Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
K-3-Rh Protects Against Cerebral Ischemia/Reperfusion Injury by Anti-Apoptotic Effect Through PI3K-Akt Signaling Pathway in Rat
Authors Sun J, Wang J, Hu L, Yan J
Received 6 October 2019
Accepted for publication 14 April 2020
Published 12 May 2020 Volume 2020:16 Pages 1217—1227
DOI https://doi.org/10.2147/NDT.S233622
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Yuping Ning
Juan Sun, Jian Wang, Luoman Hu, Jinfeng Yan
Rehabilitation Department, The Affiliated Hospital of Qingdao University, Qingdao City, Shandong Province 266000, People’s Republic of China
Correspondence: Juan Sun
Rehabilitation Department, The Affiliated Hospital of Qingdao University, No. 1677 Wutaishan Road, Qingdao City, Shandong Province 266000, People’s Republic of China
Email [email protected]
Background/Aims: Ischemic stroke is the main cause of nerve damage and brain dysfunction, accompanied by strong brain cell apoptosis. This study aimed to investigate the effect of kaempferol-3-O-rhamnoside (K-3-rh) on cerebral ischemia-reperfusion (I/R) injury.
Methods and Materials: A rat model of cerebral I/R injury was established. The effects of K-3-rh on cerebral infarction size, brain water content and neurological deficits in rats were evaluated. Apoptosis of ischemic brain cells after mouse I/R was observed by TUNEL staining and flow cytometry. Western blot and qRT-PCR were used to detect the effect of K-3-rh on the expression of apoptosis-related proteins.
Results: K-3-rh can improve the neurological deficit score, reduce the infarct volume and brain water content, and inhibit cell apoptosis. In addition, K-3-rh significantly downregulated the expression of Bax and p53 and upregulated the expression of Bcl-2, and the phosphorylation level of Akt. Blockade of PI3K activity by the PI3K inhibitor wortmannin not only reversed the effects of K-3-rh on infarct volume and brain water content but also reversed the expression level of p-Akt.
Conclusion: K-3-rh had obvious neuroprotective effects on brain I/R injury and neuronal apoptosis, and its mechanism may be related to activation of PI3K/Akt signaling pathway.
Keywords: kaempferol-3-O-rhamnoside, PI3K; Akt, apoptosis, cerebral ischemia/reperfusion injury
Introduction
Ischemic cerebrovascular disease is the most common disease in neurology and its prevalence, disability, and mortality are very high.1 The main treatment is to dredge the blocked blood vessels and improve the blood supply of the blocked parts.2 However, when the blood supply to the ischemic site is improved.3 Cerebral ischemia/reperfusion (I/R) injury is a complex pathological and physiological process involving multiple mechanisms.4 The initiating factor of I/R is ischemia and hypoxia. Under reperfusion, brain cells undergo multiple processes, such as the increase of oxygen free radicals, calcium overload, inflammation, excitatory amino acid toxicity, apoptotic gene activation, and so on.5 The induction and regulation of apoptosis is complex, and this process involves a series of apoptosis-related factors and signal transduction pathways.6,7 Therefore, it is important to reveal the mechanisms of neuronal apoptosis and reduce the apoptosis of neurons after cerebral I/R.
Acute brain damage in the brain is caused by central necrosis, while secondary or late death is mainly caused by neuronal apoptosis in the ischemic penumbra.8,9 The progression of apoptosis involves the regulation, expression, and activation of a series of genes.10 Current understandings of the molecular mechanism of apoptosis in cerebral I/R injury are that after the ischemia and hypoxia of the cerebral nerve cells, the energy metabolism of the cells is impaired, and various apoptotic stimulator free radicals, nitric oxide, excitatory amino acid toxicity and calcium overload factors are produced.11,12 The PI3K/Akt signaling pathway plays important roles in promoting cell survival. This signal transduction pathway is involved in the regulation of cell growth, proliferation and differentiation.13 In cerebral ischemia, the PI3K/Akt signaling pathway is a critical signaling pathway that promotes the survival of ischemic nerve cells.14 Therefore, the search for effective PI3K/Akt activators may be a future research direction for the treatment of ischemic cerebrovascular disease.
