Back to Journals » OncoTargets and Therapy » Volume 11
Knockdown of long noncoding RNA 00152 (LINC00152) inhibits human retinoblastoma progression
Authors Li S , Wen D , Che S, Cui Z, Sun Y, Ren H, Hao J
Received 20 December 2017
Accepted for publication 12 March 2018
Published 6 June 2018 Volume 2018:11 Pages 3215—3223
DOI https://doi.org/10.2147/OTT.S160428
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Ingrid Espinoza
This paper has been retracted.
Songhe Li,1 Dacheng Wen,2 Songtian Che,3 Zhihua Cui,1 Yabin Sun,1 Hua Ren,1 Jilong Hao1
1Department of Ophthalmology, The First Hospital of Jilin University, Changchun, People’s Republic of China; 2Department of Gastrointestinal Nutrition and Hernia Surgery, The Second Hospital of Jilin University, Changchun, People’s Republic of China; 3Department of Ophthalmology, The Second Hospital of Jilin University, Changchun, People’s Republic of China
Background: A growing body of evidence supports the involvement of long noncoding RNA 00152 (LINC00152) in the progression and metastasis of multiple cancers. However, the exact roles of LINC00152 in the progression of human retinoblastoma (RB) remain unknown. We explored the expression and biological function of human RB.
Materials and methods: The expression level of LINC00152 in RB tissues and cells was analyzed using quantitative real-time PCR. The function of LINC00152 was determined using a series of in vitro assays. In vivo, a nude mouse model was established to analyze the function of LINC00152. Gene and protein expressions were detected using quantitative real-time PCR and Western blot assays, respectively.
Results: The expression of LINC00152 mRNA was upregulated in RB tissues and cell lines. Knockdown of LINC00152 significantly inhibited cell proliferation, colony formation, migration, and invasion and promoted cell apoptosis and caspase-3 and caspase-8 activities in vitro, as well as suppressing tumorigenesis in vivo. We identified several genes related to proliferation, apoptosis, and invasion including Ki-67, Bcl-2, and MMP-9 that were transcriptionally inactivated by LINC00152.
Conclusion: Taken together, these data implicate LINC00152 as a therapeutic target in RB.
Keywords: retinoblastoma, LINC00152, proliferation, invasion
Introduction
Retinoblastoma (RB) is the most common primary intraocular malignant tumor in infants and children.1 Patients with RB suffer multiple life-threatening risks that include RB cell invasion, malignant transformation of intracranial neuroblastoma, and heterochronogenous tumor.2 Despite substantial improvements in therapeutic techniques, the survival rate is still poor for RB patients, mainly because of its limited sensitivity to chemotherapy and radiotherapy.3 Although a number of studies have addressed genome alterations and splicing defects in RB, the details of pathogenesis in RB remain largely unclear. Understanding the molecular events involved in the initiation and progression of RB and developing molecular targeted therapeutic strategies are urgently required to treat this disease.
Long noncoding RNAs (lncRNAs) are a class of noncoding RNA transcripts that exceed 200 nucleotides in length. Most lncRNAs lack protein-coding capability.4 lncRNAs are reportedly involved in a variety of biological processes such as cell apoptosis, proliferation, metastasis, chemotherapy drug resistance, and differentiation.5,6 Accumulating evidence indicates that the dysregulation of lncRNAs plays a crucial role in cancer initiation and progression, including in RB,7–9 suggesting that lncRNAs might be useful as a diagnostic marker and a therapeutic agent for RB.
Long intergenic noncoding RNA (lincRNA) 152 (LINC00152) is an 828 base pair lncRNA located on chromosome 2p11.2. It is highly conserved among mammals.10 LINC00152 is reportedly upregulated and is closely associated with the progression and prognosis of multiple cancers.11–19 However, the role and underlying molecular mechanism of LINC00152 in RB remain unclear. In this study, we measured the expression of LINC00152 in RB tissues and cell lines, the effect of LINC00152 on cell proliferation, colony formation, migration, and invasion in RB, and examined the mechanisms of action underlying the effects of LINC00152 in RB.
