Back to Journals » OncoTargets and Therapy » Volume 7
MiR-206 functions as a tumor suppressor and directly targets K-Ras in human oral squamous cell carcinoma
Authors Lin F, Yao L, Xiao J, Liu D, Ni Z
Received 12 May 2014
Accepted for publication 26 June 2014
Published 11 September 2014 Volume 2014:7 Pages 1583—1591
DOI https://doi.org/10.2147/OTT.S67624
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Feiou Lin,1 Linjie Yao,2 Jin Xiao,3 DengFeng Liu,3 Zhenyu Ni1
1Department of Orthodontics, 2Department of Pedodontics, 3Department of Oral and Maxillofacial Surgery, School and Hospital of Stomatology, Wenzhou Medical University, Wenzhou, People’s Republic of China
Purpose: MicroRNA-206 (miR-206) has been proven to be downregulated in many human malignancies and is correlated with tumor progression. However, the roles of miR-206 and its related molecular mechanisms in oral squamous cell carcinoma (OSCC) are still unclear. Thus, the aim of this study was to explore the effects of miR-206 in OSCC tumorigenesis and development.
Methods: Quantitative real-time polymerase chain reaction was used to detect miR-206 expression in OSCC cell lines and primary tumor tissues. The association of miR-206 expression with clinicopathological factors and prognosis was also analyzed. In addition, the effects of miR-206 on the biological behavior of OSCC cells were investigated. Lastly, the potential regulatory function of miR-206 on K-Ras expression was confirmed.
Results: MiR-206 expression was significantly downregulated in OSCC tissue samples and cell lines (both P<0.001). Decreased miR-206 expression was significantly associated with advanced tumor node metastasis (TNM) stage, advanced T classifications (ie, size and/or extent of the primary tumor), positive N classification (ie, spread to regional lymph nodes), and shorter overall survival. In addition, upregulation of miR-206 in Tca8113 cells was able to reduce cell proliferation, invasion, and migration and promote cell apoptosis in vitro. Further, K-Ras was confirmed as a direct target of miR-206 by using luciferase reporter assay.
Conclusion: These findings indicate that miR-206 may act as a tumor suppressor in OSCC and could serve as a novel therapeutic agent for miR-based therapy.
Keywords: miR-206, oral squamous cell carcinoma, prognosis, proliferation, apoptosis, invasion
Introduction
Oral squamous cell carcinoma (OSCC) is the most common malignant neoplasm of the oral cavity and represents about 90% of all oral malignancies.1 Despite recent advances in multimodality treatments, more than 50% of OSCC patients experience relapse, including local recurrence, regional lymph node metastasis, and distant spread. The 5-year overall survival rate in OSCC is less than 50%, which has not significantly increased in the last three decades.2 Previous studies have demonstrated diverse genetic alterations in OSCC,3,4 but the highly complex molecular mechanisms underlying OSCC carcinogenesis and progression remain obscure. Therefore, it is necessary to search for novel markers for OSCC that can accurately identify biological characteristics of tumors, improve therapeutic strategies, and predict clinical outcome.
MicroRNAs (miRs) are a class of naturally occurring small (21–25 nucleotides) noncoding RNAs. They bind through partial sequence homology to the 3′ untranslated region of target messenger RNAs and either block translation or promote messenger RNA degradation.5,6 Beyond their involvement in diverse biological processes, including cell growth, apoptosis, development, differentiation, and endocrine homeostasis,6 emerging evidence strongly suggests that the deregulation or dysfunction of miRs contributes to human carcinogenesis and cancer progression.7–9 MiRs can function as either oncogenes or tumor suppressors according to the roles of their target genes. In terms of miR-206, it has been reported to be downregulated and exert tumor suppressive function in several malignancies such as breast cancer,10 lung cancer,11 gastric cancer,12 colorectal cancer,13 renal cell carcinoma,14 endometrioid adenocarcinoma,15 glioma and neuroblastoma,16 rhabdomyosarcoma,17 and osteosarcoma.18 In addition, downregulation of miR-206 was associated with tumor progression and worse prognosis.12,18–20 However, the role of miR-206 and its direct functional targets in human OSCC are still not completely clear.