Schima wallichii Korth is a traditional Chinese medicine with important application values.15 Studies have revealed different pharmacological effects of different chemical constituents of Schima wallichii Korth, which have different therapeutic or auxiliary therapeutic effects on different diseases.16 Novel chemical composition and new pharmacological effects of Schima wallichii Korth are constantly being discovered.17 Studies have found that the leaves of Schima wallichii Korth contain active ingredients such as flavonoids, proteins, polysaccharides, lignans and volatile oils.16,18 Schima wallichii Korth leaves can play a better role in the treatment of tumors, liver diseases, cerebrovascular diseases, etc.19 Kaempferol-3-O-rhamnoside (K-3-rh) is an active ingredient extracted from Schima wallichii Korth leaves.20 It has many biological functions such as antihypertensive, anti-fatigue and anti-tumor effects.21 In recent years, its regulation of antagonizing excitatory neurotransmitters, anti-oxidative damage, and secretion of neuroprotective factors during neurological damage has received increasing attention.22 The chemical structure of K-3-rh is shown in Figure 1A. However, there are no reports about its effects on cerebral I/R injury. This study was carried out using the animal model of cerebral I/R to investigate whether K-3-rh can inhibit apoptosis by activating the expression of PI3K/Akt signaling pathway, and the pharmacological mechanism of cerebrovascular diseases was explored. It would provide new research directions for finding new drugs for the treatment of cerebral I/R injury.
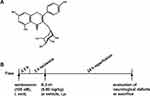 |
Figure 1 (A) Chemical structure of K-3-rh. (B) Experimental design. |
Methods and Materials
Experimental Animals
Healthy male Sprague-Dawley rats that were 7–8 weeks old and weighing 200 −220 g were provided by the Experimental Animal Center of the affiliated Hospital of Qingdao University. They had free access to drinking water at room temperature 20 −25 °C. All experiments were approved by the affiliated Hospital of Qingdao University Animal Care and Use Committee and conducted in accordance with the National Institutes of Health Laboratory Animal Care and Use Guidelines.
Focal Cerebral Ischemia/Reperfusion (I/R) Model
Transient focal ischemia was induced in rats by right middle cerebral artery (MCA) occlusion (MCAO). Briefly, the rats were anesthetized by inhalation of a 69%/30% (vol/vol) mixture of nitrous oxide/oxygen and 1% halothane using a face mask. A midline neck incision was made and the right common carotid artery was exposed. The right MCA was occluded by insertion of 4–0 surgical nylon thread with a silicone coating through the common carotid artery. Using this technique, the tip of the thread occludes the origin of the right MCA. Successful stroke was achieved in nearly all animals. During these procedures, body temperature of rats was monitored with a rectal probe and was maintained at 37 ± 0.3 °C using a heating pad. The surgical incision was then closed and the animals were allowed to recover at room temperature. After 90 min of transient MCA occlusion (tMCAO), the suture was removed to restore blood flow (reperfusion). Sham control animals were treated with cervical surgery but without insertion of the nylon thread. The animals were kept at ambient temperature until sampling, with free access to water and food.23
Groups and Drug Administration
Rats were randomly divided into sham-operated group, I/R group, kaempferol-3-O-rhamnoside group, wortmannin group, and K-3-rh + wortmannin group. In the sham group (n = 60), rats were treated with middle cerebral artery occlusion (MCAO), except for non-inserted CCA. In the I/R group (n = 6), reperfusion was performed at 2 h after cerebral ischemia for 24 h in rats. In the kaempferol-3-O-rhamnoside (K-3-rh) group (n = 6), 5–80 mg/kg K-3-rh (Sigma, purity ≥98%) was dissolved in 0.9% NaCl and injected slowly in the rat peritoneum at 2 h after cerebral ischemia. In the wortmannin group (n = 6), it was slowly injected intraperitoneally in rats for 24 h.24 In the K-3-rh (80 mg/kg) + wortmannin (100 nM) group (n = 6), the method was the same as K-3-rh and wortmannin group. The sham group and the I/R group received an equal 0.9% NaCl at the same time. Experimental design is shown in Figure 1B.