Materials and methods
Human tissue samples and cell lines
A total of 24 human RB specimens were obtained from patients (mean age 5.5 years) with RB at the Department of Ophthalmology, The First Hospital of Jilin University (Changchun, People’s Republic of China). Retina tissues from the ruptured globes of 6 patients (mean age 8.3 years) were used as the control. All tissue samples were stored in liquid nitrogen immediately after resection and were maintained there until use. All patients were diagnosed based on a postoperative pathological analysis by three pathologists. The study protocol was approved by the Ethics Committee of The First Hospital of Jilin University (Changchun, People’s Republic of China), and was verified to be in accordance with the guidelines of the Helsinki Declaration. Written informed consent was obtained from all participants for the use of their tissues for this research.
The human retinal pigmented epithelial cell line ARPE-19 and 2 RB cell lines (Y79 and SO-RB50) were obtained from the Institute of Biochemistry and Cell Biology of the Chinese Academy of Sciences (Shanghai, People’s Republic of China). ARPE-19 cells were cultured in Dulbecco’s Modified Eagle’s Medium/F12 Medium (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco, Grand Island, NY, USA). Y79 and SO-RB50 cells were maintained in Roswell Park Memorial Institute-1640 medium (Gibco) supplemented with 10% FBS. All cells were cultured in a humidified atmosphere of 95% air and 5% CO2 at 37°C.
Cell transfection and generation of stably transfected cells
A positive short hairpin RNA (shRNA) was designed to target different regions of the LINC00152 gene. Corresponding scramble negative control (NC) shRNA was designed and synthesized by Ribobio (Guangzhou, People’s Republic of China). The shRNAs were inserted into the pGenesil-1 vector (Ribobio) and were used to transform competent Escherichia coli DH5α (Tiangen, Beijing, People’s Republic of China). The recombinant plasmids were determined by enzyme identification and sequencing by Sangon Biotech Co. (Shanghai, People’s Republic of China). The plasmids were designated sh-LINC00152 and sh-NC. The constructed shRNAs plasmids were transfected into Y79 cells in 6-well plates using Lipofectamine™ 2000 (Invitrogen) according to the manufacturer’s protocol. Forty-eight hours after transfection, complete medium containing 800 μg/mL Geneticin (G418; Invitrogen) was added and the cells were cultured for 2 weeks to allow the growth of positive colonies. Stable cell lines were established by expanded culture and were designated Y79/sh-LINC00152 and Y79/sh-NC.
RNA extraction and quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNA was extracted from tissues or cultured cells using TRIzol Reagent (Invitrogen) according to the manufacturer’s protocol. The extracted RNA was reverse-transcribed into complementary DNA (cDNA) using the Prime-Script RT reagent Kit (Takara, Dalian, People’s Republic of China). qRT-PCR was performed using SYBR® Premix Ex Taq™ II (Takara) using an Applied Biosystems 7500HT system (Applied Biosystems, Foster City, CA, USA). β-actin was used as endogenous control for the quantification of LINC00152. The relative expression levels of LINC00152 were calculated using the 2−ΔΔCt method. Primer sequences were: β-actin, 5′-AGCGAGCATCCCCCAAAGTT-3′ (forward) and 5′-GGGCACGAAGGCTCATCATT-3 (reverse); and LINC00152, 5′-TGAGAATGAAGGCTGAGGTGT-3′ (forward) and 5′-GCAGCGACCATCCAGTCATT-3′ (reverse).
Cell proliferation and colony formation assay
Transfected cells were seeded into 96-well plates at a density of 5×103 cells per well and cultured for 24–96 h. Cell proliferation was determined at 24, 48, 72, and 96 h using a CCK-8 assay (Dojindo Laboratories, Rockville, MD, USA) following the manufacturer’s instructions. The absorbance value of each sample was recorded at 450 nm using a Benchmark Plus™ microplate spectrometer (Bio-Rad, Hercules, CA, USA).
For the colony formation assay, the 6-well plate was pretreated with 1,000 μL of 0.1 mg/mL Poly-L-Lysine hydrobromide (Sigma-Aldrich, St Louis, MO, USA) according to manufacturer’s instructions. Then Y79 cells stably expressing sh-LINC00152 (Y79/sh-LINC00152) or sh-NC (Y79/sh-NC) were seeded into 6-well plates at a density of 500 cells per well and cultured for 10 days. The culture medium was replaced with fresh medium every 24 h. After incubation for 10 days, the cells were fixed with methanol and Giemsa stained. The fixed colonies were photographed and counted.
Apoptosis assay
Cell apoptosis was determined using Annexin V-APC/7-AAD (BD Biosciences, Santa Clara, CA, USA) following the instructions of the manufacturer. Cell samples were analyzed using a FACScan (Becton-Dickinson) and apoptotic fractions were recorded.