Ras is the first human oncogene discovered by Shih and Weinberg in the early 1980s.21 K-Ras is one of the three members of the Ras oncogene family. Activation of the K-Ras oncogene has been implicated in the carcinogenesis of many human cancers, including OSCC,22 and downregulation of K-Ras could suppress cancer cell growth and increase chemosensitivity.23,24 Recently, several miRs were reported to suppress K-Ras expression and function as tumor suppressors.25–28 These findings support the importance of miRs in regulation of K-Ras activity, but the potential regulatory effect of miR-206 on K-Ras expression has not yet been confirmed.
The aim of this study was to examine miR-206 expression in OSCC tissues and cell lines using real-time polymerase chain reaction (PCR). The association of miR-206 levels with clinicopathologic features and prognosis was also analyzed. Furthermore, the effects of miR-206 on the proliferation, apoptosis, invasion, and migration of OSCC cells were investigated. Finally, K-Ras was identified as a direct target of miR-206 by luciferase reporter assay.
Materials and methods
Tissue samples and cell lines
One-hundred and nine pairs of primary OSCC and adjacent noncancerous tissues were collected from the Department of Oral and Maxillofacial Surgery, School and Hospital of Stomatology, Wenzhou Medical University (Wenzhou, People’s Republic of China) between January 2005 and December 2008. All tissues were immediately frozen in liquid nitrogen and stored at −80°C until analysis. None of the patients had undergone chemotherapy or radiotherapy before surgery. The patients’ information is summarized in Table 1. All of the patients received follow-up. Overall survival was defined as the time from primary surgery to death of the patient or, for living patients, the date of last follow-up. This study was approved by the Research Ethics Committee of the hospital, and all patients gave informed consent.
Human OSCC cell lines (SCC-4, SCC-9, SCC-25, and Tca8113), and a human normal oral keratinocyte cell line were obtained from the Beijing Institute for Cancer Research (Beijing, People’s Republic of China) and maintained in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with Invitrogen® 10% fetal bovine serum (Thermo Fisher Scientific, Waltham, MA, USA), 100 U/mL penicillin, and 100 μg/mL streptomycin. All the cells were incubated at 37°C in a humidified atmosphere with 5% CO2.
RNA extraction and quantitative real-time PCR
Total RNA was isolated using the TRIzol® reagent (Thermo Fisher) according to the manufacturer’s instructions. Reverse transcription reaction was carried out starting from 100 ng of total RNA using the looped primers. Real-time PCR was performed using the standard TaqMan® MicroRNA assays protocol on the Applied Biosystems® 7500 real-time PCR detection system (Thermo Fisher), with cycling conditions of 95°C for 10 minutes followed by 40 cycles of 95°C for 15 seconds and 60°C for 60 seconds. U6 small nuclear RNA was used as an internal control. The RT primers were 5′-GCGCGTGAGCAGGCTGGAGAAATTAACCACGCGC-3′ for miR-206 and 5′-TGGTGTCGTGGAGTCG-3′ for U6. The PCR primers for mature miR-206 or U6 were designed as follows: miR-206 sense 5′-GGAATGTAAGGAAGTGTG-3′ and miR-206 reverse 5′-GAGCAGGCTGGAGAA-3′; U6 sense 5′-CTCGCTTCGGCAGCACA-3′ and U6 reverse 5′-AACGCTTCACGAATTTGCGT-3′. The threshold cycle was defined as the fractional cycle number at which the fluorescence passed the fixed threshold. Each sample was measured in triplicate, and the relative amount of miR-206 to U6 was calculated using the following equation:
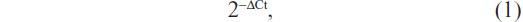
where Ct is the threshold cycle and ΔCt is the threshold cycle of miR-206 minus the threshold cycle of U6 (ie, CtmiR-206 − CtU6).
Cell transfection
For RNA transfection, the cells were seeded into each well of a 24-well plate and incubated overnight and then transfected with either miR-206 mimics (Shanghai GenePharma Co, Ltd, Shanghai, People’s Republic of China) or negative control (NC) RNA-oligonucleotides (GenePharma) using Lipofectamine® 2000 (Thermo Fisher) in accordance with the manufacturer’s procedure. The transfection efficiency of the miR-206 mimics was confirmed by real-time PCR analysis.
