Back to Journals » Drug Design, Development and Therapy » Volume 17
Macro, Micro, and Nano-Inspired Bioactive Polymeric Biomaterials in Therapeutic, and Regenerative Orofacial Applications
Authors Atia GA , Shalaby HK, Roomi AB, Ghobashy MM, Attia HA, Mohamed SZ, Abdeen A , Abdo M , Fericean L, Bănățean Dunea I , Atwa AM, Hasan T, Mady W, Abdelkader A , Ali SA, Habotta OA , Azouz RA, Malhat F, Shukry M , Foda T, Dinu S
Received 19 May 2023
Accepted for publication 12 August 2023
Published 27 September 2023 Volume 2023:17 Pages 2985—3021
DOI https://doi.org/10.2147/DDDT.S419361
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Tuo Deng
Gamal A Atia,1 Hany K Shalaby,2 Ali B Roomi,3,4 Mohamed M Ghobashy,5 Hager A Attia,6 Sara Z Mohamed,7 Ahmed Abdeen,8 Mohamed Abdo,9,10 Liana Fericean,11 Ioan Bănățean Dunea,11 Ahmed M Atwa,12 Tabinda Hasan,13 Wessam Mady,14 Afaf Abdelkader,15 Susan A Ali,16 Ola A Habotta,17 Rehab A Azouz,18 Farag Malhat,19 Mustafa Shukry,20 Tarek Foda,21 Stefania Dinu22
1Department of Oral Medicine, Periodontology, and Diagnosis, Faculty of Dentistry, Suez Canal University, Ismailia, Egypt; 2Department of Oral Medicine, Periodontology and Oral Diagnosis, Faculty of Dentistry, Suez University, Suez, Egypt; 3Department of Quality Assurance, University of Thi-Qar, Thi-Qar, Iraq; 4Department of Medical Laboratory, College of Health and Medical Technology, National University of Science and Technology, Thi-Qar, Iraq; 5Radiation Research of Polymer Chemistry Department, National Center for Radiation Research and Technology (NCRRT), Atomic Energy Authority, Cairo, Egypt; 6Department of Molecular Biology and Chemistry, Faculty of Science, Alexandria University, Alexandria, Egypt; 7Department of Removable Prosthodontics, Faculty of Dentistry, Suez Canal University, Ismailia, Egypt; 8Department of Forensic Medicine and Toxicology, Faculty of Veterinary Medicine, Benha University, Toukh, Egypt; 9Department of Animal Histology and Anatomy, School of Veterinary Medicine, Badr University in Cairo (BUC), Badr City, Egypt; 10Department of Anatomy and Embryology, Faculty of Veterinary Medicine, University of Sadat City, Sadat, Egypt; 11Department of Biology and Plant Protection, Faculty of Agriculture. University of Life Sciences “King Michael I” from Timișoara, Timișoara, Romania; 12Department of Pharmacology and Toxicology, Faculty of Pharmacy, Egyptian Russian University, Cairo, Egypt; 13Department of Basic Sciences, College of Medicine, Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia; 14Center of Excellence in Genomic Medicine Research, King Abdulaziz University, Jeddah, Saudi Arabia; 15Department of Forensic Medicine and Clinical Toxicology, Faculty of Medicine, Benha University, Benha, Egypt; 16Department of Radiodiagnosis, Faculty of Medicine, Ain Shams University, Abbassia, 1181, Egypt; 17Department of Forensic Medicine and Toxicology, Faculty of Veterinary Medicine, Mansoura University, Mansoura, Egypt; 18Department of Forensic Medicine and Toxicology, Faculty of Veterinary Medicine, Cairo University, Giza, Egypt; 19Department of Pesticide Residues and Environmental Pollution, Central Agricultural Pesticide Laboratory, Agricultural Research Center, Giza, Egypt; 20Department of Physiology, Faculty of Veterinary Medicine, Kafrelsheikh University, Kafrelsheikh, Egypt; 21Oral Health Sciences Department, Temple University’s Kornberg School of Dentistry, Philadelphia, PA, USA; 22Department of Pedodontics, Faculty of Dental Medicine, Victor Babes University of Medicine and Pharmacy Timisoara, Timisoara, 300041, Romania
Correspondence: Gamal A Atia, Department of Oral Medicine, Periodontology, and Diagnosis, Faculty of Dentistry, Suez Canal University, Ismailia, 41522, Egypt, Tel +201026961611, Email [email protected] Liana Fericean. Department of Biology and Plant Protection, Faculty of Agriculture, University of Life Sciences “King Michael I” from Timișoara, Calea Aradului 119, CUI 3487181, Romania, Tel +40723085385, Email [email protected]
Abstract: Introducing dental polymers has accelerated biotechnological research, advancing tissue engineering, biomaterials development, and drug delivery. Polymers have been utilized effectively in dentistry to build dentures and orthodontic equipment and are key components in the composition of numerous restorative materials. Furthermore, dental polymers have the potential to be employed for medication administration and tissue regeneration. To analyze the influence of polymer-based investigations on practical medical trials, it is required to evaluate the research undertaken in this sector. The present review aims to gather evidence on polymer applications in dental, oral, and maxillofacial reconstruction.
Keywords: polymers, regeneration, oral, dental, maxillofacial
Introduction
Numerous conditions, including infections, genetic disorders, malignancies, and trauma, can lead to dental, oral, and maxillofacial tissue defects. The Global Burden of Diseases, Injuries, and Risk Factors Study (GBD 2017) demonstrated that oral problems had the most significant occurrence and frequency rates on a global scale.1 In this context and thanks to recent advancements in biomaterials and production technologies, natural and synthetic polymeric biomaterials have been introduced as viable therapeutic alternatives for various defects and abnormalities in dental, oral, and maxillofacial structures.2 The therapeutic application of polymeric scaffolds has been extensively studied in regenerative therapies for several tissues. Numerous tissue engineering methods and surgical interventions have been introduced with an emphasis on comprehending the unique features of a biomaterial at cellular contact. Accordingly, this review provides an overview of the most important polymeric scaffolds utilized for therapeutic, and regenerative purposes in dental, oral and maxillofacial tissues, as well as information on their characteristics, formulations, manufacturing techniques, advantages, disadvantages, and clinical uses.
Overview of Biomaterials in Dental Applications
Polymers are macromolecules made up of repeated monomeric subunits that have outstanding characteristics. Innovation in biomaterials research encompasses applications in tissue engineering, nanotechnology, and the administration of bioactive molecules for the healing and regeneration of diverse tissues. The new generation of polymers was eminently distinguished by the development of resorbable, biodegradable polymers that displayed controlled degradation of the polymer chain3.Various biomaterials have been implemented in the dental field (Table 1).4 Naturally, materials (eg, collagen) attracted attention; synthetically derived polymeric biomaterials were later introduced. Chitosan and collagen are naturally occurring polymers with high biocompatibility, bone conductivity, and minimal immunological reactions.5 Nevertheless, disadvantages include a slow degradation rate and inferior mechanical qualities.
 |
Table 1 Various Biomaterials Employed in the Dental Field |
Breakthroughs in the composition of synthetic biopolymers have paved the way for the development of further novel materials with excellent tissue responsiveness and integration, and capable of being degraded and removed from the system, permitting correct tissue integration as the material breaks down, without exerting any harmful influence on the biological systems.7 Further investigations are underway to develop scaffold formulations appropriate for biological systems with little or no negative impact on living systems.
Polymers are macromolecules made up of repeated monomeric subunits that are covalently bound. They can be amorphous or crystalline, having linear, branching, or cross-linked chains. Their mechanical characteristics could be extremely well regulated, allowing for the acquisition of desirable features based on application requirements. Polymers have lately been used to construct microneedles (MNs), hydrogels, microcapsules, microspheres, and fibers. These frameworks can further be designed to be biocompatible and biodegradable, eliminating possible immune reactions and allowing for simple body disposal (Figure 1 shows the advantages of polymeric scaffolds).6 Furthermore, their hydrophilicity may give them biologically advantageous properties, especially as reservoirs for biological compounds in various medical applications.6
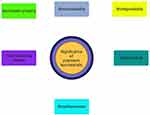 |
Figure 1 Advantages of polymeric scaffolds. |
Biological Requirements
The essential component in applying biomaterial in situ tissue engineering frameworks is biocompatibility. The scaffold is biocompatible, does not elicit an immune response, generates no hazardous byproducts, and permits cells to attach, proliferate, reproduce, and survive on its surface.8,9
Structural Features
Scaffolds must have high porosity to allow cell growth and movement, essential micronutrients, angiogenesis, and spatial layout. They should be customized to fit the regenerated tissue. They should be strong enough to endure biomechanical loads until restored tissue can withstand forces.10 Another critical factor is architecture, which could be altered by adjusting artificial ECM and/or biomolecules to be administered in the microenvironment.11
Biomaterial Composition
Depending on their architecture and intended use, they can be injectable or rigid.12 Polymers may be both natural and synthetic. Chitosan and collagen are naturally occurring polymers with high biocompatibility, bone conductivity, and minimal immunological reactions. Nevertheless, disadvantages include a slow degradation rate and restricted mechanical qualities.5
Polymeric-Based Biomaterials
Nowadays, polymer-based methods can be used as a tactic to enhance tissue regeneration. Examples of polymeric materials implemented in tissue engineering applications, as represented in Table 2.10,13–15 Figure 2 represents natural and synthetic polymers in dental applications. Many polymeric formulations, including dendrimers, nano gels, microneedles, and nanocapsules, have been studied as prospective approaches for dental applications.
 |
Table 2 Polymeric Biomaterials, Benefits, and Limitations in Biomedical Applications |
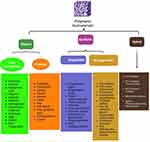 |
Figure 2 Natural and synthetic polymers in dental applications. |
Natural Polymers
Natural polymers originate from natural substances and guaranteed natural restructure biomimetic character and biocompatibility. Table 3 shows the advantages, and disadvantages of some natural biopolymers.16 Polysaccharide and protein-based polymers are roughly classified based on their monomeric units and structure.17
 |
Table 3 Advantages and Limitations of Some Natural Polymers |
Polysaccharide-Based Polymers
Thanks to their resemblance to the extracellular matrix, polysaccharides have great degradability, bioactivity, ease of chemical modification, and low manufacturing costs.25 Even with their numerous advantages, natural polymers have some downfalls related to their branching, dispersion, pattern, and molecular mass, which results in the preparation of unreliable scaffolds, which harms biological recognition events and rheology.26
Chitosan
Chitosan is the world’s second most common natural polysaccharide, following cellulose. Several studies have investigated the biological applications of chitin and chitosan.27 Chitosan is principally a chitin metabolite that has been deacetylated.28 Because of its unique features of bioactivity, good biocompatibility, and non-immunogenicity, chitosan has become increasingly popular in the biomedical field.20 Chitosan-derived metabolites have been employed in several applications.24
Dextran
Scheible in 1874 created the term Dextran.27 It is an extracellular microbial carbohydrate derived from sucrose-derived bacterial lactic acid.29 Because of their nontoxicity, hydration, bio-compatibility, and degradability, dextran polymers are frequently employed in the food sector, personal care products, wastewater treatment, and biomedical purposes.30,31
Hyaluronic Acid
HA contributes significantly to the environment’s extracellular matrix and synovial fluids.32 The molecular weight of HA governs numerous biomedical actions. High molecular weight HA has been discovered to have anti-inflammatory and immune-modulatory properties.33 A few high molecular weight HA metabolites have also been proven to show anti-angiogenic characteristics and the capability to suppress cell growth.34 Low molecular weight HA promotes cell motility and vasculogenesis.35 HA is classified as a naturally existing negatively charged polysaccharide because of a carboxyl group in its backbone.36 This feature allows HA to maintain a substantial amount of water, leading to bulging behavior of HA up to 1000 times its solid dimensions, which several investigators have used to develop HA pharmacological carriers,37 and numerous biological implementations.38,39
Heparin
While researching cephalin in 1916, Jay McLean observed anticoagulant phosphatide fractions independent from it. Emmett Holt and William Henry Howell later called Heparin after another anti-coagulant molecule obtained from the liver.40 Heparin is a natural glycosaminoglycan produced by mast cells in the Golgi apparatus and the endoplasmic reticulum. It is derived from various origins for pharmaceutical and industrial use.41 Heparin has been employed as a transporter of growth factors.42 Heparin is also anticoagulant since its penta saccharide specifically reacts with anti-thrombin.43 These features emphasize the ubiquitous utilization of heparin-based carriers, like micelles and nano gels, as drug delivery vehicles.44
Alginate
E. C. C. Stanford revealed alginic acid in 1881, detailing the isolation of alginate with sodium carbonate and its deposition in an acidic solution.45 Species of brown seaweeds like Laminaria and nereocystis, along with bacterial strains like Azotobacter and Pseudomonas, are significant sources of alginates.46 Because of its unique characteristics, alginate-based biomaterials are utilized in many biomedical applications.18 The thickness of polymeric alginate impression materials should be reduced into minimal as it greatly influences the tear strength of the polymer after setting.47
Pullulan
Pullulan is a structurally intermediate polysaccharide containing matotriose particles between amylose and dextran.48 Pullulan is hydrophilic and moderately soluble in alkaline solutions, whereas inorganic solvents, including formamide and dimethyl sulphoxide, are completely insoluble (DMSO).49 Pullulan is an intriguing material for the nutraceutical and pharmaceutical sectors, as well as medication delivery, biomedical engineering, gene therapy, and so on, due to its biodegradability, non-toxicity, and non-mutagenicity.50
Carrageenan
Carrageenan is an anionic, sulfated polygalacton discovered in the cell walls of red algae that is comparable to ECM-derived glycosaminoglycans. Carrageenan’s structure promotes osteoblastic growth and attachment.51 It increases osteoblastic activity when combined with hydroxyapatite.52 Carrageenan was mixed with other chemicals in bone regeneration tissue engineering investigations to generate hybridized bio-scaffolds.53
Chondroitin Sulphate
Chondroitin Sulphate (CoS) is an essential cartilage component connected with compression resistance. CoS originated from chondrocytes and has a crucial influence in preserving the physiological functions of cartilages.54 CoS absorbs water and has a cushion-like effect on cartilage.55 CoS’s polyanionic nature offers a foundation for electrostatic interactions with positively charged moieties, which could be employed to deliver medications and growth nutrients. CoS is anti-inflammatory, anti-apoptotic, antioxidant, and anti-cancer.56
Cellulose
Cellulose is often recognized as the most widely recognized polysaccharide on the planet.57 Cellulose derived from bacteria, termed bacterial cellulose (BC), is highly pure, but cellulose derived from plants is documented to include trace levels of contaminants such as lignin, pectin, and hemicellulose.58 Because of its nontoxicity, degradability, biocompatibility, wide availability, and remarkable mechanical qualities, cellulose, and its variants are valuable materials for drug administration, wound healing, and other biological purposes.19 Using oral thin film made from a blend of hydroxyethyl-cellulose (HEC) and cellulose nanofibers (CNF), loaded with nepheline fluorapatite glass powder as a remineralizing agent for prevention of carious lesion by remineralization was tested. Significant quantities of calcium and fluoride ions discharged and the pH values were practically neutral. The arrangement of glass powder particles was consistent, according to SEM. In comparison to the demineralized samples, the findings of enamel micro-hardness (VHN) and ultra-morphology showed a considerable rise in mean VHN following remineralization for 15 and 30 days.59
Agar
Agar is another polysaccharide biopolymer that is commonly recognized for its strong bioactivity and biocompatibility.36 It can generate a gel that resembles the physical properties of the ECM and tissues essential for transferring growth factors to wounds.60 Previous studies proved its non-toxicity and successful cell proliferation. Consequently, it can be employed in healing and skin regeneration.61
Starch
Due to starch’s degradability, non-cytotoxicity, and processability, it is an excellent example of a storing polysaccharide.62 Polymers made from starch have shown promise in a variety of biological applications. Starch hydrogels fortified with zeolite NPs and chamomile preparations successfully treated chronic ulcers with no hypersensitivity reaction to the scaffold.61
Gum Tragacanth
This is a polysaccharide originating from the Astragalus plant.63 Gum Tragacanth is widely utilized in tissue engineering to create biodegradable frameworks and medicine administration mechanisms. This scaffold has demonstrated encouraging outcomes in terms of osseous regeneration.64
Natural Proteins
Collagen
Collagen is one of the most widely utilized protein biopolymers. It possesses a significant conjunction effectiveness with other compounds, which may be changed to achieve improved biological and mechanical qualities.22 Over 28 isomers of collagen have been extracted, each with a different helix length and the type and dimensions of the non-helical parts.65 Type I collagen is the most prevalent kind that constitutes the ECM of several oral tissues, such as periodontal ligament, enamel, etc.66–70 As an ECM structural element, it has strong biocompatibility, low immunogenicity, and significant mechanical integrity, and it assists in growth, differentiation, cell attachment, and ECM formation.71 It is an attractive natural resource due to the ease with which it may be modified.72 Tannic acid cross-linked collagen frameworks made by casting have been used to improve wound healing qualities.
