Back to Journals » Journal of Hepatocellular Carcinoma » Volume 9
Macrotrabecular-Massive Hepatocellular Carcinoma: What Should We Know?
Authors Li X, Yao Q, Liu C , Wang J, Zhang H , Li S, Cai P
Received 5 March 2022
Accepted for publication 23 April 2022
Published 5 May 2022 Volume 2022:9 Pages 379—387
DOI https://doi.org/10.2147/JHC.S364742
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Ahmed Kaseb
Xiaoming Li,1,2,* Qiandong Yao,3,* Chen Liu,1,* Jian Wang,1 Huarong Zhang,4 Shiguang Li,2,5 Ping Cai1
1Department of Radiology, Southwest Hospital, Third Military Medical University (Army Military Medical University), Chongqing, People’s Republic of China; 2Department of Radiology, The First People’s Hospital of Zunyi, The Third Affiliated Hospital of Zunyi Medical University, Zunyi, People’s Republic of China; 3Department of Radiology, Sichuan Science City Hospital, Mianyang, People’s Republic of China; 4Institute of Pathology and Southwest Cancer Center, Third Military Medical University (Army Military Medical University), Chongqing, People’s Republic of China; 5The Second People’s Hospital of Guiyang (Jinyang Hospital), Guiyang, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Shiguang Li; Ping Cai, Email [email protected]; [email protected]
Abstract: Hepatocellular carcinoma is one of the most common malignancies globally. Recently, a newly identified histological subtype, designated as “macrotrabecular-massive hepatocellular carcinoma” (MTM-HCC), has been associated with an aggressive phenotype and has received extensive attention. MTM-HCC was a strong independent prognostic predictor of early and overall recurrence because it is closely related to tumor molecular subclass, gene mutation, carcinogenesis pathways, and immunohistochemical markers. In addition, preoperative imaging examination can potentially provide an essential clue for diagnosing MTM-HCC, intratumor necrosis or ischemia is an independent predictor for MTM-HCC on Gd-EOB-DTPA enhanced MRI or CT. Early diagnosis and appropriate treatment of MTM-HCC could prove beneficial for preventing early recurrence and could improve outcomes.
Keywords: hepatocellular carcinoma, macrotrabecular-massive, molecular subclass, gene mutation, magnetic resonance imaging
Introduction
Hepatocellular carcinoma (HCC) is the most common primary malignant tumor of the liver, accounting for approximately 85–90% of all malignant tumors. The most common causes of HCC are hepatitis B, excessive alcohol intake, and metabolic syndrome.1–3 Other rare causes include long-term intake of moldy food, hereditary diseases (hemochromatosis and hepatic glycogen deposition) and the malignant transformation of hepatic adenoma.4,5 Currently, there are multiple treatment options available for HCC, including radiofrequency ablation, surgical resection, and liver transplantation. However, the five-year recurrence rate of this tumor remains as high as 70%.6 Moreover, an increased risk of recurrence remains even if early-stage HCC is resected using radical surgery.7
The poor prognosis of patients with HCC is related to the pathological type and molecular subclass of HCC.8 The common histopathological patterns include macrotrabecular, microtrabecular, pseudoglandular, and compact. Approximately 35% of the pathological subtypes of HCC are unique, including steatohepatitis, clear cell, macrotrabecular-massive, and scirrhous et al.2 The macrotrabecular-massive hepatocellular carcinoma (MTM-HCC) is characterized by macrotrabecular >6 cells thick that account for more than 50% of the tumor, MTM-HCC demonstrates a relatively advanced preoperative Barcelona stage, high alpha-fetoprotein (AFP) load (>400 ng/mL), large tumor volume, peritumoral satellite nodules, macro- or microvascular invasion, and biliary tract invasion.9 These features make MTM-HCC highly prone to early recurrence and poor prognosis, and small-sample data suggest the MTM-HCC subtype is the most common subtype with a potential of metastasis.10 In addition, MTM-HCC has received extensive attention from clinicians and researchers in recent years and was officially included in the new classification of HCC by the World Health Organization (WHO) in 2019.11 The best clinical diagnosis and treatment plan can be provided if MTM-HCC is diagnosed accurately before surgery,12 because liver transplantation13,14 or radiofrequency ablation15 is not recommended for such patients. Surgeons should perform resection with wide margins or anatomical hepatectomy,16 and shorter follow-up intervals may be recommended for surveillance.
