Back to Journals » International Journal of Nanomedicine » Volume 15
Low-Dose Exposure of Silica Nanoparticles Induces Neurotoxicity via Neuroactive Ligand–Receptor Interaction Signaling Pathway in Zebrafish Embryos
Authors Wei J, Liu J, Liang S, Sun M, Duan J
Received 18 March 2020
Accepted for publication 30 May 2020
Published 19 June 2020 Volume 2020:15 Pages 4407—4415
DOI https://doi.org/10.2147/IJN.S254480
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Lei Yang
Jialiu Wei,1 Jianhui Liu,2 Shuang Liang,2 Mengqi Sun,2 Junchao Duan2
1Key Laboratory of Cardiovascular Epidemiology & Department of Epidemiology, Fuwai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, People’s Republic of China; 2Department of Toxicology and Sanitary Chemistry, School of Public Health, Capital Medical University, Beijing, People’s Republic of China
Correspondence: Junchao Duan
Department of Toxicology and Sanitary Chemistry, School of Public Health, Capital Medical University, Beijing 100069, People’s Republic of China
Email [email protected]
Objective: Silica nanoparticles (SiO2 NPs) have been extensively employed in biomedical field. SiO2 NPs are primarily designed to enter the circulatory system; however, little information is available on potential adverse effects of SiO2 NPs on the nervous system.
Methods: The neurotoxicity of SiO2 NPs at different concentrations (3, 6, 12 ng/nL) on zebrafish embryos was determined using immunofluorescence and microarray techniques, and subsequently confirmed by qRT-PCR.
Results: SiO2 NPs disrupt the axonal integrity and decrease the length of axons in Tg (NBT: EGFP) transgenic lines. The number of apoptotic cells in the brain and central nervous system of zebrafish embryos was increased in the presence of 12 ng/nL of SiO2 NPs, but the difference did not reach statistical significance. Screening for changes in the expression of genes involved in the neuroactive ligand–receptor interaction pathway was performed by microarray and confirmed by qRT-PCR. These analyses demonstrated that SiO2 NPs markedly downregulated genes associated with neural function (grm6a, drd1b, chrnb3b, adrb2a, grin2ab, npffr2.1, npy8br, gabrd, chrma3, gabrg3, gria3a, grm1a, adra2b, and glra3).
Conclusion: The obtained results documented that SiO2 NPs can induce developmental neurotoxicity by affecting the neuroactive ligand–receptor interaction signaling pathway. This new evidence may help to clarify the mechanism of SiO2 NPs-mediated neurotoxicity.
Keywords: silica nanoparticles, neurotoxicity, neuroactive ligand–receptor interaction signaling pathway, zebrafish
Introduction
Silica nanoparticles (SiO2 NPs), one of the most effective nano-powders, are widely applied in various fields.1–3 Natural SiO2 NPs have been identified as the main inorganic constituent of particulate matter (PM).4,5 Given the widespread use of SiO2 NPs, their potentially harmful impact on human health has gained significant attention. It has been shown that SiO2 NPs exert significant toxic effects on the respiratory,6 cardiovascular7 and reproductive systems.8 In zebrafish embryos, SiO2 NPs can induce blood hypercoagulability by reducing the rate of blood flow.9 Importantly, the neurotoxic effects of SiO2 NPs have also been documented. For instance, in adult zebrafish, SiO2 NPs can disturb light/dark preference, inhibit exploratory behavior, and impair memory capability.10 In vitro, SiO2 NPs exhibit size-dependent toxicity in N9 cells, which represent the residential macrophages of the central nervous system. Additionally, SiO2 NPs induce the formation of mitochondrial ROS, secretion of IL-1β, phosphorylation of GSDMD, and development of pyroptosis in N9 cells;11 and upregulate apoptosis-promoting genes in Neuro-2a cells.12 However, the impact of SiO2 NPs on neurodevelopment in vivo has not yet been fully elucidated.
