Back to Journals » Integrated Blood Pressure Control » Volume 15
Low Subjective Cardiovascular Disease Risk Perceptions among Hypertensive Patients in Addis Ababa, Ethiopia
Authors Bekele DM , Goshu DY, Yalew AW, Higgins MK, Gary RA
Received 4 May 2022
Accepted for publication 27 July 2022
Published 5 August 2022 Volume 2022:15 Pages 81—96
DOI https://doi.org/10.2147/IBPC.S370838
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Turgay Celik
Daniel Mengistu Bekele,1 Dejuma Yadeta Goshu,2 Alemayehu Worku Yalew,3 Melinda K Higgins,4 Rebecca A Gary4
1Department of Nursing, School of Nursing and Midwifery, College of Health Sciences, Addis Ababa University, Addis Ababa, Ethiopia; 2Department of Internal Medicine, School of Medicine, College of Health Sciences, Addis Ababa University, Addis Ababa, Ethiopia; 3Department of Epidemiology and Biostatistics, School of Public Health, College of Health Sciences, Addis Ababa University, Addis Ababa, Ethiopia; 4Nell Hodgson Woodruff School of Nursing, Emory University, Atlanta, GA, USA
Correspondence: Daniel Mengistu Bekele, Department of Nursing, School of Nursing and Midwifery, College of Health Sciences Addis Ababa University, P. O. Box 4412, Addis Ababa, Ethiopia, Tel +251-911119597, Email [email protected]
Background: Accurate cardiovascular disease (CVD) risk appraisal is essential for hypertensive patients to identify correctly their risk status and take efficient behavioral measures timely to avoid major adverse outcomes. However, hypertensive patients’ risk perceptions of CVD events in Ethiopia are unknown. Thus, the study aimed to compare the subjective CVD risk perception level of patients with the nonlaboratory Framingham Risk Score (nl-FRS).
Methods: A cross-sectional design was used. The Attitudes and Beliefs about Cardiovascular Disease Risk Questionnaire and the nl-FRS were used to compare subjective versus objective measures of CVD risk. Agreement between participants’ risk perceptions and the nl-FRS were examined using the kappa statistic. Bivariate chi-square test and multinomial logistic regression analyses were run to identify factors associated with risk perceptions. The statistical significance was set at a p-value < 0.05 level.
Results: Participants (n=377) had a mean age of 53.61 ± 12.80-years, range (18– 82 years), 51.2% were males, 42.7% had less than high school education, 45.1% achieved target BP control, and mean HTN duration was 8.01 ± 6.07 years. The majority (58.62%) of the participants had a low subjective risk perception of CVD events (mean 17.79, 95% CI: 17.43– 18.15). Approximately three-fourths (72.4%) had a moderate nl-FRS risk calculation (mean, 13.84, 95% CI: 13.36– 14.33). Agreement between participants perceived-risk and the nl-FRS was poor (kappa = 0.0002, standard error = 0.023, p =0.99). Participants’ CVD risk-perception inaccuracy was also high (76%) primarily due to underestimation. Hypertension duration, frequency of physician visits, and level of diabetes control were significant predictors of CVD risk underestimation.
Conclusion: Hypertensive patients had inaccurate and low subjective risk perceptions of CVD events compared to moderate objective risks identified using the nl-FRS. Planned education on HTN and CVD risk factors is essential to improve patients’ CVD risk perception to reduce adverse CVD events.
Keywords: hypertension, risk-perception, CVD
Background
Adverse cardiovascular disease (CVD) outcomes such as stroke and acute myocardial infarction (AMI) are serious and often life-threatening occurrences related to uncontrolled hypertension (HTN). Cardiovascular disease remains the single largest cause of death and disabilities worldwide with over 17 million deaths each year, representing nearly 30% of all deaths and 10% of the global disease burden (GDB).1–4 Over 80% of these deaths occur in low and middle-income countries (LMICs).3,5,6 Of the 17 million CVD worldwide deaths each year, HTN is estimated to cause more than half (9.54 million) of these deaths.4,6–8 Hypertension is the strongest contributor to adverse CVD outcomes (such as stroke, coronary heart disease [CHD], heart failure [HF], end-stage renal disease, and peripheral vascular disease.9–15 Of the major cardiovascular (CV) events; 51% and 45% of stroke and CHD deaths, respectively, occurred due to HTN.6 The prevalence of HTN has remained high and approximately 30% of adults worldwide currently live with HTN.16 In Ethiopia, the prevalence of HTN is estimated to be between 13% - 30%,17–25 which would likely be higher if the more recent blood pressure (BP) risk classification 130/80 by the Joint National Committee (JNC 8) was applied.26
According to the Framingham Heart Study, individuals who are normotensive at age 55 years (men) and 65 years (women) have a 90% lifetime risk for developing HTN.27 Because longevity is increasing in LMICs28 due to several factors, it is expected that will be a dramatic rise in the number of persons with hypertension who are at risk for adverse CVD events and outcomes over the next several decades.29–32
Studies have consistently reported that hypertensive patients often have inaccurate subjective risk perceptions of adverse CVD event33,34 and underestimate their risk when compared to objective measures such as the FRS.35–37 For example, despite an increased risk of CVD events from comorbidities such as diabetes and unhealthy lifestyle behaviors (smoking), hypertensive patients were unable to provide an accurate risk estimate33,38,39 even in high-income countries (HICs) such as the Netherlands,33 US,40 and Switzerland.41
Research on CVD risk perceptions in Africa is very limited. A study by Ajayi et al in Nigeria showed the poor perception of stroke risk among hypertensive patients37 which is similar to several studies conducted in HICs.33,35,42 Studies examining subjective self-risk perceptions of CVD outcomes among hypertensive patients are currently non-existent in Ethiopia and are also scarce in Africa. Inaccurate CVD risk perceptions may also hinder behavioral strategies shown to be effective for high BP control resulting in higher CVD mortality rates. The primary aim of the current study was to compare hypertensive patients’ subjective risk perceptions with an objective measure of CVD risk using the nl-FRS 10-year CVD risk score. A secondary aim was to determine the accuracy of subjective CVD risk perceptions among adult hypertensive participants attending follow-up care at a cardiology tertiary care outpatient clinic in Addis Ababa, Ethiopia.
