Back to Journals » International Journal of Women's Health » Volume 11
Management of acute abdomen in pregnancy: current perspectives
Authors Zachariah SK , Fenn M, Jacob K, Arthungal SA , Zachariah SA
Received 8 July 2018
Accepted for publication 25 November 2018
Published 8 February 2019 Volume 2019:11 Pages 119—134
DOI https://doi.org/10.2147/IJWH.S151501
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Elie Al-Chaer
Sanoop Koshy Zachariah,1 Miriam Fenn,2 Kirthana Jacob,2 Sherin Alias Arthungal,1 Sudeeptha Anna Zachariah3
1Department of General, Gastrointestinal & Laparoscopic Surgery, MOSC Medical College, Kolenchery Cochin, Cochin 682311, India; 2Department of Obstetrics and Gynecology, MOSC Medical College, Kolenchery Cochin, Cochin 682311, India; 3Department of Obstetrics and Gynecology, Sultan Qaboos Hospital, Salalah PO 601, Oman
Abstract: Acute abdomen in pregnancy represents a unique diagnostic and therapeutic challenge. Acute abdominal pain in pregnancy can occur due to obstetric factors as well for reasons that are unrelated to pregnancy. The diagnostic approach of acute abdomen during pregnancy can be tricky owing to the altered clinical presentations brought about by the anatomical and physiological changes of gestation along with the reluctance to use certain radiological investigations for fear of harming the fetus. Delay in diagnosis and treatment can lead to adverse outcomes for both the mother and fetus. In this article, we attempt to review and discuss the various etiologies, the current concepts of diagnosis, and treatment, with a view to developing a strategy for timely diagnosis and management of pregnant women presenting with acute abdominal pain.
Keywords: ectopic pregnancy, rupture uterus, abdominal pain, appendicitis, cholecystitis, acute abdomen, pregnancy
Introduction
Acute abdomen in pregnancy (AAP) represents a unique diagnostic and therapeutic challenge. The term acute abdomen refers to any serious acute intra-abdominal condition accompanied by pain, tenderness, and muscular rigidity, for which emergency surgery should be contemplated.1 It is often indicative of a clinical course of abdominal symptoms that can range from minutes to hours to weeks and is commonly used synonymously for a condition that requires immediate surgical intervention.2 The wide range of causes and varied spectrum of clinical presentations pose a formidable diagnostic and therapeutic challenge.
Acute abdominal pain in pregnancy can be due to obstetric as well as non-obstetric etiologies. The physiological changes of pregnancy increase the risk of developing an acute abdomen. As for non-obstetric causes, any gastrointestinal (GI) disorder can occur during pregnancy. About 0.5%–2% of all pregnant women require surgery for non-obstetric acute abdomen.3,4
The diagnostic approach of AAP can be tricky owing to the anatomical as well as the dynamic physiological changes brought about by gestation and the reluctance to use radiological diagnostic modalities such as X-ray or computed tomography (CT) scan and a low threshold to subject the patient to an emergency surgical procedure. Physical examination of the abdomen itself can be difficult in the pregnant state. Consequently, this has a bearing on clinical presentations, interpretation of physical findings, as well as a shift in the normal range of laboratory parameters. For example, even in the absence of any infection, pregnancy alone can usually produce white blood cell counts ranging from 6,000 to 30,000/μL, thus mimicking an acute infection.5
The need for a systematic approach is necessary for an accurate and timely diagnosis of potentially life-threatening conditions, which otherwise could be precarious for both the mother and fetus. We, therefore, attempt to review and discuss the various etiologies, the current concepts of diagnosis, and treatment, with a view to developing a strategy for timely diagnosis and management of pregnant women presenting with acute abdominal pain.
Anatomical and physiological changes in pregnancy
Anatomical considerations
The uterus, usually a pelvic organ, enlarges to become an intra-abdominal organ around 12 weeks of gestation. During pregnancy, the uterus can increase from a mere 70 to 1,110 g with a resultant intrauterine volume of at least 5 L.6 During the early phase of gestation, the growth is due to hyperplasia and hypertrophy of the muscle fibers, with subsequent transformation of the uterus into a thick-walled muscular organ. By the 20th week, the uterus can be felt at the umbilicus and the intrinsic growth almost ceases.
Further increase in uterine size occurs due to expansion by distension and mechanical stretching of the muscle fibers by the growing fetus. At 36 weeks, the uterus reaches the costal margin. The uterine blood vessels also undergo significant hypertrophy to adapt to the increasing demands.
The adjacent intra-abdominal viscera tend to get displaced from their normal position to accommodate the enlarging uterus (Figure 1). The stomach, omentum, and intestines are displaced upward and laterally, and the colon can get narrowed due to mechanical compression.7
As the displaced omentum might fail to wall off peritonitis and the relaxed and stretched abdominal wall can mask guarding, the underlying peritoneal inflammation may be missed. The enlarged uterus can compress the ureters, causing hydroureter and hydronephrosis, thereby mimicking urolithiasis.
These alterations of anatomical and topographical landmarks can make the diagnosis difficult in case of acute abdominal emergencies. Detailed knowledge of anatomical variations can help in arriving at an early diagnosis. Prompt early diagnosis and timely surgical intervention have shown to have a significantly better perinatal outcome.
Physiological considerations
Physiological changes are brought about by an orchestrated interplay of hormones, especially progesterone, leading to a generalized change in milieu by involving almost every organ system. These include endocrine, metabolic, cardiovascular, GI, renal, musculoskeletal, respiratory, and behavioral changes. GI changes such as delayed gastric emptying, increased intestinal transit time, gastroesophageal reflux, abdominal bloating, nausea, and vomiting can occur in 50%–80% of pregnant females.8–10 Constipation occurring in the last trimester is attributed to the mechanical compression of the colon along with increase in water and sodium absorption due to increased aldosterone levels. Lawson et al observed that there was a significant increase in the mean small bowel transit time during each trimester (first trimester, 125±48 minutes; second trimester, 137±58 minutes; third trimester, 75±33 minutes).11
The physiological leukocytosis of pregnancy can mimic an acute intra-abdominal inflammatory process. The white blood cell counts usually revert to the nonpregnant levels by the sixth postpartum day. The physiological increase in plasma volume in proportion to the red cell volume produces physiological anemia. Also, the relatively decreased hemoglobin concentration along (physiological anemia) with a physiological increase in the heart rate can make the assessment challenging in case of hemorrhage. The cardiovascular and respiratory changes in pregnancy are summarized in Table 1.
Hepatic transaminases and serum bilirubin levels are decreased, while serum ALP level is increased due to placental contribution. Also, the pharmacokinetics of drugs can be altered due to changes in GI transit time.
Etiology of AAP
This can be categorized into the following:
- Obstetric (pregnancy-related causes)
- Non-obstetric (non-pregnancy-related causes)
- Extra-abdominal causes
- Causes exacerbated by pregnancy.
The various causes are listed in Table 2.