Fibronectin
Fibronectin is a glycoprotein with a high molecular mass that is extracted from plasma. Its major purpose as a scaffold is to facilitate cell attachment and motility, influencing cell division and growth.73 Fibronectin nano fibers produced by rotary jet spinning demonstrated faster wound closure.74
Gelatin
Gelatin is an organic protein generated from collagen commonly employed in biomedicine thanks to its poor immunogenicity, bioactivity, and good biocompatibility.75 Because of its gelation properties and simplicity of processing, it may be processed into numerous formulations such as injectable hydrogels, sponges, and microspheres.76 Gelatin has also been combined with copper-activated faujasite to generate composites with high anti-microbial action using the freeze-drying process.77 Li et al established a straightforward strategy for creating gelatin-based scaffolds with hollow alginate fibers to create a vascular network.78
Elastin
Elastin is a hydrophobic and insoluble extracellular matrix (ECM) protein. Collagen-elastin frameworks have been used to treat full-thickness skin abnormalities such as burn wounds as dermal replacement.79
Keratin
Keratin is a protein in the skin, fingernails, and hair.80 This protein is found in the skin, nails, and hair. It is biocompatible and capable of building frameworks that coordinate cellular activities.61 By lyophilization, Keratin has also been used with chitosan to create a porous keratin/chitosan scaffold. The antimicrobial activity and cell proliferation rates of keratin/-chitosan scaffolds used for wound dressings were increased.81
Fibrin
Fibrin is a naturally degradable matrix composed of fibrinogen, created soon after trauma.82 The fibrin-based scaffolds promote growth by allowing the attachment of various biologically active biomolecules, promoting specific cell-matrix interactions and boosting tissue regeneration.83 They allow enough time to form neo matrix and progressive re-assimilation via protease activity.84 Fibrin can be employed as a biological framework with other elements to regenerate primary cells, skin, bones, and so on.85 Nevertheless, it has the disadvantage of quick degradation, which makes it unsuitable for creating particular scaffolds.86
Soy
Soy protein extract is a sustainable and readily accessible protein with more than 90% polypeptide. It has a macromolecular structure equal to the naturally generated proteins in bone.23 Wu et al employed a comparable bi-component scaffold for bone tissue creation in experiment.23
Silk Fibroin
Silk fibroin is a significant constituent with a unique amino acid composition that encapsulates sericin obtained from Bombyx mori cocoons.87 Because the protein resembles the stroma of bone, it is synthesized into frameworks for regenerating bone tissue.21 The addition of fibroin enhances the hydrophilicity and mechanical properties of the scaffolds, allowing human osteoblast-like cells to survive.88 A two-stage functionalization procedure generated silk fibroin nano-fibers containing hydroxyapatite particles.89
Poly Glutamic Acid
Polyglutamic acid is a biopolymer made up of carboxy-linked glutamate residues released by Bacillus subtilis.90 This polymer is edible and has great biocompatibility, nontoxicity, and biodegradability. These characteristics make it a desirable material for tissue engineering,91 biodegradable wrappers,92 wound bandages,92 and pharmaceutical administration.93
Polydopamine (PDA)
PDA is a darkish, opaque polymer generated by dopamine autoxidation.94 Thanks to its intriguing features and simplicity of manufacture, PDA has recently attracted significant attention in various biological applications, including medication delivery, photodynamic therapy, skeletal tissue engineering, cell attachment and imprinting, and antibacterial use.95
Lignin (LIG)
LIG is a recyclable, sustainable, safe, economical, and natural biomaterial that has grabbed the spotlight of research scientists thanks to its distinctive characteristics and environmentally friendly nature.96 LIG is antibacterial, antioxidant, and antiviral, with UV protection, biocompatibility, and minimal cytotoxicity.97 LIG has been poorly investigated in the biomedical area until now, with just a few research reporting on its use in healthcare applications.98
Synthetic Polymers
In the research laboratories, synthetic polymers are created using hydrocarbon-building components.99 They have the benefit of infinite shapes and well-established structures. They are mechanically stable and free of contaminants.100 They enable convenience and administration of manufacturing via polymerization, interlinkage, and productivity due to their molecular weight, molecular weight, and physicochemical characteristics. These polymers are classified according to their hydrophobicity and hydrophilicity101 and can also be categorized according to degradability.102 They are instrumental since their features may be easily modified for various and specialized purposes.14 However, their fundamental drawback is the lack of cell attachment sites and the need for chemical functionalization for cellular growth.103
Degradable Polymers
As the term indicates, biodegradable polymers may be rapidly decomposed or converted into simpler molecules or by-products.104
Polyethylene Glycol
Polyethylene glycol (PEG) is a commercially important polyether among degradable polymers (PEG). It is an ethylene oxide polymer called polyethylene oxide (PEO) or polyoxyethylene.105 It is non-ionic, biocompatible, and has excellent biological and physicochemical qualities. The cross-linking techniques used to create hydrophilic PEG hydrogels can impact scaffolds’ physicochemical characteristics and degradation rates.106 Architecture’s ease of manipulation and chemical makeup makes it an appealing scaffold material.107
Polyethylene Oxide (PEO)
PEO is a hydrophilic polymer with minimal pathogenicity, cell attachment, immunogenicity, and the ability for protein binding.108 PEG’s smaller molecular structure became well-known when its ability to block protein absorption was discovered.109 More research is needed to modify these gels to include places for cell attachment, which will help cells penetrate the scaffolds.110 When combined with electro-spun scaffolds made from other biodegradable polymers, PEO microparticles generated utilizing electro spraying approach enhanced the porosity and cellular penetration of scaffolds.111
Poly-β-Hydroxybutyrate (PHB)
Poly-β-hydroxybutyrate (PHB) is a linear biodegradable homopolymer of -hydroxybutyric acid that is found in many bacteria as an energy storage molecule.112
PPF (Poly (Propylene) Fumarate)
PPF (poly (propylene) fumarate) is a linear polyester with fumaric acid as the repeating unit. It is biodegradable, cross-linkable, and powerful.113 Cross-linking of fumarate double bonds results in creating a polymeric network.114 Because of its high mechanical strength, it is predominantly employed in orthopedic surgery.115 It is also employed with other polymers116,117 to improve their hydrophobicity. To study bone formation, pre-treated PPF scaffolds were placed into the craniums of rabbits with cranial abnormalities.118 The PPF scaffold covered with rh TGF- (PPF-TGF-) performed better regarding bone area%, bone surface area, and gap-filling percentage.61
Polycaprolactone (PCL)
Polycaprolactone (PCL) is a robust aliphatic polymer with good biocompatibility.119 It is a polyester with remarkable elasticity, physical qualities, minimal breakdown levels, non-toxicity, and low price.120 The capacity of PCL as a biologically friendly material has been demonstrated for several biomedical purposes.121 The fundamental drawback of PCL is its weak bioactivity and excessive hydrophobicity, which leads to poorer cell adhesion and restricted tissue regeneration.122 Polylactic acid is mixed with PCL to produce a less hydrophobic structure with increased processability and mechanical behavior.123
Polyglycolic Acid
Sekiya et al used polyglycolic acid to build a nano-fiber with collagen and test it’s in vivo potential to induce neovascularization and granulation histology. The study’s outcomes revealed strong cellular migration with excellent neovascularization.124
Polyvinyl Alcohol (PVA)
Polyvinyl alcohol (PVA) is a commonly utilized polymer employed in numerous biomedical applications thanks to its favorable features such as biodegradability, biocompatibility, and non-toxicity.125 It is frequently used with other polymers to create nano-fibers for wound healing and other tissue engineering applications.126
Poly (Lactic-Co-Glycolic Acid) (PLGA)
PLGA is an artificial, biodegradable, and FDA-approved polymer.127 Because of its degradability, simplicity of modification, superior mechanical properties, potential for continuous drug delivery, and sustained clinical applicability, PLGA has been extensively researched in the realm of nanomaterials for creating pharmaceutical delivery systems.128
Polyethylenimine (PEI)
PEI is an artificial, water-soluble, positively charged, linear, or branching polymer amino group. PEI has been employed in various biomedical applications such as medicine administration, gene transfer, antimicrobials, and multimodal imaging. Due to its amino groups, PEI has been extensively utilized to fabricate various organic/inorganic hybrid nano materials for various biological purposes.129
Poly (Vinylpyrrolidone) (PVP)
PVP is an FDA-approved, artificial, neutral, water-soluble polymer widely used in biologics, personal care products, and nutritional supplements. Because of its advantageous properties, including bioactivity, biodegradability, and environmental friendliness, PVP is frequently employed as a protective coating.130
Non-Degradable Polymers
Non-biodegradable polymers are generally utilized in wound dressings because they are neither destroyed nor dissolved into smaller molecules by biochemical processes.131
Polyurethane (PU)
Polyurethane (PU) is characterized by its high flexibility and capacity to degrade into degradable forms such as poly(ester-urethane) urea.132 It is used in various preparations, such as mixes with olive oil that contain antioxidant properties and a photoprotective mechanism.133 Dextran fibers electro-spun with PU showed intense angiogenic activity and significant anti-inflammatory activity, accelerating dermatological lesion repair.134
Poly Methyl Methacrylate (PMMA)
PMMA is a common biocompatible polymer that is used alone or in composites to produce frameworks for biomedical applications.135
Polystyrene (PS)
Polystyrene (PS) is a crystalline, colorless polymer with excellent water vapor permeability and high electrical resistance.136 In one study, combining PS with chamomile extract and poly (-caprolactone) expedited wound healing by increasing collagen fiber build-up, improving re-epithelialization, and increasing granulation tissue development.137 Porous PS scaffolds were created to allow fibroblast cells to construct a network of indigenous extracellular proteins.138 This enables the establishment of a sub-epithelial 3D microenvironment rich in chemical and mechanical signals, which promotes epithelial cell functionalization.139
Polyethylene Terephthalate (PET)
PET is a robust synthetic fiber that is extensively utilized in biomedical engineering as an implanted biomaterial.140 PET nano-fibers made with honey have shown astonishing outcomes as a functional wound dressing, with enhanced ability of the mats to absorb water, which is essential for wound healing.141
Polyether Sulfone (PES)
PES is a polymer with high porosity and a large surface area.142 The electro-spun nano-fibers generated from PES displayed enhanced adsorption efficiency, which is crucial for optimal wound healing.143
Poly (Di (Ethylene Glycol) Methyl Ether Methacrylate) (PDEGMA)
Nanofibers composed of PDEGMA and poly (l-lactic acid-co-caprolactone (PLLA-CL) have been utilized for controlled drug delivery, specifically for the antimicrobial ciprofloxacin.144 Because they are thermo-sensitive, they facilitate cell dissociation and ciprofloxacin distribution to wound-infected regions when the temperature is reduced. In vivo, testing on mice with excision wounds revealed that the composite scaffold had superior healing properties to commercial gauzes.145
Zwitterionic Polymers
Zwitterionic Polymers gained so much interest thanks to their remarkable features. Despite their excellent biocompatibility and stimulus reactivity, zwitterionic hydrogels need mechanical properties to be suitable tissue-promoting material in direct blood contact. This issue was overcome in a 2020 study by adding Electro spun fiber scaffold to zwitterionic hydrogels for biocompatibility and mechanical strength, allowing long-term blood contact devices to be developed.146 Over the past ten years, the combination of zwitterionic polymers’ increased hemocompatibility, outstanding non-fouling characteristics, charge-switching qualities, and resistance to protein adsorption has made them appealing in biomedical applications, particularly in tissue engineering.146 Other zwitterion therapeutic applications include implantable medical devices, tissue repair bandages and tissue frameworks, drug delivery vehicles, and biosensors.147
Hybrid Scaffolds
Hybrid scaffolds are one of the most exciting topics of study due to improved characteristics that are more acceptable for diverse tissue engineering applications resulting from combining numerous materials.15
Different Polymeric Formulations
Different formulations of polymeric biomaterials are currently employed in different biomedical applications, as shown in Figure 3.
 |
Figure 3 Different polymeric formulations. |
Micro Needles
MNs are collections of tiny needles up to 1 mm in length that enable medicinal materials to pass through without triggering tissue injury or patient pain.148 Cells are among the therapeutic materials given through MN-based medication carriers,149 tiny substances,150 and macromolecules.151–153 MNs might be hard, disintegrating, porous, covered, or hydrogel-forming.154 Although no MN-based medicines are commercially available, various clinical studies using MN-based pharmaceutical delivery methods have already been accomplished.155 Moreover, MN-based drug delivery systems are feasible for meeting the demands of vulnerable groups receiving biological therapy, including older people and children.156 Due to favorable characteristics, including improved bioactivity, better drug loading potential, a divalent affinity for their specialized biochemical implementations, versatility, wettability, and low cost of production, polymer-based MN materials are the most notable topic for researchers in drug delivery systems.157
Microrobots
Micro robots have shown enormous promise for doing micro-scale activities such as medicine delivery, cell handling, micro construction, and bio detection 158–160 using manual control. Microrobots might be employed for minimally invasive procedures as well.161 A micro-robot may perform surgical procedures such as vein or channel opening, electrocautery, hyperthermia treatments, diagnostics, electrotherapy, chopping, drilling, or biomaterial elimination.162
Nano Fibers
Nanofibers have sizes ranging from 1 to 1000 nm and give advantages, including a large surface area, permeability that provides effective mechanical action, pore connectivity, and flow properties.163,164 Nanofibers are used in medical services such as inserts for longer drug release, pharmaceutical items, healing dressings, and tissue engineering scaffolding.165 Table 4 shows the characteristics of the various production processes of nanofibers.
 |
Table 4 Features of Nanofiber Manufacturing Strategies |
Micelles
They are amphipathic particles with a hydrophobic tail that faces the core and an external hydrophilic spike that links to the fluids. An amphoteric chemical can also form an inverted micelle in a neutral medium, with the spike towards the core and the tail pointing outward. Two copolymers mix with chemicals in some solvents to form polymeric micelles. The solvent dissolves one copolymer but not the others. The inner component comprises insoluble copolymers, whereas the outside part comprises soluble copolymers, from which the micellar assembly is made. This polymeric micelle is helpful in a variety of applications.166
Polymersome
Polymersomes are synthetic vesicles with an aqueous cavity formed by amphiphilic copolymer self-assembly, as shown in Figure 4.167 Furthermore, vesicular membrane improvement can aid in the adaptation of polymersomes for several purposes, such as drug delivery systems168 or artificial organelles.169 Polymersomes are excellent antibiotic delivery vehicles for drugs that do not ordinarily enter host cells.170
 |
Figure 4 Polymersome. |
Dendrimers
D.A.Tomalia coined the name Dendrimer from two Greek words: dendrons (trees) denotes their morphology, and meros (parts) indicate their chemical structure, which is made up of continuous monomers, as represented in Figure 5. Even though dendrimers are not polymerized, they are classified as polymers due to their repetitive pattern.171 Dendrimers can function as biomimetic materials because of their globular design with less than 10nm dimensions. This results in constant dimensions and morphologies that can be changed to develop new dendrimer populations. When employed as scaffolding, their low polydispersity ensures polymeric agent cellular absorption repeatability.172 Dendrimers can be utilized for several functions, including target-specific medication delivery, neovascularization, and gene delivery.173–175
 |
Figure 5 Structure of Dendrimer. |
Nano Capsules
Polymeric nanocapsules have recently received much attention as a possible medication delivery mechanism. They are nanoparticles with a distinct architecture comprising a liquid/solid core surrounded by a polymeric shell, as represented in Figure 6. They can increase payload bioavailability and provide prolonged and localized distribution as medication delivery devices. Similarly, they can substantially decrease the adverse effects of payload and tissue conditions. By encapsulating the medicine in them, it is possible to protect the drug against breakdown or disintegration induced by the biological microenvironment. Furthermore, it can help lessen drugs’ adverse effects on healthy tissue.176
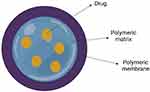 |
Figure 6 Structure of drug loaded nano capsule. |
Aerogels
Aerogels are polymeric materials with high porosity, large surface area, a large pore volume, and varied chemical structures. They are frequently used in aircraft, chemical engineering, building, electronics, and bioengineering.177 In the past few years, there has been a significant increase in the production of innovative aerogels with unique physicochemical features and functionalities. However, native aerogels with a single element typically suffer from substantial issues like poor structural rigidity and a shortage of functionalities. Building hybrid aerogels is one solution to the challenges. The initial aerogel was produced by Kistler in 1931, and he characterized aerogels as “gels where the liquid has been substituted by air, with the modest shrinking of the solid network”. There is, nevertheless, no commonly recognized description of porous materials. The most widely recognized report is that “aerogels are materials in which the usual pore patterns and connections are amazingly preserved when the liquid of a gel is displaced by air”, as stated by Hüsing and Schubert.178
Nano Gels
Nano gels are created by cross-linkers with hydrophilic or amphiphilic groups, resulting in a three-dimensional architecture with particle sizes ranging from 1–200 nm, shared by hydrogels and nanoparticles.179 Nano gels are now recognized as a new class of sophisticated delivery vehicles. This unique capacity also allows Nano gels can absorb bioactive compounds into their network, reducing bioactive component degradation due to external factors.180 Furthermore, nano gels have smaller particle sizes and a larger specific surface area, allowing for greater bio-coupling. Nano gels have the potential to significantly increase the beneficial in vivo content of bioactive compounds, making clinical or daily usage of a wide range of bioactive substances more convenient.181 Moreover, it is reported that addition of nanogel reduce the polymerization shrinkage.182
Photopolymerizable Polymers
Photopolymerizable and biodegradable polymers are increasingly used in dental applications, as shown in Figure 7. Photo-triggered polymerization technique has enabled the development of injectable photopolymerizable biomaterials for dentistry and other purposes.183 Consequently, the procedure leads to the formation of cells and a conductive microenvironment for development, which aids the manufacture of scaffold materials and, thus, the formation of complex systems. The ingredients produced by this technique vary from totally synthetic goods like polyethylene glycol to those formed entirely of natural polymers like hyaluronic acid.184 Photopolymerizable biomaterials are utilized to create injectable biomaterials that may be utilized to create multifunctional scaffolding and distribute growth factors, cells, chemicals and pharmaceuticals.