Given the prominent properties and genetic homology with most human genes, zebrafish has been employed as an efficient system for the assessment of SiO2 NPs neurotoxicity.10 Additionally, zebrafish is frequently used in neurotoxicity studies due to the conserved nervous systems in comparison with mammals.13–15 Due to its high fecundity, cost-effectiveness, well-characterized and developmental stages, zebrafish has huge potential in high-throughput nanotoxicity screening.16 Moreover, because zebrafish embryos are transparent, the use of fluorescently tagged transgenes allows the visualization of the development of the neurological systems and the monitoring of neurological disorders in a living organism.17
The current study aimed to document the potential developmental neurotoxicity of SiO2 NPs in vivo by taking advantage of the unique features of the zebrafish model. Specifically, the hypothesis was raised that SiO2 NPs would lead to the underdevelopment of neurological systems in zebrafish by perturbing a specific stage in neurodevelopment. To test this possibility, experiments were performed to determine SiO2 NPs-induced changes in 1) axonal integrity and pattern, 2) apoptosis of brain and central cells, and 3) the neuroactive ligand–receptor interaction signaling pathway. It was expected that the study would identify the mechanism of developmental neurotoxicity of SiO2 NPs in zebrafish embryos.
Materials and Methods
SiO2 NPs Preparation
SiO2 NPs were produced by using the Stöber method from Jilin University, China. Briefly, 2.5mL of tetraethylorthosilicate (TEOS, Sigma, St. Louis, MO) was transferred into a flask with 50 mL premixed ethanol, 2 mL ammonia and 1 mL water. The mixed solution was sustained at 40°C with sustained stirring (150 r/min) for 12h. After that, the resulting suspension was centrifugated at a speed of 12,000 r/min for 15min, aiming at isolating the particles. The detached particles were then rinsed with deionized water and dispersed in 50 mL deionized water as a stock medium. In addition, the suspensions were sonicated (160 W, 20kHz, 5min) (Bioruptor UDC-200; Diagenode, Liège, Belgium) prior to the experiments for avoiding possible aggregation of particles.
Zebrafish Husbandry
The zebrafish lines include Albino, Tg (fli-1: EGFP) and Tg (NBT: EGFP), which have been declared in our previous study.18 The certification number (International Association for Assessment and Accreditation of Laboratory Animal Care, 001458) was obtained from Hunter Biotechnology, Inc. The zebrafish were raised in a specific system (28°C, 80% humidity) accompanied with a round of 14 h light and 10 h dark. The collection of fertilized eggs was assisted by a stereomicroscope (Nikon, SMZ645, Japan) within 4 h post fertilization (hpf) for the following research. No less than 30 embryos were distributed for each group. Newly fertilized embryos were administrated with SiO2 NPs and those dead were excluded every day. All experiments were strictly implemented according to relevant laws and institutional guidelines.
Intravenous Microinjection
The doses of SiO2 NPs were selected according to morphologic measurements with the no observed adverse effect level in zebrafish embryos as previous descriptions.18 The no observed adverse effect level of SiO2 NPs is about 3 ng/nL under morphologic assessments by 15 parameters. Various doses of SiO2 NPs (0, 3, 6 and 12 ng/nL) were injected with 10 nL at Duct of Cuvier (DC) by microinjector (PCO1500, Zgenebio, Taipei, Taiwan) with 30 zebrafish embryos for each well for 24 h. By contrast, the zebrafish embryos of control group were received ultrapure water by injection.
Neurotoxicity Measurement
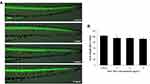
After the Tg (NBT:GFP) transgenic zebrafish was treated with 3, 6 and 12 ng/nL of SiO2 NPs for 24 h, neuron development and injury levels were measured. Images were captured under a fluorescence microscope (Nikon, AZ100, Japan).
Immunofluorescence
The immunofluorescence protocol has been stated in a previous study.19 Briefly, after being exposed to SiO2 NPs for various doses, zebrafish was fixed with paraformaldehyde for 24 h. Zebrafish was then permeated with trypsin and fixed with paraformaldehyde, and immersed in the mouse antibody against sea urchin acetylated-tubulin (α-AT) (Sigma, USA) for 24 h. After washing for 3 times, the zebrafish was incubated with Rhodamine Red-X-AffiniPure goat anti-mouse IgG þ IgM (H þ L) (1:1000, Jackson ImmunoResearch, USA) for 4 h, which is considered as a secondary antibody. Finally, zebrafish was observed under a fluorescence microscope. NIS-Elements D 3.10 software was employed for analysis of fluorescence intensity.