Currently, there are few published studies related to MTM-HCC, and only 20 related articles were retrieved from PubMed using the keywords “macrotrabecular-massive” and “hepatocellular carcinoma”. Table 1 summarizes the associated research on the evaluation of MTM-HCC from 2019 to 2021 in PubMed. Herein, we review the latest progress in this field combined with previous research reports.
 |
Table 1 The Researches on the Evaluation of MTM-HCC from 2019 to 2021 |
Molecular and Histological Features Related to MTM-HCC
MTM-HCC is a special heterogeneous solid tumor. Such tumors with a diameter >5 cm are prone to early recurrence after radical resection or radiofrequency ablation.17 The aggressive biological behaviour of MTM-HCC is attributed to its ability to activate angiogenesis18 and is closely related to tumor molecular subclass, gene mutation, carcinogenesis pathways,17 and immunohistochemical markers (Figure 1).
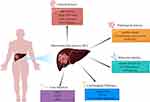 |
Figure 1 Integration of HCC clinical features, pathological features, molecular subclass, carcinogenic pathway, and gene mutation. |
Molecular Subclass of MTM-HCC
In 2004, in a study comprising 91 HCC patients, Lee et al classified HCC into two categories (Cluster A and Cluster B) by analyzing tumor gene expression with statistically different influences on survival time. Cluster A had robust cellular proliferation, anti-apoptosis ability, and poor prognosis. AFP (>300 ng/mL) and Edmonson Grade III accounted for 62.5% and 77%, respectively;19 MTM-HCC belonged to Cluster A. In 2007, Boyault et al further studied the correlations among transcriptome-genotype-phenotype in HCC. Based on 57 cases of HCC and 3 cases of hepatic adenoma, the whole genome was sequenced and verified on another 63 cases of HCC. Based on the clinical and genetic characteristics, six groups of HCC subtypes (G1-G6) were obtained.20 MTM-HCC was classified as G3 and was characterized by TP53 mutation and overexpression of cell cycle regulatory genes. Recently, Zhongshan Hospital and Fudan University determined the protein genome of hepatitis B-related HCC in China and found that HCC had metabolic reprogramming, immune microenvironment changes, and increased cell proliferation. Herein, HCC was divided into three subtypes, namely, S-Mb (Metabolism), S-Me (Microenvironment), and S-Pf (Proliferation). Compared with the first two subtypes, the S-Pf subtype was more likely to present with larger tumors, tumor thrombi, and progressive HCC.21 MTM-HCC may be related to S-Pf.
Gene Mutation Related to MTM-HCC
MTM-HCC has a very strong invasion and metastasis potential, closely related to multiple gene mutations, including TP53 mutation, fibroblast growth factor 19 (FGF19) amplification, and ataxia-telangiectasia mutated protein (ATM).18
TP53 is the most commonly mutated gene in HCC and plays an important role in its pathogenesis. The average incidence of TP53 mutation is 30%22 and maybe as high as 60% in HBV-related HCC.23 TP53 plays a crucial role in cell cycle regulation and apoptosis after DNA damage. Tumor suppression is reportedly lost with TP53 gene mutations that promote tumor progression.24 These mutations primarily occur in poorly differentiated tumors. These tumors frequently invade blood vessels, activate cell proliferation, and promote angiogenesis and epithelial-mesenchymal transition. They are significantly related to the clinical (HBV infection, early recurrence, poor prognosis, and high AFP load) and pathological (satellite lesions and vascular invasion) manifestations of MTM-HCC.
FGF19 amplification is related to the malignant biological behaviour of MTM-HCC. FGF19 is a metabolic regulatory gene belonging to the family of hormone-like FGF signalling molecules.25 FGF19 amplification occurs in approximately 14% of HCC cases.26 Amplified FGF19 reportedly plays an essential role in regulating liver cells function and development, and binds to the specific receptor fibroblast growth factor receptor 4 (FGFR-4) to drive the carcinogenesis in HCC. FGF19 promotes the proliferation and anti-apoptosis ability of tumor cells and increases the risk of HCC.27,28 Moreover, the amplification of FGF19 increases the activity of FGF19 in tumor cells, thus maintaining the gene expression mode of hepatocyte differentiation.29 Calderaro et al found that MTM-HCC was related to FGF19 amplification (P=0.02)18 and was prone to early recurrence and poor prognosis (P=0.05, 0.03).