Assessment of Infarct Volume, Neurological Deficit, and Brain Water Content
Rats were cerebral ischemia-reperfusion injury for 24 h, decapitated and removed the olfactory bulb after being quickly frozen. The cerebellum and the lower brainstem were sliced and placed in TTC dye. The staining result in white was for infarction and red was for a normal brain. An image analysis system was used to detect the cerebral infarction volume to the total brain volume.
After 24 h of cerebral I/R in rats, neurological deficits were evaluated by an observer blinded to the treatment of animals (n = 6) after 24 h reperfusion according to the methods previously described: 0 point, rats behave normally; 1 point, rats cannot fully stretch their left front legs; 2 points, rats turn around into a circle; 3 points, rats fall down to the left side; 4 points, rats cannot move by themselves and lose their consciousness.25
After 24 h of cerebral I/R injury, the rats were decapitated. The olfactory bulb, the cerebellum and the lower brainstem were removed, and the wet mass of the brain was weighed. Dry mass was weighed after baking.
TUNEL Staining
Cells were stained with TUNEL assay kit (Qianchen, Shanghai, China). The percentage of TUNEL-positive cells was counted by a fluorescence microscopy (Shimei, Shanghai, China).
Apoptosis Was Detected by Flow Cytometry
Enzymatic-mechanic method was used to isolate cells.26 Briefly, the brains of anaesthetized rats were harvested and hippocampi were completely removed on ice quickly. The tissues were rinsed in PBS at 4°C to discharge blood. Then, each hippocampus was transferred into a 1.5 mL centrifuge tube filled with PBS at 4 °C. The tissues were minced with ophthalmic scissors until no large pieces were observed and then centrifuged to remove supernatants. Subsequently, 1 mL pre-warmed 0.25% Trypsin-EDTA was added into the tube to resuspend the tissue masses. After incubation at 37 °C for 15 min, the mixture was filtered through a cell strainer, and the filtrate was centrifuged at 300× g for 5 min to remove supernatants. The precipitations were gently washed with precooled PBS twice and resuspended in 1 mL PBS. The cell quantities of all samples were counted. The suspensions were labeled with Annexin V-FITC and PI according to the protocol and analyzed by FCM.
Real-Time Quantitative RT-PCR
Total RNAs in cells were extracted using TRIzol reagent (Invitrogen, Carlsbad, CA). The qRT-PCR reactions were performed using a ViiATM 7 real-time PCR system (Jinuo, Shanghai, China). GAPDH and U6 were used as internal references. The expression levels of P53 were detected using SYBR Premix Ex Taq II (Takara Biotechnology). The specific experimental methods of qRT-PCR were performed with reference to the literature.27 Primer sequences are listed in Table 1.
 |
Table 1 Sequences of Primers Used in RT-qPCR |
Western Blot Analysis
The right hippocampus and cortical tissues were quickly extracted on ice and weighed. The cortex was ground and then weighed to get about 80 mg homogenate. The brain tissue was weighed in a fine balance, and total proteins were extracted. The protein concentration was quantified by the BCA Protein Assay Kit and transferred to the PVDF membrane by electrophoresis. The membrane was incubated with rabbit anti-Bcl-2 (1:1000), Bax (1:1000), p-Akt (Ser 473) (1:500), Akt (1:500) and β-actin (1:1000, Santa Cruz Biotechnology) for overnight, followed by incubation with 1:5000 labeled anti-rabbit secondary antibody for 1 h. Western blot analysis was performed as described in the literature.28
Statistical Analysis
Data were analyzed by SPSS19.0 statistical software. The results were shown as mean ± standard deviation (mean ± SD). Multigroup comparisons were conducted based on one-way ANOVA. LSD test was used for subsequent analysis. P < 0.05 was considered as significant difference.
