Back to Journals » Cancer Management and Research » Volume 13
Low-Grade Nasopharyngeal Papillary Adenocarcinoma: A Review of 28 Patients in a Single Institution
Authors Lai Y , Li W, Zhai C, Song X, Yang J, Sun X , Wang D
Received 27 October 2020
Accepted for publication 24 January 2021
Published 10 February 2021 Volume 2021:13 Pages 1271—1278
DOI https://doi.org/10.2147/CMAR.S288007
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Xueqiong Zhu
Yuting Lai,1 Wanpeng Li,1 Changwen Zhai,2 Xiaole Song,1 Jingyi Yang,1 Xicai Sun,1 Dehui Wang1
1Department of Otolaryngology, Eye and ENT Hospital of Fudan University, Shanghai, 200031, People’s Republic of China; 2Department of Clinical Pathology, Eye and ENT Hospital of Fudan University, Shanghai, 200031, People’s Republic of China
Correspondence: Dehui Wang; Xicai Sun
Department of Otolaryngology, Eye and ENT Hospital of Fudan University, No. 83 Fenyang Road, Shanghai, 200031, People’s Republic of China
Tel +86 13701852008
; +86 13681751601
Fax +86 21 64377151
Email [email protected]; [email protected]
Purpose: Low-grade nasopharyngeal papillary adenocarcinoma (LGNPPA) is a rare nasopharyngeal tumor. This study aimed to analyze the clinical and histopathological features of the disease, and to share our experience of its treatment.
Patients and Methods: We collected demographic data, clinical symptoms, tumor location, pathological features, immunohistochemical results, treatments, and outcomes of 28 patients with pathologically confirmed LGNPPA between 2009 and 2019.
Results: The median age of the 28 patients was 41.5 years, with a female: male ratio of 1.5:1 (17 females, 11 males). The most common symptom was blood-stained rhinorrhea. The neoplasms were located on the roof of the nasopharynx (RON) in 13 patients, the posterior margin of the nasal septum (PMONP) in 12 patients, the lateral wall of the nasopharynx in one case, and both the RON and PMONP in two patients. Fourteen patients were diagnosed with thyroid-like LGNPPA. Immunohistochemically, the tumors were uniformly positive for cytokeratin 7, cytokeratin 8, vimentin, epithelial membrane antigen, and pan-cytokeratin, and negative for thyroglobulin. Twenty-three patients underwent pure endoscopic surgery, three patients underwent preoperative radiotherapy, and two patients underwent radiotherapy postoperatively. All patients were alive without evidence of lymphatic or distant metastases in the follow-up period (range: 7 to 121 months). Two patients (7%, 2/28) experienced disease recurrence.
Conclusion: LGNPPA is an indolent tumor with an excellent prognosis. Endonasal endoscopic excision was an effective treatment. It is important to distinguish thyroid-like LGNPPA from metastatic papillary thyroid carcinoma because these diseases have similar microscopic features but different prognoses.
Keywords: nasopharyngeal adenocarcinoma, thyroid transcription factor-1, endoscopic surgery, papillary thyroid carcinoma
Introduction
According to the current World Health Organization classification, malignant epithelial tumors of the nasopharynx consist of nasopharyngeal carcinomas (NPCs), nasopharyngeal papillary adenocarcinomas (NPACs), and salivary gland carcinomas.1,2 Nasopharyngeal adenocarcinomas, including NPACs and salivary gland carcinomas, account for 0.38% to 0.48% of all malignant nasopharyngeal neoplasms.3 Low-grade nasopharyngeal papillary adenocarcinoma (LGNPPA) is a rare malignant neoplasm. Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma (TL-LGNPPA) accounts for a small proportion of LGNPPA and is pathologically characterized by papillary growth, abnormal expression of thyroid transcription factor 1 (TTF-1) as well as negative expression for thyroglobulin (TG), mimicking papillary thyroid carcinoma (PTC).4
Because of the scarcity of these neoplasms, few studies have been published about them, and most have been single-case reports.4,5 Herein, we summarize the clinical characteristics, pathological features, immunohistochemical results, treatments, and outcomes of 28 patients of LGNPPA in a single institution over a 10-year period.