ATM belongs to the family of signal-sensing molecule phosphoinositide 3-kinase-related protein kinase (PIKK), which is extremely sensitive to radiation.30 ATM kinase plays a central role in mediating DNA damage repair and short cell cycle arrest, and regulating cell survival, proliferation, metabolism, and differentiation to ensure genomic stability and maintain cell activity.31 However, the targets of these kinases can affect the progression of the cell cycle, DNA damage, and epithelial-mesenchymal transition and can participate in tumor progression by mediating the invasion, apoptosis, and migration of tumor cells.32 ATM mutation is reportedly related to the pathological subtypes of MTM-HCC (P=0.03),18 indicating that this kinase plays a vital role in the progression of HCC.
MTM-HCC-Related Carcinogenic Pathways
Gene expression profiling has shown that angiogenesis activation is the principal carcinogenesis pathway of MTM-HCC, with overexpression of angiogenesis-2 (Ang-2) and VEGF,12 which coordinate with each other to promote angiogenesis.33 Ang-2 mainly disrupts the interaction between endothelial and para-endothelial cells, thus increasing the sensitivity to VEGF that induces blood vessel germination and activates angiogenesis.34 In addition, Ang-2 allows circulating tumor cells to extravasate and is considered an essential regulator of the pre-metastasis state.35 It is speculated that Ang-2 transiently expressed by MTM-HCC directly invades the surrounding normal liver parenchyma and forms peritumoral satellite lesions.17 TGF-β, RAS/MAPK, and PI3K/AKT, are also important carcinogenic pathways of MTM-HCC.12 TGF-β is an effective immunosuppressant that has been associated with inflammation, fibrogenesis, and immunomodulation in the HCC microenvironment.36 RAS/MAPK is driven by an acquired gene mutation, which leads to cell proliferation and survival, resulting in the development and progression of HCC.37 The PI3K/AKT signalling pathway is an important pathway of HCC development and progression, as an imbalance of this pathway leads to the slow growth and accelerated apoptosis of normal cells, which can mediate downstream biological effects, such as NF-κB and VEGF.38
Immunohistochemical Markers Related to MTM-HCC
MTM-HCC highly express neoangiogenesis-related genes, which lead to the discovery of Endothelial-Specific Molecule-1 (ESM-1) and Vessels encapsulating tumor clusters (VETC) as robust immunostaining markers, the ESM and VETC pattern has been suggested as a possible route for HCC metastasis.35,39
Endothelial-specific molecule-1 (ESM-1) is expressed by interstitial endothelial cells lining the macrotrabecular. Using separate training and verification groups, Calderaro et al showed that the sensitivity of ESM-1 was 97% and 93%, and its specificity was 92% and 91%, respectively. Thus, ESM-1 can be used as a reliable immunohistochemical marker.39 Vessels encapsulating tumor clusters (VETC) enter the circulatory system through anastomosis with peritumoral veins and then release into the blood, leading to metastasis and recurrence of HCC. The overexpression of Ang-2 can promote VETC-mediated metastasis.40,41 Research has shown that compared with its expression in non-MTM-HCC, VETC is more easily expressed in MTM-HCC (P=0.006) and is prone to recurrence at an early stage, with lower disease free survival and overall survival.42
Imaging Findings of MTM-HCC
The gold standard for MTM-HCC diagnosis is pathology. Although needle biopsies can diagnose MTM-HCC before surgery, they are limited by the risk of puncture site bleeding, implantation metastasis, and sampling error. With technological developments, imaging has gradually become more mainstream for preoperative tumor assessment. Preoperative imaging can provide an essential clue for individualized diagnosis and treatment for such patients. However globally, there are few reports on the imaging manifestations of MTM-HCC, and only seven articles related to the imaging manifestations of MTM-HCC could be retrieved from PubMed, among which only one is from China.