Methods and Materials
Study Area, Design, and Period
A cross-sectional study was conducted between February and August 2020 among 384 adult hypertensive participants attending follow-up care at a large, academic, and tertiary care outpatient clinic in Addis Ababa, Ethiopia.
Eligibility Criteria
The inclusion criteria were: 1) 30 years of age or older, 2) a minimum of three-follow-up visits in the hypertension clinic, 3) able to hear and respond appropriately to questions in the local language (Amharic), 4) not pregnant, 5) no history of CVD event, 6) no major active Axis I psychiatric condition or cognitively impaired, and 7) no history of a terminal illness such as cancer.
Sample Size Determination and Sampling Technique
The sample size was determined based on one sample t-test (as the below formula) with the assumptions of observations are independent; data fit the normal distribution; homogeneity of variance and the power set at 0.80 (β=0.2) and α (two-sided) set at 0.05 and standardized effect size of 0.15 (small)43 and considering a further 10% non-response rate for potential missing data due to the interview format.
Where Zα/2 = 1.96 (from Z table) at type 1 error of 5%
Zβ = Z0.20 = 0.842 (from Z table) at 80% power
ES2 = effect size (small, 0.15)
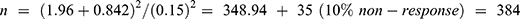 We used the hypertensive patient’s clinic follow-up registry that had the list of 1200 patients on follow-up to obtain the representative sample and recruited 384 participants for the study using the systematic random sampling technique.
We used the hypertensive patient’s clinic follow-up registry that had the list of 1200 patients on follow-up to obtain the representative sample and recruited 384 participants for the study using the systematic random sampling technique.
Data Collection Technique and Procedure
Potential hypertensive participants were approached by Bachelor’s degree qualified trained data collectors after receiving their routine follow-up care from their clinic providers. Two data collectors and one supervisor, who were trained for two days on data collection and mock exercised the interview technique and physiologic measurement procedures collected the data in a prearranged private room by a face-to-face interviewer-administered using the standardized instruments. To ensure the data quality, the instruments were translated from their original English language to the local language (Amharic) and then back to English by professional translators; the translations were very consistent. The Amharic version was used for the data collection. The instruments were piloted on 10% of the Ethiopian hypertensive patient sample at a similar tertiary level hospital to determine potential participants’ understanding of questionnaire contents. Additionally, the principal investigator monitored the data collection process and checked for data completeness, consistency, and accuracy daily.
Ethical Clearance and Consent of Participants
The study was approved by the institutional review board (IRB) of the College of Health Sciences of Addis Ababa University (AAU) (IRB protocol # 09/81) and complied with the Declaration of Helsinki. All participants reviewed and provided written consent before any study activities.
Measurements and Operational Definition
Sociodemographic and clinical variables included: age, gender, marital status, educational status, and income. The clinical and health status profile included: a history of the previous hospitalization related to CVD, co-morbidities, family history of HTN, antihypertensive and other medications, lifestyle factors such as smoking, dietary intake (number of daily fruits and vegetables), and physical activity (average time spent in moderate-intensity activities). Physiological measurements included BP measurement, weight, and height.
ABCD Risk Questionnaire: Participants’ subjective self-risk perception of CVD events was measured by the ABCD Risk Questionnaire, an 8-item questionnaire that has internal consistency reliability of 0.85.44–46 Permission to use the questionnaire was obtained from the British Medical Journal Publishing Group Ltd. The scores on the questionnaire range from 0-to 32, with higher scores showing that patients perceive a greater risk of having an adverse CVD event (a heart attack and stroke). For interpretations, participants’ total scores were categorized based on percentiles using the SPSS statistical software as low (0–17.00); moderate (17.10–19.00); and high (19.10 −32.0) for their subjective risk perceptions.
Non-laboratory-based risk estimations (nl-FRS): The Framingham 10-year CVD risk estimation uses both laboratory and non-laboratory-based risk estimations. Studies have shown that the two methods are highly correlated (r = 0.88 to 0.986) in terms of risk assessment and that the nl-FRS predicted CVD outcomes as accurately as laboratory tested.45,46 Although no studies have validated the nl-FRS in the Ethiopian population, it has been validated in South Africa.46 The nl-FRS uses the same clinical measurement parameters as the FRS (sex, age, SBP, diabetes, current smoking, antihypertensive medication), and the lipid laboratory values are replaced by BMI. In the present study, we calculated the nl-FRS based on sex, age, SBP (treated), presence or absence of diabetes, smoking status, and BMI.47 Then, we categorized participants’ CVD event risk using the conventional threshold of CVD risk stratifications as low (<10%), moderate (10–19%), and high risks (≥20%),47,48 which is consistent with the original FRS instrument.
Data Analysis
We analyzed the data after entering coded data into Epidata version 3.1 and cleansed and then exported it to SPSS version 25.00 statistical software package. Descriptive univariate analysis was run to examine the data and its plausible values, and assumptions of the logistic regression model. Perceived subjective risk and objective nl-FRS were examined using descriptive statistics, computed for the composite scores, and percentiles determined. Accordingly, patients were placed on three ordinal risk statuses as low, moderate, and high CVD risk. The levels of agreement between subjective perceptions and objective nl-FRS risk scores were calculated using Cohen’s Kappa statistics. Participants were categorized as: underestimation (low subjective CVD risk compared to the nl-FRS), agreement (subjective CVD risk agrees to the nl-FRS), and overestimation (subjective CVD risk is higher than nl-FRS). Bivariate chi-square test and multinomial logistic regression analyses were used to identify predictor variables associated with the risk perceptions (outcome variable). Variables with p-values < 0.3 in the bivariate chi-square test were considered in the final multinomial logistic regression model, and finally variables with a p-value of < 0.05 level were considered statistically significant. Odds ratio (OR) at 95% CI was considered to declare that there exist the predictor variable effects on the outcome variable. The findings are summarized below.