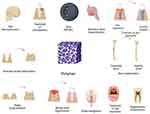 |
Figure 7 Polymeric biomaterials in oral, dental, and maxillofacial reconstruction. |
Hydrogels
Hydrogels are three-dimensional frameworks of different polymers, such as chitosan, alginate, casein, and others, with increased wettability and excellent permeability for several molecules. They have features that are pretty useful for a variety of purposes. Furthermore, the hydrogel architecture and characteristics may be dramatically altered by a reversible sol-gel phase transformation induced by temperature variations or the existence of certain chemicals reacting with the polymer network and causing their collapse or swelling.185,186
Membranes
Barrier membranes are utilised in dentistry to enhance the chances of successful periodontal tissue regeneration, particularly in the bifurcation area and during implant therapy.187 The treatment of periodontal and peri-implant abnormalities has been made possible by the development of a number of resorbable membranes that have proven clinically successful. The resorbable nature of these membranes eliminates the need for additional surgery. Patients favour them over non-resorbable membranes as a result. Depending on the epithelial exclusion concept, commercially accessible resorbable membranes have been employed over the past ten years to address periodontal and peri-implant abnormalities by GTR.188
Films
Thanks to their simple manufacturing process, polymer-based films were originally brought to the field of tissue engineering. In the realm of wound treatment, the polymeric films have gradually proven to be effective for application as occlusive wound dressings. Wound dressing materials made of natural and synthetic polymer-based films have been widely researched.189 Natural polymer-based films have been demonstrated for possessing lower durability and more readily degradable characteristics. Chemical cross-linkers like glutaraldehyde were first utilised to enhance their physical qualities. Researchers have begun to look towards non-cytotoxic cross-linkers since chemical cross-linkers have shown to be toxic to growing cells in recent years.190 A polymeric film was prepared for biological purposes using several cross-linking techniques, including physical cross-linking and ionic cross-linking.191 There have been reports of increased antibacterial and healing characteristics in polymeric films loaded with bioactive molecules.192–194
Micro-Particles
Microspheres and are other names for microparticles. They are tiny, irregularly shaped polymeric particles having a range of sizes. Micrometres, generally between 1 and 1000, are the range in which microparticle sizes vary. Typically, a polymer matrix where a lower quantity of an active ingredient may be immobilised forms polymeric microparticles.195 Natural or synthetic polymers, that might be biodegradable or nondegradable, can be used to create micro-spheres. Emulsification-solvent evaporation, coacervation, spray drying, electrospraying, and phase separation are a few techniques for producing the micro-particles.196 Interleukin-1 receptor antagonist (IL-1ra) was added to dextran/poly (d,l-lactide-co-glycolide) microspheres in an experiment conducted by Lu et al, and the system was then tested for its physicochemical and anti-inflammatory capabilities.197
Methods of Scaffold Fabrication
Common approaches for creating polymeric scaffolds involve solvent casting/salt leaching, phase separation, gel casting, precipitation, and emulsion freeze-drying.198,199 Despite the idea that traditional fabrication methods can integrate pores of the necessitated surface topography by influencing numerous variables, the frameworks generated from these strategies can be made from a single polymer. They could result in inaccurate and unexpected amorphous morphological features.200 Furthermore, these procedures need an organic solvent purification phase, which is time-consuming and difficult for quick adoption.
Electro Spinning
The electrospinning technology was utilized to develop 3D nanofibrous scaffolds. This technology is essential to build a supporting scaffold for cell proliferation by mimicking the precise nanoarchitecture of an extracellular matrix. Electrospun scaffolds have a controlled fiber diameter and are ideal for constructions constructed in sheets and layers.201 Despite the small number of in vivo uses, multiple research has demonstrated the bioactivity of electro-spun scaffolds in regenerative endodontics, demonstrating the diversity of scaffold synthesis for pulpal regeneration.202,203
Supercritical Fluid-Gassing
Maspero et al proposed a unique approach for rapidly fabricating a net-shaped porous scaffold.200 This method, which comprised the rapid centralization of PLGA particles in a mould using sub-critical CO2, enabled the rapid manufacture of a precise porous copy of a tooth root without chemicals. In this method, a mould was created using sterile polyvinylsiloxane, recreating the meticulous architecture of the tooth by inserting the dental root into the polyvinylsiloxane polymer. After the imprint had been set, the root was drained, and the mould was filled with sterile PLGA particles ranging in size from 700 to 1400 m, resulting in an indiscernible structure. Open porous scaffolds with the required form were made using the demonstrated moulding procedure.200 Tai et al found limitations and improvement information.204 They revealed that PLGA structures are brittle and that their pore size decreases as glycolic acid concentration increases. A nonporous layer may emerge when the depressurization phase is finished too quickly. They pushed for a lesser speed of pressure loss to develop more uniform and broader pores. This technique is slower; it may take an hour or more instead of a few minutes, but it may provide better outcomes.
Three-Dimensional Bio Printing
The most recent advancement in tissue engineering methodologies is the incremental pouring of a cell-containing hydrogel using inkjet equipment and CAD/CAM technology.205 The exact placement of various cells in 3D printing is useful, like the implantation of odontoblasts in the scaffold’s perimeter and angiogenic fibroblasts in the core, which will preserve a fundamental matrix of blood vessels and nerves in the substituted pulp tissue.206 Bioprinting techniques can result in readily customizable cell orientation inside a scaffold, improving cell adhesion and regeneration. It can also generate functioning small-diameter blood arteries local to the pulp chamber.207 Nevertheless, in vivo, models continue to have constraints.
Self-Assembling
Moreover, innovative regenerative approaches were developed using a tissue regeneration technique that utilized a hydrogel scaffold implanted with SHED and DPSCs in addition to peptide-amphiphile (PA). Cell-matrix relationships can then be carried out by including the cell adhesion sequence, RGD, and an enzyme-cleavable region. SHED and DPSCs implanted in PA hydrogels were grown for four weeks under varied osteogenic induction settings. With the hydrogel scaffolds, these cells differentiated and proliferated typically. This technology might also be used to create 3D PAs self-assembly structures of Nanofibers and tissues. Moreover, due to the hydrogels’ physicochemical qualities, they may be inserted into microscopic and randomized faults. The suggested approach is ideal for creating soft and hard mineralized dental/pulp tissue regeneration frameworks.208
Clinical Applications
Polymeric biomaterials are extensively utilized in oral, dental, and maxillofacial reconstruction,209 as depicted in Figure 7.
Alveolar Ridge Preservation
The goal of alveolar ridge maintenance following tooth extraction is to prevent or eliminate resorptive bone remodeling and to optimize tissue distribution before fitting the final prosthetic replacement; Table 5 represents the application of biopolymers in alveolar ridge preservation.210–213 Socket preservation treatments employ biomaterials and barrier materials to refill the socket. Salamanca et al found that hydroxyapatite/b-TCP ceramic combined with homogeneous collagen solution had slightly greater effectiveness in osteogenesis and similar osteoconductivity to bovine-derived bone (Bio-Oss®) with collagen membrane in a canine investigation.214
 |
Table 5 Implementation of Polymeric Materials in Ridge Preservation |
Vertical and Horizontal Ridge Augmentation
Guided bone regeneration (GBR) utilizing a membrane is the most often used surgical approach to regenerate atrophic residual ridges. GBR depends on three primary criteria: (1) cell occlusivity, (2) wound integrity, and (3) space formation and preservation. Resorbable polymeric membranes are preferred because they can regenerate noncritical-size defects continuously, promote the healing process and economical price, and reduce surgical discomfort and consequences.
Membrane permeability impacts vasculature and bone progenitor cell growth over competitive soft tissue cells.215 Composite grafts of bone allografts or xenografts with a resorbable membrane provided clinical outcomes comparable to autologous bone transplants.216 These findings support that composite grafts might be a viable alternative to autogenous bone harvesting, overcoming key obstacles such as donor site complications. Differences in surgical procedures and operator skills should be addressed when estimating clinical outcomes of various combinations.
Maxillary Sinus Augmentation
Maxillary sinus augmentation can be accomplished using the lateral window method or the transrectal approach to create a gap between the sinus floor and the Schneiderian membrane to fill with biomaterials that encourage the creation of new osseous tissue.217 Several bone fillers have demonstrated effectiveness in increasing vertical bone height for future implant implantation. Table 6 depicts the implementation of polymers in sinus lifting.218–220 A comprehensive review discovered no statistically significant variations in implant longevity between bone auto grafts and bone replacement materials.221
 |
Table 6 Various Polymeric Scaffolds in Sinus Lifting |
Temporomandibular Joint Reconstruction
The TMJ’s two interacting structural constituents are the temporal bone and the mandibular condyle. TMJ deterioration can damage the disc, the surrounding fibrous tissue, the proliferative and hypertrophic fibrocartilage layers, or the condylar bone. Regenerative TMJ therapies have been offered to enhance clinical outcomes, long-term ossification, and adherence prevention. To reconstruct the TMJ, biphasic cartilage and bone engineering employing ex-vivo cell seeding and bioactive chemicals on an acellular scaffold is being employed. Among the several multipotent cell types studied for condylar cartilage regeneration, DPSCs have been extensively researched for their high reliability and multi-lineage development into chondrogenic cells, osteoblasts, and other important cell forms.222,223
Natural materials have been widely employed for TMJ disc regeneration. They have mechanical characteristics comparable to those of the natural disc, like cell adherence and penetration, cell growth, and proteoglycan accumulation.224
Synthetic polymers have been presented to produce synthetic ECM for TMJ fibrocartilage regeneration that outperforms natural materials in terms of mechanical strength and biodegradability.225 Poly-L-lactic-glycolic acid is one example the FDA has authorized for clinical use (PLGA). PLGA promotes mesenchymal stem cell colonization and proliferation while interacting with chondrocytes and other TMJ discal cells.226 PCL fibers are a form of pro-regenerative biomimetic nanofiber that the FDA has cleared for clinical use.227
Periodontal Tissue Regeneration, and Engineering
Periodontal regeneration attempts to heal the alveolar bone, periodontal ligament, and cementum that have been damaged by periodontitis. Scaling and root planning, curettage, and open flap cleaning are current therapy techniques with minimal success. Periodontal regeneration using GTR permits bone cells, fibroblasts, and PDL cells to be chosen to fill the periodontal defect. Melcher was the first to utilize a barrier membrane to control the natural wound-healing process in 1976. Bioactive substances and nanoparticles recently incorporated into polymeric scaffolds in order to create a smart scaffold that respond to internal signals.228 The use of regenerating biomaterials in GTR is widely established.229 A systematic review of GTR results with a collagen membrane revealed that pocket depth reduction is predictable with or without a bone replacement.230 Scaffolding materials, in addition to membranes, are commonly used in fault areas. On the other hand, polymeric scaffold materials are often osteoconductive, maintaining room for cells to migrate into the defect location; Figure 8 represents optimal characteristics for periodontal engineering scaffolds.
 |
Figure 8 Optimal characteristics for periodontal engineering scaffolds. |
Different polymer matrices and scaffolds have been employed for periodontal regeneration of intraosseous abnormalities since the 1980s, as shown in Table 7.231–246 Likewise, 3D-printed scaffolds have recently been designed to improve on current supporting matrices. In contrast to traditional grafts’ brittleness, poorly processed porosities, and generic shapes, 3D scaffolds may be adapted to the unique demands of patients. To regenerate each periodontium tissue type ideally, compartmentalization, internal topographies, and pore diameters and angulations can be precisely engineered.13
 |
Table 7 Different Polymeric Biomaterials in Periodontal Tissue Regeneration and Engineering |
A case study proved the effectiveness of using a 3D-printed PCL scaffold for periodontal regeneration. The graft failed because of the slow breakdown rate of PCL in comparison to surrounding tissue, resulting in graft exposure.34 Recent advancements in additive manufacturing technology enable the creation of nanoscale scaffolds with adjustable parameters such as fiber diameter, porosity, shape, and surface properties.
Growth factors, proteins, and medications can also be added to these polymeric matrices to regulate cellular responses and adjust the local inflammatory milieu to enhance periodontal tissue engineering, as seen in Figure 9.
 |
Figure 9 Requirements of tissue engineering. |
Implant Coating
Dental implants are often used to replace missing teeth, restore function, and enhance patients’ life satisfaction.247,248 The long-term durability of dental implants may malfunction because of biological peri-implantitis (20%) caused by microbial infections and mechanical issues, including stress shielding, which may cause osteopenia and clenching-bruxism.249 Several investigators156,234,250–253 have assessed the effectiveness of these biopolymer-based hydrogels as coating materials for enhancing the characteristics of dental implants. In addition to surface changes, the coating of dental implants with various materials and biomolecules to accomplish particular advantages has been studied in the past few years.254,255
Ansari et al used the in-situ sol-gel approach to create polycaprolactone (PCL) and PCL/fluoride substituted HA (PCL-FHA) composites as a coating material for alkali-treated Ti6Al4V platform to increase protection against corrosion and in vitro biocompatibility.250 Joo et al251 synthesized and employed a hydrogel generated from hyaluronic acid and gelatin for the surface modification of polydimethylsiloxane (PDMS), frequently employed as an implant-supported covering material. In another study, Yang et al used carboxymethyl cellulose-dopamine (CMC-DA) hydrogel as a stimulator to cover the surface of magnesium alloys with HA.
The surface-modified alloys’ corrosion resistance and biocompatibility were then assessed. The outcomes showed that corrosion resistance of CMC-DA/HA coated AZ31 alloy (AZ31/CMC-DA/HA) enhanced 18.9-fold compared to naked AZ31 alloy. Furthermore, biocompatibility experiments revealed that AZ31/CMC-DA/HA induced a 21% boost in the multiplication of MC3T3-E1 cells following 5-day incubation compared to the raw AZ31 alloy, showing the coating’s superior cytocompatibility.153 Table 8 shows polymers as coating materials around implants210,250,251,256–260.
 |
Table 8 Polymeric Coating Materials Around Implants |
Oral Mucositis
Oral mucositis (OM) is a classic side event in oncological patients. It results from pathogenesis is a direct and indirect effect of tumor therapeutics that suppress physiologic cellular pathways and results in epithelial apoptosis.261 In vivo testing revealed that benzydamine hydrochloride-loaded lyophilized mucoadhesive displayed consistent structural, mechanical, and biological properties. It might be effective carrier for drug distribution for OM262 —carboxymethyl cellulose is functionalized with N-(2-aminoethyl) maleimide.