Computed Tomography Findings of MTM-HCC
To date, only Feng et al43 reported the computed tomography (CT) findings of MTM-HCC. These researchers found that compared with non-MTM-HCC, MTM-HCC is more likely to show signs of necrosis and hemorrhage on contrast-enhanced CT scanning. Combining the radiological signs with AFP level (>100 ng/mL) establishes an ANH scoring system (in which A represents AFP, N represents necrosis, and H represents hemorrhage). The areas under the receiver operating characteristic curve in the training and verification groups were 0.77 and 0.73, respectively. The probability of early recurrence rises concomitant to increases in the ANH score.44 Renne et al also found that compared with non-VETC-HCC, VETC-HCC was often larger (>5 cm) and was more prone to necrosis.42 However, that study did not elaborate on whether there is any difference in CT imaging manifestations between MTM-HCC (VETC+) and MTM-HCC (VETC-).
Magnetic Resonance Imaging Findings of MTM-HCC
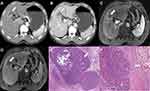
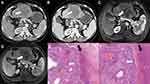
In 2019, Rhee et al45 proposed irregular rim-like arterial phase enhancement (IRE) as an imaging sign based on extracellular contrast-enhanced magnetic resonance imaging (MRI). IRE is more common in pathological subtypes of HCC with a stronger invasion ability, especially MTM-HCC. In 2020, Rhee et al46 collected multicentre data to study the Gd-EOB-DTPA MRI findings of MTM-HCC and recommended the following MRI diagnostic criteria for MTM-HCC: 1) The proportion of hypo-enhanced components inside the tumor in the arterial phase accounts for more than 20% of the tumor, and the sensitivity and negative predictive value in the verification group were 88% and 97% respectively; and 2) the proportion of hypo-enhanced components inside the tumor in the arterial phase accounts for more than 50%, and the tumor contains two or more additional image features (intra-tumoral arteries, abnormal enhancement around the tumor in the arterial phase, and irregular tumor margin), which have relatively low sensitivity and very high specificity (96% in the verification group). The mechanism of tumor necrosis or ischemia is mainly related to the expression of ESM-1 in the tumor microenvironment, which activates the regeneration of blood vessels around the tumor; this causes the tumor to grow fast associated with hypoxia, resulting in the necrosis seen in advanced HCC.39,47 Therefore, to ensure relatively high sensitivity, for the Gd-EOB-DTPA MRI manifestations of MTM-HCC, substantial intratumor ischemia or necrosis has been defined as the proportion of hypo-enhanced areas within the tumor in the arterial phase accounting for ≥20%. Mulé et al48 reported that substantial intra-tumoral ischemia or necrosis is an independent predictor of MTM-HCC (sensitivity 65%, specificity 93%). Chen et al49 also agreed with the above view and held that this sign could help diagnose 86% of MTM-HCC cases (specificity 66%). In our studies,50 we concluded that MTM-HCC could be effectively diagnosed by intratumor ischemia or necrosis combined with intratumor fat deficiency (Figures 2 and 3). However, a study by Kang et al51 showed inconsistency with the above results because most patients in their study had large tumors (≥5 cm), and the authors defined substantial necrosis as ≥50%. Zhu et al52 diagnosed MTM-HCC using radiomics based on the hotness of artificial intelligence and found that radiomics Rad-score and intratumor fat were independent predictors, while intratumor ischemia or necrosis showed a statistical difference only in univariate analysis. In addition, their research results related to intratumor fat were contrary to our study. We believe that intratumor fat mainly occurs in well-differentiated liver cancer and that fat deposition is caused by transient hypoxia in arterial angiogenesis in liver cancer.53,54 However, a study found intratumor fat, intratumor hyperenhancement in the arterial phase, and “rapid wash-out” mostly occur in advanced liver cancers.55 In addition, a study showed that MTM-HCC was more commonly seen in noncirrhotic livers,56 which needs to be confirmed by multi-center studies.