Results
Participants enrolled (n= 377) were predominately male (51.2%, n= 193) with a mean age of 53.61 ± 12.80 years, (range, 30–82 years) (Table 1). Three-fourths (75.3%) were married, 42.7% had less than 8th-grade education, 47.5% had a monthly income between 3001–6000 ETB (~85–171 USD), and 38.15% had a family history of HTN. Nearly three-quarters (72.10%) of them were attending follow-up care every two months for HTN. Only less than half (45.1%) achieved target BP control though the mean (134.18 ± 16.02 mmHg) SBP measurement showed patients had a BP level within the recommended range based on the current local and international HTN management guidelines.49–51 Over half (56.23%) of the participants had comorbidities; diabetes was the most common (48.11%). Over three-fourths (76.7%) reported their physician providers were the main source of information regarding the seriousness of HTN. Overweight and obesity were common with 60% having a mean BMI of 26.02 ± 4.09 Kg/M2. The mean and median duration of HTN was 8.01 ± 6.07 and 6.00 years, respectively (range, 1–30 years). The sociodemographic and clinical characteristics of participants are shown in Table 1 below.
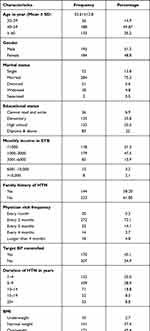 |
Table 1 Sociodemographic and Clinical Characteristics of Adult Hypertensive Patients Attending CV Follow-Up at a Tertiary Outpatient Clinic in Addis Ababa, Ethiopia, 2020 (n=377) |
The mean CVD risk perception was 17.79 (range 8–32), 95% CI: 17.43–18.15, showing the majority (58.62%) of the participants had a low subjective self-risk perception of having an adverse CVD event. In addition, based on the nl-FRS, 72.4% were at moderate risk (mean 13.84, 95% CI: 13.36–14.33, range 1–25) for experiencing an adverse CVD event over the next 10 years. The nl-FRS risk estimates (low, moderate, and high) are depicted in Figure 1.
The inter-observation reliability of Cohen’s Kappa statistics agreement (Table 2) between participants’ perceived and the nl-Framingham risk estimate showed no agreement between them (k= 0.0002, SE=0.023, p = 0.99). Overall, over three-quarters of the participants had inaccurate subjective perceptions of CVD risk levels, with most underestimating their risk. The objective risk using the nl-FRS, however, showed a moderate level of risk was present.
The bivariate chi-square test analyses of independent variables, the frequency of physician visits for HTN, and the duration of HTN were significantly associated with risk perceptions (Table 3). However, there were no observed associations between risk perceptions and sociodemographic or clinical variables.
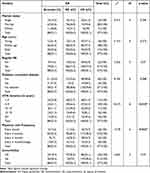 |
Table 3 Bivariate Chi-Square Test Showing the Association Between Risk Perception Category (Kappa Agreements: Accurate, OE, and UE) and Independents Variables N = 377 |
All independent variables with p-values less than 0.3 were considered in the final multinomial logistic regression analysis model to test their association and strength of the association with the risk perception category. All the model data assumptions were checked with no violations identified. Multinomial logistic regression analyses (Table 4) showed inaccurate (overestimated) risk perception that was significantly associated with frequency of physician visits every two and three months, controlled diabetes, and HTN duration of 10–14 years. Similarly, inaccurate (underestimated) risk perception was significantly associated with frequency of physician visits every three months, not engaged in PA, HTN duration of 5–9 years, and the HTN duration of 10–14 years.
Patients who had the clinic follow-up every two and three months overestimated their CVD risk four to six-fold (OR: 4.34, 95% CI: 1.29–14.57, p = 0.018 and OR: 6.33, 95% CI: 1.48–27.03, p = 0.013), respectively compared to those who had the four months or longer. Patients with controlled diabetes were 90% less likely to overestimate their CVD risk (OR: 0.10, 95% CI: 0.01–0.86, p = 0.036) compared to those with no history of diabetes. Physician visits every three months were associated with a three times greater likelihood (OR: 3.21, 95% CI: 1.03–10.02, p = 0.045) to perceive lower CVD risk compared to those with the frequency of visits every four months or longer.
Patients who did not engage in regular PA tended to underestimate almost three times (OR: 2.58, 95% CI: 1.13–3.94, p = 0.025) their CVD risk compared to those engaged in regular PA. Patients with HTN duration of 5–9 years and 10–14 years tend to underestimate their CVD risk about three times and six times (OR: 3.17, 95% CI: 3.18–8.59, p = 0.023 and OR: 6.35, 95% CI: 2.00–20.14, p = 0.002, respectively) compared to those with HTN duration of 20 years or more.
Discussion
The primary aim of the study was to compare subjective perceptions with an objective measure of CVD risk using the nl-FRS among adult hypertensive patients attending follow-up care at a tertiary outpatient clinic in Addis Ababa, Ethiopia. The major findings of the study were low subjective risk perception (underestimation) of CVD events, moderate risk of future CVD adverse events based on the nl-FRS, and lack of agreement between participants’ subjective perceptions and an objective nl-FRS measure.
The mean CVD risk perception suggests most participants had low subjective risk perceptions. Studies measuring risk perceptions among hypertensive patients are limited in Ethiopia. Several studies that have examined hypertensive patients’ risk perceptions in other countries, however, have reported similar findings with 46%52 and 41%53 of participants having low CVD risk perceptions of adverse events. Several other HTN studies also reported comparable findings (64–65%) of low CVD risk perceptions.54,55 Although plenty of studies since Weinstein (1980)56 refer risk underestimation to as “optimistic bias”,53,56–58 which is an error in an individual”s judgment tending to rate oneself above average for positive events and below average for the negatives,56 risk perceptions are threat specific, rather than reflecting a general sense of optimism or pessimism.59 The finding of low CVD risk perception in this population in Ethiopia is very concerning since all patients were receiving care at a large academic tertiary care level clinic where they are more likely to be informed about risk factors being referred from smaller, community-level hospitals.
The finding that many participants had low rates of BP control (45.1%) and were not engaged in CVD reduction behaviors (52.52%) may be attributed to a lack of knowledge. Only a small proportion of participants perceived their CVD risk at moderate (14.6%) and high (26.8%) levels. This differed from the study of Lefler et al who showed rural, lower socioeconomic residents had higher CVD risk perceptions53 in a small sample of older age patients. Participants in the current study lived in a large urban area and were younger but most also had a lower education level, which may be a key factor in patients being unable to correctly perceive their CVD risks. Further studies are needed to determine which strategies work best among individuals with low educational levels to improve CVD risk knowledge and perceptions.
