agreeing to be accountable for all aspects of the work.
Disclosure
The authors declare no competing financial interest and no conflicts of interest for this work.
References
1. Lu H, Stratton CW, Tang YW. Outbreak of pneumonia of unknown etiology in Wuhan, China: the mystery and the miracle. J Med Virol. 2020;92(4):401–402. doi:10.1002/jmv.25678
2. Kostarelos K. Nanoscale nights of COVID-19. Nat Nanotechnol. 2020;15(5):343–344.
3. WHO Coronavirus Disease (COVID-19) Dashboard. 2020.
4. de Wilde AH, Snijder EJ, Kikkert M, van Hemert MJ. Host Factors in Coronavirus Replication. Curr Top Microbiol Immunol. 2018;419:1–42.
5. Wu A, Peng Y, Huang B, et al. Genome Composition and Divergence of the Novel Coronavirus (2019-nCoV) Originating in China. Cell Host Microbe. 2020;27(3):325–328.
6. Sun Z, Thilakavathy K, Kumar SS, He G, Liu SV. Potential Factors Influencing Repeated SARS Outbreaks in China. Int J Environ Res Public Health. 2020;17:5.
7. Kaushik A. Manipulative magnetic nanomedicine: the future of COVID-19 pandemic/endemic therapy. Expert Opin Drug Deliv. 2020;1–4.
8. van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N Engl J Med. 2020;382(16):1564–1567.
9. Abdullahi IN, Emeribe AU, Mustapha JO, et al. Exploring the genetics, ecology of SARS-COV-2 and climatic factors as possible control strategies against COVID-19. Infez Med. 2020;28(2):166–173.
10. Kaushik AK, Dhau JS, Gohel H, et al. Electrochemical SARS-CoV-2 Sensing at Point-of-Care and Artificial Intelligence for Intelligent COVID-19 Management. ACS Applied Bio Materials. 2020;3(11):7306–7325. doi:10.1021/acsabm.0c01004
11. Shibata A, McMullen E, Pham A, et al. Polymeric nanoparticles containing combination antiretroviral drugs for HIV type 1 treatment. AIDS Res Hum Retroviruses. 2013;29(5):746–754. doi:10.1089/aid.2012.0301
12. Destache CJ, Belgum T, Christensen K, Shibata A, Sharma A, Dash A. Combination antiretroviral drugs in PLGA nanoparticle for HIV-1. BMC Infect Dis. 2009;9(1):198. doi:10.1186/1471-2334-9-198
13. Jayant RD, Tiwari S, Atluri V, et al. Multifunctional Nanotherapeutics for the Treatment of neuroAIDS in Drug Abusers. Sci Rep. 2018;8(1):12991. doi:10.1038/s41598-018-31285-w
14. Lurie N, Saville M, Hatchett R, Halton J. Developing Covid-19 Vaccines at Pandemic Speed. N Engl J Med. 2020;382(21):1969–1973.
15. Amanat F, Krammer F. SARS-CoV-2 Vaccines: status Report. Immunity. 2020;52(4):583–589.
16. Loutfy MR, Blatt LM, Siminovitch KA, et al. Interferon alfacon-1 plus corticosteroids in severe acute respiratory syndrome: a preliminary study. JAMA. 2003;290(24):3222–3228.
17. Kranz LM, Diken M, Haas H, et al. Systemic RNA delivery to dendritic cells exploits antiviral defence for cancer immunotherapy. Nature. 2016;534(7607):396–401.
18. Kanasty R, Dorkin JR, Vegas A, Anderson D. Delivery materials for siRNA therapeutics. Nat Mater. 2013;12(11):967–977.
19. Chauhan G, Madou MJ, Kalra S, Chopra V, Ghosh D, Martinez-Chapa SO. Nanotechnology for COVID-19: therapeutics and Vaccine Research. ACS Nano. 2020;14(7):7760–7782.
20. Mousavizadeh L, Ghasemi S. Genotype and phenotype of COVID-19: their roles in pathogenesis. J Microbiol Immunol Infect. 2020.