Patients and Methods
We retrospectively collected data for 28 patients presenting with pathologically confirmed LGNPPA. The patients were retrieved from the Department of Otorhinolaryngology of the Affiliated Eye and ENT Hospital (AEENTH), Fudan University, Shanghai, China over a 10-year period between 2009 and 2019. This retrospective study was reviewed and approved by the institutional review board of AEENTH and conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from each patient and was provided by the parents of the patient under the age of 18. The following information was retrieved from the medical records: demographic data, tumor location, sinonasal surgical history, clinical symptoms, histological tumor type, pathological features, immunohistochemical results, treatment, and outcome. The duration of follow-up for each patient was calculated as the time from the date of primary surgery to the date of the outcome or last follow-up. Tumor sections from formalin fixation and paraffin-embedded tissues were stained with H&E and reviewed by two experienced pathologists independently.
Immunohistochemistry
The tissues were fixed in formalin, dehydrated, and embedded in paraffin, after which 4-μm sections were cut. The En Vension method was used to perform the immunohistochemical staining. The information of antibodies for the antigens was as follows: chromogranin (CHG, clone SP12, Gene Tech Company Limited), cytokeratin 5/6 (CK5/6, clone MX0-40, Maixin, Fuzhou), cytokeratin 7 (CK7, clone OV-TL12/30, Maixin, Fuzhou), cytokeratin 8 (CK8, clone TS1, Gene Tech Company Limited), cytokeratin 19 (CK19, clone RCK108, Gene Tech Company Limited), P63 (clone 4A4, Gene Tech Company Limited), S-100 (clone 4c4.9, Gene Tech Company Limited), vimentin (Vim, clone V9, Gene Tech Company Limited), epithelial membrane antigen (EMA, clone E29, Gene Tech Company Limited), pan-cytokeratin (CKpan, clone AE1/AE3, Maixin, Fuzhou), TG (clone 2H11/6E1, Gene Tech Company Limited), thyroid transcription factor 1 (TTF-1,clone SPT24, Gene Tech Company Limited) and Ki-67 (clone GM001, Gene Tech Company Limited).
Results
Demographic Data and Clinical Features
The demographic data and clinical features of the patients in this series are shown in Table 1. No patients had history of thyroid neoplasms or sinonasal surgery. No lymphatic or distant metastases were found before surgery. Of the 28 patients, 17 were females and 11 were males. The age at the time of surgery ranged from 7 to 77 years, with a median age of 41.5 years. The tumors were located on the roof of the nasopharynx (RON) in 13 patients, on the posterior margin of the nasal septum (PMONP) in 12 patients, on the lateral wall of the nasopharynx in one case, and on both the RON and the PMONP in two patients. The volume of all the tumors was limited in the nasopharynx, without lymphatic and distant metastasis. According to the 8th AJCC staging system, all tumors were T1N0M0. Three tumors were found during physical examination. The other 25 patients had one or more symptoms. The most common symptom was blood-stained rhinorrhea. The distribution of symptoms in the patients is shown in Figure 1.
 |
Table 1 A Summary of Demographic Data, Clinical Features, Treatments and Outcomes of the Patients |
 |
Figure 1 The distribution of symptoms for all patients. |
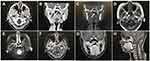
Nasopharyngeal endoscopy typically revealed polypoid or pedunculated masses on the RON or PMONP without adhesion to adjacent tissue (Figure 2), or shallow pharyngeal crypts. Sinus computed tomography (CT) images showed diffuse thickened soft tissue without a clear margin, and a circular or nodular mass at the nasopharynx or the edge of the nasal septum. Enhanced CT images showed that the tumors were moderately to strongly enhanced with no bone destruction (Figure 3A–C). Sinus magnetic resonance imaging revealed masses that had moderate signals in plain T1-weighted images, were enhanced in contrast-enhanced images, and had strong signals in T2-weighted images (Figure 3D–H).