Non-obstetric causes of AAP
Acute appendicitis
In 1848, Hancock reported the first case of acute appendicitis complicating pregnancy.12 The currently reported incidence of acute appendicitis in pregnant women is between 0.04% and 0.2%.13 Acute appendicitis remains the most common non-obstetric surgical emergency during pregnancy (being suspected in 1/800 pregnancies and confirmed in ∼1 in 1,000–2,000 pregnancies). This is followed by cholecystitis, pancreatitis, and bowel obstruction.14,15 Also, it is the most common cause for non-obstetrical surgical intervention performed during pregnancy, accounting for 25% of the non-obstetric surgical interventions done during pregnancy.16
Acute appendicitis can present in any of the three trimesters, with the highest incidence found in the second trimester. However, appendicular perforation has been reported to be more frequent in the third trimester. Clinical presentation may be ambiguous. Classically, acute appendicitis presents as an initial periumbilical pain, which later shifts and localizes to the right lower quadrant with maximal tenderness at the McBurney’s point. Peritoneal irritation due to the inflamed appendix may produce guarding and even rebound tenderness (Rovsing’s sign). These classical clinical signs may be altered, especially in advanced pregnancy. Guarding or rebound tenderness may not be as pronounced as in the nonpregnant state. Pain in the right lower quadrant is the most common symptom. Fever may be present in some patients.
A meticulous physical examination is the key. It has been traditionally accepted that the appendix tends to get displaced from its normal position by the enlarging uterus. However, this view has been challenged by a few researchers.17 More recently, Hodjati and Kazerooni in their comparative study on 291 women showed that there was no significant change in the position of the appendix during pregnancy.18 The Alder’s sign can be useful in differentiating uterine pain from appendicular pain in pregnancy.19
Presence of leukocytosis may be physiological and may not necessarily indicate appendicitis, but a shift to left and presence of band forms may point toward an underlying inflammatory pathology.
Imaging is the obvious next step. The role of imaging is to reduce delay in surgical intervention and negative appendectomy rates.20 Ultrasonography (USG) has a reported sensitivity of 67%–100% and specificity of 83%–96% for appendicitis in pregnancy.21 CT has a reported sensitivity of 86% and specificity of 97% in such patients. However, there may be a reluctance to perform CT due to the inherent risk of radiation exposure.
In pregnant women with suspected appendicitis, magnetic resonance imaging (MRI) has a high sensitivity and specificity coupled with the benefit of avoiding radiation exposure. This is probably the reason that MRI has been recommended by The American College of Radiology as the second line of imaging in case of inconclusive USG for suspected appendicitis in pregnancy.22 MRI is most useful in identifying a normal appendix, thereby ruling out inflammation. Unfortunately, it is less reliable in detecting the presence of extraluminal air in perforated viscus.
The definitive treatment for acute appendicitis is surgery. The decision to operate or not is crucial. The decision to operate depends on the clinical condition of the patient and investigatory findings. Delay in diagnosis is associated with increased risk of perforation, peritonitis, and septicemia, leading to adverse maternal and fetal outcomes such as miscarriage, preterm labor, and intrauterine death. The fetal loss rate has been reported to be in the range of 3%–5% in cases of unruptured appendix without significant effect on maternal mortality. However, in perforated appendicitis, the fetal loss rate increases to 20%–25% and maternal mortality rate escalates to around 4%.23
A few studies have shown antibiotic therapy to be successful in nonpregnant adults. Vons et al, in their randomized control trial, showed that antibiotics as the first-line therapy were not inferior to surgery in acute appendicitis.24 Furthermore, specific meta-analyses and Cochrane reviews have demonstrated primary antibiotic treatment to be safe in uncomplicated appendicitis.25,26 Young et al, in their small case series, showed successful management of perforated appendix with antibiotic therapy.27 As for now, there is insufficient evidence to recommend conservative management for acute appendicitis in pregnancy. However, for uncomplicated appendicitis, just like in nonpregnant patients, a trial of antibiotic therapy can be tried, provided proper monitoring is performed while maintaining a low threshold for surgery. Hence, it would be sensible to consider surgical intervention if the diagnosis of acute appendicitis is for sure, keeping in mind that the fetal mortality is close to 36% if the appendix perforates. Moreover, perforation can lead to sepsis and increase the risk of preterm delivery.28 Considering the significant risk of fetal loss due to delay in diagnosis, it is justifiable to consider early surgical intervention. Even though the negative appendectomy rate is around 35%, it is still justifiable.29
A concomitant cesarean section is rarely indicated at the time of appendectomy unless the gestation is above 37 weeks and already a caesarian is anticipated. As a dictum, only the appendicitis is managed while the pregnancy is left undisturbed.30 Traditionally, open appendectomy was the surgical procedure of choice for pregnant women.31 Currently, laparoscopy is being proposed as a diagnostic tool for suspected appendicitis and therapeutic method for confirmed cases of appendicitis in pregnancy.
Gallbladder disease
Gallstone disease is the second most common indication for surgical intervention during pregnancy. Acute cholecystitis may affect up to 20% of women by the age of 40 years.32
Elevated serum cholesterol and lipid levels in pregnancy coupled with decreased gallbladder motility and delayed emptying can predispose the formation of gallstones. Another mechanism suggested is that while estrogen increases cholesterol secretion, progesterone reduces soluble bile acid secretion, and this favors insoluble bile acid accumulation predisposing to stone formation.33
Symptomatic gallstones may present with typical features of biliary colic with postprandial abdominal discomfort, bloating, nausea, and mild to severe colicky pain in the right upper quadrant or epigastrium.34
Acute cholecystitis is usually related to the presence of gallstones. Previously, asymptomatic gallstones were reported to be found in up to 3.5% of pregnant women, and 90% of the cases were responsible for cholecystitis presenting during pregnancy. Since there are no obvious anatomical changes in position, the clinical features are similar to nonpregnant adults and diagnosis is usually straightforward. Murphy’s sign is typically positive. Even though there is a predisposition to formation of biliary sludge and stones, cholecystitis does not occur more frequently during pregnancy, affecting only 1% of the pregnant women.35 Serious complications include cholangitis, sepsis, jaundice, gallstone pancreatitis, perforation, and abscess formation.
USG is the investigation of choice with a sensitivity of >95%. Currently, high-resolution USG can detect gallstones as small as 2 mm.36
Pregnant patients with cholecystitis need to be admitted. In the absence of serious complications, conservative treatment is an initial option with bowel rest, intravenous hydration, analgesics (opioids or nonsteroidal anti-inflammatory drugs), and broad-spectrum antibiotics.
Extensive data about the optimum management of gallbladder disease in pregnant women are not available. Nonsteroidal anti-inflammatory drugs should be used with caution after 32 weeks due to the risk of developing oligohydramnios and narrowing of ductus arteriosus. Preferred antibiotics include cephalosporins and clindamycin. Traditionally, definitive surgery is usually deferred in uncomplicated cases. However, some researchers are of the opinion that a conservative approach is associated with higher relapse rates in the range of 40%–70%.37
A decision analysis study performed by Jelin at al showed that there was a higher risk of fetal death (7%) among those patients who underwent nonoperative management than in those who underwent laparoscopic cholecystectomy (2.2%).38 They were of the opinion that in pregnant women with biliary tract disease, laparoscopic cholecystectomy was superior to nonoperative management during the first and second trimesters.
In case of complications due to common bile duct stones, such as cholangitis or pancreatitis, endoscopic retrograde cholangiopancreatography with sphincterotomy and stone extraction to relieve pancreatitis can be safely performed with minimal risk of ionizing radiation exposure to the fetus. Elective cholecystectomy can then be performed postpartum.39
Acute pancreatitis
Acute pancreatitis complicating pregnancy is rare with a reported rate of 1/10,000 pregnancies and most frequently occurs in the third trimester.40 Most often, it is a self-limiting disease, but can progress to a more severe condition associated with multi-organ failure, shock, and death. The maternal mortality rate is <1%, and the rate of preterm delivery is about 20%.
