Back to Journals » Journal of Multidisciplinary Healthcare » Volume 9
Managing the pediatric patient with celiac disease: a multidisciplinary approach
Authors Isaac DM, Wu J, Mager DR, Turner JM
Received 15 August 2016
Accepted for publication 7 September 2016
Published 13 October 2016 Volume 2016:9 Pages 529—536
DOI https://doi.org/10.2147/JMDH.S95323
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Daniela Migliarese Isaac,1 Jessica Wu,2 Diana R Mager,3,4 Justine M Turner1
1Department of Pediatric Gastroenterology and Nutrition, Faculty of Medicine and Dentistry, University of Alberta; 2Alberta Health Services–Child Health Nutrition Services, Stollery Children’s Hospital; 3Department of Agriculture, Food and Nutritional Science; 4Department of Pediatrics, University of Alberta, Edmonton, AB, Canada
Abstract: Celiac disease (CD) is an autoimmune reaction to gluten, leading to intestinal inflammation, villous atrophy, and malabsorption. It is the most common autoimmune gastrointestinal disorder, with an increasing prevalence. A life-long gluten-free diet (GFD) is an effective treatment to alleviate symptoms, normalize autoantibodies, and heal the intestinal mucosa in patients with CD. Poorly controlled CD poses a significant concern for ongoing malabsorption, growth restriction, and the long-term concern of intestinal lymphoma. Achieving GFD compliance and long-term disease control poses a challenge, with adolescents at particular risk for high rates of noncompliance. Attention has turned toward innovative management strategies to improve adherence and achieve better disease control. One such strategy is the development of multidisciplinary clinic approach, and CD is a complex life-long disease state that would benefit from a multifaceted team approach as recognized by multiple national and international bodies, including the National Institutes of Health. Utilizing the combined efforts of the pediatric gastroenterologist, registered dietitian, registered nurse, and primary care provider (general practitioner or general pediatrician) in a CD multidisciplinary clinic model will be of benefit for patients and families in optimizing diagnosis, provision of GFD teaching, and long-term adherence to a GFD. This paper discusses the benefits and proposed structure for multidisciplinary care in improving management of CD.
Keywords: celiac disease, multidisciplinary care, team, pediatric
Introduction
Celiac disease (CD) is an autoimmune reaction to gluten, leading to intestinal inflammation, villous atrophy, and malabsorption.1,2 The prevalence of CD is 1% in the general population,2,3 making it the most common autoimmune gastrointestinal disorder. CD is triggered by the ingestion of gluten present in cereals such as wheat, barley, and rye in genetically susceptible individuals.1,4 Patients are screened for CD using serological testing for anti-tissue transglutaminase (atTG) antibodies, followed most commonly in North America by biopsy confirmation with characteristic intestinal histopathology.1 The cornerstone of management of CD is life-long adherence to a gluten-free diet (GFD), which has been shown to alleviate gastrointestinal and other associated symptoms, normalize serological markers, and heal intestinal mucosa.4–6 Despite widespread recognition of the importance of the GFD and consensus on the goals of treatment, achieving compliance and long-term disease control remains a challenge. Estimates of the rate of compliance vary in the literature from 45% to 60%, many of which are likely overestimates.3,7–10 Adolescents have been identified as a subgroup with a particularly high rate of noncompliance.11
Thus, much attention in the literature has turned toward new and innovative management and follow-up strategies to improve adherence and achieve better disease and symptom control. One such strategy that deserves greater attention is the development of the multidisciplinary clinic (MDC) approach. The North American Society for Pediatric Gastroenterology, Hepatology and Nutrition (NASPGHAN) has published clinical guidelines for the management of CD in children and adolescents, and recommends multidisciplinary collaboration and follow-up.3 However, there exist no published recommendations on the utility of MDC for pediatric CD, nor guidelines on how such a clinic should be structured. The purpose of this article is to present the evidence for the use of an MDC for pediatric CD, as well as to review the current state of the pediatric CD MDCs in Canada, and to propose a structure and strategy for the creation and maintenance of such a clinic.
