Back to Journals » Journal of Pain Research » Volume 13
Medicolegal Consideration to Prevent Medical Malpractice Regarding Opioid Administration: An Analysis of Judicial Opinion in South Korea
Authors Kim J , Shin S , Jeong Y, Kim SY, Lee HJ
Received 3 April 2020
Accepted for publication 3 June 2020
Published 25 June 2020 Volume 2020:13 Pages 1525—1532
DOI https://doi.org/10.2147/JPR.S256759
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Robert B. Raffa
Jeongsoo Kim,1 SuHwan Shin,2,3 YoungHyun Jeong,4 So Yoon Kim,5,6 Ho-Jin Lee4,7
1Department of Anesthesiology and Pain Medicine, SMG-SNU Boramae Medical Center, Seoul, Republic of Korea; 2Department of Medical Law and Ethics, Graduate School, Yonsei University, Seoul, Republic of Korea; 3Blue Urology Clinic, Seoul, Republic of Korea; 4Department of Anesthesiology and Pain Medicine, Seoul National University Hospital, Seoul, Republic of Korea; 5Division of Medical Law and Bioethics, Department of Medical Humanities and Social Sciences, Yonsei University College of Medicine, Seoul, Korea; 6Asian Institute for Bioethics and Health Law, Yonsei University, Seoul, Korea; 7Department of Anesthesiology and Pain Medicine, Seoul National University College of Medicine, Seoul, Republic of Korea
Correspondence: Ho-Jin Lee
Department of Anesthesiology and Pain Medicine, Seoul National University College of Medicine, Seoul, Republic of Korea
Tel +82-2-2072-0039
Fax +82-2-747-8363
Email [email protected]
Purpose: Although the use of opioids is increasing in South Korea, there have been no studies on the serious complications caused by the opioids. The aim of this study was to investigate the rare but serious complications through medicolegal analysis.
Materials and Methods: From January 1994 to December 2019, we retrospectively reviewed the closed cases of lawsuits involving the complications of opioids using the database of judgments of the Supreme Court of Korea. General characteristics, opioid-induced complications, and judicial characteristics were analyzed.
Results: Of the 46 cases, 31 cases of complications were finally included in the analysis. There were 28 (90.3%) cases of opioid administration for acute pain and 3 (9.7%) cases for chronic pain. The most commonly prescribed opioid was pethidine (n = 13, 41.9%), and the most common complication was respiratory depression (n = 17, 54.8%). All except two cases were associated with permanent injuries, including 18 (58%) deaths. Twelve (38.7%) cases were ruled in favor of the plaintiff in the claims for damages, with a median payment of United States dollar (USD) 126,346 (IQR: USD 77,275– 379,219). Of these cases, the most frequently admitted complaint by the court was the neglect of observation (n = 10, 32.3%), followed by the inappropriate drug choice (n = 4, 12.9%). Eleven (36.7%) cases were plaintiffs’ claims for violating explanation obligations, of which 2 (6.7%) were recognized in the court.
Conclusion: Our results suggest that physicians must be aware of the serious complications related to opioids and health policies to prevent such complications and malpractice should be adopted.
Keywords: acute pain, adverse drug events, complications, legislation and jurisprudence, medical liability, opioids
Introduction
Opioid analgesics have played a pivotal role in the management of acute pain and chronic cancer pain since the 1990s.1,2 More recently, it has also played an important role in the management of chronic non-cancer pain.3,4 Opioids have strong analgesic potency without a ceiling effect unlike other analgesics, and it can be administered through various routes such as transdermal, submucosal, intranasal, oral, neuraxial, or intravenous routes.4 As a result, opioids are now being widely used for pain management, and their use has increased dramatically in South Korea.5 According to the Adequacy of Consumption Measure (ACM) of opioids in 2010, opioid usage in South Korea has grown exponentially, with a 77-fold increase in ACM ratio between 2006 and 2010.6 In addition, a recent study conducted in South Korea also showed a dramatic increase in opioid use.7 Understandably, with the increase in opioid use, there is growing concern about the side effects of opioids in South Korea.8,9
Opioids can cause serious side effects despite its many advantages, so physicians need to be careful when prescribing them.10 Opioid usage can cause a wide range of side effects, from mild symptoms such as nausea and vomiting to fatal complications such as respiratory depression in the acute period of administration.11 Long-term use of opioids can result in addiction and abuse, with its prevalence reported to be 26% in the United States.12 In addition, chronic opioid use has recently been reported to be associated with long-term mortality.7 As opioid prescription increases rapidly, the serious complications caused by opioids are expected to increase as well.