The most frequent cause of pancreatitis in pregnancy is cholelithiasis and congenital or acquired hypertriglyceridemia (hyperlipidemic pancreatitis: blood triglyceride level >26.54 mmol/L). Even though hypertriglyceridemia can occur in any trimester, pancreatitis commonly occurs in the third trimester. As such, gallstones are responsible for 70% of cases of pancreatitis in pregnancy. Very rarely, pancreatitis can be associated with preeclampsia–eclampsia or hemolysis, elevated liver enzymes, and low platelet count (HELLP) syndrome.
The classical presentation is similar to that in nonpregnant adults. The presence of sudden-onset nausea, vomiting, and mild to severe upper boring abdominal pain radiating to the back and sometimes relieved on leaning forward should raise the suspicion of acute pancreatitis. However, many times, the symptoms may only be upper abdominal pain. The diagnosis involves demonstration of elevated serum pancreatic amylase and lipase levels. USG is useful, but may not always be accurate in visualizing the pancreas as it may be obscured by the presence of bowel gas, but its role is in identifying the presence of gallstones. CT scan is rarely required.
Management is usually conservative with adequate bowel rest, nasogastric aspiration, proper hydration, electrolyte correction, and analgesics. Meperidine (Pethidine) is the analgesic of choice, and short-term administration is relatively safe in pregnancy.41 In majority of the patients, clinical improvement occurs in about 5 days. The role of antibiotics, radiological aspiration, parenteral nutrition, and surgical intervention should be considered in case of complications such as abscess, hemorrhage, necrosis, or sepsis.
Intestinal obstruction
Small bowel obstruction in pregnancy is extremely rare to the extent that an individual general surgeon is likely to observe only one to two cases during his or her career. It is a potentially severe non-obstetric surgical condition which can be associated with adverse maternal and fetal outcomes.
Webster et al recently reviewed all the literature published between 1992 and 2014.42 According to them, the overall rate of fetal loss was 17% and the maternal mortality rate was 2%. Adhesions remain the single most common cause for intestinal obstruction. Adhesions usually occur due to previous abdominal surgeries including previous cesarean section.
Usually, there are three time periods associated with the increased frequency of developing an intestinal obstruction in pregnancy, that is, 16th–20th week, the 36th week, and immediate puerperium.
Adhesive obstruction occurs more commonly in advanced pregnancy. Reported rates are 6%, 28%, 45%, and 21% during the first, second, third trimesters, and puerperium, respectively. The other causes include volvulus (25%), intussusceptions (5%), hernia (3%), carcinoma (1%), and idiopathic “ileus” (8%). Volvulus occurs more commonly during pregnancy (23%–25%) than in the nonpregnant state (3%–5%).43
The symptoms include nausea, vomiting (82%), abdominal pain (98%), and absolute constipation (30%). Abdominal distension may be difficult to assess, especially late in pregnancy. Bowel sounds may be hyperperistaltic to start with and progressively become hypoperistaltic, which is an ominous sign denoting the onset of strangulation.
Initially, a trial of conservative approach (ie, nonoperative management) with bowel rest, intravenous hydration, and nasogastric aspiration with close monitoring. Urgent surgical intervention is mandatory in case of failure of conservative therapy as denoted by signs of impending bowel strangulation or symptoms of fetal distress.
Plain abdominal radiographs have been reported to be positive in 82%–100% of pregnant women with intestinal obstruction and, therefore, may provide necessary information when there is a high clinical suspicion.44–46 CT and MRI are also being increasingly used to diagnose small bowel obstruction in pregnancy.
Laparotomy is best done through a midline incision to allow for a detailed exploration of the coelomic cavity, with minimal handling of the uterus. As for now, laparoscopy cannot be recommended.
Other GI conditions
Peptic ulcer
Peptic ulcer disease and its complications are usually not common in pregnancy, ranging from 1 to 6 in every 23,000 pregnancies. Possible mechanisms involve reduced gastric secretion along with increased placental histaminase (diamine oxidase) secretion. In the majority of cases, remission occurs with diet modification, histamine receptor antagonists, or proton pump inhibitor.
Life-threatening complications of peptic ulcer are perforation and bleeding. The diagnosis of perforation is predominantly clinical, with signs of guarding, rigidity, and tachycardia. Plain erect radiographs are valuable in detecting free intraperitoneal air. However, in 10%–20% of the patients, pneumoperitoneum may not be seen on X-ray. In such situations, CT may be more sensitive to detect free air. A timely diagnosis of perforation within the first 6 hours carries excellent prognosis, while a delay of 12 hours or more is linked with poorer outcomes. Perforation requires laparotomy.
Papa Essilfie et al47 and Amdeslasie et al48 have shown that in the case of perforation, timely diagnosis and prompt surgical intervention can prevent maternal and fetal mortality. In cases of massive peptic ulcer hemorrhage, endoscopic control should be attempted initially, and if this fails, laparotomy should be performed without delay.
Urolithiasis
Symptomatic urolithiasis is not common during pregnancy. Theoretically, the physiological changes leading to an increase in glomerular filtration proportional to an increase in blood volume coupled with urinary stasis can predispose to formation of urinary calculi.49 Symptoms include severe colicky flank pain, nausea, and vomiting in the absence of peritoneal signs. The gravid uterus may obscure adequate visualization of the ureters on USG. In such instances, a single-shot intravenous pyelogram can be performed. Spontaneous passage of obstructing calculi occurs in 85% of cases. In the case of ongoing obstruction, the best possible options should be considered in close consultation with the urology team.
Intra-abdominal hemorrhage
Hemorrhage into the peritoneal cavity is a life-threatening condition, but it is fortunately rare. It can occur during pregnancy and also postpartum. The more common causes include rupture of splenic artery aneurysm and rupture of the dilated high-pressure veins of the ovary and broad ligaments at the time of labor. It can present with sudden-onset severe localized or diffuse abdominal pain and rapid progression to circulatory shock. USG and guided aspiration of frank blood might clinch the diagnosis.
Splenic artery aneurysm rupture usually occurs in the third trimester.50 According to Trimble and Hill’s hypothesis, two factors, namely, weakness in the arterial wall and an increase in blood pressure, are contributory for development of aneurysms.51 These factors are known to be augmented in pregnancy.
The mortality following rupture is 25% in nonpregnant adults, and it drastically increases to 75% in pregnant women and is associated with a fetal mortality of 95%.52
Management is most often surgical. Urgent laparotomy, evacuation of hematoma, ligation of offending veins, and splenectomy with ligation of the splenic artery are the options. At times, a concomitant cesarean section may be needed to identify the source of bleed. As for now, embolization techniques have a success rate of <85%.53
Gastroesophageal reflux disease
The incidence of gastroesophageal reflux disease (GERD) in pregnancy is around 80%. GERD can cause upper abdominal pain, which may at times be severe enough to seek urgent medical attention. The high incidence is due to hypotonic lower esophageal sphincter, delayed GI transit, and also due to the mechanical compression by the gravid uterus.54 The symptoms of GERD are exacerbated by recumbency. Hemorrhage from esophagitis is a serious complication.