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay
Cells were seeded into 96-well culture plates at a density of 2,000 cells in 200 μL/well and incubated at 37°C after transfection. Then, 100 μL of MTT solution (0.5 mg/mL; Sigma-Aldrich Co, St Louis, MO, USA) was added to each well, and the cells were incubated for another 4 hours. The medium was then replaced with 150 μL of dimethyl sulfoxide. Spectrometric absorbance at 490 nm was measured using a microplate reader (BIO-RAD 680). Cell proliferation was assessed daily for four consecutive days, and the MTT assay was repeated three times.
Detection of apoptosis by flow cytometry
Apoptosis was detected by flow cytometric analysis. Briefly, the cells were washed and resuspended at a concentration of 1×106 cells/mL. Then, the cells were stained with Annexin V and propidium iodide, using the Annexin V Apoptosis Detection Kit (BD Biosciences, San Jose, CA, USA). After incubation at room temperature in the dark for 15 minutes, cell apoptosis was analyzed on a BD FACSCalibur™ (BD Biosciences).
Transwell migration and invasion assays
The migration and invasion assays were performed using 24-well Transwell® chambers (8 μm; Corning Incorporated, Corning, NY, USA). For the migration assay, tumor cells were resuspended in DMEM and 2×105 cells were seeded into the upper chambers. Then, 0.5 mL DMEM containing 10% FBS was added to the bottom chambers. Following a 24-hour incubation, cells on the upper surface of the membrane were scrubbed off, and the migrated cells were fixed with 95% ethanol, stained with 0.1% crystal violet, and counted under a light microscope (Olympus Corp., Tokyo, Japan). The invasion assay protocol was similar to that of the migration assay except that the upper chambers were first covered with 1 mg/mL Matrigel® (Corning).
Luciferase reporter assays
The pGL3 luciferase reporter vector (Promega Corporation, Fitchburg, WI, USA) was used for the construction of the pGL3–K-Ras and pGL3–K-Ras–mut vectors. The pGL3–K-Ras–mut vector was built with K-Ras that had undergone site-directed mutagenesis of the miR-206 target site using the QuikChange® Site-Directed Mutagenesis Kit (Agilent Technologies, Santa Clara, CA, USA). For the luciferase reporter assay, cells were cultured in 24-well plates and transfected with the plasmids and miR-206 mimics using Lipofectamine 2000. Twenty-four hours after transfection, luciferase activity was measured using the Dual-Luciferase® Reporter Assay System (Promega). Firefly luciferase activity was normalized to renilla luciferase activity for each transfected well.
Western blot analysis
Protein lysates were separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to the nitrocellulose membrane. After blocking, the membranes were incubated with purified rabbit anti-K-Ras antisera at 4°C overnight. The next day, the membranes were washed with phosphate-buffered saline and then incubated with peroxidase-conjugated goat anti-rabbit immunoglobulin G (Santa Cruz Biotechnology, Texas, USA). Immunodetection was conducted with Pierce® ECL chemiluminescence reagents (Thermo Fisher) and exposed on an X-ray film. β-actin was used as an internal reference for relative quantification.
Statistics
All statistical analyses were carried out using SPSS® 16.0 software package (SPSS Inc., Chicago, IL, USA). Data were presented as mean ± standard deviation (SD). Differences between groups were analyzed using Student’s t-test or the chi-square test. Relationships between miR-206 expression and K-Ras protein levels were explored by Pearson’s correlation analysis. Survival curves were constructed with the Kaplan–Meier method and compared by log-rank tests. To evaluate independent prognostic factors associated with survival, a multivariate Cox proportional hazards regression analysis was used. P<0.05 was considered to be statistically significant.
Results
Decreased expression of miR-206 in OSCC and its correlation with K-Ras levels
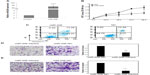
The expression levels of miR-206 in OSCC tissues, corresponding noncancerous samples, human OSCC cell lines SCC-4, SCC-9, SCC-25, and Tca8113, and human normal oral keratinocyte cells were detected by quantitative real-time PCR and normalized to U6 small nuclear RNA. As in Figure 1A, the results showed that the expression levels of miR-206 were significantly lower in OSCC specimens (mean ± SD 8.2±2.1) than those in the corresponding adjacent noncancerous tissues (mean ± SD 19.4±4.1; P<0.001). The miR-206 expression in four OSCC cell lines was also clearly downregulated (Figure 1B). The Tca8113 cell line, which possessed the lowest levels of miR-206 expression among all tested cell lines, was selected for further studies.
