Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 16
Longitudinal Relationship Between Growth Differentiation Factor 11 and Physical Activity in Chronic Obstructive Pulmonary Disease
Authors Tanaka R, Koarai A ![]() , Yamada M
, Yamada M ![]() , Fujino N, Ichikawa T, Numakura T, Onodera K, Kyogoku Y, Tamada T, Miura M, Minakata Y
, Fujino N, Ichikawa T, Numakura T, Onodera K, Kyogoku Y, Tamada T, Miura M, Minakata Y ![]() , Ichinose M, Sugiura H
, Ichinose M, Sugiura H ![]()
Received 19 January 2021
Accepted for publication 21 March 2021
Published 15 April 2021 Volume 2021:16 Pages 999—1006
DOI https://doi.org/10.2147/COPD.S301690
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Richard Russell
Rie Tanaka,1 Akira Koarai,1 Mitsuhiro Yamada,1 Naoya Fujino,1 Tomohiro Ichikawa,1 Tadahisa Numakura,1 Katsuhiro Onodera,2 Yorihiko Kyogoku,1 Tsutomu Tamada,1 Motohiko Miura,3 Yoshiaki Minakata,4 Masakazu Ichinose,5 Hisatoshi Sugiura1
1Department of Respiratory Medicine, Tohoku University Graduate School of Medicine, Sendai, Japan; 2Department of Internal Medicine, Mizusawa Hospital, Oshu, Japan; 3Department of Respiratory Medicine, Tohoku Rosai Hospital, Sendai, Japan; 4Department of Respiratory Medicine, National Hospital Organization Wakayama Hospital, Wakayama, Japan; 5Academic Center, Osaki Citizen Hospital, Osaki, Japan
Correspondence: Akira Koarai
Department of Respiratory Medicine, Tohoku University Graduate School of Medicine, Sendai, Japan
Tel +81-22-717-8539
Fax +81-22-717-8549
Email [email protected]
Background: Daily physical activity is reduced in patients with chronic obstructive pulmonary disease (COPD) and a reduced level of physical activity has been shown to be an important predictor for the prognosis, such as increased risk of exacerbation and mortality. However, there has not yet been a useful biomarker of the physical activity. In our previous cross-sectional study, we showed that the level of one of the possible myokines, which is an anti-aging factor, growth differentiation factor 11 (GDF11), was decreased in the plasma from patients with COPD and correlated with the physical activity. To clarify this relationship, we conducted a longitudinal evaluation of such factors.
Patients and Methods: Twenty-four COPD patients were enrolled and prospectively followed. We measured the levels of plasma GDF11 and systemic inflammatory markers with immunoblotting or ELISA, respectively. We also evaluated lung function and daily physical activity using a triaxial accelerometer and the incidence of exacerbation.
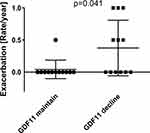
Results: The change in the plasma level of GDF11, but not systemic inflammatory markers, was positively correlated with the change in the physical activity in an intensity-dependent manner (between the change in the number of steps and GDF11; r = 0.41, p = 0.047). In the multiple regression analysis, the relationship was confirmed (β = 0.93, p < 0.001). In addition, patients who maintained their plasma level of GDF11 showed a significantly lower incidence in exacerbations of COPD than those with decreased levels of GDF11 (p = 0.041).
Conclusion: The longitudinal change in the plasma level of GDF11 was positively correlated with the change in the daily physical activity in COPD. GDF11 could be a useful humoral factor that reflects the physical activity in COPD.
Keywords: chronic obstructive pulmonary disease, physical activity, growth differentiation factor 11, exacerbation, longitudinal study
Introduction
Daily physical activity is reduced in patients with chronic obstructive pulmonary disease (COPD).1–3 The reduced level of physical activity has been reported to be associated with lower lung function3–5 and more frequent hospitalizations caused by exacerbations.6,7 In addition, a low level of physical activity has been reported to be associated with increased mortality risk in patients with COPD,6,8,9 and the level of physical activity has been shown to be an important predictor of the prognosis.10 Thus, it is recommended to encourage patients with COPD to maintain their level of daily physical activity.11 At present, the daily physical activity is evaluated using motion sensors and self-reported questionnaires,4–8,12 but the assessment is not simple. Therefore, a specific biomarker of the physical activity is needed.