The case for multidisciplinary care
Multidisciplinary patient care is a rapidly growing feature of modern health care delivery. As the complexity of modern medicine and the needs of patients with chronic diseases increase, it becomes ever more important to involve diverse health care disciplines in the care of complex patients. Much of this is a result of the intense subspecialization that has become a prominent feature of health care in North America. The detailed knowledge and skills required to master a particular discipline makes collaboration with other disciplines critical in providing holistic, well-rounded patient-centered care. This is especially important in the management of patients with complex life-long illnesses such as cardiovascular disease, chronic kidney disease, diabetes, and many others.12–16 Many authors echo the sentiment of Komenda and Levin who argue that “traditional physician-based models of care delivery may no longer be feasible given the growth and complexity” of patients with chronic diseases.12 MDCs are logical not only from a health care professional perspective, but also the increasing scarcity of health care resources demand more streamlined and cost-effective care for patients with chronic diseases, as they represent a large proportion of any health care budget. Coupled with the increasing demand from patients for more holistic, patient-centered, and coordinated care, the MDC has been gaining traction both in the adult and pediatric literature across a widening disease spectrum.
The MDC was first utilized in the management of adults with cardiovascular and kidney disease, where its efficacy and importance was noted. Several studies reported improved outcomes, including blood pressure targets and overall survival in patients managed through an MDC.12,13,17 One randomized controlled trial comparing an MDC with traditional care for the management of congestive heart failure identified fewer hospital admissions, fewer days in hospital, and better overall quality of life for the group managed via the MDC.14 In the pediatric age group, MDCs have been used for the management of patients with spina bifida, chronic kidney disease, diabetes, inflammatory bowel disease (IBD), and functional constipation.15,18–20 With respect to children with kidney disease, one study reported improved clinical markers, including hemoglobin and parathyroid hormone levels, as well as less frequent unplanned initiation of dialysis after the introduction of an MDC at a tertiary care center.15
In addition to improved patient outcomes and clinical indicators, MDCs can also be beneficial in reducing health care costs. Levin et al reported an overall cost savings of USD $4,000 per patient enrolled in an MDC for adults with chronic kidney disease.13 The cost savings associated with MDCs are a result of fewer admissions and, in the case of kidney disease, fewer urgent dialysis interventions. Although some argue against the MDC, quoting the high cost of initiation and maintenance, several studies have shown that a well-planned MDC can more than offset its own cost.13,21 Finally, the majority of studies that have evaluated the use of an MDC have shown high rates of patient satisfaction, which is an important marker of success.14,21,22 This is especially true in pediatrics, where parent and patient satisfaction can be a major determinant in treatment adherence.11,23
The multidisciplinary clinic for celiac disease
Available literature supports the use of MDCs for the long-term management of CD patients. As Komenda and Levin argued, “the basic premise of the MDC model is that complex disease states require multifaceted teams to improve patient outcomes. The model makes intuitive sense in health care environments with an ever-increasing need to provide complicated and efficient care with limited resources.”12 CD fulfills the tenets of a complex life-long disease state that would benefit from a multifaceted team approach. Poorly controlled CD leads to ongoing intestinal inflammation and villous atrophy, thereby increasing the risk of malabsorption, growth restriction, diminished bone health, and the long-term concern of intestinal lymphoma.3 For pediatric patients in particular, critical periods of growth and development can be significantly and negatively impacted by malabsorption due to poorly controlled CD. Adherence to a GFD can prevent or improve the risk of these negative consequences. Many CD patients may face the additional management complexity of being affected by associated health conditions, such as type 1 diabetes mellitus, autoimmune hypothyroidism, Down syndrome, and Turner syndrome.2–4 The increased financial burden of a GFD can create additional challenges for families dealing with CD, with 21% of adult patients reporting the cost of a GFD to be problematic.24 A 2016 study examining the impact of CD on health-related quality of life found that 68% of CD patients felt that a GFD reduced their enjoyment of food, and 54% participated less frequently in enjoyable activities, particularly eating out.24 Sociocultural and language barriers can also lead to adherence concerns and longer time to normalization of atTG antibody titers, as illustrated by a recent Canadian study.25