K-Ras protein levels were detected by using Western blot analysis in clinical specimens and cell lines. The results showed that K-Ras protein levels in tumor samples were higher than in the adjacent normal tissues (P<0.001; Figure 1C). K-Ras levels in OSCC cells were also higher than in human normal oral keratinocyte cells (Figure 1D). In addition, an obvious inverse correlation (R=−0.4758; P=0.0002) was obtained between K-Ras levels and miR-206 expression in OSCC tumor tissues (Figure 1E).
MiR-206 expression and clinicopathologic features in OSCC
The associations of miR-206 expression with various clinicopathological parameters of OSCC tissues are summarized in Table 1. Using the median miR-206 expression in all 109 OSCC patients as a cutoff, the patients were divided into a high miR-206 expression group and low miR-206 expression group. As shown in Table 1, low expression of miR-206 was significantly correlated with advanced tumor node metastasis (TNM) stage (P<0.001), advanced T classifications (ie, size and/or extent of the tumor; P=0.028), and positive N classification (ie, spread to regional lymph nodes; P=0.001). No significant difference was observed between miR-206 expression and patients’ age, sex, and tumor differentiation.
Correlation between miR-206 expression and prognosis of OSCC patients
It was then evaluated whether miR-206 expression had prognostic potential for overall survival of OSCC patients. Using the Kaplan–Meier method and log-rank test, the overall survival of patients with low miR-206 expression was significantly shorter than in those with high miR-206 expression (P<0.001; Figure 2). The survival benefits were also found in those with early T classification (P=0.008), negative N classification (P=0.006), and early TNM stage (P<0.001; Table 2).
Multivariate Cox regression analysis enrolling the abovementioned significant parameters revealed that miR-206 expression (relative risk [RR] 6.245; P=0.015), T classification (RR 3.655; P=0.032), N classification (RR 4.278; P=0.022), and TNM stage (RR 8.379; P=0.008) were independent prognostic markers for overall survival of OSCC patients (Table 2).
Effects of miR-206 on the proliferation, apoptosis, invasion, and migration of Tca8113 cells
The biological role of miR-206 in Tca8113 cells was also assessed. As shown in Figure 3A, the expression level of miR-206 in transfected cells of miR-206 mimics was significantly higher compared with noncancerous transfected cells (P<0.001). MTT assay showed that cell proliferation was significantly impaired after miR-206 mimics transfection (Figure 3B). Promoted cell apoptosis in transfected cells of miR-206 mimics was also observed (Figure 3C).
Cell invasion is a significant aspect of cancer progression and involves the migration of tumor cells into contiguous tissues and the dissolution of extracellular matrix proteins. Transwell invasion and migration assays were performed to investigate whether miR-206 had a direct influence on Tca8113 cell migration and invasion. As shown in Figure 3D and E, upregulation of miR-206 impeded cell invasion/migration compared with the control.
K-Ras is the target gene of miR-206
Using the algorithms for target gene prediction, K-Ras was identified as one of the potential targets of miR-206. The predicted binding of miR-206 with K-Ras 3′ untranslated region is illustrated in Figure 4A. To further confirm that K-Ras is the direct target of miR-206 in OSCC, miR-206 mimics were transfected into Tca8113 cells and it was found that miR-206 mimics significantly reduced K-Ras protein levels in these cells (Figure 4B). Then, pGL3–K-Ras and pGL3–K-Ras–mut plasmids were created. The reporter assay revealed that transfection of miR-206 mimics triggered a marked decrease of luciferase activity of the pGL3–K-Ras plasmid in Tca8113 cells without change in the luciferase activity of pGL3–K-Ras–mut (Figure 4C). These data indicate that K-Ras is a direct target of miR-206 in OSCC.
