Back to Journals » Journal of Multidisciplinary Healthcare » Volume 11
Long-term health complications following snake envenoming
Authors Jayawardana S, Arambepola C, Chang T, Gnanathasan A
Received 4 November 2016
Accepted for publication 10 January 2017
Published 26 June 2018 Volume 2018:11 Pages 279—285
DOI https://doi.org/10.2147/JMDH.S126648
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Subashini Jayawardana,1 Carukshi Arambepola,2 Thashi Chang,3 Ariaranee Gnanathasan3
1Department of Allied Health Sciences, Faculty of Medicine, University of Colombo, Colombo, Sri Lanka; 2Department of Community Medicine, Faculty of Medicine, University of Colombo, Colombo, Sri Lanka; 3Department of Clinical Medicine, Faculty of Medicine, University of Colombo, Colombo, Sri Lanka
Background: Snakebite is an important public health problem in tropical regions of the world. Although devastating effects of envenoming such as kidney failure, tissue necrosis, bleeding diathesis, and neurotoxicity are well known in the acute stage following a snakebite, the long-term effects of snake envenoming have not been adequately studied.
Materials and methods: A population-based study was conducted among 8707 residents in a rural district in Sri Lanka to assess the long-term sequelae following snakebite. Health-related complaints that snakebite victims had developed immediately or within 4 weeks of the bite and persisted for more than 3 months, were assessed by interviewer-administered questionnaire and in-depth interviews, and further evaluated by physical examination and relevant investigations.
Results: Of the 816 participants who were identified as ever snakebite victims, 112 (13.7%) presented with at least one snakebite-related long-term health complication. Among them, “migraine-like-syndrome” characterized by headache vertigo, and photosensitivity to sunlight was found in 46 (5.6%); musculoskeletal disorders such as pain, local swelling, muscle weakness, deformities, contractures, and amputations were found in 26 (3.2%); visual impairment in 21 (2.6%); acute kidney injury in 4 (0.5%); skin blisters at the bite site in 5 (0.6%); psychological distress in 2 (0.2%); hemiplegia in 1 (0.1%); right-side facial nerve palsy in 1 (0.1%); paresthesia over bite site in 1 (0.1%); generalized shivering in 1 (0.1%); and chronic nonhealing ulcer in 1 (0.1%). Interestingly, 31 (3.8%) reported nonspecific somatic symptoms such as abdominal colic, chest tightness, wheezing, receding gums, excessive hair loss, and lassitude with body aches following the bite. The average duration of symptoms since snakebite was 12.7 years (SD=11.7).
Conclusion: This study highlights that a significant proportion of snakebite victims suffer disabling chronic health sequelae. There is a need to place systems to address these unmet health needs.
Keywords: snakebite, population-based study, long-term health complications Sri Lanka
Introduction
There are six highly venomous and, therefore, medically important terrestrial snake species in Sri Lanka. Among them, cobra bite causes significant local envenoming; pain, swelling, blistering, and severe local necrosis as well as acute systemic envenoming; ptosis, diplopia, external ophthalmoplegia, and descending paralysis.1 Common krait and Sri Lankan krait bites cause a distinct clinical syndrome consisting of minimal or no local envenoming, abdominal pain, vomiting, ptosis, diplopia, external ophthalmoplegia, and descending paralysis that most often lead to respiratory paralysis.2 Russell’s viper bite is venomous enough to develop local envenoming, spontaneous bleeding, incoagulable blood, neurological manifestations, ptosis, ophthalmoplegia, descending paralysis, rhabdomyolysis, and acute kidney injury (AKI).3 Saw-scaled viper bite is responsible for coagulopathy and spontaneous systemic hemorrhage and local envenoming.4 Hump-nosed viper bite mostly accounts for severe local swelling, hemorrhagic blisters, local tissue necrosis, incoagulable blood, spontaneous bleeding mainly hematuria, and AKI.5
Venom toxins result in devastating effects that are usually encountered during the acute stage of snake envenoming, which will either result in death or, with timely medical management, including antivenom treatment, would resolve completely after the acute phase.6 However, given the dramatic nature and management required following snakebite, it is likely that many other venom toxin-related complications of victims, which would not amount to devastating outcomes, could go unnoticed during the acute stage owing to more attention paid by health care providers as well as patients to the fatal complications. Nevertheless, such complications that persist beyond the acute stage may unfavorably affect the day-to-day lives of snakebite victims. Other than a few studies,7–10 long-term effects of snake envenoming, as perceived by the victims, are not well documented in any population-based epidemiological studies.11
A recent national survey of snakebite with geospatial analysis estimated that the incidence of snakebite was 398 (95% CI: 356–441) per 100,000 population in Sri Lanka.12 Owing to the abundance of natural habitats in the environment, poor housing owing to poor economic status, and frequent engagement in outdoor farming occupations, rural residents in Sri Lanka are particularly at increased risk of snakebite. Furthermore, they represent the silent sufferers of envenoming effects in rural communities. In such vulnerable communities, no estimates on long-term health complications associated with snake envenoming are available. This study was conducted to identify the snakebite victims with long-term health sequelae in rural Sri Lanka.
