Back to Journals » Nature and Science of Sleep » Volume 6
Longitudinal assessment of sleep disordered breathing in Vietnam veterans with post-traumatic stress disorder
Authors Yesavage J, Kinoshita L, Noda A, Lazzeroni L, Fairchild J, Friedman L, Sekhon G, Thompson S, Cheng J, Zeitzer J
Received 28 March 2014
Accepted for publication 22 April 2014
Published 29 October 2014 Volume 2014:6 Pages 123—127
DOI https://doi.org/10.2147/NSS.S65034
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Jerome A Yesavage,1,2 Lisa M Kinoshita,1,2 Art Noda,2 Laura C Lazzeroni,2 Jennifer Kaci Fairchild,1,2 Leah Friedman,1,2 Gundeep Sekhon,1,2 Stephanie Thompson,1,2 Jauhtai Cheng,1,2 Jamie M Zeitzer1,2
1Department of Veterans Affairs Health Care System, Palo Alto, CA, USA; 2Department of Psychiatry and Behavioral Sciences, Stanford University School of Medicine, Stanford, CA, USA
Purpose: Previous work has demonstrated the relatively high prevalence of risk factors for cognitive impairment, such as sleep disordered breathing (SDB) and obesity, in Vietnam War era veterans with post-traumatic stress disorder (PTSD). No data are currently available on the longitudinal stability of SDB as a risk factor for cognitive decline in that population, which this study now reports.
Methods: Sample consisted of 48 veterans of the Vietnam War with PTSD who completed longitudinal sleep assessments over a 3-year period. The primary outcome measure, the Apnea-Hypopnea Index (AHI) indicator, was determined during standard overnight polysomnography. Body mass index (BMI) was calculated using standard measurements. Measures of cognitive function tapped auditory verbal memory as measured by the Rey Auditory Verbal Learning Test and executive functioning as measured by the Color-Word Interference Test of the Delis–Kaplan Executive Function System battery. Statistical analyses included mixed effects modeling.
Results: In this sample, AHI increased significantly by 2.19 points per year (β=2.19; P<0.005). AHI worsened over the 3-year period, increasing from a mean of 18.7±15.7 to 24.7±17.4 points. Neither BMI nor cognition showed significant change over the 3-year period.
Conclusion: SDB worsened in a group of veterans of the Vietnam War with PTSD over a 3-year period. The worsening of SDB over time suggests the need for appropriate countermeasures in populations at risk for progression of the condition.
Keywords: SDB, PTSD, sleep apnea, BMI, obesity, cognition
Introduction
Veterans of the Vietnam War have higher rates of medical and psychiatric comorbidities than similarly aged non-veterans.1 Sleep disordered breathing (SDB), as defined by the National Institutes of Health (http://www.nhlbi.nih.gov) describes a group of disorders characterized by abnormalities of respiratory pattern (pauses in breathing) or the quantity of ventilation during sleep. Obstructive sleep apnea (OSA), the most common such disorder, is characterized by the repetitive collapse or partial collapse of the pharyngeal airway during sleep and the need to arouse to resume ventilation. Previous work has demonstrated a prevalence of SDB of approximately 60% in Vietnam War veterans with post-traumatic stress disorder (PTSD), and with high rates of obesity.2,3 In a study comparing veterans with PTSD to veterans without PTSD, 82% of those with PTSD were overweight, obese, or morbidly obese.4 The average body mass index (BMI) of Vietnam War veterans with PTSD has been estimated to be 30.5±5.9 kg/m2.4 These comorbidities appear to place Vietnam veterans at an increased risk of cognitive decline.5 However, no data are currently available on the stability of SDB or obesity in these populations, or on the longitudinal relationships of these conditions and cognition. A better understanding of these conditions is necessary to develop effective countermeasures. Using data from the PTSD Sleep Apnea Clinical Study, we examined the trajectory of SDB, obesity and cognition in Vietnam veterans with PTSD. Given the relationship between each of these conditions, we hypothesized that the conditions would follow similar trajectories over the 3-year period.
Methods
Participants
Forty-eight participants were selected from a cohort of community-dwelling male veterans aged 55 years or older who were recruited into a study of the effects of PTSD, sleep apnea, and cognition.2,3 Recruitment methods included media advertisement, contact with local veteran agencies, and clinician referral from the outpatient clinics of Veterans Affairs (VA) Palo Alto Health Care System (VAPAHCS). The 48 participants selected for this analysis completed annual assessments over a 3-year period. Informed consents, approved by Stanford University and VAPAHCS Institutional Review Boards, were obtained from all participants.