21. Boopathi S, Poma AB, Kolandaivel P. Novel 2019 coronavirus structure, mechanism of action, antiviral drug promises and rule out against its treatment. J Biomol Struct Dyn. 2020:1–10.
22. Paliwal P, Sargolzaei S, Bhardwaj SK, Bhardwaj V, Dixit C, Kaushik A. Grand Challenges in Bio-Nanotechnology to Manage COVID-19 Pandemic. Front Nanotechn. 2020;2:571284.
23. Yamamoto V, Bolanos JF, Fiallos J, et al. COVID-19: review of a 21st Century Pandemic from Etiology to Neuro-psychiatric Implications. J Alzheimers Dis. 2020;77(2):459–504.
24. Kirchdoerfer RN, Cottrell CA, Wang N, et al. Pre-fusion structure of a human coronavirus spike protein. Nature. 2016;531(7592):118–121.
25. Gupta MK, Vemula S, Donde R, Gouda G, Behera L, Vadde R. In-silico approaches to detect inhibitors of the human severe acute respiratory syndrome coronavirus envelope protein ion channel. J Biomol Struct Dyn. 2020;1–11.
26. Ziebuhr J. The coronavirus replicase. Curr Top Microbiol Immunol. 2005;287:57–94.
27. Subissi L, Posthuma CC, Collet A, et al. One severe acute respiratory syndrome coronavirus protein complex integrates processive RNA polymerase and exonuclease activities. Proc Natl Acad Sci U S A. 2014;111(37):E3900.
28. Holshue ML, DeBolt C, Lindquist S, et al. First Case of 2019 Novel Coronavirus in the United States. N Engl J Med. 2020;382(10):929–936.
29. Wang M, Cao R, Zhang L, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30(3):269–271.
30. Holmes KV. Structural biology. Adaptation of SARS coronavirus to humans. Science. 2005;309(5742):1822–1823.
31. Dimitrov DS. The secret life of ACE2 as a receptor for the SARS virus. Cell. 2003;115(6):652–653.
32. Hasan A, Paray BA, Hussain A, et al. A review on the cleavage priming of the spike protein on coronavirus by angiotensin-converting enzyme-2 and furin. J Biomol Struct Dyn. 2020;1–9.
33. Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181(2):271–280.
34. Ni W, Yang X, Yang D, et al. Role of angiotensin-converting enzyme 2 (ACE2) in COVID-19. Crit Care. 2020;24(1):422.
35. Pezzini A, Padovani A. Lifting the mask on neurological manifestations of COVID-19. Nat Rev Neurol. 2020;16(11):636–644.
36. Osman Shabir MS. Emily Henderson, B.Sc. What is TMPRSS2? 2020
37. Du L, He Y, Zhou Y, Liu S, Zheng BJ, Jiang S. The spike protein of SARS-CoV–a target for vaccine and therapeutic development. Nat Rev Microbiol. 2009;7(3):226–236.
38. Yuki K, Fujiogi M, Koutsogiannaki S. COVID-19 pathophysiology: A review. Clin Immunol. 2020;215:108427.
39. Alanagreh L, Alzoughool F, Atoum M. The Human Coronavirus Disease COVID-19: its Origin, Characteristics, and Insights into Potential Drugs and Its Mechanisms. Pathogens. 2020;9:5.
40. Koyama T, Platt D, Parida L. Variant analysis of SARS-CoV-2 genomes. Bull World Health Organ. 2020;98(7):495–504.
41. Toyoshima Y, Nemoto K, Matsumoto S, Nakamura Y, Kiyotani K. SARS-CoV-2 genomic variations associated with mortality rate of COVID-19. J Hum Genet. 2020.
42. Guan WJ, Ni ZY, Hu Y, et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382(18):1708–1720.
43. Chan JF, Yuan S, Kok KH, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet. 2020;395(10223):514–523.
44. Phan LT, Nguyen TV, Luong QC, et al. Importation and Human-to-Human Transmission of a Novel Coronavirus in Vietnam. N Engl J Med. 2020;382(9):872–874.