However, to our knowledge, there have been no previous studies evaluating serious complications as a result of opioid administration in South Korea. Therefore, in this study, we attempted to investigate the very rare but serious complications associated with opioid administration through the analysis of medical malpractice lawsuits.13 Through this study, we hope to increase awareness regarding catastrophic complications of opioids and to prevent medical malpractice associated with opioid administration.
Materials and Methods
We analyzed adjudicated lawsuits that are publicly accessible in the database of the Supreme Court of Korea’s judgments. This database contains both civil and criminal proceedings sentenced from the levels of the district court to the Supreme Court. The details of each case were provided to the researcher without identifiable personal information. All medical malpractice litigations that were sentenced from January 1, 1994 to December 31, 2019 were queried using the search terms “opioid” and “narcotic analgesics”. We included the cases in which the malpractice associated with opioid administration was contained in the plaintiff’s claims. We excluded cases unrelated to opioid administration based on the pain physician’s judgment (J Kim and H-J Lee). This study was approved by the institutional review board (IRB) of Seoul National University Hospital (IRB no. 2003–048-1106).
Each lawsuit text contained a detailed description of the case, the malpractice claims of the plaintiff, and the court decisions regarding medical malpractice. Two board-certified pain physicians (J Kim and H-J Lee) reviewed the judgment texts and collected the following information: year of the event, age, sex, underlying medical diseases, causes of opioid administration, parameters related to opioid administration (type, route, number of doses), and the types and severity of the complications.
The severity of complications was evaluated using the 10-point National Association of Insurance Commissioners (NAIC) scale where 0 is “no obvious injury” and 9 is “death” (0: No obvious injury; 1: Emotional only; 2: Temporary insignificant; 3: Temporary minor; 4: Temporary major; 5: Permanent minor; 6: Permanent significant; 7: Permanent major; 8: Permanent grave; 9: Death).14
Data regarding the detailed claims of plaintiffs, the opinion of the court, and the final financial compensation awarded were also collected. The plaintiffs’ allegations that were related to opioid administration were classified into the following two categories; violation of the duty of care and violation of the duty of explanation. Each case was also investigated to determine if it was accepted by the court. After the first review, the violation of duty of care that was identified in our data was further classified into the following three categories; “Neglect of observation”, “Inappropriate drug choice”, and “Overdose”. “Neglect of observation” was defined as inappropriate monitoring after opioid administration. “Inappropriate drug choice” was defined as when the administration of opioids was not appropriate considering the patient’s condition. “Overdose” was defined as the inappropriate dosage of opioids that resulted in the occurrence of complications. The classification and judgment of medical malpractice was conducted independently by two pain physicians (J Kim and H-J Lee). In the case of a conflict between the authors, a review by an author of medical law and ethics (SH Shin) was conducted.
Descriptive statistics were conducted using the MedCalc Statistical Software version 18.6 (MedCalc Software bvba, Ostend, Belgium). Categorical data are described as percentages, and continuous data are described as medians (interquartile range [IQR]).
Results
There was a total of 46 cases (from 69 judicial precedents) during the study period. Of these cases, 15 cases were excluded and a total of 31 cases were included in the final analysis (Figure 1). Detailed information of our cases is provided in supplementary Table 1. The clinical characteristics of the patients are presented in Table 1. There were 28 (90.3%) cases of opioid administration for acute pain and 3 (9.7%) cases for chronic pain. The most common cause of opioid administration was acute abdominal pain (n = 9, 29%), followed by acute postoperative pain (n=8, 25.8%). There were two fetuses (case no. 5, 23) and one 6-month old infant (case no. 21) among those who were adversely affected by opioid use. In the case of the fetuses, the opioid was administered to the pregnant woman for labor pain relief.
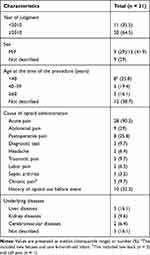 |
Table 1 General Characteristics of the Cases |
 |
Figure 1 Flowchart of the study. |
Table 2 shows the detailed information of opioid administration and opioid-induced complications. The most commonly used opioid was pethidine (n = 13, 41.9%). The most common complication following opioid administration was respiratory depression (n = 17, 54.8%). All except two were associated with permanent injuries, with a total of 18 deaths.