Cellular Apoptosis Assay
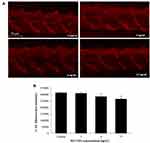
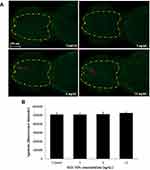
Cell apoptosis was measured in the brain and central nervous system using acridine orange (AO) staining, respectively. AO stains cells with disturbed plasma membrane permeability so it primarily stains necrotic or late apoptotic cells, however, normal cells are nonpermeable to AO. Brains of zebrafish were exposed to SiO2 NPs (3, 6, and 12 ng/nL) for 24 h. Then, brains were rinsed three times with PBS and incubated in 5 mg/mL AO for 30 min in the dark at 28°C, followed by being rinsed at PBS. Finally, brain tissues were observed under a fluorescence microscope (Olympus BX61, Tokyo, Japan). The captured fluorescence was assessed and quantified by Volocity Demo 6.1.1 software (PerkinElmer, USA).
Microarray and Bioinformatic Analysis
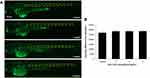
The analysis of microarray and bioinformation was executed as previous descriptions.20 The mRNA expression patterns of zebrafish embryos treated with SiO2 NPs were measured by Zebrafish Gene 1.0 ST Array (AFFY-METRIX GeneChipVR, Santa Clara, CA). The data of microarray in the current study have been stored in NCBI Gene Expression Omnibus (GEO) and the series accession number GSE73427.
Statistical Analysis
All data were analyzed using SPSS 23.0 software. Data were described as means ± S.D. The least significant difference (LSD) test was implemented to evaluate the difference between two groups. Statistical significance was at the level of *p<0.05.
Results
Characterization of SiO2 NPs
The characterization of SiO2 NPs has been reported in our previous studies.9,18 Briefly, the transmission electron microscope images showed that the SiO2 NPs exhibited a near-spherical shape and were well dispersed with an average diameter of approximately 62 nm. The hydrodynamic sizes and Zeta potentials of SiO2 NPs in ultrapure water were about 108 ± 1 nm and −38 ± 2 mV, respectively. Additionally, no endotoxin was detected in SiO2 NPs suspensions using limulus amebocyte lysate assay.
Neurotoxicity Induced by SiO2 NPs in Zebrafish Embryos
The neuron damage induced by SiO2 NPs was captured by images, which were observed under a multi-purpose zoom fluorescence microscopy (Figure 1). SiO2 NPs had a significantly depressor effect on the length of axons Tg (NBT: GFP) transgenic zebrafish lines. Axons structure also showed significant damage after exposure to SiO2 NPs, especially on the 6 and 12 ng/nL doses groups (Figure 2). The above results explain that SiO2 NPs could damage the nervous system and result in neuron injury in zebrafish model.
Cellular Apoptosis Caused by SiO2 NPs in Zebrafish Embryos
The number of apoptotic cells in the brain of zebrafish embryos was increased in the 12 ng/nL groups, but the difference did not reach statistical significance (Figure 3). Similarly, there were no significant differences between the control group and the SiO2 NPs-exposed group regarding apoptosis of central cells in zebrafish embryos (Figure 4).
The Effect of SiO2 NPs on Neuroactive Ligand–Receptor Interaction Pathway in Zebrafish Embryos
In line with gene chip high-throughput screening, the expression levels of genes in neuroactive ligand–receptor interaction pathway were detected in zebrafish embryos (Figure 5A). The related genes were verified by qRT-PCR analysis. In addition, as illustrated in Figure 5B, the genes regulating neuroactive ligand–receptor interaction (grm6a, drd1b, chrnb3b, adra2b, chrm5b, grin2ab, npffr2.1, npy8br, gabrd, chrna3, gabrg3, gria3a, adrb2a, grm1a, and glra3) were significantly decreased following SiO2 NPs treatment.