Based on the nl-FRS, nearly three-quarters of participants were at moderate CVD risk. Although the discussion of this finding was restricted by the lack of studies in Ethiopia for comparison, the findings were inconsistent with studies in Brazil and South Korea that found most participants to be at low CVD risk based on the nl-FRS (77.9% and 54.2%, respectively).55,60 The differences may be due to demographic variations of risk factors among participants across countries. For example, the mean age in the present study was higher than in Brazil and South Korean studies.55,60 Interestingly, those who were at high (10.6%) CVD risk in the present study were comparable to previous studies finding 7.7%55 and 12.6%.60 However, this is far less when compared to the proportion (41%) of patients with other chronic conditions such as rheumatoid arthritis who misperceived their CVD risk mainly of the risk overestimation,61 which may be due to the physical limitations posed on those patients by the disease.
The subjectively-perceived CVD risk kappa agreement or accuracy with the nl-FRS 10-year CVD risk estimated was poor (k = 0.002, p = 0.99) with a low rate (23.34%) of accuracy (Table 2), which is alarming for patients receiving long-term follow-up care at a tertiary clinic. In several previous studies, rates of patients who had accurate risk perceptions were consistently low and comparable with the present finding.62–65 However, some prior studies have documented higher rates of agreement between patients’ risk perceptions and their laboratory FRS52,55 than the current study findings. For instance, compared to the present study, most participants in the Katz et al 201555 study were higher in socioeconomic status and patients with lower socioeconomic status were also found to have less accurate CVD risk perceptions.66 The majority of our study participants had inaccurate risk-perception of CVD risk, mainly due to CVD risk underestimation, which is also consistent with several studies that reported poor risk-perceptions agreement between patients’ perceived and their FRS 10-year CVD risk calculated with tendencies for optimism (risk underestimation).5,52,54,61–73 The finding of consistently low agreements between patients’ subjectively perceived and objectively predicted CVD risk in the present and several previous studies’ findings may signal the importance of appraising patients’ CVD risk factors. The paramount importance of studies of risk perceptions is their ability to uncover individuals’ risk appraisal of a disease threat or a health condition and the ability to self-evaluate and construe its potential health consequences.59,74 This is apparent from the Health Belief Model (HBM), which has been widely used in health and behavioral research, that the concept of individual perception of susceptibility and seriousness of the disease threat is recognized as the key components for optimal behavior changes.75 That means perceptions of risk were conditional factors for behavioral change to occur, but alone were not sufficient for individuals to engage to the recommended actions.76,77 It is generally believed that an individual seeks preventive care including health screening if he/she has sufficient motivation for the health issue of concern. On the other hand, this motivation is enhanced if he/she believes that he/she is susceptible (perceived susceptibility) to a serious health condition or its aftermaths (perceived severity) and that undertaking the recommended preventive behavioral action will avert or mitigate the known risk, taking into account the relative advantages of the actions to be taken over barriers to actions.73,75 Thus, Lowering CVD risk through lifestyle changes and the avoidance of adverse events requires an accurate estimation of CVD risk.
The FRS laboratory and non-laboratory-based results’ comparability is limited in Africa to one study,78 but has been compared in other countries with acceptable reliability.46 Validated CVD risk tools are currently lacking in Ethiopia and the eastern sub-Saharan Africa population. Studies of CVD clinical characteristics using the FRS (including Africa) showed a high correlation between non-laboratory-based and laboratory-based scores (r = 0.88–0.986), the nl-FRS predicted CVD outcomes as accurately as a laboratory.45,46 The use of the nl-FRS is ideal in LMICs like Ethiopia where laboratory assessments are cost-prohibitive since most tests are not covered by insurance, requiring out-of-pocket payment and there is a lack of available laboratories, especially in rural geographic areas.
Participants who had the clinic visits every two and three months overestimated (Table 4) their CVD risk by four and six-fold respectively, compared to those who visited less often. Patients may perceive their HTN as more of a health threat requiring more attention when they were put on a more frequent clinic schedule. CVD risk overestimation is thought to be more health-protective and preferred compared to underestimation,79 since it may trigger patients to participate in positive healthy behaviors to reduce the likelihood of adverse CVD outcomes. It is also possible that those with more frequent clinic visits received a higher level of provider support concerning risk reduction behaviors (diet, PA, body weight, medication adherence including monitoring of side effects and dose adjustments), which may translate to the adoption of healthier behaviors, improved blood pressure control and in turn lower CVD risk.
Hypertensive patients with controlled diabetes (Table 4) were 90% less likely to overestimate their CVD risk compared to those with no history of diabetes. Previous studies have shown that patients with a history of diabetes had higher CVD risk-perception66,69 and in some studies overestimated their CVD risk.33 It is well established that Type II diabetes mellitus (T2DM) is a strong risk factor for the development of CVD. The reason for higher risk perception may be related to ongoing provider communication of the likelihood of developing heart disease in the presence of T2DM. It is also possible that family members or significant others who developed CVD because of T2DM received or requested more targeted education on risk reduction.
In the present study, hypertensive patients with a duration of 10–14 years overestimated their CVD risk as nearly four times higher than those with a 20-year or greater history. This was consistent with a study in Nepal that reported a significant association between HTN duration of 10–15 years and overestimation of CVD risk.80 In the present study, the duration of HTN exhibited a U-shaped pattern with 10–14 years as the peak, which suggests the importance of disease over time on CVD risk perceptions. In the original Framingham Heart Study, the progression of HTN to target organ diseases (TOD) such as HF and other CVD complications had a mean of 14 years.81 In comparison, over 78 months, in a retrospective follow-up study of hypertensive patients in Gondar (Ethiopia) that examined the progression of HTN, CVD complications were present at the time of diagnosis,82 which are likely due to late clinical presentation of HTN and/or late health-seeking behavior of the patients and reinforces why HTN is called “the silent killer”.83 In addition, the study showed lower rates of HTN complications in the first three years of treatment but after seven years, the risk of developing complications increased threefold.82 Poor HTN control, low treatment adherence rates, and late diagnosis in Ethiopia may explain the differences in clinical presentation and outcomes. Treatment of CVD is not necessarily associated with reduced risk. A retrospective, large cohort study (n = 3150) of adults in Brazil followed for two years to examine the progression of CVD risk using the FRS showed those classified as moderate to high risk at year one follow-up showed a 31% increase in CVD risk at year two.84 In addition, 66% of those who were classified as high CVD risk in the first assessment did not exhibit improved risk status at year two follow-up.84 Specifically, the Brazil study showed poorer serum lipid levels and no significant mean BP changes.84 Regular risk assessments that integrate patient education and self-management strategies are essential to reduce the potential adverse CVD outcomes among hypertensive patients. In the present study, the finding of significant relationships between participants’ underestimation and overestimation of subjective risk perceptions with HTN duration and frequency of physician visits was a counterintuitive finding, which may be encountered due to prevalent poor CVD risk perceptions.85
The present findings of prevalent subjective CVD risk underestimation and poor kappa agreement between the subjective and objective CVD risk measures may be related to the less common occurrence of CVD risk in the general population as CVD is in the emergent phase in Ethiopia with less coverage by media in creating public awareness on the seriousness and escalating nature of CVD, which was evidenced by participants source of information, and lack of a single lipid test for most of the participants. In this case, participants’ unrealistic CVD risk underestimation may be due to a general lack of information and awareness, which is also supported by literature that low education and literacy skills were associated with unrealistic risk underestimation.86 Regular risk assessments that integrate patient education and self-management strategies are essential to reduce the potential adverse CVD outcomes among hypertensive patients.