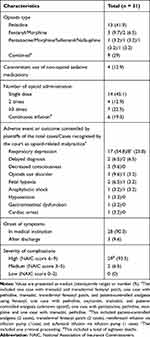 |
Table 2 Detailed Information of Opioid Administration and Opioid-Induced Complications |
Judgment statuses are shown in Table 3. One case was a criminal proceeding while the others were all civil proceedings. In the criminal proceeding, the defendant was sentenced to 10 months in a suspended sentence for the negligence of observation after opioid administration (case no. 31). This criminal proceeding did not overlap with the civil proceedings. Of the 30 civil proceedings, 15 were dismissed by the court. Three of the remaining cases were not dismissed, but the malpractice associated with opioids claimed by the plaintiff was not recognized. The remaining 12 claims (38.7%) associated with malpractice of opioid administration resulted in payments to the plaintiffs, with a median payment of United States Dollar (USD) 126,346 (IQR: USD 77,275–379,219). Violation of the duty of care related to opioid prescription was claimed by plaintiffs in 30 cases. Of these cases, 12 (38.7%) were ruled as violation of the duty of care related to opioid prescription by the court. This violation of duty that was ruled physician malpractice included neglect of observation after opioid administration (n = 10), inappropriate drug choice (n = 4), and overdose (n = 3). Violation of the physician’s duty of informed consent was claimed by plaintiffs in 11 (36.7%) cases. Of these cases, 2 (6.7%) were ruled as medical malpractice by the court. Of the cases in which the court recognized opioid-related medical malpractices, there were four deaths, three cases of vegetative states, and three cases of quadriplegia.
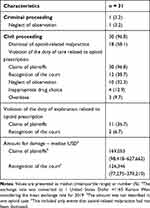 |
Table 3 Judicial Characteristics from 31 Cases Related to Opioid Administration |
Discussion
This study analyzed 31 medical malpractice lawsuits related to the complications of opioid administration in the Korean court system. Our major findings were that in the majority of cases, opioids were administered for acute pain, not chronic pain. The most common opioid encountered in this study was pethidine and the most common type of complication was respiratory depression. Neglect of observation after opioid administration was the most commonly ruled physician malpractice. Except for two, all cases were associated with permanent injuries.
Previous medicolegal studies on the complications of opioid administration have been reported. According to the analysis of the closed claims database of the American Society of Anesthesiologists (ASA) from 2005 to 2008, 48 claims were related to opioid prescriptions, which accounted for 94% of medication management claims (n = 51) in chronic pain management.15 In that study, the most common outcome was death (n = 29, 57%), followed by addiction (n = 12, 24%). In another analysis of 35 malpractice lawsuits involving opioid-related overdose in patients with chronic pain in the United States, there were a total of 20 deaths and the most commonly implicated opioid was methadone (n = 10, 50%).16 Unlike the above two studies, we did not limit the study population to chronic pain patients, and consequently, the results of our study were different from them. Until the 2000s, the use of opioids for treatment, especially in chronic non-cancer pain, was limited in South Korea.17 However, the use of opioids in patients with chronic non-cancer pain has increased recently in Korea5,7 and the guidelines for their use in these patients were recently published.4 Therefore, in our study, most cases were associated with acute pain rather than chronic pain. In addition, few cases were associated with addiction and the most commonly used opioid in our study was pethidine, and it was mainly used for the treatment of acute pain.
Although we searched for cases from 1994, there were only two cases in the 1990s and two-thirds of the cases occurred post 2010. Since we included only the closed cases, some lawsuits from recent years may not be included in our study. Therefore, there may be more cases in reality than those reported here since 2010. An analysis of the trends in pain medicine claims of the ASA closed claims database reported that only 2% of pain medicine claims in the 1980s were related to medication mismanagement; however, this has increased significantly since 2000 by 17%.18 The authors described that this finding was associated with the national trends of opioid prescriptions. As described in the introduction, opioid use in Korea has also increased steadily, and the problems related to opioid overuse are expected to increase gradually in the future. Therefore, it is meaningful to investigate the serious complications of opioid administration in Korea and to raise its awareness among pain physicians who commonly prescribe them.