Through the interaction of the maleimide moiety and mucin, the polymer demonstrated outstanding mucoadhesive potential compared to carboxymethyl cellulose. The functionalized polymer may also regulate the release of benzydamine using Higuchi’s release model, proving it to be a reliable solution in bio-adhesive drug delivery.263
Liquid oral gel based on polyacrylate silver salt/polyvinylpyrrolidone Clinical OM and oral function were examined weekly after four weeks, until 5e10 days after radiation was completed. Standard Terminology Criteria for Adverse Events (CTCAE) v3.0 was used to evaluate the data of OM grade.264
Regenerative Endodontics
In regenerative endodontics, polymeric scaffolds have been utilized to produce an appropriate microenvironment for dentin-pulp system regeneration. Various scaffolds have shown clinical effectiveness in multiple applications, as shown in Table 9.265–269
 |
Table 9 Different Polymeric Scaffolds Utilized in Regenerative Endodontics |
PRF scaffolds revealed apical closure, elimination of apical radiolucency, continued root extension, and dentinal wall thickening in permanent adolescent teeth with necrotic pulps.268
Bio-Gide collagen membranes (Geistlich, Wolhusen, Switzerland) have been found to enhance dentinal wall formation in the middle third of the root, strengthening the root and preventing cervical root fractures.270
Whole Tooth Regeneration
Whether treated or not, dental disorders such as tooth decay or periodontitis ultimately lead to tooth loss. Bioengineered edentulism management is a viable alternative to the advancement of tissue engineering.
Yang et al271 showed the reliability of incorporating fibrin glue with PRF as a matrix cultured with dental bud cells for dental regeneration purposes. PRF contains high amounts of tissue growth factor-β (TGF-β) and platelets-derived growth factor (PDGF), significantly affecting angiogenesis and complex tissue regeneration. Additionally, dental bud cells seeded on PRF/PRP frameworks were observed to regenerate an entire tooth structure.
Oral Drug Delivery Systems
Administering medicinal substances into the oral cavity is an established way of treating local inflammation, discomfort, and illnesses of the mucosa and dentition. Polymeric drug carriers have astonishing characteristics that make them widely utilized as drug delivery carriers, as shown in Figure 10.
 |
Figure 10 Benefits of polymeric biomaterials as drug delivery systems. |
An injectable chitosan sponge could be utilized for this objective because it promotes biomineralization in major-sized osseous deformities and is less expensive than growth factors such as BMPs.272,273 Bacterial cellulose is another substance that is very good for antibiotic dissemination.274 Nanoparticles or comparable technical solutions should be noted among unique formulations of different films, fibers, biodegradable matrices, local gels, or microspheres.276 Semi-solid gel preparations with antimicrobial, anti-inflammatory, and aseptic qualities commonly apply active compounds.
For the management of oral illnesses, several formulations have been developed, as represented in Table 10.275,277–281 In therapeutic strategies for oral cancer, they are frequently distinguished by bio-adhesive characteristics or response to stimuli.
 |
Table 10 Advantages of Polymeric Drug Carriers |
They could also be employed with photodynamic treatment to administer photosensitizers.276 The ability to selectively destroy neoplastic cells while reducing cytotoxicity towards non-cancerous tissues and bactericidal action against bacteria building oral biofilm are further advantages of nanocarriers in oral cancer therapy.282
Bone Tissue Engineering
Despite the endogenous reparative capacity of bone, polymeric scaffolds have a critical role in enhancing the recovery process, shortening the healing period, and thus limiting postoperative complications and maximizing treatment success, as shown in Table 11.283–291 It is critical to achieving the goals of regenerative medicine by generating specific releases of biomolecules at the specific receptors. This was associated with the balance of biological signaling pathways in tissues, particularly bone.292
 |
Table 11 Numerous Polymeric Materials in Bone Tissue Engineering |
Polyurethane foam/nano-hydroxyapatite composite was fabricated and potentially functioned as an extracellular environment in creating osteoblasts tissues.293 PLA membrane was adjusted by incorporating HAp NWs to exert barrier/-osteo induction dual roles in a rat mandibular deformity.294
Conclusions
It is necessary to acknowledge that this review can hardly avoid bias and errors to some extent, as there is no uniform evaluation of the scientific validity of the design, methodology, and results of the original literature such as a systematic review. However, it is still informative to learn about the application and research progress of hydrogels in maxillofacial and oral drug delivery. Compared with other materials such as nanoparticles, nanofibers, and thin films, many hydrogels have biocompatibility and unique stimulusresponsive properties that make them suitable as carriers or platforms for transporting drugs, cells, and others to target locations, which have unique advantages in local therapies and are therefore well suited for topical applications in the oral environment. This paper describes the application and research progress of hydrogels in maxillofacial and oral drug delivery. Different types of hydrogels have a wide range of applications in oral soft and hard tissue regeneration, antibacterial, and local drug targeting delivery due to their injectability, temperature sensitivity, pH sensitivity, biodegradability, and other properties. Many hydrogels do not have the function of repairing tissue defects themselves, but can be used as carriers for different drugs, growth factors, and even stem cells to achieve higher concentrations maintained over a long period of time. Some hydrogel systems are able to respond to stimuli such as temperature, pH, and light irradiation, which can modulate the drug release from the hydrogel on demand. In the future, how to obtain hydrogel systems with greater biocompatibility through smaller cost and simpler synthesis methods to achieve superior sustained local drug delivery efficacy are still important issues for researchers to focus on. There are many hydrogel drug delivery systems made with different materials and synthesis methods for complex oral and maxillofacial environments, but the high costs, complex synthesis steps, and toxic biodegradation by-products may be the key challenges affecting their further clinical applications.295,296 Meanwhile, in vivo studies and animal studies are relatively scarce, and the exploration of relevant hydrogels for clinical applications is even rarer. The stability of topical drug delivery and the biocompatibility of hydrogels need to be further investigated, and their use in the clinic requires more rigorous testing. It is expected that more clinical studies will be conducted in the future to verify the therapeutic effects of hydrogels as drug release platforms in different oral diseases, especially in the treatment of periodontal diseases, which currently have a relatively large number of clinical studies.
Abbreviations
AgNPs, Silver nanoparticles; AMP, Antimicrobial peptide; ATV, Atorvastatin; BBR, Berberine; BC, Bacterial cellulose; BMP, Bone morphogenic protein; CA, Cellulose acetate; CAD/CAM, Computer-Aided Design And Manufacturing; CEC, N-carboxyethyl chitosan; CMC-DA, Carboxymethyl cellulose-dopamine; COAs, Clear overlay appliances; CoS, Chondroitin sulphate; CS, Chitosan; CTCAE, Common Terminology Criteria for Adverse Events; DFO, deferoxamine; DMOG, Dimethyloxalylglycine; DMSO, Dimethyl sulfoxide; DPSCs, Dental pulp stem cells; ECM, Extracellular matrix; EPO, Erythropoietin; FDA, Food and Drug Administration; GA, Gelatin; GBD, Global Burden of Diseases; GBR, Guided bone regeneration; GP, Glycerophosphate; G-PDA, Guanidine appended polydiacetylene; GTR, Guided tissue regeneration; HA, Hyaluronic acid; HA, hydroxyapatite; HBMSCs, human bone marrow stem cells; LIG, Lignin; MC3T3-E1 cells, Osteoblast precursor cell line derived from Mus musculus (mouse) calvaria; MNs, micro needles; nSi, nano silicate; OM, Oral mucositis; PA, peptide-amphiphile; PAA, Polyacrylic acid; PCL, Polycaprolactone; PCL-FHA, fluoride substituted HA; PDA, Polydopamine; PDEGMA, Poly di ethylene glycol methyl ether methacrylate; PDGF, Platelets derived growth factor; PDL, Periodontal ligament; PEEK, Polyether ether ketone; PEG, Poly ethylene glycol; PEI, Polyethylenimine; PEO, Polyethylene oxide; PES, Polyether sulfone; PET, Polyethylene terephthalate; PETG, Poly (ethylene terephthalate)-glycol; PHB, Poly-β-hydroxybutyrate; PLGA, Poly (lactic-co-glycolic acid); PLLA-CL, Poly l-lactic acid-co-caprolactone; PMMA, Poly methyl methacrylate; PPF, Poly propylene fumarate; PRF, Platelet rich fibrin; PRP, Platelet rich plasma; PS, Polystyrene; PU, Polyurethane; PVA, Poly vinyl alcohol; PVP, Poly vinyl pyrrolidone; RGD, Arginyl-glycyl-aspartic acid; rh TGF, Recombinant Human Transforming Growth Factor; SHED, Stem cells from human exfoliated deciduous teeth; SMV, Simvastatin; SNs, Silicate nanoparticles; TGF-β, tissue growth factor-β; TMJ, Temporomandibular Joint Reconstruction; Van, Vancomycin; β -TCP, β-tricalcium phosphate.
Acknowledgment
The authors thank the resources provided by the project 6PFE of the University of Life Sciences “King Mihai I” from Timisoara and the Research Institute for Biosecurity and Bioengineering from Timisoara.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This paper is published from the project 6PFE of the University of Life Sciences “King Mihai I” from Timisoara and Research Institute for Biosecurity and Bioengineering from Timisoara.
Disclosure
The authors declare no conflicts of interest.
References
1. James SL, Abate D, Abate KH, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1789–1858.
2. Latimer JM, Maekawa S, Yao Y, Wu DT, Chen M, Giannobile WV. Regenerative medicine technologies to treat dental, oral, and craniofacial defects. Front Bioeng Biotechnol. 2021;2021:637.
3. Hamdy TM. Polymers and ceramics biomaterials in orthopedics and dentistry: a review article. Egypt J Chem. 2018;61(4):723–730.
4. Hamouda IM. Current perspectives of nanoparticles in medical and dental biomaterials. J Biomed Res. 2012;26(3):143–151. doi:10.7555/JBR.26.20120027
5. Liu S, Yu JM, Gan YC, et al. Biomimetic natural biomaterials for tissue engineering and regenerative medicine: new biosynthesis methods, recent advances, and emerging applications. Milit MedRes. 2023;10(1):16. doi:10.1186/s40779-023-00448-w
6. Shahiwala A. Formulation approaches in enhancement of patient compliance to oral drug therapy. Expert Opin Drug Deliv. 2011;8(11):1521–1529. doi:10.1517/17425247.2011.628311
7. X-F L, Zhao W-L, Zhu X-D, Zhang X-D. Fabrication methods of bioactive materials for bone regeneration. Bioact Mater Bone Regen. 2020;2020:3.
8. Sun Y, Chen L-G, Fan X-M, Pang JN. Ultrasound responsive smart implantable hydrogels for targeted delivery of drugs: reviewing current practices. Int J Nanomedicine. 2022;5001–5026. doi:10.2147/IJN.S374247
9. Jin S-E, Jin H-E, Hong S. Targeted delivery system of nanobiomaterials in anticancer therapy: from cells to clinics. Biomed Res Int. 2014;2014:1–23. doi:10.1155/2014/814208
10. Qu H, Fu H, Han Z, Sun Y. Biomaterials for bone tissue engineering scaffolds: a review. RSC Adv. 2019;9(45):26252–26262. doi:10.1039/C9RA05214C
11. Zhang X, Chen X, Hong H, Hu R, Liu J, Liu C. Decellularized extracellular matrix scaffolds: recent trends and emerging strategies in tissue engineering. Bioact Mater. 2022;10:15–31. doi:10.1016/j.bioactmat.2021.09.014
12. Gao X, Han S, Zhang R, Liu G, Wu J. Progress in electrospun composite nanofibers: composition, performance and applications for tissue engineering. J Mater Chem B. 2019;7(45):7075–7089. doi:10.1039/C9TB01730E
13. Abdel Nasser Atia G, Shalaby HK, Zehravi M, et al. Locally applied repositioned hormones for oral bone and periodontal tissue engineering: a narrative review. Polymers. 2022;14(14):2964. doi:10.3390/polym14142964
14. Gunatillake P, Mayadunne R, Adhikari R. Recent developments in biodegradable synthetic polymers. Biotechnol Annu Rev. 2006;12:301–347.
15. Khorshidi S, Solouk A, Mirzadeh H, et al. A review of key challenges of electrospun scaffolds for tissue‐engineering applications. J Tissue Eng Regen Med. 2016;10(9):715–738. doi:10.1002/term.1978
16. Negut I, Dorcioman G, Grumezescu V. Scaffolds for wound healing applications. Polymers. 2020;12(9):2010. doi:10.3390/polym12092010
17. Yi S, Ding F, Gong L, Gu X. Extracellular matrix scaffolds for tissue engineering and regenerative medicine. Curr Stem Cell Res Ther. 2017;12(3):233–246. doi:10.2174/1574888X11666160905092513
18. Zhang H, Cheng J, Ao Q. Preparation of alginate-based biomaterials and their applications in biomedicine. Mar Drugs. 2021;19(5):264. doi:10.3390/md19050264
19. Seddiqi H, Oliaei E, Honarkar H, et al. Cellulose and its derivatives: towards biomedical applications. Cellulose. 2021;28(4):1893–1931.
20. El-Sherbiny IM, El-Baz NM. A review on bionanocomposites based on chitosan and its derivatives for biomedical applications. Eco Fndly Polym Nanocom. 2015;2015:173–208.
21. Sofi HS, Ashraf R, Beigh MA, Sheikh FA. Scaffolds Fabricated From Natural Polymers/Composites By Electrospinning For Bone Tissue Regeneration. In: Cutting-Edge Enabling Technologies for Regenerative Medicine. Springer; 2018:49–78.
22. DeFrates KG, Moore R, Borgesi J, et al. Protein-based fiber materials in medicine: a review. Nanomaterials. 2018;8(7):457. doi:10.3390/nano8070457
23. Wu M, Wu P, Xiao L, et al. Biomimetic mineralization of novel hydroxyethyl cellulose/soy protein isolate scaffolds promote bone regeneration in vitro and in vivo. Int J Biol Macromol. 2020;162:1627–1641. doi:10.1016/j.ijbiomac.2020.08.029
24. Dutta PK. Chitin and Chitosan for Regenerative Medicine. Springer; 2016.
25. Russo L, Cipolla L. Glycomics: new challenges and opportunities in regenerative medicine. Chem Eur J. 2016;22(38):13380–13388. doi:10.1002/chem.201602156
26. Tiwari S, Patil R, Bahadur P. Polysaccharide based scaffolds for soft tissue engineering applications. Polymers. 2018;11(1):1. doi:10.3390/polym11010001
27. Sood A, Gupta A, Agrawal G. Recent advances in polysaccharides based biomaterials for drug delivery and tissue engineering applications. Carbohydr Polym Technol Appl. 2021;2:100067.
28. Islam S, Bhuiyan MA, Islam MN. Chitin and chitosan: structure, properties and applications in biomedical engineering. J Polym Environ. 2017;25(3):854–866. doi:10.1007/s10924-016-0865-5
29. Llamas-Arriba MG, Hernández-Alcántara AM, Mohedano ML, et al. Lactic acid bacteria isolated from fermented doughs in Spain produce dextrans and riboflavin. Foods. 2021;10(9):2004. doi:10.3390/foods10092004
30. Maia J, Evangelista MB, Gil H, Ferreira L. Dextran-based materials for biomedical applications. Res Signpost. 2014;37661:31–53.
31. Ramasundaram S, Saravanakumar G, Sobha S, Oh TH. Dextran sulfate nanocarriers: design, strategies and biomedical applications. Int J Mol Sci. 2022;24(1):355. doi:10.3390/ijms24010355
32. Selyanin MA, Boykov PY, Khabarov VN, Polyak F. The history of hyaluronic acid discovery, foundational research and initial use. HA. 2015;1–8.
33. Litwiniuk M, Krejner A, Speyrer MS, Gauto AR, Grzela T. Hyaluronic acid in inflammation and tissue regeneration. Wounds. 2016;28(3):78–88.