Growth differentiation factor 11 (GDF11), a member of the transforming growth factor (TGF) beta superfamily,13 has been reported to be an anti-aging and rejuvenating factor.14–16 GDF11 is one of the possible myokines highly detected in skeletal muscles14,17 and has been shown to increase the muscle strength and exercise endurance in aged mice.16 In our previous cross-sectional study, the plasma level of GDF11 in patients with COPD was decreased and positively correlated with the daily physical activity.18,19 However, the relationship has not been fully evaluated, especially in terms of the data stability and continuity at the individual level. Therefore, we conducted a longitudinal evaluation of the changes in the plasma level of GDF11 and the daily physical activity to clarify the relationship.
Patients and Methods
Study Design and Subjects
Patients with COPD were recruited into the current study from Tohoku University Hospital and Tohoku Rosai Hospital. The recruitment period was between 2015 and 2017. All patients with COPD were older than 40 years, had a former smoking history of more than 10 pack-years, and were diagnosed according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) documents.20 All patients with COPD had been free of infection of the respiratory tract and exacerbations due to any reasons for at least 2 months prior to the study. All patients had quit smoking at least 1 year before. Patients with pathologic conditions including cerebrovascular diseases, rheumatoid arthritis and arteriosclerosis obliterans, which affect the physical activity, were excluded from the study. All the patients underwent baseline examination and follow-up examination including lung function, daily physical activity and venous blood sampling, and the time point differed among patients. Finally, the data from twenty-four outpatients were analyzed in current study. The median (inter quartile range; IQR) time between the baseline examination and follow-up examination was 439 (245–725) days. Written informed consent was obtained from all patients. All experiments in the current study were approved by the ethics committee of Tohoku University Graduate School of Medicine (approval number: #2019-1-353) and Tohoku Rosai Hospital (approval number: #15-11). This study was conducted in accordance with the Declaration of Helsinki. Whole blood was collected into vacutainer tubes containing an anticoagulant. The plasma samples were obtained by centrifugation of the tubes at 3000 rpm for 10 minutes and then stored at −80°C until the assay. Physical activity was measured by a triaxial accelerometer for 2 weeks and the levels of physical activity were analyzed.19,21
Assessment of Physical Activity
Patients wore the triaxial accelerometer (Actimarker; Panasonic CO., Osaka, Japan) during the daytime for 14 consecutive days. From among the 14 days monitoring data, except the first and last days, the data of 5 non-rainy weekdays were analyzed according to previous studies.21 We measured the parameters of physical activity as follows: number of steps, duration of activity at ≥2.0, ≥2.5 and ≥3.0 metabolic equivalents (METs).
Definition of Exacerbation of COPD
Exacerbation was defined as an acute worsening of respiratory symptoms that led to the prescription of antibiotics and/or corticosteroids.22–24 The number of exacerbations was assessed by medical reports for 2 years from the baseline.
Immunoblot Analysis of Plasma GDF11
Immunoblot analysis of the plasma GDF11 levels was performed as described previously.18,19 The plasma samples were diluted with saline and the diluted samples were mixed with sample buffer (Bio-Rad Laboratories, Hercules, CA). The samples containing 50 µg of protein were loaded and separated by electrophoresis on 10% SDS polyacrylamide gels. In addition to the samples, in order to compare samples from separate gels, 50 µg of a particular sample was loaded. After electrophoresis, the separated proteins were transferred to a PVDF membrane (Merck Millipore Ltd., Darmstadt, Germany). We used an antibody raised against GDF11 specifically (1:1000 dilution, R&D Systems Inc., Minneapolis, MN).18 Bound antibodies were visualized using a peroxidase-conjugated anti-mouse goat antibody (1:2000 dilution, Santa Cruz Biotechnology Inc., Dallas, TX) and enhanced chemiluminescence (GE Healthcare Ltd., Buckinghamshire, UK) with a chemiluminescence imaging system (LAS-4000 mini; Fujifilm CO., Tokyo, Japan). Ponceau S staining was used to evaluate the amounts of protein. The GDF11 levels were calculated by measuring the intensity of the bands. Band intensity was quantified using ImageJ 1.52v software (National Institutes of Health, Bethesda, MD). In order to compare across blots, the protein levels were normalized against a particular sample.