Diagnosis and Treatment of MTM-HCC
Currently, the guidelines in China and other countries state that HCC can be diagnosed using advanced imaging without pathological diagnosis.2,57 In the past decade, different pathological subtypes of HCC have not exerted enough influence in clinical practice. Therefore, they have not aroused the interest of clinicians. However, for MTM-HCC, the diagnostic consistency of pathologists after core biopsy is high,58 so the utility of preoperative biopsies should be re-examined. In addition, based on its different molecular types and carcinogenic pathways, MTM-HCC may be relatively sensitive to angiogenesis inhibitors, such as anti-Ang-2 and anti-VEGFA antibody.59 Furthermore, scholars have found that CKLF-like MARVEL transmembrane domain-containing 6 (CMTM6) promotes Programmed death-1 (PD-L1) expression in tumor cells in the defense against T cells. Thus, the combination of immune status assessment with anti-CMTM6 and anti-PD-L1 therapy may be more effective in the treatment of MTM-HCC.60 Other scholars have provided mechanistic insight into the therapeutic effects of immunotherapy, anti-angiogenic therapy, and their combination.61 So, the therapies used to treat MTM-HCC should differ from those used for the more common types of HCC, and individualized treatment should be provided for patients with MTM-HCC.
Consent for Publication
All authors approved the final manuscript.
Funding
This study was supported by the National Key Research and Development Project (2016YFC0107101) and Zunyi Natural Science Foundation of Innovation Cultivation Program [Project No. Zunshirencai (2020) 6].
Disclosure
The authors declare that they have no competing interests in this work.
References
1. Papatheodoridis GV, Sypsa V, Dalekos GN, et al. Hepatocellular carcinoma prediction beyond year 5 of oral therapy in a large cohort of Caucasian patients with chronic hepatitis B. J Hepatol. 2020;72(6):1088–1096. doi:10.1016/j.jhep.2020.01.007
2. Department of Medical Administration, National Health and Health Commission of the People’s Republic of China. Guidelines for diagnosis and treatment of primary liver cancer in China (2019 edition). Chin J Hepatol. 2020;28(02):112–128. doi:10.3760/cma.j.issn.1007-3418.2020.02.004
3. Zhou J, Sun H, Wang Z, et al. Guidelines for the diagnosis and treatment of hepatocellular carcinoma (2019 Edition). Liver Cancer. 2020;9(6):682–720. doi:10.1159/000509424
4. Li X, Jing H, Cheng L, et al. A case study of glycogen storage disease type Ia presenting with multiple hepatocellular adenomas: an analysis by gadolinium ethoxybenzyl-diethylenetriamine-pentaacetic acid magnetic resonance imaging. Quant Imaging Med Surg. 2021;11(6):2785–2791. doi:10.21037/qims-20-746
5. Asfari MM, Talal Sarmini M, Alomari M, Lopez R, Dasarathy S, McCullough AJ. The association of nonalcoholic steatohepatitis and hepatocellular carcinoma. Eur J Gastroenterol Hepatol. 2020;32(12):1566–1570. doi:10.1097/meg.0000000000001681
6. Yubo Z, Yiming N, Peng Z, et al. Establishment and validation of a nomogram prediction model for early preoperative diagnosis of hepatocellular carcinoma with microvascular invasion. Chin J Hepatobiliary Surg. 2021;27(10):721–726. doi:10.3760/cma.j.cn113884-20210327-00113
7. Marrero JA, Kulik LM, Sirlin CB, et al. Diagnosis, staging, and management of hepatocellular carcinoma: 2018 practice guidance by the American Association for the Study of Liver Diseases. Hepatology. 2018;68(2):723–750. doi:10.1002/hep.29913
8. Nault JC, Martin Y, Caruso S, et al. Clinical impact of genomic diversity from early to advanced hepatocellular carcinoma. Hepatology. 2020;71(1):164–182. doi:10.1002/hep.30811