Video capsule endoscopy can be used for recurrent GI bleed after nondiagnostic esophageal gastric disorders.55 Diet modification and postural changes are beneficial for GERD. Antacids are safe, but those containing sodium bicarbonate cause fluid overload and alkalosis. Sucralfate is safe, but the aluminum content can trigger renal insufficiency for both mother and fetus. H2 receptor blockers are safe in pregnancy; ranitidine and famotidine can be used. Cimetidine has antiandrogenic effects.56 Proton pump inhibitors were initially reserved for refractory GERD, but have been used extensively in pregnancy. Lansoprazole, rabeprazole, and pantoprazole are category B drugs. Omeprazole is a category C drug.
Obstetric causes of AAP
Pregnancy-related causes for abdominal pain could be physiological or pathological. The physiological causes for abdominal pain in pregnancy may be round ligament pain caused by stretching of the round ligament, pain perceived during fetal movement, and painful Braxton Hick contractions. Round ligament pain can complicate 10%–30% of pregnancies and usually occurs during the end of the first trimester and in the second trimester. It is localized to the lower abdominal quadrants radiating to the groin; it is a cramp-like pain which is worse on movement and is more common in multiparous women. The concern with round ligament pain is that there is a trend to overdiagnose this benign condition, hence leading to failure or delay in diagnosis of significant pathology. Pathological causes which are life-threatening include ruptured ectopic pregnancy, abruption, HELLP syndrome, acute fatty liver of pregnancy, and uterine rupture.
Ectopic pregnancy
Implantation of pregnancy in any site other than the endometrial lining of the uterus is termed ectopic pregnancy. The incidence of ectopic pregnancy is 1%–2%, but ruptured ectopics are responsible for 6% of maternal deaths.57 Nearly 95% implant in the fallopian tube, whereas the remaining 5% may implant in the ovary, abdominal cavity, cesarean scar, or cervix. In heterotopic pregnancy, a twin pregnancy occurs, where one fetus implants in the normal site and the other in the fallopian tube. The incidence of heterotopic pregnancy is on the rise after assisted reproductive technology procedures. The etiology of ectopic pregnancies may be tubal surgery, previous ectopic pregnancy, history of pelvic inflammatory disease, and infertility treatment.58
The outcome of tubal pregnancies may be a tubal rupture, tubal abortion, or spontaneous resolution. Also, 60%–80% of women with ectopic pregnancies may present with some vaginal bleeding. The diagnostic modalities used to identify ectopic pregnancies are transvaginal scanning, beta-human chorionic gonadotropin hormone values, and occasionally, a dilation and curettage.
Women with ectopic pregnancies who are hemodynamically stable can be managed medically with methotrexate injections or surgically. Cesarean scar ectopic pregnancy incidence is on the rise and presents both a diagnostic and therapeutic dilemma. They can be managed medically with methotrexate, reserving surgical procedure for persistent cases.59 Patients with ruptured ectopic pregnancies presenting in hypovolemic shock have to be managed with urgent salpingectomy, drainage of hemoperitoneum, and replacement of blood and products.
Abruption
Premature separation of a normally situated placenta is abruption. This condition may present with abdominal pain, with or without vaginal bleeding. If all the blood remains behind the placenta with no revealed bleeding per vaginum, this is concealed hemorrhage and can cause a delay in diagnosis of the condition.60 Incidence of abruption is 0.6%.61
Abruptio placenta can result in perinatal as well as maternal mortality. Predisposing factors to placental abruption are hypertension or preeclampsia, preterm, premature membrane rupture, cigarette smoking, cocaine abuse, and anti-phospholipid antibodies. Abruption placenta can be clinically suspected when antepartum hemorrhage is accompanied by pain. On clinical examination, the patient will be found to have a uterus which may be larger than the period of gestation and is tense and tender. The fetal heart sounds on auscultation may show evidence of severe fetal distress or even absent fetal heart sounds. In the vast majority of patients with this condition, expeditious delivery by the quickest possible route along with an artificial rupture of membranes to reduce thromboplastin release into circulation is performed.
HELLP syndrome
HELLP syndrome refers to the condition characterized by hemolysis, elevated liver enzymes, and low platelets. Even with the passage of three decades, HELLP syndrome remains a challenge to the scientific community. The Mississippi or the Tennessee criteria have been used to categorize HELLP according to severity.62 In patients with HELLP syndrome, the incidence of subcapsular liver hematoma and rupture is increased.63 These patients can present as acute abdomen with pain localized to right upper quadrant. Other complications of HELLP syndrome include eclampsia (6%), placental abruption (10%), acute kidney injury (5%), and pulmonary edema (10%). Most women with HELLP syndrome need termination of pregnancy. Hepatic hematomas can be diagnosed by MRI. Unless there is active hemorrhage, a conservative approach is indicated. Ongoing hemorrhage requires prompt surgical intervention.64
Uterine rupture
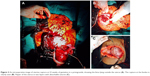
Uterine rupture can occur in a previously intact uterus when it is called primary rupture, or more frequently occurs in a previously scarred uterus when it is called secondary rupture. The scars may be due to surgery such as cesarean section or myomectomy; injury to the uterus or can occur in an anomalous uterus. Injury to the uterus may be due to prior curettage, or perforation, endometrial ablation, or hysteroscopy. The frequency of primary rupture is rare, with the incidence being 1 in 10,000–15,000 births. The relative reduction in primary rupture may be because grand multiparity is becoming less frequent and also due to more judicious use of oxytocin and prostaglandins. With the increase in cesarean section rates, secondary rupture is on the rise. Uterine rupture can be a cause for significant perinatal mortality and morbidity.65 Uterine rupture causes abdominal pain only after there is significant hemoperitoneum. The diaphragmatic irritation may cause the pain to be referred to the chest or the shoulder. If uterine rupture occurs during the trial of labor after C-section, the early signs of rupture may be fetal heart recording showing variable decelerations followed by bradycardia. Frank uterine rupture with placental expulsion will cause fetal death, hemoperitoneum, loss of uterine contour, vaginal bleeding, fetal parts being felt superficially, and occasionally, hematuria. An urgent USG in the labor room will confirm our findings, and treatment includes immediate laparotomy along with fluid and blood resuscitation and repair of the uterine rent (Figure 2) or sometimes hysterectomy. Other pregnancy-related conditions which may be painful but not life-threatening include red degeneration of fibroid, ovarian torsion, and rarely, an abdominal pregnancy.66–68
Uterine and ovarian torsion
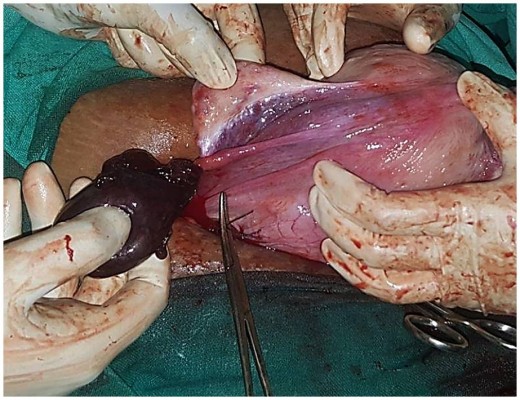
Uterine torsion is a condition where >40% axial rotation is observed. The etiology for uterine torsion may be the presence of fibroids and performance of external cephalic version to correct a malpresentation.69 Symptoms of uterine torsion may be pain, shock, and urinary or bowel complaints. Uterine torsion can cause maternal vasovagal shock and fetal asphyxia. The management options available for uterine torsion may include conservative measures, such as bed rest, analgesia, altering the maternal position, or surgical measures – laparotomy detorsion of the uterus and hoping to continue the pregnancy if the fetus is preterm or performance of a C-section after detorsion in a viable fetus. In case of ovarian/adnexal torsion, detorsion can be performed with no added risk of fetal or maternal complications.70 If the cyst is gangrenous, excision should be carried out (Figure 3).