Multiple national and international bodies, including the National Institutes of Health, have recognized the importance of addressing the complexity of CD care posed by the above challenges. In their consensus report, the National Institutes of Health argues that the six main components of CD management are consultation from a skilled dietitian, lifelong adherence to a GFD, identification and treatment of nutritional deficiencies, access to advocacy groups, education, and long-term follow-up by a multidisciplinary team.26 These last two components are critical, as one Greek study identified poor parental knowledge as the only independent predictor of nonadherence to a GFD in children.10
It is clear that the current rates of achieving GFD compliance must be improved, with some studies reporting the compliance rate to be as low as 45% in children and adolescents.8,10,27 Moreover, GFD adherence in childhood is highly predictive of maintaining follow-up and dietary compliance into adulthood.7 One study found that children who are lost to follow-up are less likely to adhere to a GFD, and have poorer vitamin and nutritional status.28 These represent gaps in the current CD management paradigm that could be addressed by an MDC. Dietary compliance has been shown to improve with regular dietetic review and intervention,29 which can be facilitated through an MDC. Although there is a paucity of studies examining multidisciplinary care in CD, one group from the UK reported an adult CD MDC reduced outpatient wait times, yielded high patient satisfaction, and saved over GBP £11,000 over a period of 2 years.21 Patients also seem to prefer multidisciplinary follow-up care, including regular consultation with a dietitian and yearly follow-up visits at minimum.22 Currently there is limited utilization of multidisciplinary care in CD management, with one adult study reporting only 15% of patients regularly meet with a dietitian.30
Multidisciplinary care beginning in childhood is likely to yield even greater benefits. However, due to the complexity of CD, difficulty in achieving GFD compliance, the negative consequences of poorly controlled CD, and the lack of multidisciplinary care currently being utilized, clinicians are in need of clear recommendations on how to integrate multidisciplinary care into the management of CD patients. Furthermore, research is needed to demonstrate the expected benefits in support of this model of care.
The current state of pediatric celiac disease care in Canada
Currently in North America, patients with CD are most commonly diagnosed based on positive screening atTG testing, followed by endoscopy with characteristic histopathology for confirmation based on Marsh scoring.3,6 Few North American centers are utilizing serologic diagnosis as outlined by the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) guidelines for the diagnosis of CD.31 The standard of care in North America at this time for patients who screen positive for CD using serology for atTG antibodies is referral to a gastroenterologist for assessment and biopsy confirmation prior to initiation of a GFD. The difference in diagnostic standards globally adds additional complexity for contemporary pediatric management of CD. In addition, the general increased uptake of GFD in the community for non-celiac dietary gluten concerns has further complicated the diagnosis, and created a component of confusion and misinformation for patients and physicians.
In 2010, Silvester and Rashid conducted a survey that included Canadian adult and pediatric gastroenterologists and found that the “involvement of multiple health care professionals in the follow-up of patients with CD is being realized to a much greater extent in pediatric than in adult clinics.”32 At that time, they had reported that 29% of Canadian pediatric CD clinics always involved a nurse, and 81% always involved a dietitian; however, pediatric clinics were much less likely to involve a family physician.32
In July 2016, we informally contacted nine Canadian tertiary care pediatric gastroenterology centers to assess the current landscape for CD care in Canada. Of the six centers that responded, four had established pediatric CD clinics in Canada (2007–2016). In contrast, five of the six centers reported having dedicated pediatric IBD clinics that were established earlier (2000–2012). This is consistent with more developed and earlier defined models of pediatric IBD care in the literature.20 Of the four centers with a dedicated CD clinic, three had registered dietitian (RD) participation during each patient visit, and the fourth was run by an RD. One of the centers without a dedicated CD clinic noted that due to resource limitations, RDs did not routinely follow-up with CD patients after initial diagnosis and counseling, unless specifically asked to do so.