Results
K-3-Rh Decreased the Infarct Volumes, Behavioral Outcomes and Water Content
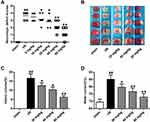
To assess the effect of K-3-rh on brain I/R injury in the I/R rat model, rats were treated with different doses of K-3-rh (5–80 mg/kg). As shown in Figure 2A, compared to the Sham group, the neurological deficit score in the I/R group was significantly increased (P < 0.05). Compared to the I/R group, the neurological deficit scores were significantly decreased in the K-3-rh group (P < 0.05, P < 0.01). The infarct size of the I/R group was significantly increased compared to that in the Sham group (P < 0.01), and the infarct size of the rats in the K-3-rh group was significantly increased compared to that in the I/R group (P < 0.05, P <0.01) (Figure 2B and C). As shown in Figure 2D, compared to the Sham group, the brain water content in the I/R group was significantly increased (P < 0.05). The brain water content in the K-3-rh group was significantly reduced compared to that in the I/R group (P < 0.05, P < 0.01). These findings demonstrated the effectiveness of K-3-rh on brain I/R injury.
K-3-Rh Attenuated I/R Injury Induced Apoptosis
As shown in Figure 3, the apoptotic cell rate of the sham group was less than 5%. Compared to the Sham group, the apoptosis rate of I/R group was as high as (41.05 ± 3.18)%, which was significantly increased compared to that in the sham group (P < 0.01). Compared to the I/R group, the proportion of apoptotic cells in the K-3-rh treatment group decreased to (32.33 ± 2.11)%, (27.45 ± 3.03)% and (17.71 ± 1.94)%, respectively, which was significantly lower compared to that in the I/R Group (P < 0.05, P < 0.01). These results indicated that K-3-rh exerted neuroprotective effects on I/R by inducing apoptosis.
 |
Figure 3 Effect of K-3-rh on apoptosis. VS, ##P < 0.01; VS the I/R group, *P < 0.05, **P < 0.01. |
In addition, as shown in Figure 4, almost no positive cells were expressed in the brain tissues of the Sham group. In contrast, a large number of TUNEL-positive cells were expressed in the brain I/R injury model group (P < 0.01). The number of TUNEL-positive cells in the ischemic penumbra of the K-3-rh group was significantly reduced compared to that in the I/R model group (P < 0.01). These results indicated that K-3-rh can effectively reduce I/R-induced neuronal death.
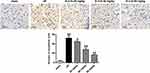 |
Figure 4 Effect of K-3-rh detected by TUNEL staining on neuronal apoptosis. Scale bar = 50 μm (×400 times). VS, ##P < 0.01; VS the I/R group, *P < 0.05, **P < 0.01. |
Effects of K-3-Rh Treatment on the Expression of Apoptosis-Related Proteins
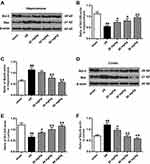
As shown in Figure 5A and B, compared to the sham group, expression levels of p53 in the hippocampus and cortex of the I/R group were significantly increased (P < 0.01). Compared to the I/R group, the expression levels of p53 in the hippocampus and cortex of the K-3-rh-treated group were significantly reduced (P < 0.05, P < 0.01). As shown in Figures 5C–F and 6, compared to the sham group, the expression of Bcl-2 in hippocampus and cortex of I/R group was significantly down-regulated, and the expression of Bax was significantly up-regulated (P < 0.01) at both mRNA and protein levels. Compared to the I/R group, the expression of Bcl-2 in hippocampus and cortex of K-3-rh treatment group was significantly up-regulated, and the expression of Bax was significantly down-regulated at both mRNA and protein levels (P < 0.05, P < 0.01).