Measurement of Plasma IL-6, hs-CRP and MDA
Plasma levels of interleukin-6 (IL-6) and high sensitivity C-reactive protein (hs-CRP) were measured with an enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems Inc., Minneapolis, MN), according to the manufacturer’s instructions. Malondialdehyde (MDA), which is a marker of lipid peroxidation, was measured as thiobarbituric acid (TBA) reactive substances according to the manufacturer’s instructions (Abcam plc., Cambridge, UK).
Statistical Analysis
The data were expressed as mean ± standard deviation (SD) or median (interquartile range [IQR]) as appropriate. Data were analysed using the Wilcoxon Rank-Sum Test. Statistical correlation analyses were performed using Spearman’s rank test. A linear regression analysis was performed using the method of least squares. Multivariable models were used to determine associations between the decline of physical activity and other variables. Multiple regression analysis was performed using the change in the number of steps as the dependent variable. The independent variables were selected using a stepwise approach and included in the model (ie, change in GDF11). Potential confounders such as age, body mass index (BMI), lung function, inflammatory markers were also included in the model. Statistical analysis was performed using the GraphPad Prism 6 (GraphPad Software Inc., San Diego, CA) and JMP v15 Pro (SAS Institute Inc., Cary, NC). P values less than 0.05 were considered significant.
Results
Characteristics at Baseline
Twenty-four patients with COPD (GOLD stage I, n= 2; GOLD stage II, n =11; GOLD stage III, n =8; GOLD stage IV, n =3) took part in the current study. The characteristics of the study subjects are presented in Table 1. The mean age of participants was 72.2 years and the mean % predicted of FEV1 was 54.4%. Concerning the basal level of physical activity, the number of median steps per day was 2846.
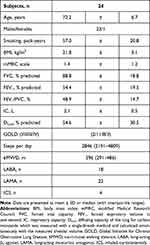 |
Table 1 Characteristics of the Study Subjects |
Longitudinal Association Between Plasma Level of GDF11 and Physical Activity
At first, there was no significant difference in the median number of steps (IQR) in the patients with COPD between at baseline and the follow-up in the current study [2846 (2191–4809) at baseline vs 2551 (1687–5052) at the follow-up, p = 0.751] (Supplementary Figure S1). Also, there was no significant difference in the mean level of plasma GDF11 between the two time points (Supplementary Figure S2). When we evaluated the longitudinal association between the plasma level of GDF11 and physical activity, the changes in the plasma levels of GDF11 in the patients with COPD had a significant positive correlation with the changes in the number of steps (r = 0.41, p = 0.047, Figure 1A). In the sub-analysis with the intensity of the physical activity, the changes in the plasma levels of GDF11 had significant positive correlations with changes in the duration ≥3.0 METs (r = 0.57, p = 0.004, Figure 1B) and ≥2.5 METs (r = 0.50, p = 0.013, Figure 1C), but not with those of a duration ≥2.0 METs (r = 0.39, p = 0.056, Figure 1D). We also evaluated whether the severity of COPD affect the changes of physical activity, but there was no significant correlation between the basal % predicted of FEV1 and the changes in the number of steps (r = 0.28, p = 0.181).
Longitudinal Relationship Between Systemic Inflammatory Markers and Physical Activity
Since the daily physical activity has also been shown to be associated with systemic inflammation in COPD,25,26 we investigated the longitudinal relationship between the number of steps and systemic inflammatory markers including IL-6, hs-CRP and MDA. However, there were no correlations between the changes in physical activity and the changes in the values of plasma IL-6 (r = −0.06, p = 0.787, Figure 2A), hs-CRP (r = −0.23, p = 0.325, Figure 2B) or MDA (r = −0.35, p = 0.135, Figure 2C). In the sub-analysis with the intensity of physical activity, there were no relationships between the changes in the duration of METs and the systemic inflammatory markers (data not shown).