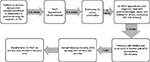
The Stollery Children’s Hospital Celiac Clinic in Edmonton, Alberta was established in 2007 to address the increasing incidence of CD and demand for specialized CD care, as well as the complexity of diagnosis with the intent to improve long-term dietary adherence.33 The Stollery Celiac Clinic structure (Figure 1) includes initial referral from a primary care provider (most commonly a general practitioner or general pediatrician in the community) or specialist of a patient who has screened positive for CD through atTG serology, or a patient with a strong family history of CD with consistent symptoms and immunoglobulin A deficiency. Patients are initially assessed by a pediatric gastroenterologist, a gastroenterology registered nurse (RN), and an expert pediatric RD. After the initial clinical assessment and diagnosis (via biopsy confirmation or a serologic approach), patients receive expert teaching by the RD on the GFD. Further follow-up appointments are facilitated by the gastroenterology RN and RD; the pediatric gastroenterologist does not typically continue to see patients in follow-up unless specific concerns arise as a means of optimizing limited resource allocation. After diagnosis, patients are followed every 6–12 months until their atTG serology normalize. Once the atTG serology has normalized, patients are followed up on an as-needed basis until 17 years of age, at which point they are transitioned to the care of their general practitioner or, in more complex cases, referred to an adult gastroenterologist. The RD is available for all pediatric patients until transition as they require.
Highlighting the importance of the dietitian in celiac multidisciplinary care
Multidisciplinary care that includes an RD is critically important in the care of patients with CD.29,34,35 However, the key role for involvement of an RD in the ongoing care of CD patients may be underappreciated. With the recent increase in the popularity of the GFD among those not diagnosed with CD, there has been an accompanying rise in the available information regarding gluten-free food, especially on the internet. Hence, it can be argued that much of this external dietary information is unverified and of questionable accuracy. The role of the RD in providing reliable and accurate information about the GFD is important, especially for children and their families, to ensure that nutritional needs of the child with CD is met during critical growth periods and to ensure long-term disease remission.
Several studies have examined different types of patient-based education for the GFD and their effectiveness in CD; however, limited knowledge is available regarding the effect of RD counseling on overall patient knowledge around CD and the GFD.36 Addressing this research gap may help increase access to an RD as a crucial member of multidisciplinary teams caring for CD patients. For this reason, a local study (unpublished) was completed between January and June 2016 to determine whether dietetic counseling by the RD improved knowledge about CD and the GFD in primary caregivers (parents or legal guardians) of children newly diagnosed with CD. A 22-item survey was administered to primary caregivers of children with newly diagnosed CD. Surveys were completed both before and after a dietetic counseling session by one of the two expert clinic RDs at the Stollery Children’s Hospital Celiac Clinic.
In our CD MDC, after children are diagnosed with CD, they meet with an RD for a 60- to 90-minute dietary counseling session. At the same time, written information is provided to each family. Topics covered in the education session include: explanation of CD and the appropriate treatment (GFD), how CD is diagnosed, tips for reducing cross-contamination at home, grocery shopping advice, label reading, eating out at restaurants, gluten-free and gluten-containing foods within each food group, ways to add fiber to the GFD, how to help children cope socially and emotionally with a GFD, and how to claim gluten-free foods on taxes specific to the Canadian system. Contact information for the RD was provided to each family after teaching in the event they had further questions after the clinic visit.
The “before” survey was administered to a caregiver at the clinic via an electronic tablet prior to meeting with an RD. The “after” survey was sent 1 week later by email to be completed online by the same caregiver. Both surveys were administered using REDcap software. Survey questions were adapted from Tomlin et al,37 and included questions regarding: caregiver demographic information, their general knowledge of CD and treatment, and information regarding the gluten content of 13 common foods identified as foods most often asked about during teaching sessions (Figure 2). Caregivers were also asked which sources of information about a GFD they utilized prior to dietetic counseling, and were asked to rate the usefulness of those sources of information. Twenty-six caregivers (80% mothers, aged 30–39 years) completed the before and after survey between January and June 2016.