34. Longinotti C. The use of hyaluronic acid based dressings to treat burns: a review. Burns Trauma. 2014;2(4):2321–3868. doi:10.4103/2321-3868.142398
35. Bouvard C, Galy-Fauroux I, Grelac F, et al. Low-molecular-weight fucoidan induces endothelial cell migration via the PI3K/AKT pathway and modulates the transcription of genes involved in angiogenesis. Mar Drugs. 2015;13(12):7446–7462. doi:10.3390/md13127075
36. Debele TA, Mekuria SL, Tsai H-C. Polysaccharide based nanogels in the drug delivery system: application as the carrier of pharmaceutical agents. Mater Sci Eng. 2016;68:964–981. doi:10.1016/j.msec.2016.05.121
37. Larrañeta E, Henry M, Irwin NJ, Trotter J, Perminova AA, Donnelly RF. Synthesis and characterization of hyaluronic acid hydrogels crosslinked using a solvent-free process for potential biomedical applications. Carbohydr Polym. 2018;181:1194–1205. doi:10.1016/j.carbpol.2017.12.015
38. Gupta KC, Haider A, Choi Y, Kang IK. Nanofibrous scaffolds in biomedical applications. Biomater Res. 2014;18(1):1–11. doi:10.1186/2055-7124-18-5
39. Bhatia S. Systems for Drug Delivery. Springer; 2016.
40. Wardrop D, Keeling D. The story of the discovery of heparin and warfarin. Br J Haematol. 2008;141(6):757–763. doi:10.1111/j.1365-2141.2008.07119.x
41. Laremore TN, Zhang F, Dordick JS, Liu J, Linhardt RJ. Recent progress and applications in glycosaminoglycan and heparin research. Curr Opin Chem Biol. 2009;13(5–6):633–640. doi:10.1016/j.cbpa.2009.08.017
42. Joung YK, Bae JW, Park KD. Controlled release of heparin-binding growth factors using heparin-containing particulate systems for tissue regeneration. Expert Opin Drug Deliv. 2008;5(11):1173–1184. doi:10.1517/17425240802431811
43. Rezaie AR, Giri H. Anticoagulant and signaling functions of antithrombin. J Thromb Haemost. 2020;18(12):3142–3153. doi:10.1111/jth.15052
44. Suhail M, Rosenholm JM, Minhas MU, et al. Nanogels as drug-delivery systems: a comprehensive overview. Ther Deliv. 2019;10(11):697–717. doi:10.4155/tde-2019-0010
45. Pereira L, Cotas J. Introductory chapter: alginates-A general overview. Alginates Recent Uses Nat Polymer. 2020;2020:1.
46. Sabra W. The Promise and Challenge of Microbial Alginate Production: a Product with Novel Applications. In: Polysaccharides of Microbial Origin: Biomedical Applications. Springer; 2022:79–98.
47. Abdelraouf RM, Bayoumi RE, Hamdy TM. Effect of powder/water ratio variation on viscosity, tear strength and detail reproduction of dental alginate impression material (In vitro and clinical study). Polymers. 2021;13(17):2923. doi:10.3390/polym13172923
48. Ambrosetti R. Pullulan as a release modifier for drug delivery systems: effect of rheological behaviour of its solutions on the processability by spray drying; 2019.
49. Pacheco MS, Barbieri D, da Silva CF, de Moraes MA. A review on orally disintegrating films (ODFs) made from natural polymers such as pullulan, maltodextrin, starch, and others. Int J Biol Macromol. 2021;178:504–513. doi:10.1016/j.ijbiomac.2021.02.180
50. Agrawal S, Budhwani D, Gurjar P, Telange D, Lambole V. Pullulan based derivatives: synthesis, enhanced physicochemical properties, and applications. Drug Deliv. 2022;29(1):3328–3339. doi:10.1080/10717544.2022.2144544
51. Mihaila SM, Popa EG, Reis RL, Marques AP, Gomes ME. Fabrication of endothelial cell-laden carrageenan microfibers for microvascularized bone tissue engineering applications. Biomacromolecules. 2014;15(8):2849–2860. doi:10.1021/bm500036a
52. Ocampo JIG, de Paula MMM, Bassous NJ, Lobo AO, Orozco CPO, Webster TJ. Osteoblast responses to injectable bone substitutes of kappa-carrageenan and nano hydroxyapatite. Acta biomater. 2019;83:425–434. doi:10.1016/j.actbio.2018.10.023
53. Pacheco-Quito E-M, Ruiz-Caro R, Veiga M-D. Carrageenan: drug delivery systems and other biomedical applications. Mar Drugs. 2020;18(11):583. doi:10.3390/md18110583
54. Garnjanagoonchorn W, Wongekalak L, Engkagul A. Determination of chondroitin sulfate from different sources of cartilage. Chem Eng Process. 2007;46(5):465–471. doi:10.1016/j.cep.2006.05.019
55. Manivong S, Cullier A, Audigie F, et al. New trends for osteoarthritis: biomaterials, models and modeling. Drug Discov Today. 2023;28:103488. doi:10.1016/j.drudis.2023.103488
56. Pal D, Saha S. Chondroitin: a natural biomarker with immense biomedical applications. RSC Adv. 2019;9(48):28061–28077. doi:10.1039/C9RA05546K
57. Li T, Chen C, Brozena AH, et al. Developing fibrillated cellulose as a sustainable technological material. Nature. 2021;590(7844):47–56. doi:10.1038/s41586-020-03167-7
58. Akram M, Taha I, Ghobashy MM. Low temperature pyrolysis of carboxymethylcellulose. Cellulose. 2016;23:1713–1724. doi:10.1007/s10570-016-0950-x
59. Zaki DY, Safwat EM, Nagi SM, et al. A novel dental re-mineralizing blend of hydroxyethyl-cellulose and cellulose nanofibers oral film loaded with nepheline apatite glass: preparation, characterization and in vitro evaluation of re-mineralizing effect. Carbohydr Polym Technol Appl. 2021;2:100035. doi:10.1016/j.carpta.2021.100035
60. Upadhyay RK. Role of biological scaffolds, hydro gels and stem cells in tissue regeneration therapy. Adv Tissue Eng Regen Med Open Access. 2017;2(1):121.
61. Suamte L, Tirkey A, Babu PJ. Design of 3D smart scaffolds using natural, synthetic and hybrid derived polymers for skin regenerative applications. Smart Mater Med. 2022;2022:1.
62. Bhatt P, Kumar V, Goel R, et al. Structural modifications and strategies for native starch for applications in advanced drug delivery. Biomed Res Int. 2022;2022:1–14. doi:10.1155/2022/2188940
63. Zhang B, Liu N, Hao M, Zhou J, Xie Y, He Z. Plant-derived polysaccharides regulated immune status, gut health and microbiota of broilers: a review. Front Veter Sci. 2022;8:791371. doi:10.3389/fvets.2021.791371
64. Taghavizadeh Yazdi ME, Nazarnezhad S, Mousavi SH, et al. Gum tragacanth (GT): a versatile biocompatible material beyond borders. Molecules. 2021;26(6):1510. doi:10.3390/molecules26061510
65. Silva TH, Moreira-Silva J, Marques ALP, Domingues A, Bayon Y, Reis RL. Marine origin collagens and its potential applications. Mar Drugs. 2014;12(12):5881–5901. doi:10.3390/md12125881
66. Sapna G, Gokul S, Bagri‐Manjrekar K. Matrix metalloproteinases and periodontal diseases. Oral Dis. 2014;20(6):538–550. doi:10.1111/odi.12159
67. Gursoy UK, Könönen E, Huumonen S, et al. Salivary type I collagen degradation end‐products and related matrix metalloproteinases in periodontitis. J Clin Periodontol. 2013;40(1):18–25. doi:10.1111/jcpe.12020
68. McGuire JD, Walker MP, Dusevich V, Wang Y, Gorski JP. Enamel organic matrix: potential structural role in enamel and relationship to residual basement membrane constituents at the dentin enamel junction. Connect Tissue Res. 2014;55(sup1):33–37. doi:10.3109/03008207.2014.923883
69. Tjäderhane L, Carrilho MR, Breschi L, Tay FR, Pashley DH. Dentin basic structure and composition—an overview. Endod Topics. 2009;20(1):3–29. doi:10.1111/j.1601-1546.2012.00269.x
70. Gonçalves PF, Sallum EA, Sallum AW, Casati MZ, De toledo S, Junior FHN. Dental Cementum Reviewed: Development, Structure, Composition, Regeneration and Potential Functions. Piracicaba Dental School-UNICAMP; 2005.
71. Naomi R, Bahari H, Ridzuan PM, Othman F. Natural-based biomaterial for skin wound healing (Gelatin vs. collagen): expert review. Polymers. 2021;13(14):2319. doi:10.3390/polym13142319
72. Thombare N, Jha U, Mishra S, Siddiqui MZ. Guar gum as a promising starting material for diverse applications: a review. Int J Biol Macromol. 2016;88:361–372. doi:10.1016/j.ijbiomac.2016.04.001
73. Stoffels JMJ, Zhao C, Baron W. Fibronectin in tissue regeneration: timely disassembly of the scaffold is necessary to complete the build. Cell Mol Life Sci. 2013;70(22):4243–4253. doi:10.1007/s00018-013-1350-0
74. Chantre CO, Campbell PH, Golecki HM, et al. Production-scale fibronectin nanofibers promote wound closure and tissue repair in a dermal mouse model. Biomaterials. 2018;166:96–108. doi:10.1016/j.biomaterials.2018.03.006
75. Su K, Wang C. Recent advances in the use of gelatin in biomedical research. Biotechnol Lett. 2015;37(11):2139–2145. doi:10.1007/s10529-015-1907-0
76. Chen S, Zhang Q, Nakamoto T, Kawazoe N, Chen G. Gelatin scaffolds with controlled pore structure and mechanical property for cartilage tissue engineering. Tissue Eng Part C Methods. 2016;22(3):189–198. doi:10.1089/ten.tec.2015.0281
77. Ninan N, Muthiah M, Park I-K, Wong TW, Thomas S, Grohens Y. Natural polymer/inorganic material based hybrid scaffolds for skin wound healing. Polymer Rev. 2015;55(3):453–490. doi:10.1080/15583724.2015.1019135
78. Cui X, Li J, Hartanto Y, et al. Advances in extrusion 3D bioprinting: a focus on multicomponent hydrogel‐based bioinks. Adv Healthcare Mater. 2020;9(15):1901648. doi:10.1002/adhm.201901648
79. Philandrianos C, Andrac-Meyer L, Mordon S, et al. Comparison of five dermal substitutes in full-thickness skin wound healing in a porcine model. Burns. 2012;38(6):820–829. doi:10.1016/j.burns.2012.02.008
80. Parry DAD, Squire JM. Fibrous Proteins: Structures and Mechanisms. Springer; 2017.
81. Shanmugasundaram OL, Ahmed KSZ, Sujatha K, et al. Fabrication and characterization of chicken feather keratin/polysaccharides blended polymer coated nonwoven dressing materials for wound healing applications. Mater Sci Eng. 2018;92:26–33. doi:10.1016/j.msec.2018.06.020
82. Yasuda H, Kuroda S, Shichinohe H, Kamei S, Kawamura R, Iwasaki Y. Effect of biodegradable fibrin scaffold on survival, migration, and differentiation of transplanted bone marrow stromal cells after cortical injury in rats. J Neurosurg. 2010;112(2):336–344. doi:10.3171/2009.2.JNS08495
83. Noori A, Ashrafi SJ, Vaez-Ghaemi R, Hatamian-Zaremi A, Webster TJ. A review of fibrin and fibrin composites for bone tissue engineering. Int J Nanomedicine. 2017;12:4937. doi:10.2147/IJN.S124671
84. Khanna A, Oropeza BP, Huang NF. Engineering spatiotemporal control in vascularized tissues. Bioengineering. 2022;9(10):555. doi:10.3390/bioengineering9100555
85. Ziv-Polat O, Skaat H, Shahar A, Margel S. Novel magnetic fibrin hydrogel scaffolds containing thrombin and growth factors conjugated iron oxide nanoparticles for tissue engineering. Int J Nanomedicine. 2012;7:1259. doi:10.2147/IJN.S26533
86. Egorikhina MN, Bronnikova II, Rubtsova YP, et al. Aspects of in vitro biodegradation of hybrid fibrin-collagen scaffolds. Polymers. 2021;13(20):3470. doi:10.3390/polym13203470
87. Wang H-Y, Zhang Y-Q. Effect of regeneration of liquid silk fibroin on its structure and characterization. Soft Matter. 2013;9(1):138–145. doi:10.1039/C2SM26945G
88. Bhattacharjee P, Naskar D, Maiti TK, Bhattacharya D, Kundu SC. Investigating the potential of combined growth factors delivery, from non-mulberry silk fibroin grafted poly (ɛ-caprolactone)/hydroxyapatite nanofibrous scaffold, in bone tissue engineering. Appl Mater Today. 2016;5:52–67. doi:10.1016/j.apmt.2016.09.007
89. Ko E, Lee JS, Kim H, et al. Electrospun silk fibroin nanofibrous scaffolds with two-stage hydroxyapatite functionalization for enhancing the osteogenic differentiation of human adipose-derived mesenchymal stem cells. ACS Appl Mater Interfaces. 2017;10(9):7614–7625. doi:10.1021/acsami.7b03328
90. Wang S, Cao X, Shen M, Guo R, Bányai I, Shi X. Fabrication and morphology control of electrospun poly (γ-glutamic acid) nanofibers for biomedical applications. Colloids Surf B Biointerfaces. 2012;89:254–264. doi:10.1016/j.colsurfb.2011.09.029
91. Sofi HS, Ashraf R, Khan AH, Beigh MA, Majeed S, Sheikh FA. Reconstructing nanofibers from natural polymers using surface functionalization approaches for applications in tissue engineering, drug delivery and biosensing devices. Mater Sci Eng. 2019;94:1102–1124. doi:10.1016/j.msec.2018.10.069
92. Najar IN, Das S. Poly-glutamic acid (PGA)-Structure, synthesis, genomic organization and its application: a Review. Int J Pharm Sci Res. 2015;6(6):2258.
93. Wang G, Liu Q, Yang YM, et al. Applications and functions of γ-poly-glutamic acid and its derivatives in medicine. Curr Pharm Biotechnol. 2021;22(11):1404–1411. doi:10.2174/1389201021999201118161155
94. d’Ischia M, Napolitano A, Ball V, Chen C-T, Buehler MJ. Polydopamine and eumelanin: from structure–property relationships to a unified tailoring strategy. Acc Chem Res. 2014;47(12):3541–3550. doi:10.1021/ar500273y
95. Manivasagan P, Kim J, Jang E-S. Recent progress in multifunctional conjugated polymer nanomaterial-based synergistic combination phototherapy for microbial infection theranostics. Coord Chem Rev. 2022;470:214701. doi:10.1016/j.ccr.2022.214701
96. Ralph J, Lapierre C, Boerjan W. Lignin structure and its engineering. Curr Opin Biotechnol. 2019;56:240–249. doi:10.1016/j.copbio.2019.02.019
97. Sugiarto S, Leow Y, Tan CL, Wang G, Kai D. How far is Lignin from being a biomedical material? Bioact Mater. 2022;8:71–94. doi:10.1016/j.bioactmat.2021.06.023
98. Domínguez-Robles J, Martin NK, Fong ML, et al. Antioxidant PLA composites containing lignin for 3D printing applications: a potential material for healthcare applications. Pharmaceutics. 2019;11(4):165. doi:10.3390/pharmaceutics11040165
99. Pillai CKS. Challenges for natural monomers and polymers: novel design strategies and engineering to develop advanced polymers. Des Monomers Polym. 2010;13(2):87–121. doi:10.1163/138577210X12634696333190
100. Zhu J. Bioactive modification of poly (ethylene glycol) hydrogels for tissue engineering. Biomaterials. 2010;31(17):4639–4656. doi:10.1016/j.biomaterials.2010.02.044
101. Geckil H, Xu F, Zhang X, Moon S, Demirci U. Engineering hydrogels as extracellular matrix mimics. Nanomedicine. 2010;5(3):469–484. doi:10.2217/nnm.10.12
102. Bacakova L, Zikmundova M, Pajorova J, et al. Nanofibrous scaffolds for skin tissue engineering and wound healing based on synthetic polymers. Appl Nanobiotechnol. 2019;2019:1.
103. Kluge JA, Mauck RL. Synthetic/biopolymer nanofibrous composites as dynamic tissue engineering scaffolds. Biomed Appl Polymer Nanofibers. 2011;2011:101–130.