Multiple Regression Analysis of the Changes in Physical Activity in the Patients with COPD
Next, we investigated factors that influence the changes in physical activity by using multiple regression analysis in the patients with COPD (Table 2). In the current study, there was no relation between the changes in the daily activity and various variables including age, BMI, mMRC scale and changes in FEV1 or inspiratory capacity (IC). Concerning the humoral factors, the change in the plasma level of GDF11, but not IL-6, was significantly related to the change in number of steps per day independently of the inflammation markers and lung function (β = 0.93, p < 0.001, Table 2).
 |
Table 2 Multiple Regression Analysis of the Changes in Physical Activity in the Patients with COPD |
Relationship Between Decreased Level of GDF11 and the Exacerbations of COPD
The decline of daily physical activity was reported to be associated with more frequent hospitalization caused by exacerbations of COPD.6,7 Therefore, we also investigated whether the change in the plasma level of GDF11 was associated with the exacerbation of COPD. Firstly, since there remained no suitable cut-off value for GDF11, we divided the patients into a GDF11 high group (ie, plasma level of GDF11 ≥ mean at baseline) and GDF11 low group (ie, plasma level of GDF11 < mean at baseline) as a matter of convenience and compared the incidence of exacerbations of COPD. However, the incidence of exacerbations was not significantly different between the two groups at baseline (data not shown). Therefore, we re-defined the groups as a GDF11 maintained group (ie, the amount of plasma level of GDF11 after the follow up was same or higher than that at baseline) and a GDF11 decline group (ie, the amount of plasma level of GDF11 after the follow up was lower than that at baseline) and compared the frequency of exacerbations between the two groups. Patients who maintained their plasma level of GDF11 showed a significantly lower incidence in exacerbations of COPD than those with a decreased level of GDF11. (p = 0.041, Figure 3).
Concerning the relationship between GDF11 and exacerbations, there was also a possibility that physical activity decreased due to exacerbations and GDF11 decreased as a result. Although we evaluated whether exacerbations affected the changes of physical activity, the changes in the number of steps were not significantly different between the groups with or those without exacerbations (Supplementary Figure S3).
Discussion
In the present study, we demonstrated that a longitudinal change in the plasma level of GDF11 in patients with COPD was positively correlated with changes in the daily physical activity. Especially, in those with higher exercise intensity, a stronger positive correlation was shown in the longitudinal changes between the plasma level of GDF11 and the physical activity. Multiple regression analysis also showed that changes in the plasma level of GDF11 were related to changes in the daily physical activity independently of the lung function. In addition, we demonstrated that the patients who maintained their plasma level of GDF11 showed a significantly lower incidence of exacerbations of COPD than those with decreased levels of GDF11. From these results, we confirmed that GDF11 could be a useful humoral factor that reflects the physical activity in COPD.
GDF11 has been shown to be one of the possible myokines from mice studies,14,16,17 which suggest that the plasma level of GDF11 might be associated with the physical activity, especially in higher exercise intensity. In our previous cross-sectional study, we firstly showed that the plasma level of GDF11 in patients with COPD was decreased and positively correlated with the daily physical activity.19 In the current study, we demonstrated that longitudinal changes in the plasma level of GDF11 in the patients with COPD were positively correlated with changes in the daily physical activity with an exercise intensity-dependent manner. This result further supports that GDF11 could act as a myokine in the human body same as in mice. However, it remains unclear whether GDF11 secretion is promoted by exercise or rehabilitation. Further studies are needed to clarify this issue.
Systemic inflammation and oxidative stress in COPD have been reported to be associated with reduced levels of physical activity.25–27 Therefore, we examined whether changes in physical activity are related to systemic inflammatory markers including IL-6, hs-CRP and MDA in the current longitudinal study. However, no significant correlations were found between them in this study. Also, there is a previous study consistent with our current results, which reported that the decline in physical activity was not in parallel with a worsening of the inflammation in patients with COPD.5 Concerning the correlation between systemic inflammation and the physical activity, there is an inconsistency between the studies. This difference might be due to the relatively small number of patients and short follow-up period in our current study. A larger study population and longer period might be needed for evaluating the relationship between systemic inflammatory markers and the physical activity.
In the current study, we demonstrated that the plasma level of GDF11 in the patients with COPD was associated with the amount of high intensity physical activities. However, a recent report has shown that high intensity physical activity was associated with increased mortality in patients with severe COPD, suggesting that high intensity physical activity may be harmful to such patients.28 Although there is a possibility that high intensity physical activity in severe COPD patients causes systemic inflammation and increased the level of IL-6, our current study did not show apparent changes in the plasma level of IL-6. This might be due to differences in the severity of the patients with COPD. In our current study, the severity of the participants was mainly GOLD grade II or III, but not grade IV. High intensity physical activity in less severe patients with COPD may not induce plasma level of IL-6.