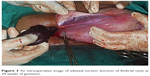  | Figure 3 An intraoperative image of adnexal torsion (torsion of fimbrial cyst) at 34 weeks of gestation. |
Abdominal pregnancy
Advanced abdominal pregnancies are rare events, with the incidence being 10 in 10,000 births. Abdominal pregnancy has a perinatal mortality of 50% and a maternal mortality of 5 in 1,000 cases with catastrophic effects to mother and fetus.71 Persistent abdominal pain is a feature in 80% of the cases and painful fetal movements. Clinical examination may reveal abdominal tenderness, easily palpable fetal parts, and absence of contraction after oxytocin or prostaglandin induction. The USG reveals fetal head and body parts localized outside the uterus and an ectopic placenta, along with failure to demonstrate a uterine wall between the fetus and the urinary bladder. MRI has high sensitivity in diagnosis.
Management options include planning the timing and nature of the intervention. If the fetus is dead, surgery is indicated after waiting for a few weeks, to allow atrophy of placental vessels. If the fetus is alive and the period of gestation is <24 weeks, a conservative approach after careful counseling can be recommended.71,72
Imaging in pregnancy
The use of radiological procedures during pregnancy is often viewed with undue fear. The ideal imaging modality in pregnancy has always been a subject of discussion. Since ionizing radiation is a carcinogen,73 the fear of exposing the growing fetus to radiation has always been the main cause for concern. Ratnapalan et al showed in their study that obstetricians and physicians had an unrealistically high perception of the risk of fetal teratogenicity with respect to CT scan and plain radiographs ordered during pregnancy.74 Up to 6% of them recommended that abortion should be carried out if the mother had been exposed to CT scan during early pregnancy.
Ultrasonography
USG is universally accepted as the first radiological modality for the evaluation of acute abdomen in pregnant women. It is the imaging technique of choice in pregnancy due to its availability, portability, and lack of ionizing radiation. It has high sensitivity and specificity for diagnosing the common non-obstetric causes of acute abdomen during pregnancy, namely, appendicitis and cholecystitis. However, the efficacy of USG can decline after the 32nd week of gestation due to the technical difficulties resulting from the mechanical effects of the enlarging uterus. Hence, adequate evaluation of the pancreas, bowel, and ureters may be impaired due to the narrow field of view owing to the presence of overlying structures.75
Ionizing radiation: X-ray, CT scan, contrast agents, and radionucleotide isotope scans
Ionizing radiation is known to have harmful effects on the living tissue. The developing fetus is more sensitively vulnerable to the injurious effects of ionizing radiation. The risks include genetic mutations, smaller head circumference, neurologic abnormalities, mental retardation, childhood cancer (leukemia), organ malformations, fetal death, and also, an increased lifetime risk of developing cancer.
A clear understanding of the safe limits of radiation dose is, therefore, essential. It has to be understood that two factors, namely, 1) the cumulative radiation dosage and 2) the fetal age at exposure are the significant risk factors for causing fetal harm.82
Fetal mortality is most significant when the exposure occurs within the first 2 weeks of conception. The most vulnerable period for central nervous system teratogenesis is between 10 and 17 weeks of gestation during organogenesis.83 In late pregnancy, the concern shifts from teratogenicity to the risk of childhood malignancy.
The National Council of Radiation Protection and Measurements (1977),84 The American Council of Obstetricians and Gynecologists (2004),85 and the American College of Radiology86 (2008) have unanimously opined that the risk of ionizing radiation-induced fetal harm is considered negligible at 50 mGy or less and the risk of malformations increases significantly only at doses above 150 mGy. Thus, fetal radiation of <1 mGy is not significant, and counseling is not required.87 Plain radiographs deliver only a negligible dose of ionizing radiation to the fetus, especially when the fetus is not in the field of view. The role of shielding the gravid uterus is controversial; it may not help reduce the dose, but can undoubtedly provide reassurance to the mother. Even in the presence of shielding, the fetus can be exposed to some background radiation.
CT scan is recommended as the first-line imaging modality in pregnant women who have sustained major trauma and in suspected cases of pulmonary embolism.78 As CT delivers a collimated dose on the target organs, here too, the radiation dose is negligible if the fetus is not in view. The usual fetal radiation dose for a routine CT of the abdomen and pelvis is around 25 mGy, which can be reduced to about 13 mGy with the use of automated exposure control facility in modern CT scanners.
Oral and intravenous iodinated contrast agents are US Food and Drug Administration category B and, to date, there is no reported fetal teratogenicity risk from their use. They can be given to pregnant women if valuable information cannot be acquired without contrast agent.
Radioisotope imaging can be safely used in pregnancy for ventilation–perfusion lung scan to identify pulmonary embolism. The fetal exposure is usually <5 mGy. Technetium 99 m is the isotope of choice during pregnancy.
Exposure depends on the number of films. The safe upper limit of exposure to ionizing radiation for the fetus is 5 rads (50 mGy) during the first trimester and up to 15 rads (150 mGy) in the second and third trimesters. When the fetal dose exceeds 150 mGy (eg, diagnostic imaging for major trauma), therapeutic abortion should be considered. Hence, if there exists a need for the use of CT or X-ray to evaluate a potentially life-threatening condition in the mother, the benefits of imaging definitely outweigh the risks.
Magnetic resonance imaging
MRI is preferable to CT during pregnancy as it provides excellent soft tissue imaging without the risk of ionizing radiation. MRI at 1.5 T or less has been shown to be safe in all trimesters of pregnancy. Pregnant women should, therefore, be imaged at 1.5 T or less.88 The safety of MRI at 3 T for pregnant women has not yet been proven. Rapid sequence MRI is preferable to conventional MRI. The possibility of adverse effects to the fetus due to the acoustic noise has not been established.
The American Academy of Pediatrics recommends an upper limit of 90 dB to be observed, beyond which permanent damage can occur to the fetal ear.89
Intravenous gadolinium, the contrast medium used for MRI, is a US Food and Drug Administration class C agent which can cross the placenta and circulate indefinitely in fetal circulation. Studies in animals have shown that these agents have the potential of inducing congenital anomalies. Although there is no such significant evidence in humans, intravenous gadolinium is preferably avoided during pregnancy. A comparison of the commonly used imaging modalities during pregnancy is shown in Table 3.