Materials and methods
A population-based cross-sectional study was conducted in the district of Ampara that typically represents the rural population in Sri Lanka. The sample included the chief householder and all eligible members belonging to the nuclear family of 2500 households that were selected using a systematic random sampling method (50 GN divisions [the smallest administrative division in a district] × 50 households per GN division). Residents of not more than 1 year, semipermanent residents in commercial/work sites, permanent residents in religious institutes or elderly homes, and infants were excluded from the sample. An interviewer-administered questionnaire was developed for the study, which consisted of sociodemographic data, information on snakebite, health-seeking behavior, and long-term persisting health complications. The questionnaire was tested for its validity and reliability by conducting a pilot test in one of the GN divisions in the district of Ampara, which was not included in the current study.
Being an ever snakebite victim was ascertained based on history obtained from the participant. It was confirmed by eye witness accounts and documental evidence (diagnosis cards, clinic records, etc.). Health complications that had arisen immediately or within 4 weeks of the bite and had persisted for more than 3 months were defined as “long-term complications following snake envenoming”, which were initially identified using an interviewer-administered questionnaire. Those who reported such complaints underwent an in-depth interview to obtain information on the progression of long-term complications and their health-seeking behavior. This initial screening for long long-term complications was carried out by two graduates well trained in quantitative and qualitative data collection techniques. It was followed by a detailed history and physical examination by a physician and a neurologist for confirmation. Clinical diagnosis cards and other relevant documents issued by state hospitals were also perused. Investigations such as thyroid function test, ECG, genetic studies, and nerve conduction tests were performed, wherever relevant. Furthermore, those with musculoskeletal complaints were assessed by experienced physiotherapists by conducting comprehensive physical assessment with calibrated instruments (goniometer, handheld dynamometer, etc.).
Ethical considerations
Ethics approval was obtained from the ethics review committee, Faculty of Medicine, University of Colombo. Written informed consent was obtained from the patients for publication of case reports and images.
Results
Among the 8707 rural residents recruited for the study, 816 snakebite victims were identified. Among them, 112 victims (13.7%) presented with at least one snakebite-related long-term health complication (Figure 1). Their average age was 42.8 years (SD=15.1) while the duration of complications was 12.7 years (SD=11.7), ranging from 3 months to more than 30 years. In 55 (49.1%), the complications were attributed to venomous snakes, predominantly following Russell’s viper and cobra bites. Sociodemographic characteristics of the victims are shown in Table 1. Majority of them were males, aged ≥40 years, and of low socioeconomic status.
  | Figure 1 Distribution of long-term specific and nonspecific complications following snakebite. |
  | Table 1 Characteristics of the snakebite victims with persisting health complications following snakebite (N=112) Abbreviation: Rs., Sri Lankan Rupees. |
The most unique health complications persisting among the majority of victims (N=46; 5.6%) were a tendency to develop headache, faintness, vertigo, and photosensitivity to sunlight, which were not present prior to the bite. This was perceived by most victims as an obstacle for engaging in their outdoor farming occupations, to the extent that some had to alternatively work in the shade, take frequent rests from work, or give up their outdoor occupations. Russell’s viper, cobra, and krait were the offending snakes in 47.8%.