The decline of daily physical activity has been shown to be associated with more frequent hospitalization caused by exacerbation of COPD.6,7 Therefore, we also investigated whether the plasma GDF11 is related to the exacerbations. In the current study, we first demonstrated a significantly lower rate of exacerbations in the patients who maintained their plasma level of GDF11 compared to those with decreased levels of GDF11. This result could suggest the possibility that the plasma level of GDF11 is correlated with the incidence of exacerbation. This correlation could be explained by the anti-inflammatory effect of GDF-11 that we have previously reported.18 However, we could not demonstrate a relationship between the basal level of GDF 11 and the incidence of the exacerbation in the current study. This might be due to the small sample size and the short follow-up duration. On the other hand, there is also a possibility that the physical activity decreases due to exacerbations leading to the decreased level of GDF11. However, in the current study, exacerbations did not appear to influence the changes in the number of steps. Therefore, the influence of exacerbations on the changes in physical activity in our current study could be small. This might be due to the relatively low rate of exacerbations in our current study. Further study is necessary to confirm the relationship between GDF11 and exacerbations of COPD.
There are several limitations in the present study. First, the sample size of this study was relatively small, especially in the severe grade of COPD. The current results should be verified in another study with larger cohorts to confirm the findings. Second, the follow-up duration before the re-evaluation in the plasma sample, physical activity and lung function was not equal among the patients, which might have caused lower sensitivity.
In conclusion, we demonstrated that the longitudinal change in the plasma level of GDF11 was positively correlated with the change in the daily physical activity in patients with COPD. This result suggests that GDF11 could be a useful humoral factor that reflects the daily physical activity in COPD.
Summary at a Glance
This is the first study to investigate the longitudinal association between the plasma level of GDF11 and physical activity in patients with COPD. We showed that the amount of GDF11 was positively correlated with the physical activity in COPD, which also had a possible relationship with exacerbations.
Acknowledgments
We thank Mr. Brent Bell for reading this manuscript. This study was supported by grants from the Japan Society for the Promotion of Science (grant number: #18K15916), a fund from the Japanese Respiratory Foundation, and supported by the Environmental Restoration and Conservation Agency of Japan.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Pitta F, Troosters T, Spruit MA, Probst VS, Decramer M, Gosselink R. Characteristics of physical activities in daily life in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2005;171:972–977. doi:10.1164/rccm.200407-855OC
2. Watz H, Waschki B, Meyer T, Magnussen H. Physical activity in patients with COPD. Eur Respir J. 2009;33:262–272. doi:10.1183/09031936.00024608
3. Troosters T, Sciurba F, Battaglia S, et al. Physical inactivity in patients with COPD, a controlled multi-center pilot-study. Respir Med. 2010;104:1005–1011. doi:10.1016/j.rmed.2010.01.012
4. Clarenbach CF, Sievi NA, Haile SR, et al. Determinants of annual change in physical activity in COPD. Respirology. 2017;22:1133–1139. doi:10.1111/resp.13035
5. Waschki B, Kirsten AM, Holz O, et al. Disease progression and changes in physical activity in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2015;192:295–306. doi:10.1164/rccm.201501-0081OC
6. Garcia-Aymerich J, Lange P, Benet M, Schnohr P, Antó JM. Regular physical activity reduces hospital admission and mortality in chronic obstructive pulmonary disease: a population-based cohort study. Thorax. 2006;61:772–778. doi:10.1136/thx.2006.060145
7. Albarrati AM, Gale NS, Munnery MM, Cockcroft JR, Shale DJ. Daily physical activity and related risk factors in COPD. BMC Pulm Med. 2020;20(1):60. doi:10.1186/s12890-020-1097-y