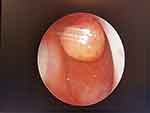 |
Figure 2 Nasopharyngeal endoscopy revealed a pedicled polypoid mass was located on the root of the nasopharynx. |
Pathological Features
Microscopic examination showed that the nasopharyngeal neoplastic tissue consisted of papillary structures with a fibrovascular and hyalinized core lined with crowded tumor epithelial cells (Figure 4A). The tumor cells were cuboidal, oval, or spindle-shaped, with nuclear overlapping and optically clear nuclei, as seen in PTC (Figure 4B). The size of the tumor cells was relatively uniform. Histological features, such as perineural invasion, lymphovascular invasion, or marginal involvement, were not observed.
Immunohistochemical Findings
Of the 28 patients, 20 patients had immunohistochemical analysis results, of which 14 patients were confirmed to have TL-LGNPPA (Table 2). Immunohistochemical data showed the tumors were uniformly positive for CK7, CK8, Vim, EMA, and CKpan, and negative for TG (Figure 4C and E). The reactivities for CK5/6, CK19, P63, S-100 and CHG were not uniform. All TL-LGNPPA tumors were positive for TTF-1 (Figure 4D). The Ki-67 proliferation value ranged from 1% to 20% (Figure 4F). Of 20 patients, the Ki-67 proliferation value was ≤ 10% in 18 patients (90%) and > 10% in the other two patients (10%) (Table 1).
 |
Table 2 Summary of Immunohistochemistry Data of the Patients in This Series |
Treatment and Follow-Up
All patients underwent endoscopic surgery. Of 28 patients, 23 patients underwent endoscopic surgery alone, three patients underwent preoperative radiotherapy, and two patients underwent radiotherapy after primary surgery. The volume of the three tumors decreased after preoperative radiotherapy. The follow-up time for all patients ranged from 7 to 121 months. All patients were alive at the time of follow-up. Two patients (7%) experienced disease recurrence, and underwent surgery to resect the recurrent LGNPPA. No lymphatic and distant metastases were found after surgery in either case. The details of all patients are summarized in Table 1.
Discussion
LGNPPA is a rare malignant tumor in the nasopharynx and few studies have reported this neoplasm. To the best of our knowledge, our study is the largest case series focusing on this rare disease in a single institution. In this study, the median age was 41.5 years, ranging from 7 to 77 years. The incidence of LGNPPA was higher in females than in males, with a female-male ratio of 1.5:1 (17 females, 11 males). The most common symptoms were blood-stained rhinorrhea, consistent with previous reports.2,6 The neoplasms were mostly localized on the RON and PMONP, which is also consistent with prior literature.6,7 Some studies have reported neoplasms located in nasopharyngeal wall.8 In our study, we also found a case in which the tumor was located on the lateral wall of the nasopharynx.
Microscopically, the continuity of papillary adenocarcinoma tumor cells with positive cytokeratin staining indicated that the papillary adenocarcinoma arose from the surface mucosal epithelium rather than from submucosal seromucous glands.9 The following microscopic features have been reported: papillary architecture with fibrovascular cores lined with a cuboidal to columnar epithelium; eosinophilic cytoplasm and epithelial cells with round to oval bland nuclei; tiny nucleoli and a vesicular or relatively clear chromatin pattern; and scattered psammoma bodies.8,10 Our microscopic findings were consistent with these features. In some patients, a transition from the normal surface respiratory epithelium to tumor cells has been identified.7 A novel histological finding in one report was scattered squamous cell foci intermixed with glandular tumor cells.11 Several patients with a spindle tumor cell component have also been reported,7–9 with some patients observed in our study.