Discussion
Dysregulation of miRs has been demonstrated to be involved in tumorigenesis and progression in various types of tumor; however, elucidation of their potential roles in OSCC remains in the early stage of development. In this study, it was found that miR-206 was downregulated in OSCC cell lines and primary tumor samples. Decreased miR-206 expression was significantly correlated with aggressive clinicopathological features and poor survival. Moreover, transfection of miR-206 mimics in Tca8113 cells was able to reduce cell proliferation, invasion, and migration and promote cell apoptosis. Finally, K-Ras was identified as a direct target of miR-206. To the authors’ knowledge, this study reported the prognostic significance of miR-206 in OSCC and confirmed K-Ras as a functional target of miR-206 for the first time.
Previous research reported dysregulated miR-206 expression and its tumor suppressive function in many human malignancies. In vitro, ectopic miR-206 expression inhibits cell growth and induces apoptosis in breast cancer,29 lung cancer,11 gastric cancer,12 glioma and neuroblastoma,16 rhabdomyosarcoma,17 and endometrial endometrioid carcinoma cells.15 Upregulation of miR-206 also reduces cell invasion and migration in lung cancer,11 rhabdomyosarcoma,17 and endometrial endometrioid carcinoma cells.15 In vivo, Bao et al revealed decreased miR-206 expression and its correlation with a worse stage, poor histological differentiation, and metastasis in patients with osteosarcoma.18 Yang et al reported that miR-206 downregulation occurs more frequently in gastric cancer patients with lymph node metastasis, along with the presence of venous invasion, hematogenous recurrence, and advanced tumor stage.19 Moreover, lower expression of miR-206 indicated worse prognosis of patients suffering gastric cancer or colorectal carcinoma.19,20 In xenotransplanted models, miR-206-treated nude mice showed smaller tumor sizes and lower tumor weights compared with the control group.12,30 Taken together, these findings suggest that miR-206 might play an important role not only in tumor initiation and progression but also in the molecular targeted therapy of human malignancies.
It is now clear that miRNAs execute their oncogenic or tumor suppressor functions by regulating the expression of target genes.31 With regard to miR-206, several targets have been reported in recent research including CCND2,12 Otx2,16 MET,30 Pax7,32 NOTCH3,33 ERα,34 and VEGF.35 Ras is involved in the execution of important steps in tumorigenesis and miR-induced K-Ras dysregulation is frequently observed in human cancers. MiR-18a* repression upregulated K-Ras expression, increased cell proliferation, and promoted anchorage-independent growth in soft agar of human squamous carcinoma A431 cells, colon adenocarcinoma HT-29 cells, and fetal hepatic WRL-68 cells.36 MiR-96 decreased pancreatic cancer cell invasion and migration and slowed tumor growth in a manner associated with K-Ras downregulation.26 MiR-143 suppressed K-Ras and functioned as a tumor suppressor in colorectal cancer.27 In OSCC, miR-181a reduced cell proliferation by depressing K-Ras.25 The current study also demonstrated that K-Ras is a functional target of miR-206 in OSCC. The overexpression of miR-206 in Tca8113 cells decreased K-Ras protein levels. After cotransfection of the pGL3 reporter vectors and miR-206 mimics, upregulation of miR-206 resulted in a significant decrease in the luciferase activity of the wild-type pGL3–K-Ras plasmid, whereas mutation of the miR-206 binding site blocked this effect. However, there is no “one-to-one” connection between miRs and target messenger RNAs. An average miR can have more than 100 targets.37 Conversely, several miRs can converge on a single transcript target.38 Previous studies have proven that some other target genes of miR-206, such as VEGF and Notch,39,40 also modulate OSCC pathogenesis. Therefore, the potential regulatory circuitry afforded by miR-206 is enormous, and the accurate mechanisms on how miR-206 influences OSCC progression need further clarification.
There are some limitations in this work. First, the clinical part was a retrospective study, and the tumor sample size was relatively small. Second, the effects of miR-206 on the biological behavior of OSCC cells were only observed in one cell line, although Tca8113 cells possessed the lowest miR-206 expression among all tested cell lines in this study. Further experimental validation using a large number of tumor samples and more cell lines will be performed in the future.
Conclusion
The results revealed that miR-206 was downregulated in OSCC cell lines and clinical samples. Restored miR-206 expression in Tca8113 cells exhibited antitumor effects in vitro. In addition, K-Ras was confirmed as a direct target of miR-206. These findings indicate that miR-206 may act as a tumor suppressor in OSCC and could serve as a novel therapeutic agent for miR-based therapy.