Strengths of the Study
To our knowledge, this is the first study to examine subjective CVD risk perceptions using self-report measures compared to an objective, nl-FRS calculation among hypertensive patients receiving outpatient tertiary level care in Ethiopia. The findings provide important insights and directions for further research and the need for large-scale national campaigns on HTN and CVD risk reduction. The sample size was large and adequately powered. The random selection of participants for the study reduced the likelihood of selection bias.
Limitations
The study was conducted at one academic referral center which limits generalization to other populations in Ethiopia. The cross-sectional design also limited the causality of the association between low-risk perceptions and objective nl-FRS measures.
Conclusions and Implications
In summary, in the present study, higher proportions of participants had low-risk perceptions despite having moderate to high objective CVD risks as determined by the nl-FRS. This shows a need for greater CVD risk factor knowledge and awareness as a national priority. In addition, primary care and community clinics are important venues to educate the public on blood pressure awareness since most of the population in Ethiopia uses these services versus academic tertiary care centers. Incorporating risk reduction strategies and the importance of managing HTN to prevent CVD adverse outcomes should begin early with risk assessment beginning in adolescents and young adults.
The findings suggest that most hypertensive patients underestimated their CVD risk and were at moderate risk for CVD events when compared to an objective measure, the nl FRS. The study established a clear need for education on CVD risk reduction strategies that will enable hypertensive patients to more accurately recognize CVD risk factors that would improve outcomes in this rapidly emerging population in Ethiopia.
Abbreviations
AAU, Addis Ababa University; ABCD, attitude and beliefs about cardiovascular disease; AMI, acute myocardial infarction; AOR, adjusted odds ratio; BMI, body mass index; BP, blood pressure; CHD, coronary heart disease; CI, confidence interval; CKD, chronic kidney disease; CV, cardiovascular; CVD, cardiovascular disease; ETB, Ethiopian Birr; FRS, Framingham Risk Score; GDB, global disease burden; HF, heart failure; HHD, hypertensive heart disease; HICs, high income countries; HTN, hypertension; IRB, institutional review board; JNC, Joint National Committee; K, Kappa; KA, kappa statistics; Kg, kilogram; LMICs, low and middle-income countries; M2, meter square; mmHg, millimeter of mercury; MNLR, multinomial logistic regression; ni-FRS, non-laboratory Framingham Risk Score; OE, overestimation; OR, odds ratio; AOR, adjusted odds ratio; PA, physical activity; SBP, systolic blood pressure; SE, standard error; T2DM, type 2 diabetes; TOD, target organ disease; UE, underestimation; US, United States; USD, United States” Dollar.
Data Sharing Statement
The original data supporting the findings of this manuscript can be accessed from the primary author upon a reasonable request.
Ethical Approval and Informed Consent
The study was approved by the institutional review board (IRB) of the College of Health Sciences of Addis Ababa University (AAU) (IRB protocol # 09/81) and complied with the Declaration of Helsinki. All participants reviewed and provided a signed consent before any study activities.
Consent for Publications
All the study participants have approved reporting the results of the research to be published in journals and conference presentations.
Acknowledgments
We thank the BMJ Publishing Group Ltd for permitting us to use the Attitudes and Beliefs about Cardiovascular Disease (ABCD) risk questionnaire. The research team would like to acknowledge Addis Ababa University for funding for the data collection of this research. We thank the study participants for their time.
Funding
Addis Ababa University has funded for data collection of this research.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bloom D, Cafero E, Jané-Llopis E, Abrahams-Gessel S, Bloom L, Fathima S. The global economic burden of non-communicable diseases: world economic forum and Harvard school of public health; 2011.
2. Bromfield S, Mounter P. High blood pressure: the leading global burden of disease risk factor and the need for worldwide prevention programs. NIH Public Access. 2013;15(3):134–136. doi:10.1007/s11906-013-0340-9.High
3. Mendis S, Puska P, Norrving B. Global Atlas on Cardiovascular Disease Prevention and Control. Geneva: World Health Organization; 2011: 1–54. Available from http://whqlibdoc.who.int/publications/2011.
4. Lim SS, Vos T, Flaxman AD, Danaei G, Shibuya K, Adair-Rohani H. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: a systematic analysis for the global burden of disease study 2010. Lancet. 2012;380(9859):2224–2260. doi:10.1016/S0140-6736(12)61766-8.A
5. Lawes CM, Hoorn S, Rodgers A; International Society of Hypertension. Global burden of blood-pressure-related disease, 2001. Lancet. 2008;371(9623):1513–1518. doi:10.1016/S0140-6736(08)60655-8
6. World Health Organization. A global brief on Hypertension: silent killer, global public health crisis world health day 2013. World Heal Organ. 2013;1–40. doi:10.1136/bmj.1.4815.882-a
7. World Health Organization. Global health risks. World Health Organization; 2009.
8. Campbell N, Lackland D, Niebylski M. High blood pressure: why prevention and control are urgent and important. A 2014 fact sheet from the world hypertension league and the international society of hypertension increased blood pressure is the leading risk factor for death and for disability. Int Soc Hypertens. 2014;16(1):5.