Caspase-3 and -8 activities were determined by an enzyme-linked immunosorbent assay (ELISA). Briefly, cells were collected by centrifugation at 3,000 ×g for 10 min at 4°C at 48 h posttransfection. Caspase-3 and -8 activities in the supernatant were determined using the caspase-3 and -8 ELISA kit (Thermo Fisher Scientific, Waltham, MA, USA), respectively, following per manufacturer’s protocol.
Cell migration and invasion assays
To explore the effect of LINC00152 on migration in Y79 cells, we performed a wound-healing assay in 24-well dishes. The 24-well plate was pretreated with 500 μL of 0.1 mg/mL Poly-L-Lysine hydrobromide (Sigma-Aldrich) according to manufacturer’s instructions. At 24 h post-transfection, an artificial homogeneous wound was created on the monolayer using a sterile pipette tip, followed by growth in serum-free medium for 24 h. The mobilized cells were observed under a light microscope (Olympus, Tokyo, Japan). The migration distance was measured using NIS-Element Basic Research v3.2 software (Nikon, Tokyo, Japan).
The cell matrigel invasion assays were performed using 24-well chemotaxis matrigel chambers (BD BIOCOAT Matrigel chambers, BD Biosciences, 8 μm pore size). The 24-well plate was pretreated with 500 μL of 0.1 mg/mL Poly-L-Lysine hydrobromide. 1×105 transfected cells were resuspended in 200 μL serum-free medium and seeded into the upper chambers of a Transwell apparatus with Matrigel™ Basement Membrane Matrix (BD Biosciences). Medium (600 μL) containing 10% FBS was added to the lower chambers of each Transwell apparatus to stimulate cell invasion. After incubation for 24 h, cells in the upper chambers were removed, while cells that migrated to the lower surface were fixed with 70% ethanol and stained with 0.1% crystal violet. Stained cells were observed in 5 randomly selected fields using a light microscope (200× magnification, Olympus).
Western blotting
The total cell lysates were collected with RIPA buffer (Beyotime, Jiangsu, People’s Republic of China) and quantified using the Bradford method. Proteins were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and subsequently transferred to polyvinylidene difluoride membranes (Merck Millipore, Darmstadt, Germany). The membranes were blocked with 5% milk in Tris-buffered saline containing Tween 20 and probed with the following primary antibodies: anti-Ki-67, anti-Bcl-2, anti-matrix metalloproteinase-2 (MMP-2), and anti-β-actin (all from Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA). After incubation with corresponding horseradish peroxidase-conjugated secondary antibodies, protein bands were visualized using an enhanced chemiluminescence system (Thermo Fisher Scientific). The relative protein intensities were quantified using Gel-pro Analyzer® software (Media Cybernetics, Rockville, MD, USA).
Xenograft tumor model
BALB/c-nu mice (5–6 weeks of age, 20–25 g) were obtained from the Experimental Animal Center of Jilin University. All animals were maintained and used in accordance with the guidelines of the Institutional Animal Care and Use Committee of the Jilin University (Changchun, People’s Republic of China). All experimental protocols were approved by the Institutional Animal Care and Use Committee of the Jilin University.
Approximately 3×106 Y79/sh-LINC0015 or Y79/sh-NC cells in 200 μL of serum-free medium were injected directly into the left dorsal flank of a mouse. Tumor volume was measured with calipers every 5 days from the first injection until sacrifice, and the tumor volumes were calculated as 1/2 (length × width2). Mice were euthanized and the wet weight of each tumor was examined 30 days after the injections. Tumor tissues were stored at −80°C until use.
Statistical analyses
SPSS statistical software for Windows Version 19 (IBM Corporation, Armonk, NY, USA) was used for statistical analysis. All quantitative data are presented as the mean ± standard deviation from at least three independent replicates. Statistical significance was tested using the Student’s t-test or one-way analysis of variance. Differences were considered to be statistically significant at P<0.05.
Results
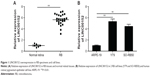
LINC00152 is overexpressed in RB specimens and cell lines
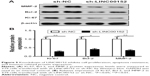
To investigate the potential role of LINC00152 in RB, we first analyzed LINC00152 mRNA expression in RB tissues. LINC00152 expression was significantly upregulated in RB tissues compared with retina tissues (Figure 1A). Consistent with this result, LINC00152 expression was aberrantly upregulated in 2 RB cell lines, as compared to the ARPE-19 human retinal pigmented epithelial cell line (Figure 1B). These results implied that LINC00152 overexpression may be influential in the progression of RB.