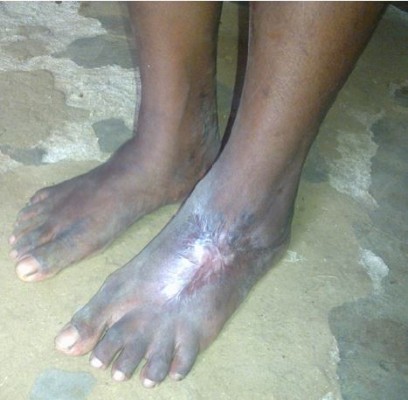
The next most frequent health complication was musculoskeletal complications on the affected limb (N=26; 3.2%), ranging from local pain, swelling, muscle weakness and wasting to chronic nonhealing ulcers, soft tissue contractures (Figure 2), deformities, and amputations (Figure 3). Lower limbs (61.5%) were the most commonly affected body part.
  | Figure 2 Soft tissue contracture of the left foot following snakebite. |
  | Figure 3 Amputation of ring finger of the right hand due to snakebite. |
Another common complication encountered was reduced vision of both eyes (N=21; 2.6%) noticed after the snakebite (none were using spectacles or having refractory errors prior to the bite). The impairment had no relationship with direct exposure to sunlight. In one, loss of vision was apparent. Russell’s viper, cobra, and krait were the offending snakes in 42.9%. The complication was disabling with difficulty in reading and recognition of persons.
Four victims developed AKI in the acute phase of envenoming and two of them were progressed to develop chronic kidney failure within 3 months and required long-term dialysis as renal replacement therapy. The offending snake was identified in three of them as Russell’s viper.
Hemiplegia due to ischemic stroke, isolated facial nerve palsy following Russell’s viper bite, paresthesia over the bite site, and generalized shivering of the body were the neurological complications other than the “migraine-like syndrome” that were attributed to snakebite. These had developed within the first 2 weeks of the bite and had persisted throughout.
Five victims showed skin blisters on the bite site, which had appeared on and off since the bite (0.6%). Chronic nonhealing ulcer following cobra bite was observed in one victim, who also showed extensive gum recession. Psychological distress was noted in two middle-aged female victims (0.2%) who had been bitten by Russell’s viper.
Interestingly, there were 31 victims who complained of nonspecific somatic symptoms (3.8%) following venomous (35.5%), nonvenomous (9.7%), and unidentified (54.8%) bites. The symptoms included chest tightness with no identifiable triggers (cardiac cause excluded by ECG; N=14), abdominal colic (abdominal cause excluded on history and examination; N=7), difficulty in breathing or short spells of “wheezing” that resolve on its own (respiratory cause excluded on history and examination; N=4), recession of gums exposing more of the teeth (N=3), lassitude with body aches and pains (N=3), and excessive hair loss (N=2). Of all the victims with long-term complications, 14 complaining of only nonspecific symptoms were noteworthy (12.5%).
The qualitative enquiry revealed that all these victims had sought western treatment at some point in time after the bite. However, other than those with renal failure, none had been followed up after hospital discharge for more than 3 months. For the complaints that were persisting, some had sought treatment from traditional medical practitioners while the majority had not. They claimed that they were unaware of any specialized care being offered in hospitals that would specifically address their disabling health issues. Further, although they firmly believed that their symptoms were attributed to snakebite, they appeared to accept them as incurable, thus not made an attempt to seek health care.
Patients showed varied health-seeking behavior following the health complications. A few examples are given below.