TL-LGNPPA is a special type of LGNPPA and its microscopic features strongly resemble those of metastatic PTC; thus, accurate identification is crucial for providing an accurate diagnosis, treatment, and prognosis. TTF-1, a tissue-specific transcription factor, is selectively expressed during the embryological development of the thyroid, lungs and part of the diencephalon, and is expressed in all thyroid-like NPACs, but not in other types of NPAC.10 Therefore, TTF-1 could be used to differentiate TL-LGNPPA from LGNPPA, but it might not be suitable for distinguishing primary TL-LGNPPA from metastatic PTC. Pineda-Daboin et al suggested the following features that can distinguish between TL-LGNPPA and metastatic PTC: evidence of a transition from the mucosal surface to the tumor, the predominance of stratified nuclei, the absence of TG, and the absence of a thyroid tumor in imaging studies.2 However, a case of TL-LGNPPA with focal TG expression has been reported.12 Negativity for TG, CK20, CD15, S-100, P40, P63, CK5/6, CDX-2, prostate-specific antigen, and glial fibrillary acidic protein, and positivity for CK7, CK19, vimentin, and EMA on immunohistochemistry were reported to support the diagnosis of TL-LGNPPA in some studies.3,5,12 Huang et al reported that S-100 was focally positive at a moderate intensity, especially in the glandular structures in three patients, but was negative in two patients.6 In our study, immunohistochemical analysis showed that tumor cells were uniformly positive for CK7, Vim, EMA, CKpan, and TTF-1, and negative for TG, which supported the diagnoses of TL-LGNPPA. The immunoreactivities of S-100, CK19, P63 and CK5/6 were not uniform, indicating that these proteins may not be appropriate markers for identifying TL-LGNPPA. Immunohistochemical staining for PAX-8 has been reported to be useful for identifying TL-LGNPPA from metastatic PTC because it is positive in thyroid tumors.6,7,11,13 In our patients, PAX-8 immunohistochemical staining was not performed.
Surgical excision is reported to be the most appropriate treatment method for LGNPPA, and endoscopic resection is an effective modality with low morbidity.14,15 The prognosis is excellent, with most patients remaining free of recurrent or metastatic tumors after simple surgical excision.2,4,6,7,14 In our study, the surgical outcome revealed that all patients were alive and no lymphatic or distant metastases were found in the follow-up period (7 to 121 months), supporting the excellent prognosis of LGNPPA. Two patients with recurrent disease underwent endoscopic surgery and had an uneventful survival.
The role of radiotherapy in the treatment of LGNPPA has been emphasized in several studies. Wang et al reported a case without locoregional recurrence or distant metastasis after postoperative adjuvant photodynamic therapy.16 In our study, three patients underwent radiotherapy before primary surgery and the volume of the neoplasm decreased. Additionally, two patients underwent radiotherapy after radical surgery. There was no recurrence in these five patients. These outcomes might indicate that the tumor cells reacted to radiation to some extent and combined radiotherapy could help improve the disease-free survival time. Because of the limited number of patients who underwent radiotherapy, further study is needed to clarify the role of radiotherapy in treating LGNPPA.
Most nasopharyngeal tumors originating from the nasopharyngeal epithelium are often associated with Epstein–Barr virus. However, a close relationship between LGNPPA and Epstein–Barr virus has not been confirmed.5 Negative reactivity for Epstein–Barr virus encoded RNA (EBER)3,4,6,7,9,17,18 and latent membrane protein (LMP1)9 tumor cells was reported using in situ hybridization. KRAS, NRAS, and BRAF (V600E) gene mutations were not found in most previous studies.6,11,13 EGFR and ALK gene mutations have also been reported to be negative.11 However, due to the limited number of studies, further investigation is needed to elucidate the pathogenesis of LGNPPA.
In conclusion, LGNPPA is a rare indolent nasopharyngeal malignant tumor. We found that endonasal endoscopic excision was an effective treatment with an excellent prognosis. Because of the resemblance of TL-LGNPPA to metastatic PTC, pathologists and clinicians should take care to distinguish these types of neoplasm accurately to provide accurate diagnoses and choose the right treatment modality.
Disclosure
The authors do not have any potential conflicts of interest to disclose.