The presence of PTSD was determined by the Clinician Administered PTSD Scale for Diagnostic and Statistical Manual, Fourth Edition (DSM-IV) (CAPS),6 with the entry criteria of a CAPS current and/or lifetime score ≥40. Participants were excluded if they: 1) had a current or lifetime history of any other psychiatric disorder with primary psychotic features or symptoms that met the DSM-IV as determined by the Structured Clinical Interview for DSM-IV TR Axis I Disorders, Research Version, Non-patient Edition. (SCID-I/NP);7 2) had a current or recent exposure to trauma in the past 3 months (chart review, interview); 3) met DSM-IV criteria for drug and/or alcohol abuse or dependence (except nicotine) within the last 30 days as determined by the SCID;7 4) were alcohol intoxicated (by breathalyzer), in alcohol withdrawal (by physical exam) during testing, or exhibited toxicology evidence of illicit substance use prior to exam (72-hour metabolite toxicology screen); 5) showed evidence of probable or possible dementia (Mini-Mental State Examination8 score of <23); 6) reported a history of seizure or other neurologic disorder or system illness affecting central nervous system function (chart review, interview); 7) suffered a head injury within 1 year or had a history of loss of consciousness >24 hours or; 8) used systemic steroid medication (with the exception of estrogen replacement therapy). Participants were not recruited on the basis of presence or absence of sleep or SDB complaints.
Patients were informed of a suspected SDB finding when the Apnea-Hypopnea Index (AHI) was ≥10. Referral to Pulmonary Medicine specialist was facilitated. At baseline, seven subjects reported a prior apnea diagnosis but only two were using continuous positive airway pressure (CPAP) machines. At the final visit, the number of subjects using CPAP had increased to 13, of which only five reported nightly use.
Measures
Cognitive assessments and overnight polysomnography were performed by trained clinicians or technicians.
Body mass index
BMI was determined by VA staff using the standard formula (weight in pounds ×703)/(height in inches)2.
Polysomnography
SDB was assessed using unattended, overnight polysomnography (Safiro Ambulatory Polysomnography System; Compumedics USA Inc., Charlotte, NC, USA). The data were scored for sleep staging and respiratory events by a registered polysomnographic technologist9 and were reviewed by a diplomat of the American Board of Sleep Medicine (Dr Cheng). The primary outcome measure was the AHI, a descriptor of the severity of SDB. Respiratory events were defined as cessation (apnea) or discrete reduction (hypopnea) in airflow or chest wall impedance, lasting at least 10 seconds and associated with oxygen desaturation level of at least 3%. The AHI represents “the number of apneas and hypopneas divided by the number of hours of sleep”.10 All subjects with clinically significant SDB (defined as AHI ≥10) agreed to have their primary medical doctors notified of this finding. Mean pulse oximeter oxygen saturation (mean SpO2) was also measured during polysomnography.
Cognitive assessments
Auditory verbal memory was assessed with the Rey Auditory Verbal Learning Test (RAVLT).11 The RAVLT is a reliable and valid measure of verbal learning and memory. Note that a higher RAVLT score indicates higher levels of cognitive functioning. For the purposes of these analyses, we used the sum of trials I through V of the RAVLT.
Executive function was assessed with the Color-Word Interference Test (CWIT) from the Delis–Kaplan Executive Function System battery.12 For this study, we used a CWIT time score, which was calculated by subtracting the average time it took each participant to complete the color and word naming tasks from the time it took each participant to complete the CWIT [interference time – ((color-naming time + word-naming time)/2)]. Higher CWIT scores indicate lower levels of cognitive functioning.
Statistical analyses
We used mixed effects modeling to examine the change in the variables of interest (ie, AHI, BMI, and cognition) over the 3-year period. Estimation of our model was performed using the SAS PROC MIXED procedure, version 9.3 (SAS Institute, Cary, NC, USA). In the analysis of our primary outcome measure, AHI was the dependent variable and year (0, 1, 2, or 3) was the predictor. In our secondary analyses, we ran three additional mixed models where BMI, the sum of trials I through V of the RAVLT, and the CWIT time score were dependent variables, with year as the predictor variable for each model.
Results
Table 1 contains a description of our sample of Vietnam War era veterans. All are male, and the prevalence of SDB was 60% using a criterion of AHI ≥10/hour. Seventy-three percent of our sample was non-Hispanic, and the same percentage was Caucasian.
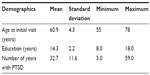  | Table 1 Description of the clinical sample (N=48 subjects) |
Table 2 contains the yearly mean AHI, mean SpO2, BMI, RAVLT, and CWIT scores, along with standard deviations and standard errors, minimums and maximums. Based on the model, AHI increased significantly by 2.19 points per year (β=2.19; P<0.005). AHI worsened over the 3-year period, increasing from a mean of 18.7±15.7 points to 24.7±17.4 points. Figure 1 shows the change of AHI over time with corresponding standard errors. Analysis of other outcomes of interest (ie, BMI and cognitive function) revealed no significant change over the 3-year period (P-values ≥0.20).
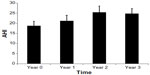  | Figure 1 Apnea-Hypopnea Index (AHI) change over time, with corresponding standard errors. |
Discussion
This study examined the 3-year trajectory of SDB, BMI, and cognitive function in a sample of older Vietnam War era veterans diagnosed with PTSD. Prior work has demonstrated high prevalence rates of both SDB and obesity in this population.2,3 While SDB was found to worsen significantly over the 3-year period, no significant change was seen in cognitive function or BMI.
In light of prior work demonstrating the negative impact that SDB has on cognition,5 the stability of cognitive functioning in the presence of worsening SDB was surprising. This observation may be due to the fact that the duration of follow-up may not have been sufficient to document change in non-demented individuals.