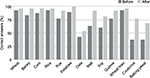  | Figure 2 Caregiver ability to correctly identify gluten-free versus gluten-containing foods before and after dietitian counseling. |
Overall caregiver knowledge improved with expert dietetics counseling. The ability of caregivers to correctly identify gluten-free or gluten-containing food was significantly improved (71.7% before versus 88.2% after, P=0.004). At baseline, 80% of the caregivers were aware that GFD was the treatment for CD, increasing to 100% post-RD counseling (P=0.03). Prior to RD counseling, only 57% of caregivers recognized the treatment requirement was an absolute GFD, and 37% reported not knowing if some gluten intake was acceptable. After counseling, 92% correctly identified that the requirement was for an absolute GFD (P=0.05).
Prior to RD counseling, caregivers were knowledgeable about gluten exposure from common grains and starches, such as wheat, corn, potatoes, and rice. Following RD counseling, there was a consistent trend toward increasing ability to correctly identify the gluten content of all foods tested, including less commonly used foods such as malt, soy, baking yeast, and couscous (Figure 2). Knowledge about the gluten content of oats continued to cause confusion for caregivers even after a dietetic counseling session, with only 54% correctly identifying it as a gluten-free food (Figure 2).
Caregivers reported utilizing 11 different sources of information about the GFD prior to dietetic counseling (Figure 3). The internet was the most utilized resource (86% of caregivers), secondary to another person with CD as a source of information (68%), followed by cookbooks (45%) and newspapers/magazines (45%) being equally accessed. Prior to meeting an RD, the internet was rated as the best resource for information about the GFD.
  | Figure 3 Caregiver sources of information about the gluten-free diet predietitian counseling. |
Given that CD diagnosis involves multiple steps from initial screening, referral for GI assessment, and potential intestinal biopsy, it is not surprising that caregivers seek sources of information about CD and the GFD prior to being seen by a specialist. As anticipated, the most common source of information on the GFD prior to attending our clinic was the internet, followed by other patients with CD (Figure 3). The internet is easy to access and readily available; RD counseling, either provided in conjunction with a pediatric gastroenterology clinic or separately through a community RD referral, can take upward of months to arrange. It is critical for CD patients to be stringent in the avoidance of gluten, as cross-contamination in even small amounts can cause clinical symptoms, villous atrophy, and malabsorption.1,2 As the quality of information available on the internet or received from other CD patients can vary, these sources should not be relied upon for teaching on the GFD. A study by Hoffman et al in 2015 showed that families found the RD to be more informative when it came to nutrient counseling compared to other sources, including the internet, with only the GI specialist being considered potentially more superior.38 Furthermore, Hoffman et al found that post-dietetic counseling, caregivers had significant concerns about the credibility of the nutritional information related to GFD found on the internet.38
Though this brief questionnaire had limitations, including the small sample size, it still provides useful information that supports the benefit of expert dietary counseling. In particular, the additional knowledge gained from counseling would be expected to improve adherence to a GFD and reduce the risk of accidental cross-contamination. Additional benefits in meeting with an RD included the opportunity for families to ask questions, practice label reading, and discuss any concerns with a nutrition expert. They were also able to receive personal advice relevant to their sociocultural and economic circumstances, and the opportunity to form a strong therapeutic relationship between the family and the RD to assist in facilitating ongoing education and adherence to a GFD.
The survey results align with another study done in 2013 by Rajani et al, wherein caregivers stated that post-dietetic counseling, the RD was their preferred educational resource for the GFD.35 Caregivers were also satisfied with the clinical care from an RD, and felt that they would benefit from additional appointments with an RD.35 In addition, patients with CD on a GFD have significantly higher glycemic index, higher glycemic load, and lower folate intake than children on a regular diet.34 This places children with CD more at risk for unintended weight gain and folate deficiency if expert dietary counseling and follow-up are not a part of their health care plan. These results further highlight the essential contribution of an RD in providing care to CD patients. Providing patients and families with multidisciplinary care that includes an expert RD ensures accurate information is provided to families, and can bolster caregiver knowledge, confidence, and satisfaction with maintaining a lifelong and nutritionally balanced GFD in the setting of a multifaceted chronic disease.