Discussion
The widespread use of SiO2 NPs in consumer products has caused controversy and concern due to its known neuro-, teratogenic, and developmental toxicity.21–23 Our previous study showed that nano-scale silica particles can induce more severe cytotoxicity and autophagy dysfunction than micro-scale silica particles in a size-dependent manner.24 Kim et al observed that approximately 60 nm SiO2 NPs affect cells differently than nanoparticles of other sizes due to enhanced uptake. Therefore, uptake of approximately 60 nm particles may be optimal to affect cellular physiology whereas larger particles exhibit more toxicity.25 SiO2 NPs are washed several times using deionised water during the preparation process, similar to the separation of water-extractable ions in PM2.5 particles.26,27 Also, SiO2 NPs are stored at low temperature until use to maintain ion release. While they are very stable at room temperature and neutral pH, SiO2 NPs may release some ions when exposed to strong acids, strong bases, or high temperatures. In this study, the internal environment of zebrafish was a neutral pH with low temperature. Therefore, SiO2 NPs were very stable and no ions were released under experimental conditions, ensuring the accuracy of our findings.
Previous experimental studies have found that SiO2 NPs can result in behavioural and morphological changes in zebrafish by modification of Parkinson-related genes.10 In addition, SiO2 NPs can interfere with the structure of the blood-brain barrier (BBB) and induce BBB inflammation in Sprague-Dawley rats.22 The purpose of the current study was to detect the developmental toxicity of SiO2 NPs and to illustrate its effect and underlying mechanism in a zebrafish model. We measured the neurotoxicity of SiO2 NPs in zebrafish by transgenosis and immunofluorescence assays. We assessed the development of axons, which is a time-efficient method to screen for neurotoxicity.28,29 Our results demonstrated that SiO2 NPs reduced the axon length and perturbed the axon pattern in zebrafish. There has been limited research on the effects of SiO2 NPs on the structure of the nervous system. Our previous study discovered that PM2.5 particles are capable of disrupting axon integrity and reducing axon length in Tg (NBT:EGFP) transgenic zebrafish lines.30 Taken together, our data demonstrated that exposure to SiO2 NPs incurred neurotoxicity in zebrafish embryos, especially on the development of axons.
Our previous studies found alterations of some signalling pathways after exposure to SiO2 NPs in zebrafish through microarray analyses. We used the KEGG database to analyse the 11 downregulated genes and found significant involvement of specific pathways. Neuroactive ligand–receptor interaction signaling pathway was directly related to neuro function.18 Neuroactive ligands influence neuronal function by binding to intracellular receptors, which has the capability of binding transcription factors and regulating gene expressions.31 Disrupting the genes involved in neuroactive ligand–receptor interaction results in decreased memory function.32 qRT-PCR based on high-throughput genetic screening in our study showed that the expression of neuroactive ligand–receptor interaction signalling pathways were significantly decreased in zebrafish treated with SiO2 NPs, which were in alignment with results from microarray analyses. Drd1b is a member of the dopaminergic receptor family, which belongs to the superfamily of G protein-coupled receptors. A recent study showed that dopaminergic receptors can modulate extracellular matrix remodelling, resulting in physiological and pathological plasticity accompanied with disturbances or malformation of the neural extracellular matrix.33 Expression levels of Gabrd, Chrna3, and Npy8br encoding for gamma-aminobutyric acid (GABA) receptor, cholinergic receptor, and neuropeptide receptor, respectively, are vital indicators for neurotoxicity.34–36 Our study previously demonstrated that the decreased expression of Chrna3 and Chrnb3b were correlated with the expression of GABA receptor genes, indicating that SiO2 NPs may inhibit the release of neurotransmitters in zebrafish brains.37 Neuropeptide FF is a member of the RF-amide peptide family, which plays an important role in controlling the feeding behaviour of vertebrates.38 It binds to neuropeptide FF receptor 2 (NPFFR2), which is mainly expressed in the hypothalamus, superficial layers of the spinal cord, and thalamic nuclei and is involved in the regulation of extracellular signal-regulated protein kinase pathway as well as pain and sensory input.39 The Adra2b gene has been shown to be associated with enhanced emotional memory40 and Li et al found that SiO2 NPs inhibits memory capability in adult zebrafish.10 In addition, Glra3 seems to play a vital role in inflammatory pain sensitisation. A previous study found that SiO2 NPs caused inflammation–coagulation response mediated by the JAK1/TF pathway.9 These results suggested that SiO2 NPs may cause neurotoxicity by initiating inflammation. In summary, these data indicate that the signaling pathways associated with neuroactive ligand–receptor interaction are indeed activated by SiO2 NPs in zebrafish.