In our study, respiratory depression was the most common complication after opioid administration. Opioids have a direct inhibitory effect on neurons expressing μ - opioid peptide receptors at the respiratory centers in the brainstem. Opioid-induced respiratory depression (OIRD) leads to decreased oxygen supply to the brain, resulting in hypoxic brain injury. An analysis of ASA closed claim database from 1990 to 2009 found that of the 357 claims associated with postoperative pain management, 92 claims were associated with OIRD, of which 77% resulted in severe brain damage or death.19 In this study, respiratory depression was the most common complication, with very poor outcomes.
To prevent catastrophic outcomes after opioid administration, physicians need to be mindful of the possible complications of opioids. In our study, negligence of observation after opioid administration was the most common opioid-related malpractice recognized by the court. In addition, despite most of these complications occurring in inpatients, there was only one instance of administration of naloxone, the only antidote for OIRD. Based on these findings, we cautiously claim that there may be a lack of vigilance and a proper monitoring system of OIRD in Korea. According to the American society for pain management nursing (ASPMN) guidelines on monitoring for opioid-induced sedation and respiratory depression, serial sedation and respiratory assessments are recommended during opioid therapy.20 The assessment of sedation is very important in patients receiving opioid administration because sedation precedes OIRD. It is also important to assess the patients’ respiratory rate periodically, because a decreased respiratory rate is a premonitory sign of OIRD. If a patient is at risk for OIRD, it is recommended that continuous pulse oximetry and capnography monitoring be performed.21 A recent multicenter study reported that the implementation of ASPMN guidelines related to OIRD had a positive effect on the management of OIRD.22
It is recommended that in the outpatient setting, opioid administration should be started with the minimum dose and gradually increased as necessary.4 In one of the cases not dismissed by the court, a transdermal fentanyl patch was prescribed to an opioid-naive patient with an initial dose of 50 mcg in an outpatient setting (case no. 18). Although the analgesic effect may be insufficient in the early stage, this should be explained to the patient and the opioid titration performed over a safe period of time. Since OIRD is not an unavoidable complication like anaphylaxis, rather a preventable one, medical staff should be watchful for this complication and a system to aid its early detection and prevention must be in place.
In addition to practices to prevent OIRD, the following precautions are necessary while administering opioids. Firstly, the patient’s complete medical history which includes pre-existing diseases should be obtained before opioid administration. In one case in our study, pethidine use in a patient with increased intracranial pressure from intracranial bleeding was recognized as a malpractice (case no. 16). Pethidine, which was most commonly implicated in this study, requires caution in its use because it has many contraindications such as bronchial asthma, heart failure, convulsive disorders, and increased intracranial pressure, different from other opioids.23 Pethidine is not recommended in both acute and chronic pain because it has a wider side-effect profile than other opioids but no specific advantage.24 In one case related to anaphylaxis, it was recognized as a malpractice to re-administer pentazocine to a patient who had previously experienced a hypersensitivity reaction to it (case no. 1). In patients with a history of hypersensitivity to a specific drug, additional exposure to that may result in anaphylaxis.25 Therefore, history taking of drug hypersensitivity is very important. In addition, patients with liver or kidney disease can overdose inadvertently if their doses are not adjusted for impaired renal or hepatic drug clearance, with side effects occurring at doses considered safe for the general population.26 Secondly, opioid administration can cause the physician to miss important diagnostic clues. In two cases of the study, the delayed diagnoses (postoperative compartment syndrome and small bowel perforation) due to opioid administration were recognized as the malpractice by the court (case no. 10, 13). In the past, the use of opioids had been discouraged in emergencies such as acute abdominal pain due to the concerns about masking important diagnostic clues. However, it has been reported that opioid analgesia is safe and does not delay diagnosis in emergencies.27 Nevertheless, accurate diagnosis should be given significance over analgesic administration during emergencies and physicians should not neglect the diagnosis of pain-causing diseases while providing adequate analgesia. Lastly, opioid abuse should be considered especially when opioids are prescribed for long-term use.4,28 There is only one such case in our study (case no. 3), and to date, Korea has seemed relatively safe from this problem.9 However, the opioid use in chronic non-cancer pain patients has recently increased in South Korea7 and the recent legalization of medical marijuana in South Korea could provoke the problem related to opioid abuse.29 A recently reported single-center study in South Korea reported that one-fourth of the study participants showed opioid use disorders.30 In the future, this problem is expected to increase in South Korea as seen in the Western countries. Therefore, Korean physician will have to pay attention to this problem.