Role of laparoscopy in pregnancy
Traditionally, open surgery used to be the preferred procedure for surgical intervention during pregnancy. The laparoscopic approach in pregnancy was contraindicated due to lack of sufficient evidence regarding its safety. The concerns included fear of trocar injury to the gravid uterus, anticipated technical difficulty due to lack of adequate space owing to the enlarged uterus, fear of adverse effects of carbon dioxide insufflation, such as fetal acidosis, and decreased maternal venous return due to raised intra-abdominal pressure.90,91
However, over the years, various studies have shown an increasing trend toward acceptance of laparoscopy during pregnancy as a feasible, safe, and effective therapeutic option.92 A report by Gurbuz and Peetz was one among initial reports demonstrating the safety of laparoscopic technique for acute non-obstetric abdominal pain during pregnancy, without additional risk to the fetus.93
Although earlier it was suggested that laparoscopic surgeries should be done preferably during the second trimester, recent evidence suggests laparoscopic surgery can be done during any trimester, with very low rates of maternal and fetal morbidity.94,95
For gaining access to the abdominal cavity, the open access is considered to be safer than the closed technique to avoid inadvertent injury to the uterus and displaced viscera.96
The preferred insufflation pressure is 8–12 mmHg as it reduces the possibility of uterine hypoperfusion and maternal pulmonary events. Therefore, laparoscopic appendectomy and cholecystectomy can be recommended during pregnancy if the need for surgery arises. The updated guidelines for the role of laparoscopy in pregnant women have been published by The Society of American Gastrointestinal and Endoscopic Surgeons.97
Abdominal trauma in pregnancy
Trauma is the leading non-obstetric cause of death in pregnancy.98 Common causes include road traffic accidents, falls, and partner violence.99,100 Fetal loss is around 70% even for minor trauma. Sudden acceleration–deceleration forces can lead to abruption of the placenta. The primary survey involves attending to airway, breathing, and circulation, and the secondary survey should focus on non-obstetric/obstetric injuries and fetal condition. Four to 24 hours tocodynamometry monitoring is recommended for minor trauma. In case of major trauma, the Kleihauer–Betke test should be performed to identify feto-maternal hemorrhage by determining the amount of fetal hemoglobin that has passed into the maternal circulation.101 Cardiopulmonary resuscitation should be performed by left-lateral displacement of the body to about 25° with manual uterine displacement. After cardiac arrest, perimortem cesarean delivery can improve neonatal and maternal outcomes.
Approach to the patient
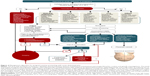
AAP can pose serious diagnostic and therapeutic challenges. Our proposal of a systematic approach to acute abdominal pain in pregnancy is outlined as an algorithm in Figure 4, and important key points are summarized in Table 4.
  | Table 4 Key practice points |
Conclusion
AAP can be due to obstetric as well as non-obstetric causes. Life-threatening pathologies may not manifest with classical presentations. Nonionizing examinations are preferred as the first line of radiological investigation. Investigations using ionizing radiations, such as X-ray and CT scan, are generally safe and should not be withheld if there is a definite clinical indication and there is no other alternative, especially in life-threatening conditions. Laparoscopic surgery is feasible and safe in selected patients. A clear understanding of the anatomical and physiological changes in pregnancy, systematic clinical evaluation, adequate knowledge of the safe limits of radiological investigations, and a multidisciplinary approach are indispensable for timely diagnosis and treatment of pregnant women presenting with acute abdomen.
Disclosure
The authors report no conflicts of interest in this work.
References
Stedman’s Medical Dictionary, 2. (2018). Stedman’s Medical Dictionary, 27th Edition Deluxe. [online] Alibris. Available from: https://www.alibris.com/Stedmans-Medical-Dictionary-27th-Edition-Deluxe-Stedmans/book/30000738. Accessed July 7, 2018. | ||
Martin RF, Rossi RL. The acute abdomen. Surg Clin North Am. 1997;77(6):1227–1243. | ||
Malangoni MA. Gastrointestinal surgery and pregnancy. Gastroenterol Clin North Am. 2003;32(1):181–200. | ||
Augustin G, Majerovic M. Non-obstetrical acute abdomen during pregnancy. Eur J Obstet Gynecol Reprod Biol. 2007;131(1):4–12. | ||
Pritchard JA, Baldwin RM, Dickey JC. Blood volume changes in pregnancy and the puerperium, II: red blood cell loss and changes in apparent blood volume during and following vaginal delivery, cesarean section, and cesarean section plus total hysterectomy. Am J Obstet Gynecol. 1962;84:1271. | ||
Campbell-Brown M, Hytten F. Nutrition. In: Chamberlain G, Broughton Pipkin F, editors. Clinical physiology in obstetrics. Oxford: Blackwell Science; 1998:165–191. | ||
Tan EK, Tan EL. Alterations in physiology and anatomy during pregnancy. Best Pract Res Clin Obstet Gynaecol. 2013;27(6):791–802. | ||
Soma-Pillay P, Nelson-Piercy C, Tolppanen H, Mebazaa A. Physiological changes in pregnancy. Cardiovasc J Afr. 2016;27(2):89–94. | ||
Conklin KA. Maternal physiology adaptations during gestation, labour, and the puerperium. Semin Anesth. 1991;X(4):221–234. | ||
Broussard CN, Richter JE. Nausea and vomiting of pregnancy. Gastroenterol Clin North Am. 1998;27(1):123–151. | ||
Lawson M, Kern F, Everson GT. Gastrointestinal transit time in human pregnancy: prolongation in the second and third trimesters followed by postpartum normalization. Gastroenterology. 1985;89(5):996–999. | ||
Hancock H. Disease of the appendix cæci cured by operation. Boston Med Surg J. 1848;39(17):331–334. | ||
Choi JJ, Mustafa R, Lynn ET, Divino CM. Appendectomy during pregnancy: follow-up of progeny. J Am Coll Surg. 2011;213(5):627–632. | ||
Andersson RE, Lambe M. Incidence of appendicitis during pregnancy. Int J Epidemiol. 2001;30(6):1281–1285. | ||
Angelini DJ. Obstetric triage revisited: update on non-obstetric surgical conditions in pregnancy. J Midwifery Womens Health. 2003;48(2):111–118. | ||
Mourad J, Elliott JP, Erickson L, Lisboa L. Appendicitis in pregnancy: new information that contradicts long-held clinical beliefs. Am J Obstet Gynecol. 2000;182(5):1027–1029. | ||
Baer JL, Reis RA, Arens RA. Appendicitis in pregnancy: with changes in position and axis of the normal appendix in pregnancy. J Am Med Assoc. 1932;98(16):1359–1364. | ||
Hodjati H, Kazerooni T. Location of the appendix in the gravid patient: a re-evaluation of the established concept. Int J Gynaecol Obstet. 2003;81(3):245–247. | ||