9. Chen G, Mcalister F, Walker R, Hemmelgarn B, Campbell N. Cardiovascular outcomes in Framingham participants with diabetes. The importance of blood pressure. Hypertension. 2011;57(5):891–897. doi:10.1161/HYPERTENSIONAHA.110.162446
10. Yau JA, Karaye KM, Okeahialam BN. Profile of cardiovascular risk factors in Nigerians with stroke. World J Cardiovasc Surg. 2016;06:125–137. doi:10.4236/wjcs.2016.610020
11. Alkali N, Bwala S, Akano A, Osi-Ogbu O, Peter Alabi P, Ayeni O. Stroke risk factors, subtypes, & 30-day case fatality in Abuja, Nigeria. Niger J Med. 2013;54(2):129–135. doi:10.4103/0300-1652.110051
12. Watila MM, Nyandaiti YW, Ibrahim A, et al. Risk factor profile among black stroke patients in Northeastern Nigeria. J Neurosci Behav Health. 2012;4:50–58. doi:10.5897/JNBH11.052
13. Obiako OR, Oparah SK, Ogunniyi A. Prognosis and outcome of acute stroke in the University College Hospital Ibadan, Nigeria. Niger J Clin Pract. 2011;14(3):5–8. doi:10.4103/1119-3077.86784
14. O’Donnell M, Xavier D, Liu L, Zhang H, Chin S, Rao-Melacini P. Risk factors for ischaemic and intracerebral haemorrhagic stroke in 22 countries (the INTERSTROKE study): a case-control study. Lancet. 2010;376(9735):112–123. doi:10.1016/S0140-6736(10)60834-3
15. World Health Organization. Clinical Guidelines for the Management of Hypertens. Cairo: World Health Organization Regional Office for the Eastern Mediterranean; EMRO Technical Publications Series 29; 2005.
16. World Health Organization. Noncommunicable diseases in the South-East Asia region: situation & response; 2011.
17. Gudina EK, Michael Y, Assegid S. Prevalence of hypertension and its risk factors in southwest Ethiopia: a hospital-based cross-sectional survey. Integr Blood Press Control. 2013;6:111. doi:10.2147/IBPC.S47298
18. Abebe SM, Berhane Y, Worku A, Getachew A. Prevalence and associated factors of hypertension: a crossectional community based study in Northwest Ethiopia. PloS one. 2015;241:1–11. doi:10.1371/journal.pone.0125210
19. Bonsa F, Gudina EK, Hajito KW. Prevalence of hypertension and associated factors in Bedele Town, Southwest Ethiopia. Ethiop J Heal Sci. 2014;24(1):21–26. doi:10.4314/ejhs.v24i1.3
20. Helelo TP, Gelaw YA, Adane AA. Prevalence and associated factors of hypertension among Adults in Durame. PloS one. 2014:1–9. doi:10.1371/journal.pone.0112790
21. Angaw K, Dadi AF, Alene KA. Prevalence of hypertension among federal ministry civil servants in Addis AbabaEthiopia: a call for a workplace-screening program. BMC Cardiovasc Disord. 2015;15(1):1–6. doi:10.1186/s12872-015-0062-9
22. Kibret KT, Mesfin YM. Prevalence of hypertension in Ethiopia: a systematic meta-analysis. Public Health Rev. 2015;36. doi:10.1186/s40985-015-0014-z
23. Tesfaye F Epidemiology of cardiovascular disease risk factors in Ethiopia: the rural-urban gradient fikru Tesfaye; 2008.
24. Ethiopian Public Health Institute. Ethiopian STEPS report on risk factors for non-communicable diseases. Ethiopia; 2016:1–13. Available from: https://www.ephi.gov.et/images/pictures/download2009/NCD.
25. World Health Organization. Non communicable diseases country profiles. WHO Press. 2014;1–210. doi:10.1111/jgs.12171
26. Whelton P, Carey RM, Aronow WS, et al. High blood pressure clinical practice guideline. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA: guideline for the prevention, detection, evaluation, and management of high blood pressure in adults. Rep Am Coll Cardiol. 2017. doi:10.1161/HYP.0000000000000065/-/DC1.The
27. Vasan RS, Beiser A, Seshadri S, et al. Residual lifetime risk for developing hypertension in middle-aged women and men the Framingham heart study. Am Med Assoc. 2002;287(8):1003–1010. doi:10.1001/jama.287.8.1003
28. Sudharsanan N, Bloom DE The demography of aging in low- and middle-income countries: chronological versus functional perspectives - nikkil sudharsanan and David e. Bloom. 2018:309–338. Available from: https://mgetit.lib.umich.edu/resolve?ctx_ver=Z39.88-2004&ctx_enc=info%3Aofi%2Fenc%3AUTF-8&rfr_id=info%3Asid%2Fsummon.serialssolutions.com&rft_val_fmt=info%3Aofi%2Ffmt%3Akev%3Amtx%3Abook&rft.genre=bookitem&rft.title=Future+Directions+for+the+Demography+of+.
29. Kulkarni A, Mehta A, Yang E, Parapid B. 2017 guideline for the prevention, detection, evaluation and management of high blood pressure in adults. Am Hear Assoc. 2008;58(10):1117–1129.