The results of our study should be interpreted cautiously for several reasons. Firstly, our study could not represent the comprehensive features of opioid-related complications. Due to the highly contentious nature of our data, the complications in our study skewed toward rare and serious complications. Further, cases that ended in agreement or arbitration have not been included here. In a previous study of medical disputes regarding pain management in Korea using insurance database, there were no cases related to opioids.31 Another recent study using the Korea Medical Dispute Medication and Arbitration Agency (KMDMAA) data from 2012 to 2016 reported that a total of four cases were associated with medical complications or drug side effects.32 According to the additional data that we directly obtained through the KMDMAA, there were a total of six cases associated with opioids from 2012 to 2018. However, we excluded this data from the analysis because of the unavailability of detailed clinical information to determine the causality between opioid use and outcomes. In addition, in Korea, the act of medical malpractice damage’s relief and medication for medical dispute resolution has been enacted since 2012.33 Considering the previous legal environment, many medical malpractices before 2012 might not lead to litigation. Secondly, the clinical features described in judicial sentences had limitations, especially in cases that were dismissed. Lastly, although medical appraisers’ opinions play an important role in judges’ malpractice decisions, a judge’s subjectivity may also affect the decision. Despite these limitations, this study provides useful information on rare adverse events that would be difficult to study prospectively without an expensive and time-consuming large multicenter design. Awareness of these rare, but critical, adverse events may help pain physicians to recognize these complications earlier, and prompt them to take remedial action before it is too late.
Conclusion
In conclusion, healthcare professionals should be acquainted with the risk of serious adverse events surrounding the prescription of opioids. In addition, national health policies to prevent such fatal complications are necessary to ensure patient safety and to mitigate medical liability.
Acknowledgments
We would like to thank Editage for English language editing.
Disclosure
The authors declare no conflicts of interest in this work.
References
1. Austrup ML, Korean G. Analgesic agents for the postoperative period: opioids. Surg Clin North Am. 1999;79(2):253–273. doi:10.1016/S0039-6109(05)70382-0
2. Moon DE. Treatment of cancer pain. Korean J Pain. 2002;15(1):1–12.
3. Park HJ, Moon DE. Pharmacologic management of chronic pain. Korean J Pain. 2010;23(2):99–108. doi:10.3344/kjp.2010.23.2.99
4. Kim ED, Lee JY, Son JS, et al. Guidelines for prescribing opioids for chronic non-cancer pain in Korea. Korean J Pain. 2017;30(1):18–33. doi:10.3344/kjp.2017.30.1.18
5. Cho YS, Lee JY, Kim HS, Kwon K. Trends in the consumption of opioid analgesics in a tertiary care hospital from 2000 to 2012. Yakhak Hoeji. 2014;58(4):268–276.
6. Duthey B, Scholten W. Adequacy of opioid analgesic consumption at country, global, and regional levels in 2010, its relationship with development level, and changes compared with 2006. J Pain Symptom Manage. 2014;47(2):283–297. doi:10.1016/j.jpainsymman.2013.03.015
7. Oh TK, Jeon YT, Choi JW. Trends in chronic opioid use and association with five-year survival in South Korea: a population-based cohort study. Br J Anaesth. 2019;123(5):655–663. doi:10.1016/j.bja.2019.08.012
8. Kim YD. Opioid: toward an effective strategy for better use. Korean J Pain. 2019;32(2):67–68. doi:10.3344/kjp.2019.32.2.67
9. Lee JH. The opioid epidemic and crisis in US: how about Korea? Korean J Pain. 2019;32(4):243–244. doi:10.3344/kjp.2019.32.4.243
10. Chapman CR. Opioid pharmacotherapy for chronic noncancer pain: the American experience. Korean J Pain. 2013;26(1):3–13. doi:10.3344/kjp.2013.26.1.3
11. Ricardo Buenaventura M, Rajive Adlaka M, Nalini Sehgal M. Opioid complications and side effects. Pain Physician. 2008;11:S105–S120.
12. Boscarino JA, Rukstalis M, Hoffman SN, et al. Risk factors for drug dependence among out patients on opioid therapy in a large US health care system. Addiction. 2010;105(10):1776–1782. doi:10.1111/j.1360-0443.2010.03052.x
13. Cho SI, Shin S, Jung H, Moon JY, Lee HJ. Analysis of judicial precedent cases regarding epidural injection in chronic pain management in Republic of Korea. Reg Anesth Pain Med. 2020;45(5):337–343. doi:10.1136/rapm-2019-101169
14. Sowka MP. The medical malpractice closed claims study. Conducted by the national association of insurance commissioners. Conn Med. 1981;45(2):91–101.