Alders N. A sign for differentiating uterine from extrauterine complications of pregnancy and puerperium. Br Med J. 1951;2(4741):1194–1195. | ||
Wallace CA, Petrov MS, Soybel DI, Ferzoco SJ, Ashley SW, Tavakkolizadeh A. Influence of imaging on the negative appendectomy rate in pregnancy. J Gastrointest Surg. 2008;12(1):46–50. | ||
Williams R, Shaw J. Ultrasound scanning in the diagnosis of acute appendicitis in pregnancy. Emerg Med J. 2007;24(5):359–360. | ||
Rosen MP, Ding A, Blake MA, et al. ACR Appropriateness Criteria® right lower quadrant pain – suspected appendicitis. J Am Coll Radiol. 2011;8(11):749–755. | ||
Doberneck RC. Appendectomy during pregnancy. Am Surg. 1985;51(5):265–268. | ||
Vons C, Barry C, Maitre S, et al. Amoxicillin plus clavulanic acid versus appendicectomy for treatment of acute uncomplicated appendicitis: an open-label, non-inferiority, randomised controlled trial. Lancet. 2011;377(9777):1573–1579. | ||
Wilms IMHA, de Hoog DENM, de Visser DC, Janzing HMJ; Cochrane Colorectal Cancer Group. Appendectomy versus antibiotic treatment for acute appendicitis. Cochrane Database Syst Rev. 2011;82(2):CD008359. | ||
Ansaloni L, Catena F, Coccolini F, et al. Surgery versus conservative antibiotic treatment in acute appendicitis: a systematic review and meta-analysis of randomized controlled trials. Dig Surg. 2011;28(3):210–221. | ||
Young BC, Hamar BD, Levine D, Roqué H. Medical management of ruptured appendicitis in pregnancy. Obstet Gynecol. 2009;114(2 Pt 2):453–456. | ||
Walsh CA, Tang T, Walsh SR. Laparoscopic versus open appendicectomy in pregnancy: a systematic review. Int J Surg. 2008;6(4):339–344. | ||
Mcgory ML, Zingmond DS, Tillou A, Hiatt JR, Ko CY, Cryer HM. Negative appendectomy in pregnant women is associated with a substantial risk of fetal loss. J Am Coll Surg. 2007;205(4):534–540. | ||
Weston P, Moroz P. Appendicitis in pregnancy: how to manage and whether to deliver. Obstet Gynaecol. 2015;17(2):105–110. | ||
Mohammed JA, Oxorn H. Appendicitis in pregnancy. Can Med Assoc J. 1975;112(10):1187–1188. | ||
Ramin KD, Ramsey PS. Disease of the gallbladder and pancreas in pregnancy. Obstet Gynecol Clin North Am. 2001;28(3):571–580. | ||
Affleck DG, Handrahan DL, Egger MJ, Price RR. The laparoscopic management of appendicitis and cholelithiasis during pregnancy. Am J Surg. 1999;178(6):523–528. | ||
Landers D, Carmona R, Crombleholme W, Lim R. Acute cholecystitis in pregnancy. Obstet Gynecol. 1987;69(1):131. | ||
Dietrich CS, Hill CC, Hueman M. Surgical diseases presenting in pregnancy. Surg Clin North Am. 2008;88(2):403–419. | ||
Borzellino G, Massimiliano Motton AP, Minniti F, Montemezzi S, Tomezzoli A, Genna M. Sonographic diagnosis of acute cholecystitis in patients with symptomatic gallstones. J Clin Ultrasound. 2016;44(3):152–158. | ||
Swisher SG, Schmit PJ, Hunt KK, et al. Biliary disease during pregnancy. Am J Surg. 1994;168(6):576–581. | ||
Jelin EB, Smink DS, Vernon AH, Brooks DC. Management of biliary tract disease during pregnancy: a decision analysis. Surg Endosc. 2008;22(1):54–60. | ||
Tham TC, Vandervoort J, Wong RC, et al. Safety of ERCP during pregnancy. Am J Gastroenterol. 2003;98(2):308–311. | ||
Pitchumoni CS, Yegneswaran B. Acute pancreatitis in pregnancy. World J Gastroenterol. 2009;15:5641–5646. | ||
Briggs GG, Freeman RK, Yaffe SJ. Meperidine. Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk. Philadelphia: Lippincott Williams & Wilkins; 2005:999–1000. | ||
Webster PJ, Bailey MA, Wilson J, Burke DA. Small bowel obstruction in pregnancy is a complex surgical problem with a high risk of fetal loss. Ann R Coll Surg Engl. 2015;97(5):339–344. | ||
Augustin G, Majerovic M. Non-obstetrical acute abdomen during pregnancy. Eur J Obstet Gynecol Reprod Biol. 2007;131(1):4–12. | ||
American College of Obstetricians and Gynecologists. Guidelines for diagnostic imaging during pregnancy. ACOG Committee Opinion 158. Washington, DC: ACOG; 1995. | ||
Zachariah SK, Fenn MG. Acute intestinal obstruction complicating pregnancy: diagnosis and surgical management. BMJ Case Rep. 2014;2014:bcr2013203235. | ||
Chiedozi LC, Ajabor LN, Iweze FI. Small intestinal obstruction in pregnancy and puerperium. Saudi J Gastroenterol. 1999;5(3):134–139. | ||
Essilfie P, Hussain M, Bolaji I. Perforated duodenal ulcer in pregnancy – a rare cause of acute abdominal pain in pregnancy: a case report and literature review. Case Rep Obstet Gynecol. 2011;2011:263016. | ||
Amdeslasie F, Berhe Y, Gebremariam TT. Perforated duodenal ulcer in the third trimester of pregnancy. Saudi J Med Med Sci. 2015;3:164–166. | ||
Drago JR, Rohner TJ, Chez RA. Management of urinary calculi in pregnancy. Urology. 1982;20(6):578–581. | ||
Sadat U, Dar O, Walsh S, Varty K. Splenic artery aneurysms in pregnancy – a systematic review. Int J Surg. 2008;6(3):261–265. | ||
Trimble WK, Hill JH. Congestive splenomegaly (Banti’s disease) due to portal stenosis without hepatic cirrhosis; aneurysms of the splenic artery. Arch Pathol Lab Med. 1942;34:423. | ||
Caillouette JC, Merchant EB. Ruptured splenic artery aneurysm in pregnancy. Twelfth reported case with maternal and fetal survival. Am J Obstet Gynecol. 1993;168(6 Pt 1):18101811–18111813. | ||
Nincheri Kunz M, Pantalone D, Borri A, et al. Management of true splenic artery aneurysms. Two case reports and review of the literature. Minerva Chir. 2003;58(2):247–256. | ||
Castro LP, de Paula Castro L. Reflux esophagitis as the cause of heartburn in pregnancy. Am J Obstet Gynecol. 1967;98(1):1–10. | ||
Figueiredo P, Almeida N, Lopes S, et al. Small-bowel capsule endoscopy in patients with suspected Crohn’s disease – diagnostic value and complications. Diagn Ther Endosc. 2010;2010(9):1–7. | ||
Matok I, Gorodischer R, Koren G. The safety of H2-blockers use during pregnancy. J Clin Pharmacol. 2013;50(1):81–87. | ||
Berg CJ, Callaghan WM, Syverson C, Henderson Z. Pregnancy-related mortality in the United States, 1998 to 2005. Obstet Gynecol. 2010;116(6):1302–1309. | ||
Clayton HB, Schieve LA, Peterson HB, Jamieson DJ, Reynolds MA, Wright VC. Ectopic pregnancy risk with assisted reproductive technology procedures. Obstet Gynecol. 2006;107(3):595–604. | ||
Jabeen K, Karuppaswamy J. Non-surgical management of cesarean scar ectopic pregnancy – a five-year experience. J Obstet Gynaecol. 2018;8:1–7. | ||
Chang YL, Chang SD, Cheng PJ. Perinatal outcome in patients with placental abruption with and without antepartum hemorrhage. Int J Gynaecol Obstet. 2001;75(2):193–194. | ||
Salihu HM, Bekan B, Aliyu MH, Rouse DJ, Kirby RS, Alexander GR. Perinatal mortality associated with abruptio placenta in singletons and multiples. Am J Obstet Gynecol. 2005;193(1):198–203. | ||