Disclosure
The authors report no conflicts of interest in this work.
References
Sharma P, Saxena S, Aggarwal P. Trends in the epidemiology of oral squamous cell carcinoma in Western UP: an institutional study. Indian J Dent Res. 2010;21(3):316–319. | |
Massano J, Regateiro FS, Januario G, Ferreira A. Oral squamous cell carcinoma: review of prognostic and predictive factors. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;102(1):67–76. | |
Rodrigo JP, Lequerica-Fernandez P, Rosado P, Allonca E, Garcia-Pedrero JM, de Vicente JC. Clinical significance of annexin A2 downregulation in oral squamous cell carcinoma. Head Neck. 2011;33(12):1708–1714. | |
Lin YT, Chuang HC, Chen CH, et al. Clinical significance of erythropoietin receptor expression in oral squamous cell carcinoma. BMC Cancer. 2012;12:194. | |
Osman A. MicroRNAs in health and disease – basic science and clinical applications. Clin Lab. 2012;58(5–6):393–402. | |
Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116(2):281–297. | |
Zhang B, Pan X, Cobb GP, Anderson TA. MicroRNAs as oncogenes and tumor suppressors. Dev Biol. 2007;302(1):1–12. | |
Dieckmann KP, Spiekermann M, Balks T, et al. MicroRNAs miR-371-3 in serum as diagnostic tools in the management of testicular germ cell tumours. Br J Cancer. 2012;107(10):1754–1760. | |
Takahashi M, Cuatrecasas M, Balaguer F, et al. The clinical significance of miR-148a as a predictive biomarker in patients with advanced colorectal cancer. PLoS One. 2012;7(10):e46684. | |
Iorio MV, Ferracin M, Liu CG, et al. MicroRNA gene expression deregulation in human breast cancer. Cancer Res. 2005;65(16):7065–7070. | |
Wang X, Ling C, Bai Y, Zhao J. MicroRNA-206 is associated with invasion and metastasis of lung cancer. Anat Rec (Hoboken). 2011;294(1):88–92. | |
Zhang L, Liu X, Jin H, et al. MiR-206 inhibits gastric cancer proliferation in part by repressing cyclinD2. Cancer Lett. 2013;332(1):94–101. | |
Parasramka MA, Dashwood WM, Wang R, et al. A role for low-abundance miRNAs in colon cancer: the miR-206/Kruppel-like factor 4 (KLF4) axis. Clin Epigenetics. 2012;4(1):16. | |
Zhou L, Chen J, Li Z, et al. Integrated profiling of microRNAs and mRNAs: microRNAs located on Xq27.3 associate with clear cell renal cell carcinoma. PLoS One. 2010;5(12):e15224. | |
Chen X, Yan Q, Li S, et al. Expression of the tumor suppressor miR-206 is associated with cellular proliferative inhibition and impairs invasion in ERα-positive endometrioid adenocarcinoma. Cancer Lett. 2012;314(1):41–53. | |
Wang R, Hu Y, Song G, et al. MiR-206 regulates neural cells proliferation and apoptosis via Otx2. Cell Physiol Biochem. 2012;29(3–4):381–390. | |
Missiaglia E, Shepherd CJ, Patel S, et al. MicroRNA-206 expression levels correlate with clinical behaviour of rhabdomyosarcomas. Br J Cancer. 2010;102(12):1769–1777. | |
Bao YP, Yi Y, Peng LL, et al. Roles of microRNA-206 in osteosarcoma pathogenesis and progression. Asian Pac J Cancer Prev. 2013;14(6):3751–3755. | |
Yang Q, Zhang C, Huang B, et al. Downregulation of microRNA-206 is a potent prognostic marker for patients with gastric cancer. Eur J Gastroenterol Hepatol. 2013;25(8):953–957. | |
Vickers MM, Bar J, Gorn-Hondermann I, et al. Stage-dependent differential expression of microRNAs in colorectal cancer: potential role as markers of metastatic disease. Clin Exp Metastasis. 2012;29(2):123–132. | |