45. Li X, Ma X. Acute respiratory failure in COVID-19: is it “typical” ARDS? Crit Care. 2020;24(1):198.
46. Inciardi RM, Lupi L, Zaccone G, et al. Cardiac Involvement in a Patient With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020.
47. Guo T, Fan Y, Chen M, et al. Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020.
48. Archie SR, Cucullo L. Cerebrovascular and Neurological Dysfunction under the Threat of COVID-19: is There a Comorbid Role for Smoking and Vaping? Int J Mol Sci. 2020;21:11.
49. Hossain M, Sathe T, Fazio V, et al. Tobacco smoke: a critical etiological factor for vascular impairment at the blood-brain barrier. Brain Res. 2009;1287:192–205.
50. Desforges M, Le Coupanec A, Dubeau P, et al. Human Coronaviruses and Other Respiratory Viruses: underestimated Opportunistic Pathogens of the Central Nervous System? Viruses. 2019;12:1.
51. Nuzzo D, Picone P. Potential neurological effects of severe COVID-19 infection. Neurosci Res. 2020.
52. Wu Y, Xu X, Chen Z, et al. Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav Immun. 2020;87:18–22.
53. Fu Y, Cheng Y, Understanding WY. SARS-CoV-2-Mediated Inflammatory Responses: from Mechanisms to Potential Therapeutic Tools. Virol Sin. 2020;35(3):266–271.
54. Peretto G, Sala S, Caforio ALP. Acute myocardial injury, MINOCA, or myocarditis? Improving characterization of coronavirus-associated myocardial involvement. Eur Heart J. 2020;41(22):2124–2125.
55. Koyuncu OO, Hogue IB, Enquist LW. Virus infections in the nervous system. Cell Host Microbe. 2013;13(4):379–393.
56. Fiani B, Covarrubias C, Desai A, Sekhon M, Jarrah R. A Contemporary Review of Neurological Sequelae of COVID-19. Front Neurol. 2020;11:640.
57. Moriguchi T, Harii N, Goto J, et al. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int J Infect Dis. 2020;94:55–58.
58. Li Y, Li H, Fan R, et al. Coronavirus Infections in the Central Nervous System and Respiratory Tract Show Distinct Features in Hospitalized Children. Intervirology. 2016;59(3):163–169.
59. Shang J, Wan Y, Luo C, et al. Cell entry mechanisms of SARS-CoV-2. Proc Natl Acad Sci U S A. 2020;117(21):11727–11734.
60. Rismanbaf A, Liver ZS. and Kidney Injuries in COVID-19 and Their Effects on Drug Therapy; a Letter to Editor. Arch Acad Emerg Med. 2020;8(1):e17.
61. Zhang C, Shi L, Wang FS. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020;5(5):428–430.
62. Xu Z, Shi L, Wang Y, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420–422.
63. Cheng Y, Luo R, Wang K, et al. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020;97(5):829–838.
64. Pan XW, Xu D, Zhang H, Zhou W, Wang LH, Cui XG. Identification of a potential mechanism of acute kidney injury during the COVID-19 outbreak: a study based on single-cell transcriptome analysis. Intensive Care Med. 2020;46(6):1114–1116.
65. Yu X, Yang RCOVID-19. transmission through asymptomatic carriers is a challenge to containment. Influenza Other Respir Virus. 2020;14(4):474–475.
66. Rahimi F. Talebi Bezmin Abadi A. Challenges of managing the asymptomatic carriers of SARS-CoV-2. Travel Med Infect Dis. 2020;101677.
67. Barlow A, Landolf KM, Barlow B, et al. Review of Emerging Pharmacotherapy for the Treatment of Coronavirus Disease 2019. Pharmacotherapy. 2020;40(5):416–437.
68. Nishiura H, Kobayashi T, Yang Y, et al. The Rate of Underascertainment of Novel Coronavirus (2019-nCoV) Infection: estimation Using Japanese Passengers Data on Evacuation Flights. J Clin Med. 2020;9:2.