104. Zhang Z, Ortiz O, Goyal R, Kohn J. Biodegradable polymers. In: Handbook of Polymer Applications in Medicine and Medical Devices. Elsevier; 2014;303–335.
105. D’souza AA, Shegokar R. Polyethylene glycol (PEG): a versatile polymer for pharmaceutical applications. Expert Opin Drug Deliv. 2016;13(9):1257–1275. doi:10.1080/17425247.2016.1182485
106. Zustiak SP, Leach JB. Hydrolytically degradable poly (ethylene glycol) hydrogel scaffolds with tunable degradation and mechanical properties. Biomacromolecules. 2010;11(5):1348–1357. doi:10.1021/bm100137q
107. Ayres CE, Jha BS, Sell SA, Bowlin GL, Simpson DG. Nanotechnology in the design of soft tissue scaffolds: innovations in structure and function. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2010;2(1):20–34. doi:10.1002/wnan.55
108. Calvo P, Remunan‐Lopez C, Vila‐Jato JL, Alonso MJ. Novel hydrophilic chitosan‐polyethylene oxide nanoparticles as protein carriers. J Appl Polym Sci. 1997;63(1):125–132. doi:10.1002/(SICI)1097-4628(19970103)63:1<125::AID-APP13>3.0.CO;2-4
109. Howard MD, Jay M, Dziubla TD, Lu X. PEGylation of nanocarrier drug delivery systems: state of the art. J Biomed Nanotechnol. 2008;4(2):133–148. doi:10.1166/jbn.2008.021
110. Wang P, Chu W, Zhuo X, et al. Modified PLGA–PEG–PLGA thermosensitive hydrogels with suitable thermosensitivity and properties for use in a drug delivery system. J Mater Chem B. 2017;5(8):1551–1565. doi:10.1039/C6TB02158A
111. Hodge J, Quint C. The improvement of cell infiltration in an electrospun scaffold with multiple synthetic biodegradable polymers using sacrificial PEO microparticles. J Biomed Mater Res A. 2019;107(9):1954–1964. doi:10.1002/jbm.a.36706
112. Trakunjae C, Boondaeng A, Apiwatanapiwat W, et al. Enhanced polyhydroxybutyrate (PHB) production by newly isolated rare actinomycetes Rhodococcus sp. strain BSRT1-1 using response surface methodology. Sci Rep. 2021;11(1):1896. doi:10.1038/s41598-021-81386-2
113. Kasper FK, Tanahashi K, Fisher JP, Mikos AG. Synthesis of poly (propylene fumarate). Nat Protoc. 2009;4(4):518–525. doi:10.1038/nprot.2009.24
114. Wang S, Yaszemski MJ, Gruetzmacher JA, Lu L. Photo-crosslinked poly(ε-caprolactone fumarate) networks: roles of crystallinity and crosslinking density in determining mechanical properties. Polymer. 2008;49(26):5692–5699. doi:10.1016/j.polymer.2008.10.021
115. Jayabalan M, Shalumon KT, Mitha MK, Ganesan K, Epple M. Effect of hydroxyapatite on the biodegradation and biomechanical stability of polyester nanocomposites for orthopaedic applications. Acta Biomater. 2010;6(3):763–775. doi:10.1016/j.actbio.2009.09.015
116. Mirmohammadi SA, Imani M, Uyama H, Atai M. In situ photocrosslinkable nanohybrids based on poly (ε-caprolactone fumarate)/polyhedral oligomeric silsesquioxane: synthesis and characterization. J Polym Res. 2013;20(12):1–13. doi:10.1007/s10965-013-0297-z
117. Abdulkareem A, Kasak P, Nassr MG, et al. Surface modification of poly (lactic acid) film via cold plasma assisted grafting of fumaric and ascorbic acid. Polymers. 2021;13(21):3717. doi:10.3390/polym13213717
118. Kim K, Fisher JP. Nanoparticle technology in bone tissue engineering. J Drug Target. 2007;15(4):241–252. doi:10.1080/10611860701289818
119. Mohamed RM, Yusoh K. A review on the recent research of polycaprolactone (PCL). Adv Mat Res. 2016;1134:249–255. doi:10.4028/www.scientific.net/AMR.1134.249
120. Yadav BK, Patel G. Mechanical properties and biodegradability of electrospun methylcellulose-polyvinylalcohol single-layered nanofibers compared to polycaprolactone/methylcellulose-polyvinylalcohol/polycaprolactone multilayered nanofibers. Int J Polym Mater Polym Biomater. 2021;2021:1–8.
121. W-J L, Tuli R, Huang X, Laquerriere P, Tuan RS. Multilineage differentiation of human mesenchymal stem cells in a three-dimensional nanofibrous scaffold. Biomaterials. 2005;26(25):5158–5166. doi:10.1016/j.biomaterials.2005.01.002
122. Malikmammadov E, Tanir TE, Kiziltay A, Hasirci V, Hasirci N. PCL and PCL-based materials in biomedical applications. J Biomater Sci Polym Ed. 2018;29(7–9):863–893. doi:10.1080/09205063.2017.1394711
123. Rasal RM, Janorkar AV, Hirt DE. Poly (lactic acid) modifications. Prog Polym Sci. 2010;35(3):338–356. doi:10.1016/j.progpolymsci.2009.12.003
124. Choudhury S, Das A. Advances in generation of three-dimensional skin equivalents: pre-clinical studies to clinical therapies. Cytotherapy. 2021;23(1):1–9. doi:10.1016/j.jcyt.2020.10.001
125. Gaaz TS, Sulong AB, Akhtar MN, Kadhum AAH, Mohamad AB, Al-Amiery AA. Properties and applications of polyvinyl alcohol, halloysite nanotubes and their nanocomposites. Molecules. 2015;20(12):22833–22847. doi:10.3390/molecules201219884
126. Rahmani Del Bakhshayesh A, Annabi N, Khalilov R, et al. Recent advances on biomedical applications of scaffolds in wound healing and dermal tissue engineering. Artif Cells, Nanomed Biotechnol. 2018;46(4):691–705. doi:10.1080/21691401.2017.1349778
127. Sharma S, Parmar A, Kori S, Sandhir R. PLGA-based nanoparticles: a new paradigm in biomedical applications. TrAC Trends Anal Chem. 2016;80:30–40. doi:10.1016/j.trac.2015.06.014
128. Alsaab HO, Alharbi FD, Alhibs AS, et al. PLGA-based nanomedicine: history of advancement and development in clinical applications of multiple diseases. Pharmaceutics. 2022;14(12):2728. doi:10.3390/pharmaceutics14122728
129. Zou Y, Li D, Shen M, Shi X. Polyethylenimine‐based nanogels for biomedical applications. Macromol Biosci. 2019;19(11):1900272. doi:10.1002/mabi.201900272
130. Husain MSB, Gupta A, Alashwal BY, Sharma S. Synthesis of PVA/PVP based hydrogel for biomedical applications: a review. Energ Sources Part A. 2018;40(20):2388–2393. doi:10.1080/15567036.2018.1495786
131. Zayed M, Baker M, Ghazal H. Journal of textiles, coloration and polymer science; 2023.
132. Gugerell A, Kober J, Laube T, et al. Electrospun poly (ester-urethane)-and poly (ester-urethane-urea) fleeces as promising tissue engineering scaffolds for adipose-derived stem cells. PLoS One. 2014;9(3):e90676. doi:10.1371/journal.pone.0090676
133. Lizundia E, Sipponen MH, Greca LG, et al. Multifunctional lignin-based nanocomposites and nanohybrids. Green Chem. 2021;23(18):6698–6760. doi:10.1039/d1gc01684a
134. Kaur G, Narayanan G, Garg D, Sachdev A, Matai I. Biomaterials-based regenerative strategies for skin tissue wound healing. ACS Appl Bio Mater. 2022;5(5):2069–2106. doi:10.1021/acsabm.2c00035
135. Mohammadalizadeh Z, Bahremandi-Toloue E, Karbasi S. Synthetic-based blended electrospun scaffolds in tissue engineering applications. J Mater Sci. 2022;34:1–60. doi:10.1007/s10856-022-06709-9
136. Bottino FA, Cinquegrani AR, Di Pasquale G, Leonardi L, Pollicino A. Chemical modifications, mechanical properties and surface photo-oxidation of films of polystyrene (PS). Polym Test. 2004;23(4):405–411. doi:10.1016/j.polymertesting.2003.10.001
137. Motealleh B, Zahedi P, Rezaeian I, Moghimi M, Abdolghaffari AH, Zarandi MA. Morphology, drug release, antibacterial, cell proliferation, and histology studies of chamomile‐loaded wound dressing mats based on electrospun nanofibrous poly (ɛ‐caprolactone)/polystyrene blends. J Biomed Mater Res B Appl Biomater. 2014;102(5):977–987. doi:10.1002/jbm.b.33078
138. Chantre CO, Gonzalez GM, Ahn S, et al. Porous biomimetic hyaluronic acid and extracellular matrix protein nanofiber scaffolds for accelerated cutaneous tissue repair. ACS Appl Mater Interfaces. 2019;11(49):45498–45510. doi:10.1021/acsami.9b17322
139. Ding X, Zhao H, Li Y, et al. Synthetic peptide hydrogels as 3D scaffolds for tissue engineering. Adv Drug Deliv Rev. 2020;160:78–104. doi:10.1016/j.addr.2020.10.005
140. Sughanthy SAP, Ansari MNM, Atiqah A. Dynamic mechanical analysis of polyethylene terephthalate/hydroxyapatite biocomposites for tissue engineering applications. J Mater Res Technol. 2020;9(2):2350–2356. doi:10.1016/j.jmrt.2019.12.066
141. Bahari N, Hashim N, Md Akim A, Maringgal B. Recent advances in honey-based nanoparticles for wound dressing: a review. Nanomaterials. 2022;12(15):2560. doi:10.3390/nano12152560
142. Rahimpour A. UV photo-grafting of hydrophilic monomers onto the surface of nano-porous PES membranes for improving surface properties. Desalination. 2011;265(1–3):93–101. doi:10.1016/j.desal.2010.07.037
143. Babaeijandaghi F, Shabani I, Seyedjafari E, et al. Accelerated epidermal regeneration and improved dermal reconstruction achieved by polyethersulfone nanofibers. Tissue Eng Part A. 2010;16(11):3527–3536. doi:10.1089/ten.tea.2009.0829
144. Li H, Williams GR, Wu J, Wang H, Sun X, Zhu L-M. Poly(N-isopropylacrylamide)/poly(l-lactic acid-co-ɛ-caprolactone) fibers loaded with ciprofloxacin as wound dressing materials. Mater Sci Eng. 2017;79:245–254. doi:10.1016/j.msec.2017.04.058
145. Farahani M, Shafiee A. Wound healing: from passive to smart dressings. Adv Healthcare Mater. 2021;10(16):2100477. doi:10.1002/adhm.202100477
146. Liu S, Ma J, Xu L, et al. An electrospun polyurethane scaffold-reinforced zwitterionic hydrogel as a biocompatible device. J Mater Chem B. 2020;8(12):2443–2453. doi:10.1039/C9TB02870F
147. Harijan M, Singh M. Zwitterionic polymers in drug delivery: a review. J Mol Recognit. 2022;35(1):e2944. doi:10.1002/jmr.2944
148. Islam S. Microneedles in pain management of Cancer; 2021.
149. Cai SS, Li T, Akinade T, Zhu Y, Leong KW. Drug delivery carriers with therapeutic functions. Adv Drug Deliv Rev. 2021;176:113884. doi:10.1016/j.addr.2021.113884
150. Tuan-Mahmood T-M, McCrudden MTC, Torrisi BM, et al. Microneedles for intradermal and transdermal drug delivery. Eur J Pharm Sci. 2013;50(5):623–637. doi:10.1016/j.ejps.2013.05.005
151. Bhatnagar S, Gadeela PR, Thathireddy P, Venuganti VVK. Microneedle-based drug delivery: materials of construction. J Chem Sci. 2019;131(9):1–28. doi:10.1007/s12039-019-1666-x
152. Avcil M, Çelik A. Microneedles in drug delivery: progress and challenges. Micromachines. 2021;12(11):1321. doi:10.3390/mi12111321
153. Yang Y, Wu Y, Wei Y, Zeng T, Cao B, Liang J. Preparation and characterization of hydroxyapatite coating on AZ31 magnesium alloy induced by carboxymethyl cellulose-dopamine. Materials. 2021;14(8):1849. doi:10.3390/ma14081849
154. Bilal M, Mehmood S, Raza A, Hayat U, Rasheed T, Iqbal HMN. Microneedles in smart drug delivery. Adv Wound Care. 2021;10(4):204–219. doi:10.1089/wound.2019.1122
155. Donnelly RF, Larrañeta E. Microarray patches: potentially useful delivery systems for long-acting nanosuspensions. Drug Discov Today. 2018;23(5):1026–1033. doi:10.1016/j.drudis.2017.10.013
156. Sheng T, Luo B, Zhang W, et al. Microneedle-mediated vaccination: innovation and translation. Adv Drug Deliv Rev. 2021;179:113919. doi:10.1016/j.addr.2021.113919
157. Ali A, Ahmed S. Recent advances in edible polymer based hydrogels as a sustainable alternative to conventional polymers. J Agric Food Chem. 2018;66(27):6940–6967. doi:10.1021/acs.jafc.8b01052
158. Kakde I, Bansode D. Microrobotics a new approach in ocular drug delivery system: a review; 2022.
159. Ceylan H, Giltinan J, Kozielski K, Sitti M. Mobile microrobots for bioengineering applications. Lab Chip. 2017;17(10):1705–1724. doi:10.1039/C7LC00064B
160. Dabbagh SR, Sarabi MR, Birtek MT, Seyfi S, Sitti M, Tasoglu S. 3D-printed microrobots from design to translation. Nat Commun. 2022;13(1):1–24. doi:10.1038/s41467-022-33409-3
161. Nelson BJ, Kaliakatsos IK, Abbott JJ. Microrobots for minimally invasive medicine. Annu Rev Biomed Eng. 2010;12:55–85. doi:10.1146/annurev-bioeng-010510-103409
162. Bunea AI, Glückstad J. Strategies for optical trapping in biological samples: aiming at microrobotic surgeons. Laser Photon Rev. 2019;13(4):1800227. doi:10.1002/lpor.201800227
163. Bhattarai DP, Aguilar LE, Park CH, Kim CS. A review on properties of natural and synthetic based electrospun fibrous materials for bone tissue engineering. Membranes. 2018;8(3):62. doi:10.3390/membranes8030062
164. Samadian H, Farzamfar S, Vaez A, et al. A tailored polylactic acid/polycaprolactone biodegradable and bioactive 3D porous scaffold containing gelatin nanofibers and Taurine for bone regeneration. Sci Rep. 2020;10(1):1–12. doi:10.1038/s41598-020-70155-2
165. Costa RG, Oliveira J, Paula G, et al. Electrospinning of polymers in solution: part II: applications and perspectives. Polímeros. 2012;22:178–185. doi:10.1590/S0104-14282012005000018
166. Lin M, Dai Y, Xia F, Zhang X. Advances in non-covalent crosslinked polymer micelles for biomedical applications. Mater Sci Eng. 2021;119:111626. doi:10.1016/j.msec.2020.111626
167. Bleul R, Thiermann R, Maskos M. Techniques to control polymersome size. Macromolecules. 2015;48(20):7396–7409. doi:10.1021/acs.macromol.5b01500
168. Ghorbanizamani F, Tok K, Moulahoum H, et al. Dye-loaded polymersome-based lateral flow assay: rational design of a COVID-19 testing platform by repurposing SARS-CoV-2 antibody cocktail and antigens obtained from positive human samples. ACS Sens. 2021;6(8):2988–2997. doi:10.1021/acssensors.1c00854
169. Hosta-Rigau L, Jensen BEB, Fjeldsø KS, et al. Surface‐adhered composite poly(vinyl alcohol) physical hydrogels: polymersome‐aided delivery of therapeutic small molecules; 2012:1.