It is known that apoptosis plays a crucial role in SiO2 NPs-induced neurotoxicity. It has been documented that SiO2 NPs at low concentrations of 10 mg/mL change cellular morphologies and induce oxidative stress and apoptosis in both mouse neuro2a and human neuroblastoma cells.41 Lee et al also reported that SiO2 NPs lead to caspase-dependent apoptosis through endoplasmic reticulum stress in neuronal cells.12 In our study, we evaluated the apoptosis of SiO2 NPs on zebrafish by acridine orange staining. Our results showed that apoptosis rates in cells of the brain and central nervous system were increased after SiO2 NPs treatment with highest dosage, although this difference was not statistically significant. The results suggest that in vivo low-dose SiO2 NPs are unable to cause apoptosis of brain and central nervous system cells.
Conclusion
In summary, our results showed that exposure to SiO2 NPs caused neurotoxicity with detrimental effects on neuron maturation processes such as axon formation in zebrafish. However, apoptosis was not significantly affected. In addition, gene expression suggested experiments suggest that the neuroactive ligand–receptor pathway played a role in SiO2 NPs-induced neurotoxicity. Our finding provides evidence to better elucidate the mechanism of neurotoxicity caused by exposure to SiO2 NPs.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Kasaai MR. Nanosized particles of silica and its derivatives for applications in various branches of food and nutrition sectors. J Nanotechnol. 2015;2015:1–6. doi:10.1155/2015/852394
2. Brinch A, Hansen SF, Hartmann NB, et al. EU regulation of nanobiocides: challenges in implementing the biocidal product regulation (BPR). Nanomaterials. 2016;6:33. doi:10.3390/nano6020033
3. Khot LR, Sankaran S, Maja JM, et al. Applications of nanomaterials in agricultural production and crop protection: a review. Crop Prot. 2012;35:64–70. doi:10.1016/j.cropro.2012.01.007
4. Dalmora AC, Ramos CG, Oliveira MLS, et al. Chemical characterization, nano-particle mineralogy and particle size distribution of basalt dust wastes. Sci Total Environ. 2016;539:560–565. doi:10.1016/j.scitotenv.2015.08.141
5. Dalmora AC, Ramos CG, Querol X, et al. Nanoparticulate mineral matter from basalt dust wastes. Chemosphere. 2016;144:2013–2017. doi:10.1016/j.chemosphere.2015.10.047
6. Guo C, Wang J, Yang M, et al. Amorphous silica nanoparticles induce malignant transformation and tumorigenesis of human lung epithelial cells via P53 signaling. Nanotoxicology. 2017;11:1176–1194. doi:10.1080/17435390.2017.1403658
7. Feng L, Yang X, Shi Y, et al. Co-exposure subacute toxicity of silica nanoparticles and lead acetate on cardiovascular system. Int J Nanomedicine. 2018;13:7819–7834. doi:10.2147/IJN.S185259
8. Zhang L, Wei J, Duan J, et al. Silica nanoparticles exacerbates reproductive toxicity development in high-fat diet-treated Wistar rats. J Hazard Mater. 2019;384:121361. doi:10.1016/j.jhazmat.2019.121361.