Proposed model for a pediatric multidisciplinary celiac clinic
A proposed model for a pediatric CD MDC would follow a similar structure to the current model utilized at the Stollery Children’s Hospital Celiac Clinic (Figure 1). The model would include initial referral from a primary care provider, who would remain a member of the team providing ongoing care once the diagnosis had been made and the atTG serology normalized. Overall, this would help to address common gaps in understanding the diagnosis and management of CD among nonspecialists. Other key team members would include a pediatric gastroenterologist (or physician with expert knowledge in CD and access to gastroenterology services), RN, and an expert RD. After diagnosis, patients should be followed up every 6 months with atTG serology testing until their atTG serology normalizes.3 Once the atTG serology has normalized, patients can be followed up in the pediatric CD MDC on an as-needed basis until adulthood.
Based on NASPGHAN guidelines, atTG titers should be followed on an annual basis once normalized to ensure continued normal atTG levels as a surrogate marker for GFD compliance and disease control.3 If no additional clinical concerns arise, atTG serology can be completed by the patient’s primary care provider on an annual basis for the remainder of the pediatric age range, and throughout adulthood. Following the atTG in the CD MDC until normalization and then switching to an as-needed basis reduces the pressures on resource limitations, such as clinic appointments and staff, but allows an avenue for patients who may need reassessment and revisionary dietary counseling again in a dedicated CD care setting. We stress that engaging and educating primary caregivers as team members is both a potential solution to the resource problem that this large patient population engenders, and will have long-term benefit for all patients with CD into adulthood. Including the family doctor as part of the health care team and for transition care has been shown to be beneficial for pediatric IBD patients.20 Regardless, there will be additional time points where there is a need for reassessment in the CD MDC, about which the primary care provider needs to be aware. A common reason would be for patients who show an increasing atTG titer despite reportedly being on a GFD, raising concern about compliance and need for additional dietary counseling.3,4 Rising atTG, or the return of symptoms despite reported compliance with the GFD, should prompt consideration of repeat gastrointestinal endoscopy.4
Adherence to the GFD can be negatively impacted by the increased cost of gluten-free foods compared to a regular diet.34 In the Canadian system, CD patients and their families are eligible for tax credits for the differential cost of the GFD. Therefore, providing families with adequate information and support about the process of applying for tax credits is not only financially beneficial for patients, but also has the capacity to improve adherence to the GFD.24 Members of a dedicated CD MDC, in particular the RD and RN, would be in an optimal position to consistently and accurately provide information about the tax credit process for families contributing to improved care for CD patients. In addition, previous studies showing improved cost-effectiveness for the health care systems could also be true for a CD MDC; however, further studies would be needed to objectively assess this claim in the CD MDC setting.13,19
While literature on patient preference for MDC care of CD patients is limited, a satisfaction survey did confirm that patients and families value MDC follow-up for children with CD in our clinic.35 In particular, two things were most highly valued: contact with an RD and repeat blood work results.35 These data and the experience of the Stollery Children’s Hospital Celiac Clinic can be utilized for CD care delivery in other regions.
Conclusion
The time is right to focus on multidisciplinary care in CD. Multidisciplinary health care teams dedicated to the care of pediatric CD patients are a beneficial way of addressing this common, multifaceted, increasingly diagnostically complex, and lifelong autoimmune condition with a proven nutritional treatment – the GFD.3,12,26,35 Utilizing the combined efforts of the pediatric gastroenterologist, RD, and RN in a CD MDC model will be of benefit for patients and families in optimizing diagnosis, provision of GFD teaching, and long-term adherence to a GFD.
Acknowledgments
We would like to acknowledge Dr Collin Barker, Dr Herbert Brill, Dr Dominica Gidrewicz, Dr Bob Issenman, Dr David Mack, and Dr Mohsin Rashid for their valuable input on pediatric CD and IBD care across Canadian pediatric gastroenterology centers. We would also like to acknowledge the work of Emily Dao and Stephanie Thornton, Dietetic Interns at the University of Alberta.
Disclosure
The authors report no conflicts of interest in this work.