170. Wayakanon K, Thornhill MH, Douglas CWI, et al. Polymersome‐mediated intracellular delivery of antibiotics to treat Porphyromonas gingivalis‐infected oral epithelial cells. FASEB J. 2013;27(11):4455–4465. doi:10.1096/fj.12-225219
171. Mittal P, Saharan A, Verma R, et al. Dendrimers: a new race of pharmaceutical nanocarriers. Biomed Res Int. 2021;2021:8844030. doi:10.1155/2021/8844030
172. Gillies ER, Frechet JM. Dendrimers and dendritic polymers in drug delivery. Drug Discov Today. 2005;10(1):35–43. doi:10.1016/S1359-6446(04)03276-3
173. Dwivedi N, Shah J, Mishra V, Tambuwala M, Kesharwani P. Nanoneuromedicine for management of neurodegenerative disorder. J Drug Deliv Sci Technol. 2019;49:477–490. doi:10.1016/j.jddst.2018.12.021
174. Bapat RA, Chaubal TV, Joshi CP, et al. An overview of application of silver nanoparticles for biomaterials in dentistry. Mater Sci Eng. 2018;91:881–898. doi:10.1016/j.msec.2018.05.069
175. Kesharwani P, Jain K, Jain PS. Dendrimer as nanocarrier for drug delivery. Prog Polym Sci. 2014;39(2):268–307. doi:10.1016/j.progpolymsci.2013.07.005
176. Madani M, Hosny S, Alshangiti DM, et al. Green synthesis of nanoparticles for varied applications: green renewable resources and energy-efficient synthetic routes. Nanotechnol Rev. 2022;11(1):731–759. doi:10.1515/ntrev-2022-0034
177. Tao Y, Endo M, Kaneko K. A review of synthesis and nanopore structures of organic polymer aerogels and carbon aerogels. Recent Pat Chem Eng. 2008;1(3):192–200. doi:10.2174/2211334710801030192
178. Liu Z, Ran Y, Xi J, Wang J. Polymeric hybrid aerogels and their biomedical applications. Soft Matter. 2020;16(40):9160–9175. doi:10.1039/D0SM01261K
179. Neamtu I, Rusu AG, Diaconu A, Nita LE, Chiriac AP. Basic concepts and recent advances in nanogels as carriers for medical applications. Drug Deliv. 2017;24(1):539–557. doi:10.1080/10717544.2016.1276232
180. Li X, Tsibouklis J, Weng T, et al. Nano carriers for drug transport across the blood–brain barrier. J Drug Target. 2017;25(1):17–28. doi:10.1080/1061186X.2016.1184272
181. Kwon GS, Kataoka KJ. Block copolymer micelles as long-circulating drug vehicles. Adv Drug Deliv Rev. 2012;64:237–245. doi:10.1016/j.addr.2012.09.016
182. Hamdy TM. Polymerization shrinkage in contemporary resin-based dental composites: a Review Article. Egypt J Chem. 2021;64(6):3087–3092.
183. Haugen HJ, Basu P, Sukul M, Mano JF, Reseland JE. Injectable Biomaterials for Dental Tissue Regeneration. Int J Mol Sci. 2020;21(10):3442. doi:10.3390/ijms21103442
184. Ifkovits JL, Burdick JA. Photopolymerizable and degradable biomaterials for tissue engineering applications. Tissue Eng. 2007;13(10):2369–2385. doi:10.1089/ten.2007.0093
185. Sosnik A, Seremeta KP. Polymeric hydrogels as technology platform for drug delivery applications. Gels. 2017;3(3):25. doi:10.3390/gels3030025
186. Ghobashy MM, El-Sawy NM, Kodous AS. Nanocomposite of cosubstituted carbonated hydroxyapatite fabricated inside Poly (sodium hyaluronate-acrylamide) hydrogel template prepared by gamma radiation for osteoblast cell regeneration. Radiat Phys Chem. 2021;183:109408. doi:10.1016/j.radphyschem.2021.109408
187. Khojasteh A, Kheiri L, Motamedian SR, Khoshkam V. Guided bone regeneration for the reconstruction of alveolar bone defects. Ann Maxillofac Surg. 2017;7(2):263. doi:10.4103/ams.ams_76_17
188. Neto AMD, Sartoretto SC, Duarte IM, et al. In vivo comparative evaluation of biocompatibility and biodegradation of bovine and porcine collagen membranes. Membranes. 2020;10(12):423. doi:10.3390/membranes10120423
189. Colobatiu L, Gavan A, Potarniche A-V, et al. Evaluation of bioactive compounds-loaded chitosan films as a novel and potential diabetic wound dressing material. React Funct Polym. 2019;145:104369. doi:10.1016/j.reactfunctpolym.2019.104369
190. Oryan A, Kamali A, Moshiri A, Baharvand H, Daemi H. Chemical crosslinking of biopolymeric scaffolds: current knowledge and future directions of crosslinked engineered bone scaffolds. Int J Biol Macromol. 2018;107:678–688. doi:10.1016/j.ijbiomac.2017.08.184
191. Wathoni N, Motoyama K, Higashi T, Okajima M, Kaneko T, Arima H. Physically crosslinked-sacran hydrogel films for wound dressing application. Int J Biol Macromol. 2016;89:465–470. doi:10.1016/j.ijbiomac.2016.05.006
192. Ahi ZB, Renkler NZ, Gul Seker M, Tuzlakoglu K. Biodegradable Polymer Films with a Natural Antibacterial Extract as Novel Periodontal Barrier Membranes. Int J Biomater. 2019;2019:7932470. doi:10.1155/2019/7932470
193. Chi M, Qi M, L A, et al. Novel bioactive and therapeutic dental polymeric materials to inhibit periodontal pathogens and biofilms. Int J Mol Sci. 2019;20(2). doi:10.3390/ijms20020278
194. Atia GA, Shalaby HK, Zehravi M, et al. Drug-loaded chitosan scaffolds for periodontal tissue regeneration. Polymers. 2022;14(15):3192. doi:10.3390/polym14153192
195. Procopio A, Lagreca E, Jamaledin R, et al. Recent fabrication methods to produce polymer-based drug delivery matrices (Experimental and In Silico Approaches). Pharmaceutics. 2022;14(4):872. doi:10.3390/pharmaceutics14040872
196. Sherstneva AA, Demina TS, Monteiro APF, Akopova TA, Grandfils C, Ilangala AB. Biodegradable microparticles for regenerative medicine: a state of the art and trends to clinical application. Polymers. 2022;14(7):1314. doi:10.3390/polym14071314
197. Lu J, Ren B, Wang L, Li M, Liu Y. Preparation and evaluation of IL-1ra-loaded dextran/PLGA microspheres for inhibiting periodontal inflammation in vitro. Inflammation. 2020;43:168–178. doi:10.1007/s10753-019-01107-w
198. Perez-Puyana V, Jiménez-Rosado M, Romero A, Guerrero A. Polymer-based scaffolds for soft-tissue engineering. Polymers. 2020;12(7):1566. doi:10.3390/polym12071566
199. Stratton S, Shelke NB, Hoshino K, Rudraiah S, Kumbar SG. Bioactive polymeric scaffolds for tissue engineering. Bioact Mater. 2016;1(2):93–108. doi:10.1016/j.bioactmat.2016.11.001
200. Zhang L, Morsi Y, Wang Y, Li Y, Ramakrishna S. Review scaffold design and stem cells for tooth regeneration. Jpn Dent Sci Rev. 2013;49(1):14–26. doi:10.1016/j.jdsr.2012.09.001
201. Albuquerque MTP, Valera MC, Nakashima M, Nör JE, Bottino MC. Tissue-engineering-based strategies for regenerative endodontics. J Dent Res. 2014;93(12):1222–1231. doi:10.1177/0022034514549809
202. Bottino MC, Kamocki K, Yassen GH, et al. Bioactive nanofibrous scaffolds for regenerative endodontics. J Dent Res. 2013;92(11):963–969. doi:10.1177/0022034513505770
203. Baylan N, Bhat S, Ditto M, Lawrence JG, Lecka-Czernik B, Yildirim-Ayan E. Polycaprolactone nanofiber interspersed collagen type-I scaffold for bone regeneration: a unique injectable osteogenic scaffold. Biomed Mater. 2013;8(4):045011. doi:10.1088/1748-6041/8/4/045011
204. Tai H, Mather ML, Howard D, et al. Control of pore size and structure of tissue engineering scaffolds produced by supercritical fluid processing. Eur Cell Mater. 2007;14:64–77. doi:10.22203/eCM.v014a07
205. Palit MC, Hedge KS, Bhat SS, Sargod SS, Mantha S, Chattopadhyay S. Tissue engineering in endodontics: root canal revascularization. J Clin Pediatr Dent. 2014;38(4):291–297. doi:10.17796/jcpd.38.4.j5285857278615r1
206. Barron JA, Krizman DB, Ringeisen BR. Laser printing of single cells: statistical analysis, cell viability, and stress. Ann Biomed Eng. 2005;33:121–130. doi:10.1007/s10439-005-8971-x
207. Jakab K, Norotte C, Marga F, Murphy K, Vunjak-Novakovic G, Forgacs G. Tissue engineering by self-assembly and bio-printing of living cells. Biofabrication. 2010;2(2):022001. doi:10.1088/1758-5082/2/2/022001
208. Galler KM, Cavender A, Yuwono V, et al. Self-assembling peptide amphiphile nanofibers as a scaffold for dental stem cells. Tissue Eng Part A. 2008;14(12):2051–2058. doi:10.1089/ten.tea.2007.0413
209. Atia GA, Shalaby HK, Ali NG, et al. New challenges and prospective applications of three-dimensional bioactive polymeric hydrogels in oral and craniofacial tissue engineering: a narrative review. Pharmaceuticals. 2023;16(5):702. doi:10.3390/ph16050702
210. Pan Y, Zhao Y, Kuang R, et al. Injectable hydrogel-loaded nano-hydroxyapatite that improves bone regeneration and alveolar ridge promotion. Mater Sci Eng. 2020;116:111158. doi:10.1016/j.msec.2020.111158
211. Chang H-C, Yang C, Feng F, Lin F-H, Wang C-H, Chang P-C. Bone morphogenetic protein-2 loaded poly (D, L-lactide-co-glycolide) microspheres enhance osteogenic potential of gelatin/hydroxyapatite/β-tricalcium phosphate cryogel composite for alveolar ridge augmentation. J Formos Med Assoc. 2017;116(12):973–981. doi:10.1016/j.jfma.2017.01.005
212. Zeng J, Mamitimin M, Song Y, Sun W, Wu Z, Qi X. Chairside administrated antibacterial hydrogels containing berberine as dental temporary stopping for alveolar ridge preservation. Eur Polym J. 2021;160:110808. doi:10.1016/j.eurpolymj.2021.110808
213. J-t G, Jiao K, Li J, et al. Polyphosphate-crosslinked collagen scaffolds for hemostasis and alveolar bone regeneration after tooth extraction. Bioact Mater. 2022;15:68–81. doi:10.1016/j.bioactmat.2021.12.019
214. Serino G, Biancu S, Iezzi G, Piattelli A. Ridge preservation following tooth extraction using a polylactide and polyglycolide sponge as space filler: a clinical and histological study in humans. Clin Oral Implants Res. 2003;14(5):651–658. doi:10.1034/j.1600-0501.2003.00970.x
215. Mancini L, Romandini M, Fratini A, Americo LM, Panda S, Marchetti E. Biomaterials for periodontal and peri-implant regeneration. Materials. 2021;14(12):3319. doi:10.3390/ma14123319
216. Wickramasinghe ML, Dias GJ, Premadasa K. A novel classification of bone graft materials. J Biomed Mater Res B Appl Biomater. 2022;110(7):1724–1749. doi:10.1002/jbm.b.35029
217. Nguyen TT, Wu DT, Ramamoorthi M, Syrbu J, Tran SD. Scaffolds for maxillary sinus augmentation. In: Handbook of Tissue Engineering Scaffolds: Volume One. Elsevier; 2019:369–386.
218. Xun X, Qiu J, Zhang J, et al. Triple-functional injectable liposome–hydrogel composite enhances bacteriostasis and osteo/angio-genesis for advanced maxillary sinus floor augmentation. Colloids Surf B Biointerfaces. 2022;217:112706. doi:10.1016/j.colsurfb.2022.112706
219. Cossellu G, Farronato G, Farronato D, Ceschel G, Angiero F. Space-maintaining management in maxillary sinus lifting: a novel technique using a resorbable polymeric thermo-reversible gel. Int J Oral Maxillofac Surg. 2017;46(5):648–654. doi:10.1016/j.ijom.2017.01.013
220. Li D, Zhao L, Cong M, et al. Injectable thermosensitive chitosan/gelatin-based hydrogel carried erythropoietin to effectively enhance maxillary sinus floor augmentation in vivo. Dent Mater. 2020;36(7):e229–e240. doi:10.1016/j.dental.2020.04.016
221. Al-Nawas B, Schiegnitz E. Augmentation procedures using bone substitute materials or autogenous bone-a systematic review and meta-analysis. Eur J Oral Implantol. 2014;7(Suppl 2):S219–S234.
222. Bousnaki M, Bakopoulou A, Papadogianni D, et al. Fibro/chondrogenic differentiation of dental stem cells into chitosan/alginate scaffolds towards temporomandibular joint disc regeneration. J Mater Sci Mater Med. 2018;29(7):1–17.
223. Van bellinghen X, Idoux-Gillet Y, Pugliano M, et al. Temporomandibular joint regenerative medicine. Int J Mol Sci. 2018;19(2):446. doi:10.3390/ijms19020446
224. Acri TM, Shin K, Seol D, et al. Tissue Engineering for the Temporomandibular Joint. Adv Healthc Mater. 2019;8(2):e1801236. doi:10.1002/adhm.201801236
225. Pina S, Ribeiro VP, Marques CF, et al. Scaffolding strategies for tissue engineering and regenerative medicine applications. Materials. 2019;12(11):1824. doi:10.3390/ma12111824
226. Uematsu K, Hattori K, Ishimoto Y, et al. Cartilage regeneration using mesenchymal stem cells and a three-dimensional poly-lactic-glycolic acid (PLGA) scaffold. Biomaterials. 2005;26(20):4273–4279. doi:10.1016/j.biomaterials.2004.10.037
227. Eap S, Morand D, Clauss F, et al. Nanostructured thick 3D nanofibrous scaffold can induce bone. Biomed Mater Eng. 2015;25(s1):79–85. doi:10.3233/BME-141248
228. Hamdy TM. Dental biomaterial scaffolds in tooth tissue engineering: a review. Curr Oral Health Rep. 2023;10(1):14–21. doi:10.1007/s40496-023-00329-0
229. Ramseier CA, Rasperini G, Batia S, Giannobile WV. Advanced reconstructive technologies for periodontal tissue repair. Periodontol 2000. 2012;59(1):185–202. doi:10.1111/j.1600-0757.2011.00432.x
230. Stoecklin-Wasmer C, Rutjes AWS, Da Costa BR, Salvi GE, Jüni P, Sculean A. Absorbable collagen membranes for periodontal regeneration: a systematic review. J Dent Res. 2013;92(9):773–781. doi:10.1177/0022034513496428
231. Das EC, Babu J, Dhawan S, et al. Synthetic osteogenic matrix using polymeric dendritic peptides for treating human periodontal defects–design and in vitro evaluation. Mater Today. 2019;15:199–216.
232. Daghrery A, Ferreira JA, Xu J, et al. Tissue-specific melt electrowritten polymeric scaffolds for coordinated regeneration of soft and hard periodontal tissues. Bioact Mater. 2023;19:268–281. doi:10.1016/j.bioactmat.2022.04.013
233. Dascălu C-A, Maidaniuc A, Pandele AM, et al. Synthesis and characterization of biocompatible polymer-ceramic film structures as favorable interface in guided bone regeneration. Appl Surf Sci. 2019;494:335–352. doi:10.1016/j.apsusc.2019.07.098
234. Shang L, Liu Z, Ma B, et al. Dimethyloxallyl glycine/nanosilicates-loaded osteogenic/angiogenic difunctional fibrous structure for functional periodontal tissue regeneration. Bioact Mater. 2021;6(4):1175–1188. doi:10.1016/j.bioactmat.2020.10.010
235. Jamalpour MR, Yadegari A, Vahdatinia F, et al. 3D-printed bi-layered polymer/hydrogel construct for interfacial tissue regeneration in a canine model. Dent Mater. 2022;38(8):1316–1329. doi:10.1016/j.dental.2022.06.020
236. Momose T, Miyaji H, Kato A, et al. Collagen hydrogel scaffold and fibroblast growth factor-2 accelerate periodontal healing of class II furcation defects in dog. Open Dent J. 2016;10:347. doi:10.2174/1874210601610010347
237. Nakamura S, Kubo T, Ijima H. Heparin-conjugated gelatin as a growth factor immobilization scaffold. J Biosci Bioeng. 2013;115(5):562–567. doi:10.1016/j.jbiosc.2012.11.011
238. Varoni EM, Vijayakumar S, Canciani E, et al. Chitosan-based trilayer scaffold for multitissue periodontal regeneration. J Dent Res. 2018;97(3):303–311. doi:10.1177/0022034517736255
239. Shang S, Yang F, Cheng X, Walboomers XF, Jansen JA. The effect of electrospun fibre alignment on the behaviour of rat periodontal ligament cells; 2010.