69. Newman SP. Drug delivery to the lungs: challenges and opportunities. Ther Deliv. 2017;8(8):647–661.
70. El-Sherbiny IM, El-Baz NM, Yacoub MH. Inhaled nano- and microparticles for drug delivery. Glob Cardiol Sci Pract. 2015;2015:2.
71. de Oliveira Cardoso VM, Moreira BJ, Comparetti EJ, et al. Is Nanotechnology Helping in the Fight Against COVID-19? 2020
72. Mainardes RM, Diedrich C. The potential role of nanomedicine on COVID-19 therapeutics. Ther Deliv. 2020;11(7):411–414.
73. Vega-Chacon J, Arbelaez MIA, Jorge JH, Marques RFC, Jafelicci MJ. pH-responsive poly(aspartic acid) hydrogel-coated magnetite nanoparticles for biomedical applications. Mater Sci Eng C Mater Biol Appl. 2017;77:366–373.
74. Weiss C, Carriere M, Fusco L, et al. Toward Nanotechnology-Enabled Approaches against the COVID-19 Pandemic. ACS Nano. 2020;14(6):6383–6406.
75. Sportelli MC, Izzi M, Kukushkina EA, et al. Can Nanotechnology and Materials Science Help the Fight against SARS-CoV-2? Nanomaterials. 2020;10:4.
76. Mehta P, McAuley DF, Brown M, et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395(10229):1033–1034.
77. Dormont F, Brusini R, Cailleau C, et al. Squalene-based multidrug nanoparticles for improved mitigation of uncontrolled inflammation in rodents. Sci Adv. 2020;6(23):eaaz5466.
78. Lembo D, Donalisio M, Civra A, Argenziano M, Cavalli R. Nanomedicine formulations for the delivery of antiviral drugs: a promising solution for the treatment of viral infections. Expert Opin Drug Deliv. 2018;15(1):93–114.
79. Siccardi M, Martin P, McDonald TO, et al. Nanomedicines for HIV therapy. Ther Deliv. 2013;4(2):153–156.
80. Karimi M, Ghasemi A, Sahandi Zangabad P, et al. Smart micro/nanoparticles in stimulus-responsive drug/gene delivery systems. Chem Soc Rev. 2016;45(5):1457–1501.
81. Steven Mufamadi LC. How Nanomedicine Could Help to Identify COVID-19 and Prevent Deaths. Int J Med. 2020;22.
82. Barar J. COVID-19 clinical implications: the significance of nanomedicine. Bioimpacts. 2020;10(3):137–138.
83. Parzych E, Weiner DB. Nucleic acid approaches to antibody-based therapeutics for COVID-19: A perspective. J Allergy Clin Immunol. 2020.
84. de la Torre Gomez C, Goreham RV, Bech Serra JJ, Nann T, Kussmann M. “Exosomics”-A Review of Biophysics, Biology and Biochemistry of Exosomes With a Focus on Human Breast Milk. Front Genet. 2018;9:92.
85. Lu M, Huang Y. Bioinspired exosome-like therapeutics and delivery nanoplatforms. Biomaterials. 2020;242:119925.
86. Hassanpour M, Rezaie J, Nouri M, Panahi Y. The role of extracellular vesicles in COVID-19 virus infection. Infect Genet Evol. 2020;85:104422.
87. Akbari A, Rezaie J. Potential therapeutic application of mesenchymal stem cell-derived exosomes in SARS-CoV-2 pneumonia. Stem Cell Res Ther. 2020;11(1):356.
88. Pinky GS, Krishnakumar V, Sharma Y, Dinda AK, Mohanty S. Mesenchymal Stem Cell Derived Exosomes: a Nano Platform for Therapeutics and Drug Delivery in Combating COVID-19. Stem Cell Rev Rep. 2020.
89. Saei AA, Sharifi S, Mahmoudi M. COVID-19: nanomedicine Uncovers Blood-Clot Mystery. J Proteome Res. 2020.
90. Jayant RD, Atluri VS, Agudelo M, Sagar V, Kaushik A, Nair M. Sustained-release nanoART formulation for the treatment of neuroAIDS. Int J Nanomedicine. 2015;10:1077–1093.