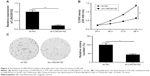
Knockdown of LINC00152 inhibits RB cell proliferation and colony formation
To investigate the biological role of LINC00152 in RB, LINC00152 expression was knocked down in Y79 cells by transfection with sh-LINC00152. qRT-PCR demonstrated that the LINC00152 expression level was significantly decreased in Y79 cells transfected with sh-LINC00152 compared to cells transfected with sh-NC (Figure 2A). The CCK8 assay showed that knockdown of LINC00152 expression in Y79 cells significantly inhibited cell proliferation compared with control cells (Figure 2B). Moreover, the colony formation ability of Y79 cells was drastically suppressed after LINC00152 was silenced compared with controls cells (Figure 2C). These data indicate that the knockdown of endogenous LINC00152 can suppress cell proliferation in vitro.
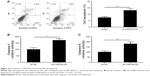
Knockdown of LINC00152 promotes RB cell apoptosis
To investigate the effect of LINC00152 on cell apoptosis, flow cytometry was performed. The downregulation of LINC00152 expression in Y79 cells significantly promoted cell apoptosis compared with control cells (Figure 3A). Moreover, the caspase-3 and -8 activities of Y79 cells were drastically increased after the knockdown of LINC00152 compared with control cells (Figure 3B and C). These results implied that the knockdown of endogenous LINC00152 promotes cell apoptosis in vitro.
Knockdown of LINC00152 inhibits cell migration and invasion in RB cells
Next, the roles of LINC00152 on RB cell migration and invasion were investigated by the wound-healing and Transwell invasion assays, respectively. The downregulation of LINC00152 significantly decreased migration and invasion of Y79 cells (Figure 4A and B). These results suggested that knockdown of endogenous LINC00152 suppresses the migration and invasion of RB cells.
Knockdown of LINC00152 inhibits cell proliferation, apoptosis, and invasion-related protein expression
To investigate the underlying mechanism of LINC00152 on proliferation, apoptosis, migration, and invasion, the expressions of the proteins related to proliferation (Ki-67), apoptosis resistance (Bcl-2), and invasion (MMP-2) were assessed in RB cells. The knockdown of endogenous LINC00152 significantly inhibited Ki-67, Bcl-2, and MMP-9 protein expression in Y79 cells (Figure 5). These data implied that knockdown of endogenous LINC00152 can inhibit the expression of proteins related to cell proliferation, apoptosis resistance, and cell invasion.
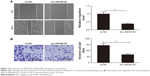
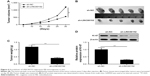
Knockdown of LINC00152 suppresses tumorigenesis of RB in vivo
To further test the effect of LINC00152 on tumorigenesis of RB in vivo, a xenograft mouse model was established by the subcutaneous injection of Y79/sh-LINC00152 or Y79/sh-NC, and tumor growth was measured. Growth curves revealed that knockdown of LINC00152 significantly reduced tumor growth compared with the Y79/sh-NC group (Figure 6A). At day 30 postinjection, the mice were killed and the tumor tissues were stripped and weighed. The Y79/sh-LINC00152 group displayed smaller tumor size (Figure 6B) and weight (Figure 6C) than the size and weight of tumors from mice in the Y79/sh-LINC00152 group. In addition, Western blot analysis demonstrated that Ki-67 expression was decreased in the tumor tissues of Y79/sh-Linc00152 compared to Y79/sh-NC group (Figure 6D). These results implied that knockdown of endogenous LINC00152 suppresses tumor growth of RB in vivo.
Discussion
Many lncRNAs have been reported to be closely related to cell proliferation, apoptosis, invasion, and metastasis in various types of tumors, including RB.7–9,20 H19 inhibits RB cell proliferation, induces RB cell cycle arrest, and induces apoptosis by competitively binding and counteracting the roles of the miR-17-92 cluster on the p21 and STAT3 signaling pathways.21 MALAT1 promotes retinoblastoma cell autophagy through miR-124-mediated STX17 regulation.22 LncRNA CCAT1 promotes proliferation, migration, and invasion, and decreased cell apoptosis of RB cells through the negative modulation of miR-218-5p.23 MEG3 suppresses proliferation, promotes apoptosis, and influences the activity of the Wnt/β-catenin pathway in RB cell lines.24 In the present study, we first evaluated lncRNA LINC00152 in RB tissues and cell lines. LINC00152 expression was significantly upregulated in RB tissues and cell lines. LINC00152 knockdown significantly suppressed cell proliferation, invasion, and migration, and promoted cell apoptosis and the activities of caspase-3 and caspase-8. In vivo, the knockdown of LINC00152 inhibited tumor growth in a nude mouse model. These results collectively indicate that LINC00152 downregulation might inhibit the origin and development of RB.