9. Duan J, Liang S, Yu Y, et al. Inflammation-coagulation response and thrombotic effects induced by silica nanoparticles in zebrafish embryos. Nanotoxicology. 2018;12:470–484. doi:10.1080/17435390.2018.1461267
10. Li X, Ji X, Wang R, et al. Zebrafish behavioral phenomics employed for characterizing behavioral neurotoxicity caused by silica nanoparticles. Chemosphere. 2020;240:124937. doi:10.1016/j.chemosphere.2019.124937
11. Du Q, Ge D, Mirshafiee V, et al. Assessment of neurotoxicity induced by different-sized Stöber silica nanoparticles: induction of pyroptosis in microglia. Nanoscale. 2019;11:12965–12972. doi:10.1039/c9nr03756j
12. Lee K-I, Lin J-W, Su -C-C, et al. Silica nanoparticles induce caspase-dependent apoptosis through reactive oxygen species-activated endoplasmic reticulum stress pathway in neuronal cells. Toxicol in Vitro. 2020;63:104739. doi:10.1016/j.tiv.2019.104739
13. Chatterjee D, Gerlai R. High precision liquid chromatography analysis of dopaminergic and serotoninergic responses to acute alcohol exposure in zebrafish. Behav Brain Res. 2009;200:208–213. doi:10.1016/j.bbr.2009.01.016
14. Shi Q, Wang M, Shi F, et al. Developmental neurotoxicity of triphenyl phosphate in zebrafish larvae. Aquat Toxicol. 2018;203:80–87. doi:10.1016/j.aquatox.2018.08.001
15. Xiaokai Chen XZ, Zhang X, Li C, Sayed SM, Sun W, Lin F. Superbright organosilica nanodots as a universal sensor for fast discrimination and accurate quantification of live/dead cells. Sens Actuators B Chem. 2019;295:49–55. doi:10.1016/j.snb.2019.05.031
16. Jia HR, Zhu YX, Duan QY, et al. Nanomaterials meet zebrafish: toxicity evaluation and drug delivery applications. J Control Release. 2019;311–312:301–318. doi:10.1016/j.jconrel.2019.08.022
17. Jia HR, Zhu YX, Xu KF, et al. Efficient cell surface labelling of live zebrafish embryos: wash-free fluorescence imaging for cellular dynamics tracking and nanotoxicity evaluation. Chem Sci. 2019;10:4062–4068. doi:10.1039/c8sc04884c
18. Duan J, Yu Y, Li Y, et al. Low-dose exposure of silica nanoparticles induces cardiac dysfunction via neutrophil-mediated inflammation and cardiac contraction in zebrafish embryos. Nanotoxicology. 2016;10:575–585. doi:10.3109/17435390.2015.1102981
19. Tilton F, Tanguay RL. Exposure to sodium metam during zebrafish somitogenesis results in early transcriptional indicators of the ensuing neuronal and muscular dysfunction. Toxicol Sci. 2008;106:103–112. doi:10.1093/toxsci/kfn145
20. Hu H, Li Q, Jiang L, et al. Genome-wide transcriptional analysis of silica nanoparticle-induced toxicity in zebrafish embryos. Toxicol Res. 2016;5:609–620. doi:10.1039/c5tx00383k
21. Liu J, Yang M, Jing L, et al. Silica nanoparticle exposure inducing granulosa cell apoptosis and follicular atresia in female Balb/c mice. Environ Sci Pollut Res Int. 2018;25:3423–3434. doi:10.1007/s11356-017-0724-5
22. Liu X, Sui B, Sun J. Blood-brain barrier dysfunction induced by silica NPs in vitro and in vivo: involvement of oxidative stress and Rho-kinase/JNK signaling pathways. Biomaterials. 2017;121:64–82. doi:10.1016/j.biomaterials.2017.01.006
23. Fu J, Gao J, Gong L, et al. Silica nanoparticle exposure during the neonatal period impairs hippocampal precursor proliferation and social behavior later in life. Int J Nanomedicine. 2018;13:3593–3608. doi:10.2147/IJN.S160828
24. Li Q, Hu H, Jiang L, et al. Cytotoxicity and autophagy dysfunction induced by different sizes of silica particles in human bronchial epithelial BEAS-2B cells. Toxicol Res. 2016;5:1216–1228. doi:10.1039/c6tx00100a