91. Kaushik A, Jayant RD, Nair M. Advances in Personalized Nanotherapeutics. Springer; 2017.
92. Kaushik A, Jayant RD, Bhardwaj V, Nair M. Personalized nanomedicine for CNS diseases. Drug Discov Today. 2018;23(5):1007–1015.
93. Tomitaka A, Kaushik A, Kevadiya BD, et al. Surface-engineered multimodal magnetic nanoparticles to manage CNS diseases. Drug Discov Today. 2019;24(3):873–882.
94. Roberto Vazquez-Munoz JLL-R. Nanotechnology as an Alternative to Reduce the Spread of COVID-19. MDPI Challenges. 2020;23.
95. Abd Ellah NH, Gad SF, Muhammad K, Hetta GEB. Nanomedicine as a promising approach for diagnosis, treatment and prophylaxis against COVID-19. Nanomedicine. 2020.
96. De Cock LJ, De Koker S, De Geest BG, et al. Polymeric multilayer capsules in drug delivery. Angew Chem Int Ed Engl. 2010;49(39):6954–6973.
97. Becker AL, Johnston AP, Caruso F. Layer-by-layer-assembled capsules and films for therapeutic delivery. Small. 2010;6(17):1836–1852.
98. Ochs CJ, Such GK, Yan Y, van Koeverden MP, Caruso F. Biodegradable click capsules with engineered drug-loaded multilayers. ACS Nano. 2010;4(3):1653–1663.
99. Remaut K, Sanders NN, De Geest BG, Braeckmans K, Demeester J, De Smedt SC. Nucleic Acid Delivery: Where Material Sciences and Bio-Sciences Meet. Elsevier; 2007.
100. Ariga K, Lvov YM, Kawakami K, Ji Q, Hill JP. Layer-by-layer self-assembled shells for drug delivery. Adv Drug Deliv Rev. 2011;63(9):762–771. doi:10.1016/j.addr.2011.03.016
101. Blacklock J, You YZ, Zhou QH, Mao G, Oupicky D. Gene delivery in vitro and in vivo from bioreducible multilayered polyelectrolyte films of plasmid DNA. Biomaterials. 2009;30(5):939–950. doi:10.1016/j.biomaterials.2008.10.012
102. Kim JY, Lee H, Oh KS, et al. Multilayer nanoparticles for sustained delivery of exenatide to treat type 2 diabetes mellitus. Biomaterials. 2013;34(33):8444–8449. doi:10.1016/j.biomaterials.2013.07.040
103. He L, Lin X, Wang Y, et al. Self-assembling nanoparticles presenting receptor binding domain and stabilized spike as next-generation COVID-19 vaccines. bioRxiv. 2020. doi:10.1101/2020.09.14.296715
104. Lin X, Choi D, Hong J. Insulin particles as building blocks for controlled insulin release multilayer nano-films. Mater Sci Eng C Mater Biol Appl. 2015;54:239–244. doi:10.1016/j.msec.2015.05.046
105. Hassanzadeh P. Nanotheranostics against COVID-19: from multivalent to immune-targeted materials. J Control Release. 2020;328:112–126. doi:10.1016/j.jconrel.2020.08.060
106. Tabish TA, Hamblin MR. Multivalent nanomedicines to treat COVID-19: A slow train coming. Nano Today. 2020;35:100962. doi:10.1016/j.nantod.2020.100962
107. Huang J, Li Y, Orza A, et al. Magnetic Nanoparticle Facilitated Drug Delivery for Cancer Therapy with Targeted and Image-Guided Approaches. Adv Funct Mater. 2016;26(22):3818–3836. doi:10.1002/adfm.201504185
108. Kaushik A, Jayant RD, Sagar V, Nair M. The potential of magneto-electric nanocarriers for drug delivery. Expert Opin Drug Deliv. 2014;11(10):1635–1646. doi:10.1517/17425247.2014.933803
109. Kaushik A, Rodriguez J, Rothen D, et al. MRI-Guided, Noninvasive Delivery of Magneto-Electric Drug Nanocarriers to the Brain in a Nonhuman Primate. ACS Applied Bio Materials. 2019;2(11):4826–4836. doi:10.1021/acsabm.9b00592