K-3-Rh Enhanced the Phosphorylation of AKt After Cerebral I/R Injury
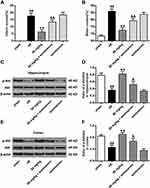
As shown in Figure 7A and B, compared to the I/R group, the cerebral infarct size and brain water content of the K-3-rh group were significantly reduced. Compared to the K-3-rh group, the cerebral infarct size and brain water content of the rats in the K-3-rh + Wortmannin group were significantly raised (P < 0.05, P < 0.01). As shown in Figure 7C–F, the expression of p-Akt was significantly increased in the K-3-rh group compared to that in the I/R group. Compared to the K-3-rh group, the expression of p-Akt was significantly inhibited in the K-3-rh + Wortmannin group (P < 0.05, P < 0.01).
Discussion
Coronary heart disease is one of the major diseases that endanger human health, and acute myocardial infarction is a common type of coronary heart disease.29 With the popularization and implementation of emergency interventional therapy or emergency thrombolysis, patients with myocardial infarction are not only saved, but the prognosis was also improved. However, I/R injury has always been a difficult problem in the treatment of acute myocardial infarction reperfusion. It often causes myocardial cell apoptosis, left ventricular remodeling, arrhythmia and heart failure. Studies have found that myocardial cells have both necrotic and apoptotic forms during I/R in acute myocardial infarction, and apoptosis can be caused by acute myocardial infarction.30,31
With the induction of external death signals, nucleated cells initiate their own internal gene expression and regulation through signal transduction pathways, leading to programmed process of cell death, which has a great effect on normal embryos and the maintenance of the stability of mature tissues and organs.32 Apoptosis can occur under many pathological conditions. Studies have shown that apoptosis is the main form of myocardial I/R injury and is one of the main factors affecting the effect of myocardial infarction reperfusion.33
Oxidative stress plays important roles in the pathogenesis of ischemia-reperfusion injury in the brain.34 Studies have shown that the leaves of Schima wallichii Korth contain the same active ingredients as in fruits.16 In addition, pharmacological experiments have also shown that the extract of the leaves of Schima wallichii Korth has multiple functions such as liver protection, antibacterial and anti-oxidation. K-3-rh is an activity extracted from the leaves of Schima wallichii Korth.16 In addition, studies have found that K-3-rh can effectively reduce dimethyl butyric acid-induced oxidative stress, restore antioxidant enzyme activity, and reduce lipid peroxidation.35 Nevertheless, the molecular mechanism of K-3-rh through ischemia-reperfusion injury has not been fully elucidated. In this study, a rat model of cerebral I/R was established to confirm that K-3-rh can alleviate cerebral ischemic neurological damage, cerebral infarction volume, and reduce brain water content. Flow cytometry, TUNEL staining and protein immunoassay showed that K-3-rh was able to reduce the expression levels of Bax in hippocampus and cortex and increase the expression levels of Bcl-2. These results indicated that K-3-rh treatment can inhibit I/R-induced apoptosis and neurons.
Upon cerebral ischemia, activated NF-κB up-regulates many proapoptotic target genes leading to neuronal apoptosis, of which p53 is one of the target genes for NF-κB-mediated neuronal apoptosis.36 P53 is an important gene regulating cell proliferation, which is involved in cell growth and plays an important role in apoptosis.37 In cerebral ischemia, p53 mediates neuronal apoptosis by transcription-dependent and transcription-independent mechanisms, aggravating ischemic brain damage.1 The results in this study showed that the expression of p53 in the brain of mice was significantly up-regulated after cerebral ischemia/reperfusion, and K-3-rh significantly inhibited the expression of p53. Therefore, we hypothesized that K-3-rh may have a neuroprotective effect on I/R by inhibiting the expression of p53 and apoptosis-related factors after cerebral ischemic injury.