References
Lebwohl B, Rubio-Tapia A, Assiri A, Newland C, Guandalini S. Diagnosis of celiac disease. Gastrointest Endosc Clin N Am. 2012;22(4):661–677. | ||
Rubio-Tapia A, Ludvigsson JF, Brantner TL, Murray JA, Everhart JE. The prevalence of celiac disease in the United States. Am J Gastroenterol. 2012;107(10):1538–1544. | ||
Hill ID, Dirks MH, Liptak GS, et al. Guideline for the diagnosis and treatment of celiac disease in children: recommendations of the North American Society for Pediatric Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr. 2005;40(1):1–19. | ||
Haines ML, Anderson RP, Gibson PR. Systematic review: the evidence base for long-term management of coeliac disease. Aliment Pharmacol Ther. 2008;28(9):1042–1066. | ||
Rostom A, Murray JA, Kagnoff MF. American gastroenterological association (AGA) institute technical review on the diagnosis and management of celiac disease. Gastroenterology. 2006;131(6):1981–2002. | ||
Rubio-Tapia A, Rahim MW, See JA, Lahr BD, Wu TT, Murray JA. Mucosal recovery and mortality in adults with celiac disease after treatment with a gluten-free diet. Am J Gastroenterol. 2010;105(6):1412–1420. | ||
O’Leary C, Wieneke P, Healy M, Cronin C, O’Regan P, Shanahan F. Celiac disease and the transition from childhood to adulthood: a 28-year follow-up. Am J Gastroenterol. 2004;99(12):2437–2441. | ||
Kumar PJ, Walker-Smith J, Milla P, Harris G, Colyer J, Halliday R. The teenage coeliac: follow up study of 102 patients. Arch Dis Child. 1988;63(8):916–920. | ||
Dewar DH, Donnelly SC, McLaughlin SD, Johnson MW, Ellis HJ, Ciclitira PJ. Celiac disease: management of persistent symptoms in patients on a gluten-free diet. World J Gastroenterol. 2012;18(12):1348–1356. | ||
Charalampopoulos D, Panayiotou J, Chouliaras G, Zellos a, Kyritsi E, Roma E. Determinants of adherence to gluten-free diet in Greek children with coeliac disease: a cross-sectional study. Eur J Clin Nutr. 2013;67(6):615–619. | ||
Fabiani E, Taccari LM, Rätsch IM, Di Giuseppe S, Coppa G V, Catassi C. Compliance with gluten-free diet in adolescents with screening-detected celiac disease: a 5-year follow-up study. J Pediatr. 2000;136(6):841–843. | ||
Komenda P, Levin A. Analysis of cardiovascular disease and kidney outcomes in multidisciplinary chronic kidney disease clinics: complex disease requires complex care models. Curr Opin Nephrol Hypertens. 2006;15(1):61–66. | ||
Levin A, Lewis M, Mortiboy P, Faber S, Hare I, Porter EC, Mendelssohn DC. Multidisciplinary predialysis programs: quantification and limitations of their impact on patient outcomes in two Canadian settings. Am J Kidney Dis. 1997;29(4):533–540. | ||
Ducharme A, Doyon O, White M, Rouleau JL, Brophy JM. Impact of care at a multidisciplinary congestive heart failure clinic: a randomized trial. CMAJ. 2005;173(1):40–45. | ||
Menon S, Valentini RP, Kapur G, Layfield S, Mattoo TK. Effectiveness of a multidisciplinary clinic in managing children with chronic kidney disease. Clin J Am Soc Nephrol. 2009;4(7):1170–1175. | ||
Haynes RB, Gibson E, Hackett B, Taylor DW, Hackett BC, Roberts RS, Johnson AL. Improvement of medication compliance in uncontrolled hypertension. Lancet. 1976;307(7972):1265–1268. | ||
Goldstein M, Yassa T, Dacouris N, McFarlane P. Multidisciplinary predialysis care and morbidity and mortality of patients on dialysis. Am J Kidney Dis. 2004;44(4):706–714. | ||
Brei TJ. The future of the multidisciplinary clinic. ScientificWorldJournal. 2007;7:1752–1756. | ||
Poenaru D, Roblin N, Bird M, et al. The pediatric bowel management clinic: Initial results of a multidisciplinary approach to functional constipation in children. J Pediatr Surg. 1997;32(6):843–848. | ||