110. Shlok Jindal PG. Nanotechnology based approaches for combatting COVID-19 viral infection. Tech Sci. 2020;25.
111. Man F, Lammers T, T. M. de Rosales R. R TMdR. Imaging Nanomedicine-Based Drug Delivery: a Review of Clinical Studies. Mol Imaging Biol. 2018;20(5):683–695. doi:10.1007/s11307-018-1255-2
112. Zavaleta C, Ho D, Chung EJ. Theranostic Nanoparticles for Tracking and Monitoring Disease State. SLAS Technol. 2018;23(3):281–293. doi:10.1177/2472630317738699
113. Yu L, Chen Y, Chen H. Chen H. H2O2-responsive theranostic nanomedicine. Chinese Chemical Letters. 2017;28(9):1841–1850. doi:10.1016/j.cclet.2017.05.023
114. Raza A, Rasheed T, Nabeel F, Hayat U, Bilal M, Iqbal HMN. Endogenous and Exogenous Stimuli-Responsive Drug Delivery Systems for Programmed Site-Specific Release. Molecules. 2019;24(6):6. doi:10.3390/molecules24061117
115. Li L, Yang WW, Xu DG. Stimuli-responsive nanoscale drug delivery systems for cancer therapy. J Drug Target. 2019;27(4):423–433. doi:10.1080/1061186X.2018.1519029
116. Al-Halifa S, Gauthier L, Arpin D, Bourgault S, Archambault D. Nanoparticle-Based Vaccines Against Respiratory Viruses. Front Immunol. 2019;10:22. doi:10.3389/fimmu.2019.00022
117. Look M, Bandyopadhyay A, Blum JS, Fahmy TM. Application of nanotechnologies for improved immune response against infectious diseases in the developing world. Adv Drug Deliv Rev. 2010;62(4–5):378–393. doi:10.1016/j.addr.2009.11.011
118. Kaushik A, Jayant RD, Nair M. Nanomedicine for neuroHIV/AIDS management. Nanomedicine. 2018;13(7):669–673. doi:10.2217/nnm-2018-0005
119. Hu TY, Frieman M, Wolfram J. Insights from nanomedicine into chloroquine efficacy against COVID-19. Nat Nanotechnol. 2020;15(4):247–249. doi:10.1038/s41565-020-0674-9
120. Group RC, Horby P, Lim WS, et al. Dexamethasone in Hospitalized Patients with Covid-19 - Preliminary Report. N Engl J Med. 2020.
121. Lammers T, Sofias AM, van der Meel R, et al. Dexamethasone nanomedicines for COVID-19. Nat Nanotechnol. 2020;15(8):622–624. doi:10.1038/s41565-020-0752-z
122. Zhang J, Leifer F, Rose S, et al. Amikacin Liposome Inhalation Suspension (ALIS) Penetrates Non-tuberculous Mycobacterial Biofilms and Enhances Amikacin Uptake Into Macrophages. Front Microbiol. 2018;9:915. doi:10.3389/fmicb.2018.00915
123. Kim E, Erdos G, Huang S, et al. Microneedle array delivered recombinant coronavirus vaccines: immunogenicity and rapid translational development. EBioMedicine. 2020;55:102743. doi:10.1016/j.ebiom.2020.102743
124. Liu J, Wan M, Lyon CJ, Hu TY. Nanomedicine therapies modulating Macrophage Dysfunction: a potential strategy to attenuate Cytokine Storms in severe infections. Theranostics. 2020;10(21):9591–9600. doi:10.7150/thno.47982
125. Weissleder R, Nahrendorf M, Pittet MJ. Imaging macrophages with nanoparticles. Nat Mater. 2014;13(2):125–138. doi:10.1038/nmat3780
126. He H, Ghosh S, Yang H. Nanomedicines for dysfunctional macrophage-associated diseases. J Control Release. 2017;247:106–126. doi:10.1016/j.jconrel.2016.12.032