Case 1
A 40-year-old woman who had a cobra bite at the age of 8 years had been treated by an indigenous medical practitioner without the need for Western medical treatment. She was again bitten by a common krait at the age of 25 years for which she sought traditional treatment for 10 days. Since then, she has been experiencing a tendency to develop recurrent headaches, vertigo, and blurred vision on exposure to sunlight. In addition, she has developed an unsteady gait and tremor. On examination, ataxia was detected; however, genetic analysis confirmed that she had spinocerebellar ataxia type 1. Her migraine-like syndrome was attributed to snake envenoming while her ataxia was attributed to her genetic disorders.
Case 2
A 48-year-old farming woman presented with a chronic nonhealing ulcer in the right foot which had developed after a cobra bite 33 years previously. She had sought Western medical treatment and has had repeated skin grafting, but with no success. She subsequently had developed toe deformities and ankylosis of the ankle joint. Skin biopsy excluded malignant transformation of the ulcer while imaging studies excluded underlying chronic osteomyelitis.
Case 3
A 52-year-old woman had a Russell’s viper bite at the age of 29 years. She had lost consciousness immediately after the bite and was taken to a traditional healer where she was treated with herbal medicine delivered via inhalation and by immersing the patient in a medicinal boat. It was only after 3 days of this intensive traditional treatment that she had been admitted to hospital for Western medical treatment and was transferred to another hospital where facilities for advance treatment were available. She also received treatment for post snakebite psychological sequelae.
Discussion
This study describes the chronic health sequelae of snake envenoming and their prevalence in a rural agricultural community in Sri Lanka, who are plagued with the risk of snakebites as an occupational hazard, and highlights the disabling nature of the snakebite-related long-term complications.
Among the chronic health sequelae, a “migraine-like syndrome” characterized by episodic headaches, vertigo, and photosensitivity, which distinctly developed after the snakebite, was the most common. The photosensitivity proved most disabling in this predominantly sunny tropical climate making most of those afflicted to give up outdoor occupations, which had the greatest economic impact for the individual and family in this farming community. This type of photosensitivity has not been reported previously. It is hypothesized that this may have been triggered by effects of vasculotoxins of snake venom on cranial blood vessels predisposing the victims to migraine-like episodes, but further investigation with functional imaging is needed to elucidate the mechanisms underlying this phenomenon.
Musculoskeletal deformities attributed to the snakebite accounted for a fifth of long-term health sequelae. The disabilities were mostly in lower limbs and ranged from swelling, muscle wasting, reduced range of motion of joints, reduced muscle power, impaired balance, chronic nonhealing ulcers, abnormal gait, and fixed deformities to amputations. Chronic musculoskeletal sequelae are likely to be related to the damaging effect of venom on tissues at and adjacent to the bite site than the effect of systemic envenoming. It is less likely to relate such chronic disabilities to degenerative changes associated with aging because most of the snakebite victims with musculoskeletal disorders were below the age of 60 years, and physical disabilities were presented directly related to the bitten site. The detailed descriptions of these musculoskeletal complications of snakebite have been recently published.13 A need for therapies to counteract local venom effects has been recognized and used against some species.14
It is curious that many patients complained of visual impairments, and clinical examination excluded evidence of cataract or retinopathy. Since ours was a community-based retrospective study, we could not conduct formal ophthalmological evaluation to determine the exact nature of the visual complaint.