30. Muli S, Meisinger C, Heier M, Thorand B, Peters A, Amann U. Prevalence, awareness, treatment, and control of hypertension in older people: results from the population-based KORA-age 1 study. BMC Public Health. 2020;20(1):1–10. doi:10.1186/s12889-020-09165-8
31. Oliveros E, Patel H, Kyung S, et al. Hypertension in older adults: assessment, management, and challenges. Clin Cardiol. 2020;43(2):99–107. doi:10.1002/clc.23303
32. Shukuri A, Tewelde T, Shaweno T. Prevalence of old age hypertension and associated factors among older adults in rural Ethiopia. Integr Blood Press Control. 2019;12:23–31. doi:10.2147/IBPC.S212821
33. Frijling B, Lobo C, Keusa M, Jenksa M, Akkermansa P, Hulscher L. Perceptions of cardiovascular risk among patients with hypertension or diabetes. Patient Educ Couns. 2004;52(1):47–53. doi:10.1016/S0738-3991(02)00248-3
34. Rodrígez García JM. Scientia potestas est – knowledge is power: Francis Bacon to Michel Foucault. Neohelicon. 2001;28(1):109–121. doi:10.1023/A:1011901104984
35. Powers BJ, Oddone EZ, Grubber JM, Olsen MK, Bosworth HB. Perceived and actual stroke risk among men with hypertension. J Clin Hypertens. 2008;10(4):287–294. doi:10.1111/j.1751-7176.2008.07797.x
36. Everett B, Salamonson Y, Rolley JX, Davidson PM. Underestimation of risk perception in patients at risk of heart disease. Eur J Cardiovasc Nurs. 2016;15(3):e2–9. doi:10.1177/1474515114556712
37. Ajayi A, Ojo O. Knowledge and perception of stroke among at risk medical out-patients in a tertiary health institution in Nigeria.Ann. Afr Med. 2007;6(2):51–53. doi:10.4103/1596-3519.55717
38. Mohd Azahar NMZ, Krishnapillai ADS, Zaini NH, Yusoff K. Risk perception of cardiovascular diseases among individuals with hypertension in rural Malaysia. Heart Asia. 2017;9(2):e010864. doi:10.1136/heartasia-2016-010864
39. Vörös K, Márkus B, Hargittay C, Sisak O, Kalabay L. Perceived cardiovascular risk and attitude towards lifestyle change. Fam Pract. 2018;35(6):712–717. doi:10.1093/fampra/cmy028
40. Barnhart JM, Wright ND, Freeman K, Correa N, Walker EA. Risk perception and its association with cardiac risk and health behaviors among urban, minority adults: the Bronx coronary risk perception study. HHS Public Access. 2015;23(5):339–342. doi:10.4278/ajhp.07072574.Risk
41. Desgraz B, Collet T-H, Rodondi N, Cornuz J, Clair C. Comparison of self-perceived cardiovascular disease risk among smokers with Framingham and PROCAM scores: a cross-sectional analysis of baseline data from a randomised controlled trial. BMJ Open. 2017;7(1):e012063. doi:10.1136/bmjopen-2016-012063
42. Foss FA, Dickinson E, Hills M. Missed opportunities for the prevention cardiovascular disease among British hypertensives in primary care. Br J General Pract. 1996;46(October1993):571–575.
43. Hulley SB, Cummings SR, Browner WS, Grady DG, Newman TB. Designing Clinical Research.
44. Woringer M, Nielsen JJ, Zibarras L, et al. Development of a questionnaire to evaluate patients’ awareness of cardiovascular disease risk in England’s National Health Service Health Check preventive cardiovascular programme. BMJ. 2017;7(9):1–10. doi:10.1136/bmjopen-2016-014413
45. Gaziano TA, Pandya A, Steyn K, et al. Comparative assessment of absolute cardiovascular disease risk characterization from non-laboratory-based risk assessment in South African populations. BMC Med. 2013;11(1):1. doi:10.1186/1741-7015-11-170
46. Gaziano TA, Young CR, Fitzmaurice G, Atwood S, Michael J. Laboratory-based versus non-laboratory-based method for assessment of cardiovascular disease risk: the NHANES I Follow-up Study cohort. Lancet. 2010;371(9616):923–931. doi:10.1016/S0140-6736(08)60418-3.Laboratory-based
47. D’Agostino RB, Vasan RS, Pencina MJ, et al. General cardiovascular risk profile for use in primary care: the Framingham heart study. Circulation. 2008;117(6):743–753. doi:10.1161/CIRCULATIONAHA.107.699579
48. Anderson TJ, Grégoire J, Pearson GJ, et al. Canadian cardiovascular society guidelines for the management of dyslipidemia for the prevention of cardiovascular disease in the adult. Can J Cardiol. 2016;32(11):1263–1282. doi:10.1016/j.cjca.2016.07.510
49. Moh E National noncommunicable diseases management protocols; 2021.
50. Unger T, Borghi C, Charchar F, et al. Clinical practice guidelines 2020 international society of hypertension global hypertension practice guidelines international society of hypertension. Hypertension. 2020;75:1334–1357. doi:10.1161/HYPERTENSIONAHA.120.15026
51. Williams B, Mancia G, Spiering W, et al. Practice guidelines for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension. Blood Press. 2018;27(6):314–340. doi:10.1080/08037051.2018.1527177
52. Barnhart JM, Wright ND, Freeman K, Correa N, Walker EA, Walker EA. Risk perception and its association with cardiac risk and health behaviors among urban, minority adults: the Bronx coronary risk perception study. Am J Heal Promot. 2009;23(5):339–342. doi:10.4278/ajhp.07072574.Risk
53. Lefler LL, Hartford JA, Fagan CM. Perceived cardiac risk among older, high-risk black and white women. Int Rev Res Open Distrib Learn. 2008;9(3):1. doi:10.19173/irrodl.v9i3.1101
54. Hussein HM, Harris-Lane P, Abdelmoula MM, Vazquez G. Accuracy of self-perception of cardiovascular risk in the community. J Vasc Interv Neurol. 2008;1(4):106–112.