127. Zhang Q, Honko A, Zhou J, et al. Cellular Nanosponges Inhibit SARS-CoV-2 Infectivity. Nano Lett. 2020;20(7):5570–5574. doi:10.1021/acs.nanolett.0c02278
128. Moitra P, Alafeef M, Dighe K, Frieman MB, Pan PD. Selective Naked-Eye Detection of SARS-CoV-2 Mediated by N Gene Targeted Antisense Oligonucleotide Capped Plasmonic Nanoparticles. ACS Nano. 2020;14(6):7617–7627. doi:10.1021/acsnano.0c03822
129. Qiu G, Gai Z, Tao Y, Schmitt J, Kullak-Ublick GA, Wang WJ. Dual-Functional Plasmonic Photothermal Biosensors for Highly Accurate Severe Acute Respiratory Syndrome Coronavirus 2 Detection. ACS Nano. 2020;14(5):5268–5277. doi:10.1021/acsnano.0c02439
130. Seo G, Lee G, Kim MJ, et al. Rapid Detection of COVID-19 Causative Virus (SARS-CoV-2) in Human Nasopharyngeal Swab Specimens Using Field-Effect Transistor-Based Biosensor. ACS Nano. 2020;14(4):5135–5142. doi:10.1021/acsnano.0c02823
131. Talebian S, Wallace GG, Schroeder A, Stellacci F, Conde J. Nanotechnology-based disinfectants and sensors for SARS-CoV-2. Nat Nanotechnol. 2020;15(8):618–621. doi:10.1038/s41565-020-0751-0
132. Wang J, Shen J, Ye D, et al. Disinfection technology of hospital wastes and wastewater: suggestions for disinfection strategy during coronavirus Disease 2019 (COVID-19) pandemic in China. Environ Pollut. 2020;262:114665. doi:10.1016/j.envpol.2020.114665
133. Balagna C, Perero S, Percivalle E, Nepita EV, Ferraris M. Virucidal effect against Coronavirus SARS-CoV-2 of a silver nanocluster/silica composite sputtered coating. Open Ceramics. 2020;1:100006. doi:10.1016/j.oceram.2020.100006
134. Germain M, Caputo F, Metcalfe S, et al. Delivering the power of nanomedicine to patients today. J Control Release. 2020;326:164–171. doi:10.1016/j.jconrel.2020.07.007
135. Patel P, Shah SJ. Safety and Toxicological Considerations of Nanomedicines: the Future Directions. Curr Clin Pharmacol. 2020;12(2):73–82. doi:10.2174/1574884712666170509161252
136. Sivasankarapillai VS, Pillai AM, Rahdar A, et al. On Facing the SARS-CoV-2 (COVID-19) with Combination of Nanomaterials and Medicine: possible Strategies and First Challenges. Nanomaterials. 2019;24(5):5. doi:10.3390/nano10050852
137. Nasrollahzadeh M, Sajjadi M, Soufi GJ, Iravani S, Varma RS. Nanomaterials and Nanotechnology-Associated Innovations against Viral Infections with a Focus on Coronaviruses. Nanomaterials. 2020;14(6):6. doi:10.3390/nano10061072
138. Sekimukai H, Iwata-Yoshikawa N, Fukushi S, et al. Gold nanoparticle-adjuvanted S protein induces a strong antigen-specific IgG response against severe acute respiratory syndrome-related coronavirus infection, but fails to induce protective antibodies and limit eosinophilic infiltration in lungs. Microbiol Immunol. 2020;64(1):33–51.
139. Borgio JF, Alsuwat HS, Al Otaibi WM, et al. State-of-the-art tools unveil potent drug targets amongst clinically approved drugs to inhibit helicase in SARS-CoV-2. Arch Med Sci. 2020;16(3):508–518.
140. Wheate NJ, Walker S, Craig GE, Oun R. The status of platinum anticancer drugs in the clinic and in clinical trials. Dalton Trans. 2010;39(35):8113–8127.