240. Batool F, Morand D-N, Thomas L, et al. Synthesis of a novel electrospun polycaprolactone scaffold functionalized with ibuprofen for periodontal regeneration: an in vitro and in vivo study. Materials. 2018;11(4):580. doi:10.3390/ma11040580
241. Reis ECC, Borges APB, Araújo MVF, Mendes VC, Guan L, Davies JE. Periodontal regeneration using a bilayered PLGA/calcium phosphate construct. Biomaterials. 2011;32(35):9244–9253. doi:10.1016/j.biomaterials.2011.08.040
242. Liu Z, Yin X, Ye Q, et al. Periodontal regeneration with stem cells-seeded collagen-hydroxyapatite scaffold. J Biomater Appl. 2016;31(1):121–131. doi:10.1177/0885328216637978
243. Liao F, Chen Y, Li Z, et al. A novel bioactive three-dimensional β-tricalcium phosphate/chitosan scaffold for periodontal tissue engineering. J Mater Sci Mater Med. 2010;21:489–496. doi:10.1007/s10856-009-3931-x
244. Costa PF, Vaquette C, Zhang Q, Reis RL, Ivanovski S, Hutmacher DW. Advanced tissue engineering scaffold design for regeneration of the complex hierarchical periodontal structure. J Clin Periodontol. 2014;41(3):283–294. doi:10.1111/jcpe.12214
245. Sowmya S, Mony U, Jayachandran P, et al. Tri‐layered nanocomposite hydrogel scaffold for the concurrent regeneration of cementum, periodontal ligament, and alveolar bone. Adv Healthcare Mater. 2017;6(7):1601251. doi:10.1002/adhm.201601251
246. Lee CH, Hajibandeh J, Suzuki T, Fan A, Shang P, Mao JJ. Three-dimensional printed multiphase scaffolds for regeneration of periodontium complex. Tissue Eng Part A. 2014;20(7–8):1342–1351. doi:10.1089/ten.tea.2013.0386
247. Zafar MS, Fareed MA, Riaz S, Latif M, Habib SR, Khurshid Z. Customized therapeutic surface coatings for dental implants. Coatings. 2020;10(6):568. doi:10.3390/coatings10060568
248. Gaviria L, Salcido JP, Guda T, Ong JL. Current trends in dental implants. J Korean Assoc Oral Maxillofac Surg. 2014;40(2):50. doi:10.5125/jkaoms.2014.40.2.50
249. Accioni F, Vázquez J, Merinero M, Begines B, Alcudia A. Latest trends in surface modification for dental implantology: innovative developments and analytical applications. Pharmaceutics. 2022;14(2):455. doi:10.3390/pharmaceutics14020455
250. Ansari Z, Kalantar M, Kharaziha M, Ambrosio L, Raucci MG. Polycaprolactone/fluoride substituted-hydroxyapatite (PCL/FHA) nanocomposite coatings prepared by in-situ sol-gel process for dental implant applications. Prog Org Coat. 2020;147:105873. doi:10.1016/j.porgcoat.2020.105873
251. Joo H, Park J, Sutthiwanjampa C, et al. Surface coating with hyaluronic acid-gelatin-crosslinked hydrogel on gelatin-conjugated poly (dimethylsiloxane) for implantable medical device-induced fibrosis. Pharmaceutics. 2021;13(2):269. doi:10.3390/pharmaceutics13020269
252. Catauro M, Bollino F, Giovanardi R, Veronesi P. Modification of Ti6Al4V implant surfaces by biocompatible TiO2/PCL hybrid layers prepared via sol-gel dip coating: structural characterization, mechanical and corrosion behavior. Mater Sci Eng. 2017;74:501–507. doi:10.1016/j.msec.2016.12.046
253. Clifford A, D’Elia A, Deering J, Lee BEJ, Grandfield K, Zhitomirsky I. Electrochemical fabrication and characterization of pectin hydrogel composite materials for bone tissue repair. ACS Appl Polymer Mater. 2020;2(8):3390–3396. doi:10.1021/acsapm.0c00480
254. Barkarmo S, Wennerberg A, Hoffman M, et al. Nano‐hydroxyapatite‐coated PEEK implants: a pilot study in rabbit bone. J Biomed Mater Res A. 2013;101(2):465–471. doi:10.1002/jbm.a.34358
255. Kulkarni Aranya A, Pushalkar S, Zhao M, LeGeros RZ, Zhang Y, Saxena D. Antibacterial and bioactive coatings on titanium implant surfaces. J Biomed Mater Res A. 2017;105(8):2218–2227. doi:10.1002/jbm.a.36081
256. Wang M, Wang C, Zhang Y, Lin Y. Controlled release of dopamine coatings on titanium bidirectionally regulate osteoclastic and osteogenic response behaviors. Mater Sci Eng. 2021;129:112376. doi:10.1016/j.msec.2021.112376
257. Zheng S, Zhong H, Cheng H, et al. Engineering multifunctional hydrogel with osteogenic capacity for critical-size segmental bone defect repair. Front Bioeng Biotechnol. 2022;10:899457. doi:10.3389/fbioe.2022.899457
258. Song W, Xiao Y. Sequential drug delivery of vancomycin and rhBMP-2 via pore-closed PLGA microparticles embedded photo-crosslinked chitosan hydrogel for enhanced osteointegration. Int J Biol Macromol. 2021;182:612–625. doi:10.1016/j.ijbiomac.2021.03.181
259. Campbell DI, Duncan WJ. The effect of a keratin hydrogel coating on osseointegration: an histological comparison of coated and non-coated dental titanium implants in an ovine model. J Maxillofac Oral Surg. 2014;13:159–164. doi:10.1007/s12663-013-0482-y
260. Cheng H, Yue K, Kazemzadeh-Narbat M, et al. Mussel-inspired multifunctional hydrogel coating for prevention of infections and enhanced osteogenesis. ACS Appl Mater Interfaces. 2017;9(13):11428–11439. doi:10.1021/acsami.6b16779
261. Naidu MUR, Ramana GV, Rani PU, Suman A, Roy P, Roy P. Chemotherapy-induced and/or radiation therapy-induced oral mucositis-complicating the treatment of cancer. Neoplasia. 2004;6(5):423–431. doi:10.1593/neo.04169
262. Mehravaran M, Haeri A, Rabbani S, Mortazavi SA, Torshabi M. Preparation and characterization of benzydamine hydrochloride-loaded lyophilized mucoadhesive wafers for the treatment of oral mucositis. J Drug Deliv Sci Technol. 2022;78:103944. doi:10.1016/j.jddst.2022.103944
263. Pornpitchanarong C, Rojanarata T, Opanasopit P, Ngawhirunpat T, Bradley M, Patrojanasophon P. Maleimide-functionalized carboxymethyl cellulose: a novel mucoadhesive polymer for transmucosal drug delivery. Carbohydr Polym. 2022;288:119368. doi:10.1016/j.carbpol.2022.119368
264. Chin H-L, Chung K-P, Liu H-C, Chen R-S, Chang -H-H, Chen M-H. Efficacy of polyacrylate silver salt/polyvinylpyrrolidone-based liquid oral gel in management of concurrence chemoradiotherapy-induced oral mucositis. J Formos Med Assoc. 2023;122:723–730. doi:10.1016/j.jfma.2022.12.007
265. Feng X, Lu X, Huang D, et al. 3D porous chitosan scaffolds suit survival and neural differentiation of dental pulp stem cells. Cell Mol Neurobiol. 2014;34:859–870. doi:10.1007/s10571-014-0063-8
266. Moussa DG, Aparicio C. Present and future of tissue engineering scaffolds for dentin‐pulp complex regeneration. J Tissue Eng Regen Med. 2019;13(1):58–75. doi:10.1002/term.2769
267. Ishimatsu H, Kitamura C, Morotomi T, et al. Formation of dentinal bridge on surface of regenerated dental pulp in dentin defects by controlled release of fibroblast growth factor–2 from gelatin hydrogels. J Endod. 2009;35(6):858–865. doi:10.1016/j.joen.2009.03.049
268. Shivashankar VY, Johns DA, Vidyanath S, Kumar MR. Platelet rich fibrin in the revitalization of tooth with necrotic pulp and open apex. J Conserv Dent. 2012;15(4):395. doi:10.4103/0972-0707.101926
269. Huang FM, Yang SF, Zhao JH, Chang YC. Platelet-rich fibrin increases proliferation and differentiation of human dental pulp cells. J Endod. 2010;36(10):1628–1632. doi:10.1016/j.joen.2010.07.004
270. Jiang X, Liu H, Peng C. Clinical and radiographic assessment of the efficacy of a collagen membrane in regenerative endodontics: a randomized, controlled clinical trial. J Endod. 2017;43(9):1465–1471. doi:10.1016/j.joen.2017.04.011
271. Yang KC, Wang CH, Chang HH, Chan WP, Chi CH, Kuo TF. Fibrin glue mixed with platelet‐rich fibrin as a scaffold seeded with dental bud cells for tooth regeneration. J Tissue Eng Regen Med. 2012;6(10):777–785. doi:10.1002/term.483
272. Wieckiewicz M, W Boening K, Grychowska N, Paradowska-Stolarz A. Clinical application of chitosan in dental specialities. Mini Rev Med Chem. 2017;17(5):401–409. doi:10.2174/1389557516666160418123054
273. Nayef L, Mekhail M, Benameur L, Rendon JS, Hamdy R, Tabrizian M. A combinatorial approach towards achieving an injectable, self-contained, phosphate-releasing scaffold for promoting biomineralization in critical size bone defects. Acta biomater. 2016;29:389–397. doi:10.1016/j.actbio.2015.10.020
274. Junka A, Bartoszewicz M, Dziadas M, et al. Application of bacterial cellulose experimental dressings saturated with gentamycin for management of bone biofilm in vitro and ex vivo. J Biomed Mater Res B Appl Biomater. 2020;108(1):30–37. doi:10.1002/jbm.b.34362
275. Pankaj D, Sahu I, Kurian IG, Pradeep AR. Comparative evaluation of subgingivally delivered 1.2% rosuvastatin and 1% metformin gel in treatment of intrabony defects in chronic periodontitis: a randomized controlled clinical trial. J Periodontol. 2018;89(11):1318–1325. doi:10.1002/jper.17-0434
276. Abrahamse H, Hamblin MR. New photosensitizers for photodynamic therapy. Biochem J. 2016;473(4):347–364. doi:10.1042/bj20150942
277. Elavarasu S, Suthanthiran TK, Naveen D. Statins: a new era in local drug delivery. J Pharm Bioallied Sci. 2012;4(Suppl 2):S248–51. doi:10.4103/0975-7406.100225
278. Javed F, Al-Askar M, Al-Rasheed A, Al-Hezaimi K. Significance of the platelet-derived growth factor in periodontal tissue regeneration. Arch Oral Biol. 2011;56(12):1476–1484. doi:10.1016/j.archoralbio.2011.06.020
279. Murakami S. Periodontal tissue regeneration by signaling molecule(s): what role does basic fibroblast growth factor (FGF-2) have in periodontal therapy? Periodontol 2000. 2011;56(1):188–208. doi:10.1111/j.1600-0757.2010.00365.x
280. Anusuya GS, Kandasamy M, Jacob Raja SA, Sabarinathan S, Ravishankar P, Kandhasamy B. Bone morphogenetic proteins: signaling periodontal bone regeneration and repair. J Pharm Bioallied Sci. 2016;8(Suppl 1):S39–s41. doi:10.4103/0975-7406.191964
281. Abourehab MAS, Rajendran RR, Singh A, et al. Alginate as a promising biopolymer in drug delivery and wound healing: a review of the state-of-the-art. Int J Mol Sci. 2022;23(16):9035. doi:10.3390/ijms23169035
282. Venkatesan J, Lee J-Y, Kang DS, et al. Antimicrobial and anticancer activities of porous chitosan-alginate biosynthesized silver nanoparticles. Int J Biol Macromol. 2017;98:515–525. doi:10.1016/j.ijbiomac.2017.01.120
283. Dong L, Wang SJ, Zhao XR, Zhu YF, Yu JK. 3D- printed poly(ε-caprolactone) scaffold integrated with cell-laden chitosan hydrogels for bone tissue engineering. Sci Rep. 2017;7(1):13412. doi:10.1038/s41598-017-13838-7
284. Ghanbari M, Salavati-Niasari M, Mohandes F. Thermosensitive alginate–gelatin–nitrogen-doped carbon dots scaffolds as potential injectable hydrogels for cartilage tissue engineering applications. RSC Adv. 2021;11(30):18423–18431. doi:10.1039/D1RA01496J
285. Santo VE, Frias AM, Carida M, et al. Carrageenan-based hydrogels for the controlled delivery of PDGF-BB in bone tissue engineering applications. Biomacromolecules. 2009;10(6):1392–1401. doi:10.1021/bm8014973
286. Kim S, Kang Y, Mercado-Pagán ÁE, Maloney WJ, Yang Y. In vitro evaluation of photo-crosslinkable chitosan-lactide hydrogels for bone tissue engineering. J Biomed Mater Res B Appl Biomater. 2014;102(7):1393–1406. doi:10.1002/jbm.b.33118
287. Kumar A, Han SS. Enhanced mechanical, biomineralization, and cellular response of nanocomposite hydrogels by bioactive glass and halloysite nanotubes for bone tissue regeneration. Mater Sci Eng C Mater Biol Appl. 2021;128:112236. doi:10.1016/j.msec.2021.112236
288. Wang K, Cheng W, Ding Z, et al. Injectable silk/hydroxyapatite nanocomposite hydrogels with vascularization capacity for bone regeneration. J Mater Sci Technol. 2020;2020:1.
289. Hou R, Zhang G, Du G, et al. Magnetic nanohydroxyapatite/PVA composite hydrogels for promoted osteoblast adhesion and proliferation. Colloids Surf B Biointerfaces. 2013;103:318–325. doi:10.1016/j.colsurfb.2012.10.067
290. Duarte Campos DF, Blaeser A, Korsten A, et al. The stiffness and structure of three-dimensional printed hydrogels direct the differentiation of mesenchymal stromal cells toward adipogenic and osteogenic lineages. Tissue Eng Part A. 2015;21(3–4):740–756. doi:10.1089/ten.TEA.2014.0231
291. Mishra D, Bhunia BK, Banerjee I, et al. Enzymatically crosslinked carboxymethyl–chitosan/gelatin/nano-hydroxyapatite injectable gels for in situ bone tissue engineering application. Mater Sci Eng. 2011;31:1295–1304. doi:10.1016/j.msec.2011.04.007
292. Qasim M, Chae DS, NYJIjon L. Advancements and frontiers in nano-based 3D and 4D scaffolds for bone and cartilage tissue engineering. Int J Nanomedicine. 2019;14:4333. doi:10.2147/IJN.S209431
293. Christy PN, Basha SK, Kumari VS. Nano zinc oxide and nano bioactive glass reinforced chitosan/poly(vinyl alcohol) scaffolds for bone tissue engineering application. Mater Today Commun. 2022;31:103429. doi:10.1016/j.mtcomm.2022.103429
294. Han J, Ma B, Liu H, et al. Hydroxyapatite nanowires modified polylactic acid membrane plays barrier/osteoinduction dual roles and promotes bone regeneration in a rat mandible defect model. J Biomed Mater Res A. 2018;106(12):3099–3110. doi:10.1002/jbm.a.36502
295. Alkhursani SA, Ghobashy MM, Alshangiti DM, et al. Plastic waste management and safety disinfection processes for reduced the COVID-19 Hazards. Int J Sustain Eng. 2023;16(1):1–14. doi:10.1080/19397038.2023.2188396
296. Alkhursani SA, Ghobashy MM, Al-Gahtany SA, et al. Application of nano-inspired scaffolds-based biopolymer hydrogel for bone and periodontal tissue regeneration. Polymers. 2022;14(18):3791. doi:10.3390/polym14183791
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