9. Yoneda N, Matsui O, Kobayashi S, et al. Current status of imaging biomarkers predicting the biological nature of hepatocellular carcinoma. Jpn J Radiol. 2019;37(3):191–208. doi:10.1007/s11604-019-00817-3
10. Kumar D, Hafez O, Jain D, Zhang X. Can primary hepatocellular carcinoma histomorphology predict extrahepatic metastasis? Hum Pathol. 2021;113:39–46. doi:10.1016/j.humpath.2021.04.008
11. Nagtegaal I, Odze R, Klimstra D, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2019;76:182–188. doi:10.1111/his.13975
12. Calderaro J, Ziol M, Paradis V, Zucman-Rossi J. Molecular and histological correlations in liver cancer. J Hepatol. 2019;71(3):616–630. doi:10.1016/j.jhep.2019.06.001
13. Wei J, Jiang H, Zeng M, et al. Prediction of microvascular invasion in hepatocellular carcinoma via deep learning: a multi-center and prospective validation study. Cancers. 2021;13(10):2368. doi:10.3390/cancers13102368
14. Aggarwal A, Te HS, Verna EC, Desai AP. A national survey of hepatocellular carcinoma surveillance practices following liver transplantation. Transplant Direct. 2021;7(1):e638. doi:10.1097/txd.0000000000001086
15. Lee S, Kang TW, Song KD, et al. Effect of microvascular invasion risk on early recurrence of hepatocellular carcinoma after surgery and radiofrequency ablation. Ann Surg. 2021;273(3):564–571. doi:10.1097/SLA.0000000000003268
16. Zhang EL, Cheng Q, Huang ZY, Dong W. Revisiting surgical strategies for hepatocellular carcinoma with microvascular invasion. Front Oncol. 2021;11:691354. doi:10.3389/fonc.2021.691354
17. Ziol M, Poté N, Amaddeo G, et al. Macrotrabecular-massive hepatocellular carcinoma: a distinctive histological subtype with clinical relevance. Hepatology. 2018;68(1):103–112. doi:10.1002/hep.29762
18. Calderaro J, Couchy G, Imbeaud S, et al. Histological subtypes of hepatocellular carcinoma are related to gene mutations and molecular tumour classification. J Hepatol. 2017;67(4):727–738. doi:10.1016/j.jhep.2017.05.014
19. Lee JS, Chu IS, Heo J, et al. Classification and prediction of survival in hepatocellular carcinoma by gene expression profiling. Hepatology. 2004;40(3):667–676. doi:10.1002/hep.20375
20. Boyault S, Rickman DS, de Reyniès A, et al. Transcriptome classification of HCC is related to gene alterations and to new therapeutic targets. Hepatology. 2007;45(1):42–52. doi:10.1002/hep.21467
21. Gao Q, Zhu H, Dong L, et al. Integrated proteogenomic characterization of HBV-related hepatocellular carcinoma. Cell. 2019;179(2):561–577.e22. doi:10.1016/j.cell.2019.08.052
22. Ally A, Balasundaram M, Carlsen R. Comprehensive and integrative genomic characterization of hepatocellular carcinoma. Cell. 2017;169(7):1327–1341.e23. doi:10.1016/j.cell.2017.05.046
23. Villanueva A, Longo DL. Hepatocellular carcinoma. N Engl J Med. 2019;380(15):1450–1462. doi:10.1056/NEJMra1713263
24. Long J, Wang A, Bai Y, et al. Development and validation of a TP53-associated immune prognostic model for hepatocellular carcinoma. EBioMedicine. 2019;42:363–374. doi:10.1016/j.ebiom.2019.03.022
25. Gao L, Wang X, Tang Y, Huang S, Hu CA, Teng Y. FGF19/FGFR4 signaling contributes to the resistance of hepatocellular carcinoma to sorafenib. J Exp Clin Cancer Res. 2017;36(1):8. doi:10.1186/s13046-016-0478-9
26. Hyeon J, Ahn S, Lee JJ, Song DH, Park CK. Expression of fibroblast growth factor 19 is associated with recurrence and poor prognosis of hepatocellular carcinoma. Dig Dis Sci. 2013;58(7):1916–1922. doi:10.1007/s10620-013-2609-x
27. Raja A, Park I, Haq F, Ahn SM. FGF19-FGFR4 signaling in hepatocellular carcinoma. Cells. 2019;8(6):536. doi:10.3390/cells8060536