Four patients developed AKI after the snakebite. Among them, three were due to Russell’s viper envenoming while in one patient the culprit was not identified confidently. After recovering from the AKI, two of them continued to have raised serum creatinine. Ultrasound scan showed bilaterally contracted kidneys with loss of corticomedullary demarcation and high echogenicity consistent with chronic kidney disease (CKD). They required long-term renal replacement therapy such as hemodialysis. Russell’s viper venom produces hypotension, venom-induced consumption coagulopathy (VICC), direct nephrotoxicity, intravascular hemolysis, and rhabdomyolysis. CKD may occur after bilateral cortical necrosis (Russell’s viper and hump-nosed pit viper bites).3,5 In another study performed in Sri Lanka, among 54 snakebite victims who had AKI developed CKD. In this study, renal histology of six patients with CKD showed predominant glomerular sclerosis and interstitial nephritis.15 CKD is a long-term complication following snake envenoming and is probably one of the causes of a large group of patients with CKD of undetermined origin in Sri Lanka. A wide range of renal histological changes has been described after snakebite. Among them, acute tubular necrosis is the most common. Acute tubular necrosis may result from prolonged hypotension and hypovolemia, VICC, direct toxic effect of the venom on the renal tubules, hemoglobinuria, myoglobinuria, and hyperkalemia. Proliferative glomerulonephritis, interstitial nephritis, toxic mesangiolysis with platelet agglutination, fibrin deposition, ischemic nephropathy, and distal tubular damage (“lower nephron nephrosis”), suggesting direct venom nephrotoxicity attributable to venom PLA2 and metalloproteases, and bilateral renal cortical necrosis with subsequent calcification were also reported following snake envenoming.16,17
In this study, about 4% of the victims complained of nonspecific somatic symptoms following venomous (35.5%), nonvenomous (9.7%), and unidentified (54.8%) snakebites. Similar unexplained nonspecific symptoms have been reported following snakebites in Tamil Nadu, India.17 Tiredness (68%) was the most commonly reported symptom requiring the victims to either give up their occupations or find substitutes to work in the fields, while numbness; swelling of the face, hands, and legs; oozing from the bite site; blurred vision; lacrimation; giddiness; shivering; and nausea were the other reported symptoms.18 Nonspecific somatic symptoms following snakebite such as poor vision, tooth decay, body aches, headaches, weakness, and tiredness have also been reported in Sri Lanka.9
In our study, one patient developed an ischemic stroke manifesting clinically as hemiplegia following a Russel’s viper bite. Ischemic stroke following Russell’s, saw-scaled, and hump-nosed viper bites have been previously reported.10,19–21 It is believed to occur due to the procoagulant properties of snake venom and its direct action on vascular endothelial cells.19 Paresthesia around the bite site reported in one patient is likely to represent direct effects of snake venom on peripheral nerves as has been previously observed.22 However, isolated facial nerve palsy following snakebite has not been previously reported.
Limitation
Owing to the snakebite episodes taking place several years back, recall of the complications following the bite as well as the reliability in attributing it entirely to the bite could have been unreliable in some cases. To overcome this bias, we used memory aids such as a diary for obtaining detailed accounts of the complaint and also excluded all complications reported to have occurred 4 weeks after the bite and those not persisting at the time of data collection.
Conclusion
This study highlights that a significant proportion of snakebite victims suffer a wide spectrum of disabling chronic health sequelae, which often go undetected and unaddressed. It is recommended that screening for such snakebite-related long-term health sequelae be established at least in regions where snakebite is a public health problem, and snakebite victims should be followed up by multidisciplinary health care team to prevent long-term suffering of the rural residents with residual disabilities of the bite. Further analytical studies are needed to verify the relationship between envenoming and the chronic health sequelae of snakebites.
Acknowledgments
We are grateful to the snakebite victims who participated in the study and administrative Officers at the Divisional Secretariat and Gramasewa officers in the district of Ampara for their support in assisting data collection. This study was funded by the Higher Education for the Twenty first Century (HETC) fund.