55. Katz M, Laurinavicius AG, Franco FGM, et al. Calculated and perceived cardiovascular risk in asymptomatic subjects submitted to a routine medical evaluation: the perception gap. Eur J Prev Cardiol. 2015;22(8):1076–1082. doi:10.1177/2047487314543074
56. Weinstein ND. Perceived probability, perceived severity, and health-protective behavior. Heal Psychol. 2000;19(1):65–74. doi:10.1037/0278-6133.19.1.65
57. Coelho MP. Unrealistic optimism: still a neglected trait. J Bus Psychol. 2010;25(3):397–408. doi:10.1007/s10869-009-9146-9
58. Harris AJL, Hahn U. Unrealistic optimism about future life events: a cautionary note. Psychol Rev. 2011;118(1):135–154. doi:10.1037/a0020997
59. Ferrer R, Klein WM. Risk perceptions and health behavior. Physiol Behav. 2017;176(1):100–106. doi:10.1016/j.copsyc.2015.03.012.Risk
60. Ko Y, Boo S. Self-perceived health versus actual cardiovascular disease risks. Jpn J Nurs Sci. 2016;13(1):65–74. doi:10.1111/jjns.12087
61. Boo S, Oh H, Froelicher ES, Suh CH. Knowledge and perception of cardiovascular disease risk among patients with rheumatoid arthritis. PLoS One. 2017;12(4):1–12. doi:10.1371/journal.pone.0176291
62. Helou TN, Santos RD, Laurinavicius AG, et al. Association between clinical factors and self-underestimation of cardiovascular risk in subjects submitted to a routine health evaluation. Clin Cardiol. 2018;41(1):28–33. doi:10.1002/clc.22841
63. Liew SM, Lee WK, Khoo EM, et al. Can doctors and patients correctly estimate cardiovascular risk? A cross-sectional study in primary care. BMJ Open. 2018;8(2):1–6. doi:10.1136/bmjopen-2017-017711
64. Petr EJ, Ayers CR, Pandey A, et al. Perceived lifetime risk for cardiovascular disease (from the Dallas heart study). Am J Cardiol. 2014;114(1):53–58. doi:10.1016/j.amjcard.2014.04.006
65. Brawarsky P, Eibensteiner K, Klinger EV, et al. Accuracy of self-perceived risk for common conditions. Cogent Med. 2018;5(1):1463894. doi:10.1080/2331205x.2018.1463894
66. Homko CJ, Santamore WP, Zamora L, et al. Cardiovascular disease knowledge and risk perception among underserved individuals at increased risk of cardiovascular disease. J Cardiovasc Nurs. 2008;23(4):332–337. doi:10.1097/01.JCN.0000317432.44586.aa
67. DeSalvo KB, Gregg J, Kleinpeter M, Pedersen BR, Stepter A, Peabody J. Cardiac risk underestimation in urban, black women. J Gen Intern Med. 2005;20(12):1127–1131. doi:10.1111/j.1525-1497.2005.00252.x
68. Claassen L, Henneman L, Nijpels G, Dekker J, Marteau T, Timmermans D. Causal beliefs and perceptions of risk for diabetes and cardiovascular disease, the Netherlands, 2007. Prev Chronic Dis. 2011;8(6):1.
69. Thakkar J, Heeley EL, Chalmers J, Chow CK. Inaccurate risk perceptions contribute to treatment gaps in secondary prevention of cardiovascular disease. Intern Med J. 2016;46(3):339–346. doi:10.1111/imj.12982
70. Stol DM, Hollander M, Damman OC, et al. Mismatch between self-perceived and calculated cardiometabolic disease risk among participants in a prevention program for cardiometabolic disease: a cross-sectional study. BMC Public Health. 2020;20(1):1–9. doi:10.1186/s12889-020-08906-z
71. Grauman Å, Veldwijk J, James S, Hansson M, Byberg L. Good general health and lack of family history influence the underestimation of cardiovascular risk: a cross-sectional study. Eur J Cardiovasc Nurs. 2021;20:676–683. doi:10.1093/eurjcn/zvab019
72. Waters EA, Klein WMP, Moser RP, et al. Correlates of unrealistic risk beliefs in a nationally representative sample. J Behav Med. 2011;34(3):225–235. doi:10.1007/s10865-010-9303-7
73. Diaz VA, Mainous AG, Williamson D, Johnson SP, Knoll ME. Cardiovascular and diabetes risk perception in a Hispanic community sample. Ethn Dis. 2012;22(1):5–11.
74. Sheeran P, Harris PR, Epton T. Does Heightening Risk Appraisals Change People’s Intentions and Behavior? Meta-Analy Exp Stud. 2014;140. doi:10.1037/a0033065
75. Jones CL, Jensen JD, Scherr CL, Brown NR, Christy K, Weaver J. The health belief model as an explanatory framework in communication research: exploring parallel, serial, and moderated mediation. Heal Commun. 2015;30(6):566–576. doi:10.1080/10410236.2013.873363
76. Janz NK, Becker MH. The Health Belief Model: a Decade Later. High Educ Q. 1984;11(1):1–47.
77. Glanz. The Health Belief Model (HBM) Health Communication | health Belief Model; 1997. Available from: https://www.utwente.nl/en/bms/communication-theories/sorted-by-cluster/Health%20Communication/Health_Belief_Model/.
78. Boateng D, Agyemang C, Beune E, et al. Cardiovascular disease risk prediction in sub-Saharan African populations — comparative analysis of risk algorithms in the RODAM study. Int J Cardiol. 2018;254:310–315. doi:10.1016/j.ijcard.2017.11.082
79. Wallis EJ, Ramsay LE, Jackson PR. Cardiovascular and coronary risk estimation in hypertension management. Heart. 2002;88(3):306–312. doi:10.1136/heart.88.3.306
80. Bashyal SP, Thapa N. Knowledge and perception regarding hypertension among hypertensive patients at a tertiary hospital in Kathmandu, Nepal. J Adv Acad Res. 2020;7(1):51–62. doi:10.3126/jaar.v7i1.35467
81. Levy D, Larson MG, Vasan RS, Kannel WB, Ho KKL. The progression from hypertension to congestive heart failure. J Am Med Assoc. 1996;275(20):1557–1562. doi:10.1001/jama.275.20.1557
82. Abegaz TM, Tefera YG, Befekadu Abebe T. Target organ damage and the long term effect of nonadherence to clinical practice guidelines in patients with hypertension: a retrospective cohort study. Int J Hypertens. 2017;2017:1–8. doi:10.1155/2017/2637051
83. Hoel D, Howard R, Gifford RW. Hypertension-stalking the silent killer. Postgrad Med J. 1997;101:116–121. doi:10.3810/pgm.1997.02.155
84. Conceição RD, Ilgueria De O, Laurinavicius Abriel AG, et al. Check-up and cardiovascular risk progression: is there a room for innovation? Einstein. 2015;13(2):196–201. doi:10.1590/S1679-45082015AO3216
85. Ross S, Walker A, MacLeod MJ. Patient compliance in hypertension: role of illness perceptions and treatment beliefs. J Hum Hypertens. 2004;18(9):607–613. doi:10.1038/sj.jhh.1001721
86. Shepperd JA, Waters EA, Weinstein ND, Klein WMP. A Primer on Unrealistic Optimism. Curr Dir Psychol Sci. 2015;24(3):232–237. doi:10.1177/0963721414568341
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.