28. Ahn SM, Jang SJ, Shim JH, et al. Genomic portrait of resectable hepatocellular carcinomas: implications of RB1 and FGF19 aberrations for patient stratification. Hepatology. 2014;60(6):1972–1982. doi:10.1002/hep.27198
29. Caruso S, Calatayud AL, Pilet J, et al. Analysis of liver cancer cell lines identifies agents with likely efficacy against hepatocellular carcinoma and markers of response. Gastroenterology. 2019;157(3):760–776. doi:10.1053/j.gastro.2019.05.001
30. Li Y, Li L, Wu Z, et al. Silencing of ATM expression by siRNA technique contributes to glioma stem cell radiosensitivity in vitro and in vivo. Oncol Rep. 2017;38(1):325–335. doi:10.3892/or.2017.5665
31. Yan X, Wu T, Tang M, et al. Methylation of the ataxia telangiectasia mutated gene (ATM) promoter as a radiotherapy outcome biomarker in patients with hepatocellular carcinoma. Medicine. 2020;99(4):e18823. doi:10.1097/md.0000000000018823
32. Qi Z, Yan F, Chen D, et al. Identification of prognostic biomarkers and correlations with immune infiltrates among cGAS-STING in hepatocellular carcinoma. Biosci Rep. 2020;40(10). doi:10.1042/bsr20202603
33. Gerald D, Chintharlapalli S, Augustin HG, Benjamin LE. Angiopoietin-2: an attractive target for improved antiangiogenic tumor therapy. Cancer Res. 2013;73(6):1649–1657. doi:10.1158/0008-5472.Can-12-4697
34. Vanderborght B, Lefere S, Vlierberghe HV, Devisscher L. The angiopoietin/Tie2 pathway in hepatocellular carcinoma. Cells. 2020;9(11):2382. doi:10.3390/cells9112382
35. Fang JH, Zhou HC, Zhang C, et al. A novel vascular pattern promotes metastasis of hepatocellular carcinoma in an epithelial-mesenchymal transition-independent manner. Hepatology. 2015;62(2):452–465. doi:10.1002/hep.27760
36. Chen J, Gingold JA, Su X. Immunomodulatory TGF-β signaling in hepatocellular carcinoma. Trends Mol Med. 2019;25(11):1010–1023. doi:10.1016/j.molmed.2019.06.007
37. Kang H, Oh T, Bahk YY, et al. HSF1 regulates mevalonate and cholesterol biosynthesis pathways. Cancers. 2019;11(9):1363. doi:10.3390/cancers11091363
38. Gong C, Ai J, Fan Y, et al. NCAPG promotes the proliferation of hepatocellular carcinoma through PI3K/AKT signaling. Onco Targets Ther. 2019;12:8537–8552. doi:10.2147/ott.S217916
39. Calderaro J, Meunier L, Nguyen C, et al. ESM1 as a marker of macrotrabecular-massive hepatocellular carcinoma. Clin Cancer Res. 2019;25(19):5859–5865. doi:10.1158/1078-0432.Ccr-19-0859
40. Zhou HC, Liu CX, Pan WD, et al. Dual and opposing roles of the androgen receptor in VETC-dependent and invasion-dependent metastasis of hepatocellular carcinoma. J Hepatol. 2021;75(4):900–911. doi:10.1016/j.jhep.2021.04.053
41. Woo HY, Rhee H, Yoo JE, et al. Lung and lymph node metastases from hepatocellular carcinoma: comparison of pathological aspects. Liver Int. 2021;42:199–209. doi:10.1111/liv.15051
42. Renne S, Woo H, Allegra S, et al. Vessels Encapsulating Tumor Clusters (VETC) is a powerful predictor of aggressive hepatocellular carcinoma. Hepatology. 2020;71(1):183–195. doi:10.1002/hep.30814
43. Feng Z, Li H, Zhao H, et al. Preoperative CT for characterization of aggressive macrotrabecular-massive subtype and vessels that encapsulate tumor clusters pattern in hepatocellular carcinoma. Radiology. 2021;300(1):219–229. doi:10.1148/radiol.2021203614
44. Yoon JH, Kim H. CT characterization of aggressive macrotrabecular-massive hepatocellular carcinoma: a step forward to personalized medicine. Radiology. 2021;300(1):230–232. doi:10.1148/radiol.2021210379