141. Pabla N, Dong Z. Cisplatin nephrotoxicity: mechanisms and renoprotective strategies. Kidney Int. 2008;73(9):994–1007.
142. Yao X, Panichpisal K, Kurtzman N, Nugent K. Cisplatin nephrotoxicity: a review. Am J Med Sci. 2007;334(2):115–124.
143. Chow EK, Ho D. Cancer nanomedicine: from drug delivery to imaging. Sci Transl Med. 2013;5(216):216rv214.
144. Albanese A, Tang PS, Chan WC. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu Rev Biomed Eng. 2012;14:1–16.
145. Wang AZ, Langer R, Farokhzad OC. Nanoparticle delivery of cancer drugs. Annu Rev Med. 2012;63:185–198.
146. Liu L, Ye Q, Lu M, et al. A new approach to reduce toxicities and to improve bioavailabilities of platinum-containing anti-cancer nanodrugs. Sci Rep. 2015;5:10881.
147. Nugent KM. Intralipid effects on reticuloendothelial function. J Leukoc Biol. 1984;36(2):123–132.
148. Vilaro S, Llobera M. Uptake and metabolism of Intralipid by rat liver: an electron-microscopic study. J Nutr. 1988;118(8):932–940.
149. Zhu MT, Wang Y, Feng WY, et al. Oxidative stress and apoptosis induced by iron oxide nanoparticles in cultured human umbilical endothelial cells. J Nanosci Nanotechnol. 2010;10(12):8584–8590.
150. Zhu MT, Wang B, Wang Y, et al. Endothelial dysfunction and inflammation induced by iron oxide nanoparticle exposure: risk factors for early atherosclerosis. Toxicol Lett. 2011;203(2):162–171.
151. Gupta AK, Wells S. Surface-modified superparamagnetic nanoparticles for drug delivery: preparation, characterization, and cytotoxicity studies. IEEE Trans Nanobioscience. 2004;3(1):66–73.
152. Chen C, Xing G, Wang J, et al. Multihydroxylated [Gd@C82(OH)22]n nanoparticles: antineoplastic activity of high efficiency and low toxicity. Nano Lett. 2005;5(10):2050–2057.
153. Chousterman BG, Swirski FK, Weber GF. Cytokine storm and sepsis disease pathogenesis. Semin Immunopathol. 2017;39(5):517–528.
154. Chen L, Xiong J, Bao L, Shi Y. Convalescent plasma as a potential therapy for COVID-19. Lancet Infect Dis. 2020;20(4):398–400.
155. Moore JB, June CH. Cytokine release syndrome in severe COVID-19. Science. 2020;368(6490):473–474.
156. Hung IF, Lung KC, Tso EY, et al. Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, Phase 2 trial. Lancet. 2020;395(10238):1695–1704.
157. Merad M, Martin JC. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat Rev Immunol. 2020;20(6):355–362.
158. Vanniasinghe AS, Bender V, Manolios N. The potential of liposomal drug delivery for the treatment of inflammatory arthritis. Semin Arthritis Rheum. 2009;39(3):182–196.
159. Bader JE, Enos RT, Velazquez KT, et al. Macrophage depletion using clodronate liposomes decreases tumorigenesis and alters gut microbiota in the AOM/DSS mouse model of colon cancer. Am J Physiol Gastrointest Liver Physiol. 2018;314(1):G22–G31.
160. Mathieu C, Rioux G, Dumas MC, Leclerc D. Induction of innate immunity in lungs with virus-like nanoparticles leads to protection against influenza and Streptococcus pneumoniae challenge. Nanomedicine. 2013;9(7):839–848.
161. Zampieri R, Brozzetti A, Pericolini E, et al. Prevention and treatment of autoimmune diseases with plant virus nanoparticles. Sci Adv. 2020;6(19):eaaz0295.
162. Mujawar MA, Gohel H, Bhardwaj SK, Srinivasan S, Hickman N, Kaushik A. Nano-enabled biosensing systems for intelligent healthcare: towards COVID-19 management. Mater Today Chem. 2020;17:100306.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.