DeFilippis EM, Sockolow R, Barfield E. Health care maintenance for the pediatric patient with inflammatory bowel disease. Pediatrics. 2016;138(3):pii: e20151971. | ||
James S, Foley C. A quantitative and qualitative review of a multidisciplinary clinic for adults with coeliac disease. Proc Nutr Soc. 2011;70:E255. | ||
Bebb JR, Lawson a, Knight T, Long RG. Long-term follow-up of coeliac disease--what do coeliac patients want? Aliment Pharmacol Ther. 2006;23(6):827–831. | ||
Zawahir S, Safta A, Fasano A. Pediatric celiac disease. Curr Opin Pediatr. 2009;21(5):655–660. | ||
White LE, Bannerman E, Gillett PM. Coeliac disease and the gluten-free diet: a review of the burdens; factors associated with adherence and impact on health-related quality of life, with specific focus on adolescence. J Hum Nutr Diet. 2016;29(5):593–606. | ||
Rajani S, Alzaben A, Rn LS, Persad R, Huynh HQ, Mager DR, Turner JM. Exploring anthropometric and laboratory differences in children of varying ethnicities with celiac disease. Can J Gastroenterol Hepatol. 2014;28(7):351–354. | ||
Suchy FJ, Brannon PM, Carpenter TO, et al. National institutes of health consensus development conference statement on celiac disease, June 28-30, 2004. Gastroenterology. 2005;128(4 Suppl 1):S1–S9. | ||
Ljungman G, Myrdal U. Compliance in teenagers with coeliac disease – a Swedish follow-up study. Acta Paediatr. 1993;82(3):235–238. | ||
Hallert C, Grant C, Grehn S, et al. Evidence of poor vitamin status in coeliac patients on a gluten-free diet for 10 years. Aliment Pharmacol Ther. 2002;16(7):1333–1339. | ||
Pietzak MM. Follow-up of patients with celiac disease: achieving compliance with treatment. Gastroenterology. 2005;128(4 Suppl 1):S135–S141. | ||
Herman ML, Rubio-Tapia A, Lahr BD, Larson JJ, Van Dyke CT, Murray JA. Patients with celiac disease are not followed up adequately. Clin Gastroenterol Hepatol. 2012;10(8):893–899. | ||
Husby S, Koletzko S, Korponay-Szabó IR, et al. European society for pediatric gastroenterology, hepatology, and nutrition guidelines for the diagnosis of coeliac disease. J Pediatr Gastroenterol Nutr. 2012;54(1):136–160. | ||
Silvester JA, Rashid M. Long-term management of patients with celiac disease: current practices of gastroenterologists in Canada. Can J Gastroenterol. 2010;24(8):499–509. | ||
Rajani S, Huynh HQ, Turner J. The changing frequency of celiac disease diagnosed at the stollery children’s hospital. Can J Gastroenterol. 2010;24(2):109–112. | ||
Alzaben AS, Turner J, Shirton L, Samuel TM, Persad R, Mager D. Assessing nutritional quality and adherence to the gluten-free diet in children and adolescents with celiac disease. Can J Diet Pract Res. 2015;76(2):56–63. | ||
Rajani S, Sawyer-Bennett J, Shirton L, et al. Patient and parent satisfaction with a dietitian- and nurse-led celiac disease clinic for children at the Stollery Children’s Hospital, Edmonton, Alberta. Can J Gastroenterol. 2013;27(8):463–466. | ||
Jacobsson LR, Friedrichsen M, Göransson A, Hallert C. Impact of an active patient education program on gastrointestinal symptoms in women with celiac disease following a gluten-free diet: a randomized controlled trial. Gastroenterol Nurs. 2012;35(3):200–206. | ||
Tomlin J, Slater H, Muganthan T, Beattie RM, Afzal NA. Parental knowledge of coeliac disease. Inform Health Soc Care. 2015;40(3):240–253. | ||
Hoffmann MR, Alzaben AS, Enns SE, Marcon MA, Turner J, Mager DR. Parental health beliefs, socio-demographics, and healthcare recommendations influence micronutrient supplementation in youth with celiac disease. Can J Diet Pract. 2016;77(1):47–53. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.