Shih C, Weinberg RA. Isolation of a transforming sequence from a human bladder carcinoma cell line. Cell. 1982;29(1):161–169. | |
Caulin C, Nguyen T, Longley MA, Zhou Z, Wang XJ, Roop DR. Inducible activation of oncogenic K-ras results in tumor formation in the oral cavity. Cancer Res. 2004;64(15):5054–5058. | |
Funato T, Ishii T, Kambe M, Scanlon KJ, Sasaki T. Anti-K-ras ribozyme induces growth inhibition and increased chemosensitivity in human colon cancer cells. Cancer Gene Ther. 2000;7(3):495–500. | |
Fleming JB, Shen GL, Holloway SE, Davis M, Brekken RA. Molecular consequences of silencing mutant K-ras in pancreatic cancer cells: justification for K-ras-directed therapy. Mol Cancer Res. 2005;3(7):413–423. | |
Shin KH, Bae SD, Hong HS, Kim RH, Kang MK, Park NH. MiR-181a shows tumor suppressive effect against oral squamous cell carcinoma cells by downregulating K-ras. Biochem Biophys Res Commun. 2011;404(4):896–902. | |
Yu S, Lu Z, Liu C, et al. MiRNA-96 suppresses KRAS and functions as a tumor suppressor gene in pancreatic cancer. Cancer Res. 2010;70(14):6015–6025. | |
Chen X, Guo X, Zhang H, et al. Role of miR-143 targeting KRAS in colorectal tumorigenesis. Oncogene. 2009;28(10):1385–1392. | |
Zhao WG, Yu SN, Lu ZH, Ma YH, Gu YM, Chen J. The miR-217 microRNA functions as a potential tumor suppressor in pancreatic ductal adenocarcinoma by targeting KRAS. Carcinogenesis. 2010;31(10):1726–1733. | |
Leivonen SK, Makela R, Ostling P, et al. Protein lysate microarray analysis to identify microRNAs regulating estrogen receptor signaling in breast cancer cell lines. Oncogene. 2009;28(44):3926–3936. | |
Taulli R, Bersani F, Foglizzo V, et al. The muscle-specific microRNA miR-206 blocks human rhabdomyosarcoma growth in xenotransplanted mice by promoting myogenic differentiation. J Clin Invest. 2009;119(8):2366–2378. | |
Liu GF, Tang D, Li P, et al. S-1-based combination therapy vs S-1 monotherapy in advanced gastric cancer: a meta-analysis. World J Gastroenterol. 2014;20(1):310–318. | |
Dey BK, Gagan J, Dutta A. MiR-206 and -486 induce myoblast differentiation by downregulating Pax7. Mol Cell Biol. 2011;31(1):203–214. | |
Song G, Zhang Y, Wang L. MicroRNA-206 targets notch3, activates apoptosis, and inhibits tumor cell migration and focus formation. J Biol Chem. 2009;284(46):31921–31927. | |
Kondo N, Toyama T, Sugiura H, Fujii Y, Yamashita H. MiR-206 expression is down-regulated in estrogen receptor α-positive human breast cancer. Cancer Res. 2008;68(13):5004–5008. | |
Zhang T, Liu M, Wang C, Lin C, Sun Y, Jin D. Down-regulation of miR-206 promotes proliferation and invasion of laryngeal cancer by regulating VEGF expression. Anticancer Res. 2011;31(11):3859–3863. | |
Tsang WP, Kwok TT. The miR-18a* microRNA functions as a potential tumor suppressor by targeting on K-Ras. Carcinogenesis. 2009;30(6):953–959. | |
Brennecke J, Stark A, Russell RB, Cohen SM. Principles of microRNA-target recognition. PLoS Biol. 2005;3(3):e85. | |
Krek A, Grun D, Poy MN, et al. Combinatorial microRNA target predictions. Nat Genet. 2005;37(5):495–500. | |
Margaritescu C, Pirici D, Simionescu C, et al. VEGF and VEGFRs expression in oral squamous cell carcinoma. Rom J Morphol Embryol. 2009;50(4):527–548. | |
Zhang T, Liu H, Liang Y, et al. [The expression and significance of the Notch signaling pathway molecules in tongue squamous cell carcinoma]. Hua Xi Kou Qiang Yi Xue Za Zhi. 2013;31(3):303–309. Chinese. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.