8. Vaes AW, Garcia-Aymerich J, Marott JL, et al. Changes in physical activity and all-cause mortality in COPD. Eur Respir J. 2014;44:1199–1209. doi:10.1183/09031936.00023214
9. Garcia-Rio F, Rojo B, Casitas R, et al. Prognostic value of the objective measurement of daily physical activity in patients with COPD. Chest. 2012;142:338–346. doi:10.1378/chest.11-2014
10. Waschki B, Kirsten A, Holz O, et al. Physical activity is the strongest predictor of all-cause mortality in patients with COPD: a prospective cohort study. Chest. 2011;140:331–342. doi:10.1378/chest.10-2521
11. Gimeno-Santos E, Frei A, Steurer-Stey C, et al. Determinants and outcomes of physical activity in patients with COPD: a systematic review. Thorax. 2014;69:731–739. doi:10.1136/thoraxjnl-2013-204763
12. Minakata Y, Sugino A, Kanda M, et al. Reduced level of physical activity in Japanese patients with chronic obstructive pulmonary disease. Respir Investig. 2014;52(1):41–48. doi:10.1016/j.resinv.2013.06.002
13. McPherron AC, Lawler AM, Lee SJ. Regulation of anterior/posterior patterning of the axial skeleton by growth/differentiation factor 11. Nat Genet. 1999;22:260–264. doi:10.1038/10320
14. Loffredo FS, Steinhauser ML, Jay SM, et al. Growth differentiation factor 11 is a circulating factor that reverses age-related cardiac hypertrophy. Cell. 2013;153:828–839. doi:10.1016/j.cell.2013.04.015
15. Katsimpardi L, Litterman NK, Schein PA, et al. Vascular and neurogenic rejuvenation of the aging mouse brain by young systemic factors. Science. 2014;344:630–634. doi:10.1126/science.1251141
16. Sinha M, Jang YC, Oh J, et al. Restoring systemic GDF11 levels reverses age-related dysfunction in mouse skeletal muscle. Science. 2014;344:649–652. doi:10.1126/science.1251152
17. Lee M, Oikawa S, Ushida T, Suzuki K, Akimoto T. Effects of exercise training on growth and differentiation factor 11 expression in aged mice. Front Physiol. 2019;10:970. doi:10.3389/fphys.2019.00970
18. Onodera K, Sugiura H, Yamada M, et al. Decrease in an anti-ageing factor, growth differentiation factor 11, in chronic obstructive pulmonary disease. Thorax. 2017;72:893–904. doi:10.1136/thoraxjnl-2016-209352
19. Tanaka R, Sugiura H, Yamada M, et al. Physical inactivity is associated with decreased growth differentiation factor 11 in chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2018;13:1333–1342. doi:10.2147/COPD.S157035
20. Global initiative for chronic obstructive lung disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease; 2021. 2021 Report. Available from: http://goldcopd.org/.
21. Sugino A, Minakata Y, Kanda M, et al. Validation of a compact motion sensor for the measurement of physical activity in patients with chronic obstructive pulmonary disease. Respiration. 2012;83:300–307. doi:10.1159/000330046
22. Hurst JR, Wedzicha JA. What is (and what is not) a COPD exacerbation: thoughts from the new GOLD guidelines. Thorax. 2007;62:198–199. doi:10.1136/thx.2007.077883
23. Wedzicha JA, Seemungal TA. COPD exacerbations: defining their cause and prevention. Lancet. 2007;370:1786–1796. doi:10.1016/S0140-6736(07)61382-8
24. Seemungal TA, Donaldson GC, Paul EA, Bestall JC, Jeffries DJ, Wedzicha JA. Effect of exacerbation on quality of life in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1998;157(5):1418–1422. doi:10.1164/ajrccm.157.5.9709032
25. Watz H, Waschki B, Boehme C, Claussen M, Meyer T, Magnussen H. Extrapulmonary effects of chronic obstructive pulmonary disease on physical activity: a cross-sectional study. Am J Respir Crit Care Med. 2008;177:743–751. doi:10.1164/rccm.200707-1011OC
26. Moy ML, Teylan M, Weston NA, Gagnon DR, Danilack VA, Garshick E. Daily step count is associated with plasma C-reactive protein and IL-6 in a US cohort with COPD. Chest. 2014;145:542–550. doi:10.1378/chest.13-1052
27. Garcia-Aymerich J, Serra I, Gómez FP, et al. Phenotype and course of COPD (PAC-COPD) Study Group. Physical activity and clinical and functional status in COPD. Chest. 2009;136:62–70.
28. Schaadt L, Christensen R, Kristensen LE, Henriksen M. Increased mortality in patients with severe COPD associated with high-intensity exercise: a preliminary cohort study. Int J Chron Obstruct Pulmon Dis. 2016;11:2329–2334. doi:10.2147/COPD.S114911
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.