15. Fitzgibbon DR, Rathmell JP, Michna E, Stephens LS, Posner KL, Domino KB. Malpractice claims associated with medication management for chronic pain. Anesthesiology. 2010;112(4):948–956. doi:10.1097/ALN.0b013e3181cdef98
16. Rich BA, Webster LR. A review of forensic implications of opioid prescribing with examples from malpractice cases involving opioid-related overdose. Pain Med. 2011;12:S59–S65. doi:10.1111/j.1526-4637.2011.01129.x
17. Han T. Opioids in cancer and non-cancer pain management in Korea: the past, present and future. Eur J Pain. 2001;5(Suppl A):73–78. doi:10.1053/eujp.2001.0284
18. Pollak KA, Stephens LS, Posner KL, et al. Trends in pain medicine liability. Anesthesiology. 2015;123(5):1133–1141. doi:10.1097/ALN.0000000000000855
19. Lee LA, Caplan RA, Stephens LS, et al. Postoperative opioid-induced respiratory depression: a closed claims analysis. Anesthesiology. 2015;122(3):659–665. doi:10.1097/ALN.0000000000000564
20. Jarzyna D, Jungquist CR, Pasero C, et al. American society for pain management nursing guidelines on monitoring for opioid-induced sedation and respiratory depression. Pain Manag Nurs. 2011;12(3):118–145. doi:10.1016/j.pmn.2011.06.008
21. Lam T, Nagappa M, Wong J, Singh M, Wong D, Chung F. Continuous pulse oximetry and capnography monitoring for postoperative respiratory depression and adverse events: a systematic review and meta-analysis. Anesth Analg. 2017;125(6):2019–2029. doi:10.1213/ANE.0000000000002557
22. Kamendat J, Dabney BW, McFarland M, Gilbert GE, Richards-Weatherby K. Implementing a clinical practice guideline on opioid-induced advancing sedation and respiratory depression. J Nurs Care Qual. 2020;35(1):13–19. doi:10.1097/NCQ.0000000000000406
23. World Health Organization. WHO model prescribing information: drugs used in anaesthesia. Geneva, World Health Organization. 1989;39–40.
24. McQuay H. Opioids and pain management. Lancet. 1999;353(9171):2229–2232. doi:10.1016/S0140-6736(99)03528-X
25. Hyeon CW, Lee JY, Jang S, et al. Medical malpractice related to drug-induced anaphylaxis: an analysis of lawsuit judgments in South Korea. Medicine. 2019;98(23):e15996. doi:10.1097/MD.0000000000015996
26. Webster LR, Cochella S, Dasgupta N, et al. An analysis of the root causes for opioid-related overdose deaths in the United States. Pain Med. 2011;12(Suppl 2):S26–S35. doi:10.1111/j.1526-4637.2011.01134.x
27. Manterola C, Vial M, Moraga J, Astudillo P. Analgesia in patients with acute abdominal pain. Cochrane Database Syst Rev. 2011;(1):CD005660. doi:10.1002/14651858.CD005660.pub3
28. Sehgal N, Manchikanti L, Smith HS. Prescription opioid abuse in chronic pain: a review of opioid abuse predictors and strategies to curb opioid abuse. Pain Physician. 2012;15:ES67–ES92.
29. Kim HJ. Korea to allow imports of medical cannabis starting in March. Yonhap news agency. Sect. 2019; 13: All Headlines.
30. Lee CS, Kim D, Park SY, Lee SC, Kim YC, Moon JY. Usefulness of the Korean version of the CAGE-adapted to include drugs combined with clinical predictors to screen for opioid-related aberrant behavior. Anesth Analg. 2019;129(3):864–873. doi:10.1213/ANE.0000000000003580
31. Kim YD, Moon HS. Review of medical dispute cases in the pain management in Korea: a medical malpractice liability insurance database study. Korean J Pain. 2015;28(4):254–264. doi:10.3344/kjp.2015.28.4.254
32. Lee JH, Song J, Kuk YH, Ha JR, Kim YD. Dispute cases related to pain management in Korea: analysis of Korea medical dispute mediation and arbitration agency data. Anesth Pain Med. 2020;15(1):96–102. doi:10.17085/apm.2020.15.1.96
33. Shin EJ. The perspective of the system on mediation in the act of medical malpractice damage’s relief and mediation for medical dispute resolution. Korean J Med Law. 2011;19:133–159.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