45. Rhee H, An C, Kim HY, Yoo JE, Park YN, Kim MJ. Hepatocellular carcinoma with irregular rim-like arterial phase hyperenhancement: more aggressive pathologic features. Liver Cancer. 2019;8(1):24–40. doi:10.1159/000488540
46. Rhee H, Cho E, Nahm J, et al. Gadoxetic acid-enhanced MRI of macrotrabecular-massive hepatocellular carcinoma and its prognostic implications. J Hepatol. 2021;74(1):109–121. doi:10.1016/j.jhep.2020.08.013
47. Tohme S, Yazdani H, Liu Y, et al. Hypoxia mediates mitochondrial biogenesis in hepatocellular carcinoma to promote tumor growth through HMGB1 and TLR9 interaction. Hepatology. 2017;66(1):182–197. doi:10.1002/hep.29184
48. Mulé S, Galletto Pregliasco A, Tenenhaus A, et al. Multiphase liver MRI for identifying the macrotrabecular-massive subtype of hepatocellular carcinoma. Radiology. 2020;295(3):562–571. doi:10.1148/radiol.2020192230
49. Chen J, Xia C, Duan T, et al. Macrotrabecular-massive hepatocellular carcinoma: imaging identification and prediction based on gadoxetic acid-enhanced magnetic resonance imaging. Eur Radiol. 2021;31:7696–7704. doi:10.1007/s00330-021-07898-7
50. Li XM, Cai P, Cheng L, et al. Clinical value of gadolinium ethoxybenzyl diethylanetriaminepentaacetic acid enhanced MRI in the preoperative diagnosis of macrotrabecular-massive hepatocellular carcinoma. Chin J Dig Surg. 2021;20(11):1218–1226. doi:10.3760/cma.j.cn115610-20210809-00385
51. Kang HJ, Kim H, Lee DH, et al. Gadoxetate-enhanced MRI features of proliferative hepatocellular carcinoma are prognostic after surgery. Radiology. 2021;300(3):572–582. doi:10.1148/radiol.2021204352
52. Zhu Y, Weng S, Li Y, et al. A radiomics nomogram based on contrast-enhanced MRI for preoperative prediction of macrotrabecular-massive hepatocellular carcinoma. Abdomin Radiol. 2021;46:3139–3148. doi:10.1007/s00261-021-02989-x
53. An C, Kim M-J. Imaging features related with prognosis of hepatocellular carcinoma. Abdomin Radiol. 2019;44(2):509–516. doi:10.1007/s00261-018-1758-y
54. Rajlawot K, Jiang T, Zhou J, et al. Accuracies of chemical shift in/opposed phase and chemical shift encoded magnetic resonance imaging to detect intratumoral fat in hepatocellular carcinoma. JMRI. 2021;53(6):1791–1802. doi:10.1002/jmri.27539
55. Rimola J, Forner A, Tremosini S, et al. Non-invasive diagnosis of hepatocellular carcinoma ≤ 2 cm in cirrhosis. Diagnostic accuracy assessing fat, capsule and signal intensity at dynamic MRI. J Hepatol. 2012;56(6):1317–1323. doi:10.1016/j.jhep.2012.01.004
56. Jain A, Mazer B, Deng Y, et al. Hepatocellular carcinoma: does the background liver with or without cirrhosis matter? Am J Clin Pathol. 2021. doi:10.1093/ajcp/aqab125
57. Heimbach JK, Kulik LM, Finn RS, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67(1):358–380. doi:10.1002/hep.29086
58. Vij M, Calderaro J. Pathologic and molecular features of hepatocellular carcinoma: an update. World J Hepatol. 2021;13(4):393–410. doi:10.4254/wjh.v13.i4.393
59. Schmittnaegel M, Rigamonti N, Kadioglu E, et al. Dual angiopoietin-2 and VEGFA inhibition elicits antitumor immunity that is enhanced by PD-1 checkpoint blockade. Sci Transl Med. 2017;9(385). doi:10.1126/scitranslmed.aak9670
60. Liu LL, Zhang SW, Chao X, et al. Coexpression of CMTM6 and PD-L1 as a predictor of poor prognosis in macrotrabecular-massive hepatocellular carcinoma. Cancer Immunol Immunother. 2021;70(2):417–429. doi:10.1007/s00262-020-02691-9
61. Kurebayashi Y, Matsuda K, Ueno A, et al. Immunovascular classification of HCC reflects reciprocal interaction between immune and angiogenic tumor microenvironments. Hepatology. 2021;75:1139–1153. doi:10.1002/hep.32201
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.