LINC00152 reportedly serves as an oncogenic lncRNA in the malignancy phenotypes of various cancers. It has been identified as an indicator of poor prognoses and is a potential therapy target in gastric cancer,10 colorectal cancer,11 breast cancer,12 lung cancer,13 glioma,14 tongue squamous cell carcinoma,16 hepatocellular carcinoma,17 gallbladder cancer,18 and clear cell renal cell carcinoma.19 However, the roles of LINC00152 with RB remain unclear. In the present study, we systemically examined the expression and biological function of LINC00152 in RB. LINC00152 was upregulated in RB tissues and cell lines relative to normal retinal tissues and normal retinal cell line, respectively. LINC00152 knockdown in Y79 cells significantly inhibited RB cell proliferation, migration, and invasion, induced cell apoptosis in vitro, and impaired tumor growth in vivo. In addition, the knockdown of LINC00152 inhibited the protein expressions of Ki-67 (a proliferation marker),25 Bcl-2 (an antiapoptosis protein),26 and MMP-2 (an invasion-related protein)27,28 in RB cells. The results suggest that LINC00152 functions as an oncogene in RB progression.
There are 3 limitations in this study. The first concerns the small number of RB samples. Future studies will require additional samples to conclusively investigate the clinical significance of LINC00152. Second, the use of 2 or more cell lines is required to validate the role of LINC00152 in the development and progression of RB. Third, lncRNA exerts its regulatory function via the regulation of multiple miRNAs or target genes. Thus, further studies on the modulation of LINC00152 expression in RB and a more thorough understanding of the underlying molecular mechanisms are needed.
Conclusion
LINC00152 is upregulated in RB tissues and cell lines. Knockdown of LINC00152 inhibits retinoblastoma cell proliferation, migration, and invasion, and enhances apoptosis and the activities of caspase-3 and caspase-8 in vitro, and suppresses tumor growth in vivo. These data imply that knockdown of LINC00152 may be a potential therapeutic strategy for RB.
Disclosure
The authors report no conflicts of interest in this work.
References
Dimaras H, Kimani K, Dimba EA, et al. Retinoblastoma. Lancet. 2012;379(9824):1436–1446. | ||
Rootman DB, Gonzalez E, Mallipatna A, et al. Hand-held high-resolution spectral domain optical coherence tomography in retinoblastoma: clinical and morphologic considerations. Br J Ophthalmol. 2013;97(1):59–65. | ||
Kivela T. The epidemiological challenge of the most frequent eye cancer: retinoblastoma, an issue of birth and death. Br J Ophthalmol. 2009;93(9):1129–1131. | ||
Cygan KJ, Soemedi R, Rhine CL, et al. Defective splicing of the RB1 transcript is the dominant cause of retinoblastomas. Hum Genet. 2017;136:1303–1312. | ||
Khalil AM, Guttman M, Huarte M, et al. Many human large intergenic noncoding RNAs associate with chromatin-modifying complexes and affect gene expression. Proc Natl Acad Sci U S A. 2009;106(28):11667–11672. | ||
Fan Q, Yang L, Zhang X, et al. The emerging role of exosome-derived non-coding RNAs in cancer biology. Cancer Lett. 2018;414:107–115. | ||
Sun W, Yang Y, Xu C, Guo J. Regulatory mechanisms of long noncoding RNAs on gene expression in cancers. Cancer Genet. 2017;216–217:105–110. | ||
Shang W, Yang Y, Zhang J, Wu Q. Long noncoding RNA BDNF-AS is a potential biomarker and regulates cancer development in human retinoblastoma. Biochem Biophys Res Commun. 2018;497(4):1142–1148. | ||
Su S, Gao J, Wang T, Wang J, Li H, Wang Z. Long non-coding RNA BANCR regulates growth and metastasis and is associated with poor prognosis in retinoblastoma. Tumour Biol. 2015;36(9):7205–7211. | ||