Dusse LM, Alpoim PN, Silva JT, Rios DR, Brandão AH, Cabral AC. Revisiting HELLP syndrome. Clin Chim Acta. 2015;451(Pt B):117–120. | ||
Haddad B, Barton JR, Livingston JC, Chahine R, Sibai BM. Risk factors for adverse maternal outcomes among women with HELLP (hemolysis, elevated liver enzymes, and low platelet count) syndrome. Am J Obstet Gynecol. 2000;183(2):444–448. | ||
Vigil-de Gracia P, Ortega-Paz L. Pre-eclampsia/eclampsia and hepatic rupture. Int J Gynaecol Obstet. 2012;118(3):186–189. | ||
Porreco RP, Clark SL, Belfort MA, Dildy GA, Meyers JA. The changing specter of uterine rupture. Am J Obstet Gynecol. 2009;200(3):269:269.e1–269.e4. | ||
Gupta S, Manyonda IT. Acute complications of fibroids. Best Pract Res Clin Obstet Gynaecol. 2009;23(5):609–617. | ||
Bassi A, Czuzoj-Shulman N, Abenhaim HA. Effect of pregnancy on the management and outcomes of ovarian torsion: a population-based matched cohort study. J Minim Invasive Gynecol. 2018;25(7):1260–1265. | ||
Cappell MS, Friedel D. Abdominal pain during pregnancy. Gastroenterol Clin North Am. 2003;32(1):1–58. | ||
Ahmed FU, Ambreen A, Zubair S, Kiran N. Torsion of a term uterus. J Coll Physicians Surg Pak. 2016;26(6 Suppl):S50–S51. | ||
Daykan Y, Bogin R, Sharvit M, et al. Adnexal torsion during pregnancy: pregnancy outcomes after surgical intervention – a retrospective case–control study. J Minim Invasive Gynecol. 2019;26(1):117–121. | ||
Dahab AA, Aburass R, Shawkat W, Babgi R, Essa O, Mujallid RH. Full-term extrauterine abdominal pregnancy: a case report. J Med Case Rep. 2011;5(1):531. | ||
Matovelo D, Ng’walida N. Hemoperitoneum in advanced abdominal pregnancy with a live baby: a case report. BMC Res Notes. 2014;7(1):106. | ||
Brenner DJ, Hall EJ. Computed tomography – an increasing source of radiation exposure. N Engl J Med Overseas Ed. 2007;357(22):2277–2284. | ||
Ratnapalan S, Bona N, Chandra K, Koren G. Physicians’ perceptions of teratogenic risk associated with radiography and CT during early pregnancy. AJR Am J Roentgenol. 2004;182(5):1107–1109. | ||
Woodfield CA, Lazarus E, Chen KC, Mayo-Smith WW. Abdominal pain in pregnancy: diagnoses and imaging unique to pregnancy – review. AJR Am J Roentgenol. 2010;194(6 Suppl):WS14–WS30. | ||
Masselli G, Brunelli R, Monti R, et al. Imaging for acute pelvic pain in pregnancy. Insights Imaging. 2014;5(2):165–181. | ||
Mccollough CH, Schueler BA, Atwell TD, et al. Radiation exposure and pregnancy: when should we be concerned? Radiographics. 2007;27(4):909–917. | ||
Sadro C, Bernstein MP, Kanal KM. Imaging of trauma: part 2, abdominal trauma and pregnancy – a radiologist’s guide to doing what is best for the mother and baby. AJR Am J Roentgenol. 2012;199(6):1207–1219. | ||
van Randen A, Bipat S, Zwinderman AH, Ubbink DT, Stoker J, Boermeester MA. Acute appendicitis: meta-analysis of diagnostic performance of CT and graded compression US related to prevalence of disease. Radiology. 2008;249(1):97–106. | ||
Dewhurst C, Beddy P, Pedrosa I. MRI evaluation of acute appendicitis in pregnancy. J Magn Reson Imaging. 2013;37(3):566–575. | ||
Ross JT, Matthay MA, Harris HW. Secondary peritonitis: principles of diagnosis and intervention. BMJ. 2018;361:k1407. | ||
Goldberg-Stein SA, Liu B, Hahn PF, Lee SI. Radiation dose management: part 2, estimating fetal radiation risk from CT during pregnancy. AJR Am J Roentgenol. 2012;198(4):W352–W356. | ||
Groen RS, Bae JY, Lim KJ. Fear of the unknown: ionizing radiation exposure during pregnancy. Am J Obstet Gynecol. 2012;206(6):456–462. | ||
National Council on Radiation Protection and Measurements. Medical radiation exposure of pregnant and potentially pregnant women. NCRP report no 54. Bethesda, MD: National Council on Radiation Protection and Measurements; 1977. | ||
ACOG Committee Opinion. Number 299, September 2004 (replaces No. 158, September 1995). Guidelines for diagnostic imaging during pregnancy. Obstet Gynecol. 2004;104:647–651. | ||
Andreotti RF, Lee SI, Choy G, et al. ACR Appropriateness Criteria on acute pelvic pain in the reproductive age group. J Am Coll Radiol. 2009;6(4):235–241. | ||
Brent R, Mettler F, Wagner L. Pregnancy and medical radiation: ICRP publication 84. International Commission on Radiological Protection Website. Available from: www.icrp.org/publication.asp?id=ICRP%20Publication%2084. Accessed July 17, 2012. | ||
Chen MM, Coakley FV, Kaimal A, Laros RK. Guidelines for computed tomography and magnetic resonance imaging use during pregnancy and lactation. Obstet Gynecol. 2008;112(2 Pt 1):333–340. | ||
Ciet P, Litmanovich DE. MR safety issues particular to women. Magn Reson Imaging Clin N Am. 2015;23(1):59–67. | ||
Kilpatrick CC, Orejuela FJ. Management of the acute abdomen in pregnancy: a review. Curr Opin Obstet Gynecol. 2008;20(6):534–539. | ||
Barber-Millet BL, Castro G, Gavara G, Pla B, Domínguez G. Update on the management of non-obstetric acute abdomen in pregnant patients. Cir Esp. 2016;9494(5):257257–265265. | ||
Palanivelu C, Rangarajan M, Senthilkumaran S, Parthasarathi R. Safety and efficacy of laparoscopic surgery in pregnancy: experience of a single institution. J Laparoendosc Adv Surg Tech A. 2007;17(2):186–190. | ||
Gurbuz AT, Peetz ME. The acute abdomen in the pregnant patient. Is there a role for laparoscopy? Surg Endosc. 1997;11(2):98–102. | ||
Rollins MD, Chan KJ, Price RR. Laparoscopy for appendicitis and cholelithiasis during pregnancy: a new standard of care. Surg Endosc. 2004;18(2):237–241. | ||
Upadhyay A, Stanten S, Kazantsev G, et al. Laparoscopic management of a nonobstetric emergency in the third trimester of pregnancy. Surg Endosc. 2007;21(8):1344–1348. | ||
Chung JC, Cho GS, Shin EJ, Kim HC, Song OP. Clinical outcomes compared between laparoscopic and open appendectomy in pregnant women. Can J Surg. 2013;56(5):341–346. | ||
Pearl JP, Price RR, Tonkin AE, Richardson WS, Stefanidis D. Guidelines for the use of laparoscopy during pregnancy. Available from: https://www.sages.org/publications/guidelines/guidelines-for-diagnosis-treatment-and-use-of-laparoscopy-for-surgical-problems-during-pregnancy/ | ||
Mendez-Figueroa H, Dahlke JD, Vrees RA, Rouse DJ. Trauma in pregnancy: an updated systematic review. Am J Obstet Gynecol. 2013;209(1):1–10. | ||
El Kady D. Perinatal outcomes of traumatic injuries during pregnancy. Clin Obstet Gynecol. 2007;50(3):582–591. | ||
El-Kady D, Gilbert WM, Anderson J, Danielsen B, Towner D, Smith LH. Trauma during pregnancy: an analysis of maternal and fetal outcomes in a large population. Am J Obstet Gynecol. 2004;190(6):1661–1668. | ||
Muench MV, Baschat AA, Reddy UM, et al. Kleihauer–Betke testing is important in all cases of maternal trauma. J Trauma. 2004;57(5):1094–1098. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.