The stability of BMI is similar to what has been reported in other studies of older adults.13 Current military personnel with PTSD also seem to suffer from an increased incidence of SDB and obesity. A recent study of 110 military personnel returned from deployment found that within 18 months of deployment, 62.7% met diagnostic criteria for OSA and had a mean BMI of 30.0±4.3 kg/m2.14 Thus the prevalence of 60% found in our veterans is similar to that reported in recently deployed military personnel.
Prior cross-sectional epidemiological work has shown that the prevalence of sleep apnea tends to increase with age but the clinical significance (severity) of apnea appeared to decrease.15 This is distinct from our current longitudinal work that shows an increase in apnea severity. One possible explanation is that subjects with the most severe apnea die at earlier ages and have not been sampled when they reached an old age, thus falsely implying a reduction of apnea symptoms in older cohorts. Although our subjects were followed for 3 years, longer follow-up would provide better mortality data.
SDB is widely accepted as having significant negative effects on health status. Our findings demonstrate that over a 3-year period, SDB was found to significantly worsen. This worsening occurred at a time of life in which there is an increased risk of cognitive impairment. Given the high prevalence rate of AHI in this sample, coupled with the increased severity of SDB symptoms over time, it is important for providers to regularly assess for SDB in veterans. Further analysis of participant characteristics in the current sample is limited due to the small sample size, thus the importance of future research with larger samples. Finally, successful treatment of SDB is challenging in civilians.16 Identification of characteristics in veterans who often have PTSD and increased SDB is necessary to develop novel veteran-centric treatment approaches.17
Acknowledgments
This research was supported by the Medical Research Service of the Department of Veterans Affairs, VA Merit Review Grant and the Sierra-Pacific Mental Illness Research, Education, and Clinical Center (MIRECC).
Disclosure
The authors declare that they have no conflict of interest in this work.
References
Agha Z, Lofgren RP, VanRuiswyk JV, Layde PM. Are patients at Veterans Affairs medical centers sicker? A comparative analysis of health status and medical resource use. Arch Intern Med. 2000;160(21):3252–3257. | |
Kinoshita LM, Yesavage JA, Noda A, et al. Modeling the effects of obstructive sleep apnea and hypertension in Vietnam veterans with PTSD. Sleep Breath. 2012;16(4):1201–1209. | |
Yesavage JA, Kinoshita LM, Kimball T, et al. Sleep-disordered breathing in Vietnam veterans with posttraumatic stress disorder. Am J Geriatr Psychiatry. 2012;20(3):199–204. | |
Vieweg WV, Julius DA, Fernandez A, Tassone DM, Narla SN, Pandurangi AK. Posttraumatic stress disorder in male military veterans with comorbid overweight and obesity: psychotropic, antihypertensive, and metabolic medications. Prim Care Companion J Clin Psychiatry. 2006;8(1):25–31. | |
Yaffe K, Vittinghoff E, Lindquist K, et al. Posttraumatic stress disorder and risk of dementia among US veterans. Archives of General Psychiatry. 2010;67(6):608–613. | |
Blake DD, Weathers FW, Nagy LM, et al. The development of a Clinician-Administered PTSD Scale. J Trauma Stress. 1995;8(1):75–90. | |
First MB, Spitzer RL, Gibbon M, Williams JBW. Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version, Non-patient Edition. (SCID-I/NP) New York: New York: Biometrics Research, New York State Psychiatric Institute; 2002. | |
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12:189–198. | |
Rechtschaffen A, Kales A. A Manual of Standardized Terminology, Techniques and Scoring System for Sleep Stages of Human Subjects. Washington, DC: US Government Printing Office; 1968. | |
International Classification of Sleep Disorders. Chicago, Illinois: American Academy of Sleep Medicine; 2005. | |
Rey A. L’examen clinique en psychologie [Clinical Examination in Psychology]. Paris: Presse Universitaire de France; 1958. French. | |
Delis DC, Kaplan E, Kramer JH, Delisle MB. Kaplan Executive Function System. San Antonio: The Psychological Corporation; 2001. | |
Botoseneanu A, Liang J. The effect of stability and change in health behaviors on trajectories of body mass index in older Americans: a 14-year longitudinal study. J Gerontol A Biol Sci Med Sci. 2012;67(10):1075–1084. | |
Mysliwiec V, Gill J, Lee H, et al. Sleep Disorders in US Military Personnel: A High Rate of Comorbid Insomnia and Obstructive Sleep Apnea. Chest. 2013;144(2):549–557. | |
Bixler EO, Vgontzas AN, Ten Have T, Tyson K, Kales A. Effects of age on sleep apnea in men: I. Prevalence and severity. Am J Respir Crit Care Med. 1998;157(1):144–148. | |
Weaver TE. Don’t start celebrating – CPAP adherence remains a problem. J Clin Sleep Med. 2013;9(6):551–552. | |
Parthasarathy S, Wendel C, Kuna S. A Pilot Study of CPAP Adherence Promotion by Peer Buddies with Sleep Apnea. J Clin Sleep Med. 2013;9:543–550. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