References
1. Liu LZ, Zhang YM, Chen Y, Li L. Spreading patterns, prognostic factors and treatment outcomes of nasopharyngeal papillary adenocarcinoma and salivary gland-type carcinomas. Clin Otolaryngol. 2016;41(2):160–168. doi:10.1111/coa.12492
2. Pineda-Daboin K, Neto A, Ochoa-Perez V, Luna MA. Nasopharyngeal adenocarcinomas: a clinicopathologic study of 44 cases including immunohistochemical features of 18 papillary phenotypes. Ann Diagn Pathol. 2006;10(4):215–221. doi:10.1016/j.anndiagpath.2005.11.002
3. Li L, Zhou F, Lin F, Han C. Clinicopathologic characteristics of thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: a case report. Appl Immunohistochem Mol Morphol. 2019;27(8):e81–e84. doi:10.1097/PAI.0000000000000545
4. Zhang WL, Ma S, Havrilla L, et al. Primary thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: a case report and literature review. Medicine. 2017;96(47):e8851. doi:10.1097/MD.0000000000008851
5. Wang X, Yan H, Luo Y, Fan T. Low-grade nasopharyngeal papillary adenocarcinoma: a case report and review of the literature. Onco Targets Ther. 2016;9:2955–2959. doi:10.2147/OTT.S100447
6. Huang F, Xiang X, Hong B, Min J, Li J. Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma. Am J Clin Pathol. 2019;152(5):582–589. doi:10.1093/ajcp/aqz082
7. Li M, Wei J, Yao X, Wang C. Clinicopathological features of low-grade thyroid-like nasopharyngeal papillary adenocarcinoma. Cancer Res Treat. 2017;49(1):213–218. doi:10.4143/crt.2016.195
8. Carrizo F, Luna MA. Thyroid transcription factor-1 expression in thyroid-like nasopharyngeal papillary adenocarcinoma: report of 2 cases. Ann Diagn Pathol. 2005;9(4):189–192. doi:10.1016/j.anndiagpath.2005.04.019
9. Fu CH, Chang KP, Ueng SH, Wu CC, Hao SP. Primary thyroid-like papillary adenocarcinoma of the nasopharynx. Auris Nasus Larynx. 2008;35(4):579–582. doi:10.1016/j.anl.2007.10.009
10. Sillings CN, Weathers DR, Delgaudio JM. Thyroid-like papillary adenocarcinoma of the nasopharynx: a case report in a 19-year-old male. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;110(3):e25–28. doi:10.1016/j.tripleo.2010.04.029
11. Oide T, Kadosono O, Matsushima J, et al. Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma with squamous differentiation: a novel histological finding. Hum Pathol. 2017;70:43–48. doi:10.1016/j.humpath.2017.05.020
12. Ozer S, Kayahan B, Cabbarzade C, Bugdayci M, Kosemehmetoglu K, Yucel OT. Thyroid-like papillary adenocarcinoma of the nasopharynx with focal thyroglobulin expression. Pathology. 2013;45(6):622–624. doi:10.1097/PAT.0b013e32836536e9
13. Oishi N, Kondo T, Nakazawa T, et al. Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: case report and literature review. Pathol Res Pract. 2014;210(12):1142–1145. doi:10.1016/j.prp.2014.04.020
14. Ryu J, Park WS, Jung YS. Exclusive endoscopic resection of nasopharyngeal papillary adenocarcinoma via combined transnasal and transoral approach. Clin Exp Otorhinolaryngol. 2013;6(1):48–51. doi:10.3342/ceo.2013.6.1.48
15. Unsaler S, Basaran B, Aslan I, Yilmazbayhan D. Endonasal endoscopic nasopharyngectomy for the treatment of nasopharyngeal papillary adenocarcinoma: report of a rare case. Int J Pediatr Otorhinolaryngol. 2018;104:51–53. doi:10.1016/j.ijporl.2017.10.041
16. Wang CP, Chang YL, Chen CT, Yang TH, Lou PJ. Photodynamic therapy with topical 5-aminolevulinic acid as a post-operative adjuvant therapy for an incompletely resected primary nasopharyngeal papillary adenocarcinoma: a case report. Lasers Surg Med. 2006;38(5):435–438. doi:10.1002/lsm.20291
17. Ohe C, Sakaida N, Tadokoro C, et al. Thyroid-like low-grade nasopharyngeal papillary adenocarcinoma: report of two cases. Pathol Int. 2010;60(2):107–111. doi:10.1111/j.1440-1827.2009.02480.x
18. Wang L, Min Q, Jiang J, Shao Y, Cao Y, Yan D. Low-grade nasopharyngeal papillary adenocarcinoma. J Coll Physicians Surg Pak. 2019;29(11):1114–1115. doi:10.29271/jcpsp.2019.11.1114
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.