Dong C, Liu S, Lv Y, et al. Long non-coding RNA HOTAIR regulates proliferation and invasion via activating Notch signalling pathway in retinoblastoma. J Biosci. 2016;41(4):677–687. | ||
Zhao J, Liu Y, Zhang W, et al. Long non-coding RNA Linc00152 is involved in cell cycle arrest, apoptosis, epithelial to mesenchymal transition, cell migration and invasion in gastric cancer. Cell Cycle. 2015;14(19):3112–3123. | ||
Bian Z, Zhang J, Li M, et al. Long non-coding RNA LINC00152 promotes cell proliferation, metastasis, and confers 5-FU resistance in colorectal cancer by inhibiting miR-139-5p. Oncogenesis. 2017;6(11):395. | ||
Wu J, Shuang Z, Zhao J, et al. Linc00152 promotes tumorigenesis by regulating DNMTs in triple-negative breast cancer. Biomed Pharmacother. 2018;97:1275–1281. | ||
Zhang PP, Wang YQ, Weng WW, et al. Linc00152 promotes cancer cell proliferation and invasion and predicts poor prognosis in lung adenocarcinoma. J Cancer. 2017;8(11):2042–2050. | ||
Yu M, Xue Y, Zheng J, et al. Linc00152 promotes malignant progression of glioma stem cells by regulating miR-103a-3p/FEZF1/CDC25A pathway. Mol Cancer. 2017;16(1):110. | ||
Yu Y, Yang J, Li Q, Xu B, Lian Y, Miao L. LINC00152: a pivotal oncogenic long non-coding RNA in human cancers. Cell Prolif. 2017;50(4):e12349. | ||
Yu J, Liu Y, Guo C, et al. Upregulated long non-coding RNA LINC00152 expression is associated with progression and poor prognosis of tongue squamous cell carcinoma. J Cancer. 2017;8(4):523–530. | ||
Deng X, Zhao XF, Liang XQ, Chen R, Pan YF, Liang J. Linc00152 promotes cancer progression in hepatitis B virus-associated hepatocellular carcinoma. Biomed Pharmacother. 2017;90:100–108. | ||
Cai Q, Wang Z, Wang S, et al. Long non-coding RNA LINC00152 promotes gallbladder cancer metastasis and epithelial-mesenchymal transition by regulating HIF-1α via miR-138. Open Biol. 2017;7(1):pii: 160247. | ||
Wu Y, Tan C, Weng WW, et al. Long non-coding RNA Linc00152 is a positive prognostic factor for and demonstrates malignant biological behavior in clear cell renal cell carcinoma. Am J Cancer Res. 2016;6(2):285–299. | ||
Zou Y, Li J, Chen Y, et al. BANCR: a novel oncogenic long non-coding RNA in human cancers. Oncotarget. 2017;8(55):94997–95004. | ||
Zhang A, Shang W, Nie Q, Li T, Li S. Long non-coding RNA H19 suppresses retinoblastoma progression via counteracting miR-17-92 cluster. J Cell Biochem. 2017;119(4):3497–3509. | ||
Huang J, Yang Y, Fang F, Liu K. MALAT1 modulates the autophagy of retinoblastoma cell through miR-124-mediated stx17 regulation. J Cell Biochem. 2018;119(5):3853–3863. | ||
Zhang H, Zhong J, Bian Z, Fang X, Peng Y, Hu Y. Long non-coding RNA CCAT1 promotes human retinoblastoma SO-RB50 and Y79 cells through negative regulation of miR-218-5p. Biomed Pharmacother. 2017;87:683–691. | ||
Gao Y, Lu X. Decreased expression of MEG3 contributes to retinoblastoma progression and affects retinoblastoma cell growth by regulating the activity of Wnt/β-catenin pathway. Tumour Biol. 2016;37(2):1461–1469. | ||
Kouzegaran S, Shahraki K, Makateb A, et al. Prognostic investigations of expression level of two genes FasL and Ki-67 as independent prognostic markers of human retinoblastoma. Oncol Res. 2017;25(4):471–478. | ||
Natalino RJ, Antoneli CB, Ribeiro KC, Campos AH, Soares FA. Immunohistochemistry of apoptosis-related proteins in retinoblastoma. Pathol Res Pract. 2016;212(12):1144–1150. | ||
Webb AH, Gao BT, Goldsmith ZK, et al. Inhibition of MMP-2 and MMP-9 decreases cellular migration, and angiogenesis in in vitro models of retinoblastoma. BMC Cancer. 2017;17(1):434. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.