Author contributions
All authors contributed toward data analysis, drafting and revising the paper and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Ariaratnam CA, Sheriff MH, Arambepola C, Theakston RD, Warrell DA. Syndromic approach to treatment of snake bite in Sri Lanka based on results of a prospective national hospital-based survey of patients envenomed by identified snakes. Am J Trop Med Hyg. 2009;81(4):725–731. | ||
Ariaratnam CA, Sheriff MH, Theakston RD, Warrell DA. Distinctive epidemiologic and clinical features of common krait (Bungarus caeruleus) bites in Sri Lanka. Am J Trop Med Hyg. 2008;79(3):458–462. | ||
Kularatne SA. Epidemiology and clinical picture of the Russell’s viper (Daboia russelii russelii) bite in Anuradhapura, Sri Lanka: a prospective study of 336 patients. Southeast Asian J Trop Med Public Health. 2003;34(4):855–862. | ||
Kularatne SA, Sivansuthan S, Medagedara SC, Maduwage K, de Silva A. Revisiting saw-scaled viper (Echis carinatus) bites in the Jaffna Peninsula of Sri Lanka: distribution, epidemiology and clinical manifestations. Trans R Soc Trop Med Hyg. 2011;105(10):591–597. | ||
Ariaratnam CA, Thuraisingam V, Kularatne SA, et al. Frequent and potentially fatal envenoming by hump-nosed pit vipers (Hypnale hypnale and H. nepa) in Sri Lanka: lack of effective antivenom. Trans R Soc Trop Med Hyg. 2008;102(11):1120–1126. | ||
Warrell DA. Snake bite. Lancet. 2010;375(9708):77–88. | ||
Abubakar SB, Habib AG, Mathew J. Amputation and disability following snakebite in Nigeria. Trop Doct. 2010;40(2):114–116. | ||
Chippaux JP. Estimate of the burden of snakebites in sub-Saharan Africa: a meta-analytic approach. Toxicon. 2011;57(4):586–599. | ||
Williams SS, Wijesinghe CA, Jayamanne SF, et al. Delayed psychological morbidity associated with snakebite envenoming. PLOS Negl Trop Dis. 2011;5(8):e1255. | ||
Gawarammana I, Mendis S, Jeganathan K. Acute ischemic strokes due to bites by Daboia russelii in Sri Lanka – first authenticated case series. Toxicon. 2009;54(4):421–428. | ||
de Silva HJ, Kasturiratne A, Pathmeswaran A, Lalloo DG. Snakebite: the true disease burden has yet to be determined. Ceylon Med J. 2013;58(3):93–95. | ||
Ediriweera DS, Kasturiratne A, Pathmeswaran A, et al. Mapping the risk of Snakebite in Sri Lanka – A National Survey with Geospatial Analysis. PLoS Negl Trop Dis. 2016;10(7):e0004813. | ||
Jayawardana S, Gnanathasan A, Arambepola C, Chang T. Chronic musculoskeletal disabilities following snake envenoming in Sri Lanka: a population-based study. PLoS Negl Trop Dis. 2016;10(11):e0005103. | ||
Gutiérrez JM, Theakston RD, Warrell DA. Confronting the neglected problem of snake bite envenoming: the need for a global partnership. PLoS Med. 2006;3(6):e150. | ||
Herath HM, Wazil AW, Abeysekara DT, et al. Chronic kidney disease in snake envenomed patients with acute kidney injury in Sri Lanka: a descriptive study. Postgrad Med J. 2012;88(1037):138–142. | ||
Sitprija V. Snakebite nephropathy. Nephrology (Carlton). 2006;11(5):442–448. | ||
Kanjanabuch T, Sitprija V. Snakebite nephrotoxicity in Asia. Semin Nephrol. 2008;28(4):363–372. | ||
Vaiyapuri S, Vaiyapuri R, Ashokan R, et al. Snakebite and its socio-economic impact on the rural population of Tamil Nadu, India. PLoS One. 2013;8(11):e80090. | ||
Jeevagan V, Chang T, Gnanathasan CA. Acute ischemic stroke following Hump-nosed viper envenoming; first authenticated case. Thromb J. 2012;10(1):21. | ||
Bashir R, Jinkins J. Cerebral infarction in a young female following snake bite. Stroke. 1985;16(2):328–330. | ||
Kularatne SA. Common krait (Bungarus caeruleus) bite in Anuradhapura, Sri Lanka: a prospective clinical study, 1996–1998. Postgrad Med J. 2002;78(919):276–280. | ||
Seneviratne U, Dissanayake S. Neurological manifestations of snake bite in Sri Lanka. J Postgrad Med. 2002;48(4):275–278. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