25. Kim IY, Joachim E, Choi H, et al. Toxicity of silica nanoparticles depends on size, dose, and cell type. Nanomedicine. 2015;11:1407–1416. doi:10.1016/j.nano.2015.03.004
26. Emma G, Snell J, Charoud-Got J, et al. Feasibility study of a candidate reference material for ions in PM2.5: does commutability matter also for inorganic matrices? Anal Bioanal Chem. 2018;410:6001–6008. doi:10.1007/s00216-018-1220-6
27. Palleschi S, Rossi B, Armiento G, et al. Toxicity of the readily leachable fraction of urban PM2.5 to human lung epithelial cells: role of soluble metals. Chemosphere. 2018;196:35–44. doi:10.1016/j.chemosphere.2017.12.147
28. Jiang F, Liu J, Zeng X, et al. Tris (2-butoxyethyl) phosphate affects motor behavior and axonal growth in zebrafish (Danio rerio) larvae. Aquat Toxicol. 2018;198:215–223. doi:10.1016/j.aquatox.2018.03.012
29. Kanungo J, Lantz S, Paule MG. In vivo imaging and quantitative analysis of changes in axon length using transgenic zebrafish embryos. Neurotoxicol Teratol. 2011;33:618–623. doi:10.1016/j.ntt.2011.08.013
30. Duan J, Hu H, Zhang Y, et al. Multi-organ toxicity induced by fine particulate matter PM 2.5 in zebrafish (Danio rerio) model. Chemosphere. 2017;180:24–32. doi:10.1016/j.chemosphere.2017.04.013
31. Xu L-M, Li J-R, Huang Y, et al. AutismKB: an evidence-based knowledgebase of autism genetics. Nucleic Acids Res. 2012;40:D1016–D1022. doi:10.1093/nar/gkr1145
32. Papassotiropoulos A, de Quervain DJF. Failed drug discovery in psychiatry: time for human genome-guided solutions. Trends Cogn Sci. 2015;19:183–187. doi:10.1016/j.tics.2015.02.002
33. Mitlohner J, Kaushik R, Niekisch H, et al. Dopamine receptor activation modulates the integrity of the perisynaptic extracellular matrix at excitatory synapses. Cells. 2020;9:260. doi:10.3390/cells9020260
34. Hong E, Santhakumar K, Akitake CA, et al. Cholinergic left-right asymmetry in the habenulo-interpeduncular pathway. Proc Natl Acad Sci U S A. 2013;110:21171–21176. doi:10.1073/pnas.1319566110
35. Raftery TD, Volz DC. Abamectin induces rapid and reversible hypoactivity within early zebrafish embryos. Neurotoxicol Teratol. 2015;49:10–18. doi:10.1016/j.ntt.2015.02.006
36. Salaneck E, Larson ET, Larsson TA, et al. Effects of a teleost tetraploidization on neuropeptide Y receptor gene repertoire in ray-finned fishes. Ann N Y Acad Sci. 2005;1040:457–459. doi:10.1196/annals.1327.089
37. Zuo L, Garcia-Milian R, Guo X, et al. Replicated risk nicotinic cholinergic receptor genes for nicotine dependence. Genes. 2016;7:95. doi:10.3390/genes7110095
38. Li Q, Wen H, Li Y, et al. Evidence for the direct effect of the NPFF peptide on the expression of feeding-related factors in spotted sea bass (Lateolabrax maculatus). Front Endocrinol. 2019;10:545. doi:10.3389/fendo.2019.00545
39. Sun YL, Zhang XY, He N, et al. Neuropeptide FF activates ERK and NF kappa B signal pathways in differentiated SH-SY5Y cells. Peptides. 2012;38:110–117. doi:10.1016/j.peptides.2012.08.019
40. Zoladz PR, Dailey AM, Nagle HE, et al. ADRA2B deletion variant influences time-dependent effects of pre-learning stress on long-term memory. Neurobiol Learn Mem. 2017;140:71–81. doi:10.1016/j.nlm.2017.02.014
41. Yang X, He C, Li J, et al. Uptake of silica nanoparticles: neurotoxicity and Alzheimer-like pathology in human SK-N-SH and mouse neuro2a neuroblastoma cells. Toxicol Lett. 2014;229:240–249. doi:10.1016/j.toxlet.2014.05.009
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.