The PI3K/Akt signaling pathway can promote cell survival.38 It is a signal transduction pathway involved in cell growth, proliferation, and differentiation regulation.39 At cerebral ischemia, the PI3K/Akt signaling pathway can promote the survival of ischemic nerve cells.40 Short-term ischemic preconditioning can alleviate ischemic brain damage and up-regulate the expression of p-Akt in brain tissues.41 Intraventricular injection of PI3K inhibitor wortmannin can restrain the expression of p-Akt and the neuroprotective effect of ischemic preconditioning, indicating that the PI3K/Akt signaling pathway takes part in the regulation of neuroprotective effects of ischemic preconditioning on cerebral ischemia.42 Our study found that the expression of p-Akt was up-regulated in the K-3-rh group. Compared with K-3-rh group, K-3-rh + Wortmannin significantly inhibited the expression of p-Akt and reversed the effects of K-3-rh on neurological deficit scores and infarct size. Therefore, it inhibited the occurrence of neuronal apoptosis after cerebral ischemia and exerted a neuroprotective effect on I/R rats. It was observed that K-3-rh attenuated nerve damage in rat cerebral ischemia by PI3K/Akt signaling pathway. In future studies, we would try to assess the effects of K-3-rh on cell apoptosis in in vitro studies (OGD/OGD/R model, etc.) and evaluate the effect of K-3-rh on blood–brain barrier systematically in further studies to confirm and expand our findings.
Conclusion
K-3-rh attenuated cerebral I/R injury by activating PI3K/Akt signaling pathway to inhibit apoptosis. And PI3K/Akt signaling pathway may be a new target to treat ischemic cerebrovascular disease in the future.
Abbreviations
K-3-rh, kaempferol-3-O-rhamnoside; I/R, ischemia/reperfusion.
Data Sharing Statement
The analyzed data sets generated during the study are available from the corresponding author on reasonable request.
Ethics Approval
All experiments were approved by the affiliated Hospital of Qingdao University Animal Care and Use Committee and conducted in accordance with the National Institutes of Health Laboratory Animal Care and Use Guidelines.
Consent for Publication
All authors have read and approved the final manuscript.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bondonno CP, Blekkenhorst LC, Prince RL, et al. Association of vegetable nitrate intake with carotid atherosclerosis and ischemic cerebrovascular disease in older women. Stroke. 2017;48(7):1724–1729. doi:10.1161/STROKEAHA.117.016844
2. Li X, Zhang Z-W, Wang Z, Li J-Q, Chen G. The role of argon in stroke. Med Gas Res. 2018;8(2):64–66. doi:10.4103/2045-9912.235129
3. Wang Q, Xin-Rui LI, Yang Z, Chang-Yin LI, Fang YI, Yang WJAAS. Mesenteric marginal vessel arch attenuates intestinal ischemia-reperfusion injury by compensating blood-supply. Acta Anat Sin. 2015;46(3):394–399.
4. Miao C, Dai LH, Fei A, Pan SM, Wang HR. Isoquercetin activates the ERK1/2-Nrf2 pathway and protects against cerebral ischemia-reperfusion injury in vivo and in vitro. Exp Ther Med. 2017;13(4):1353. doi:10.3892/etm.2017.4093
5. Chen X, Zhang X, Xue L, et al. Treatment with enriched environment reduces neuronal apoptosis in the periinfarct cortex after cerebral ischemia/reperfusion injury. Cell Physiol Biochem. 2017;41(4):1445.
6. Xiao Y, Ding L, YHJAR G. Progress of acupuncture and moxibustion research on the signal transduction pathways involved in cell apoptosis in myocardial ischemia reperfusion injury. Zhen ci yan jiu. 2017;42(5):463.
7. Garutti I, Gonzalez-Moraga F, Sanchez-Pedrosa G, et al. The effect of anesthetic preconditioning with sevoflurane on intracellular signal-transduction pathways and apoptosis, in a lung autotransplant experimental model. Rev Bras Anestesiol. 2019;69(1):48–57.
8. Fujikawa DGJC, Journal SB. The role of excitotoxic programmed necrosis in acute brain injury. Comput Struct Biotechnol J. 2015;13(C):212–221. doi:10.1016/j.csbj.2015.03.004
9. Kerr N, de Rivero Vaccari JP, Abbassi S, et al. Traumatic brain injury-induced acute lung injury: evidence for activation and inhibition of a neural-respiratory-inflammasome axis. J Neurotrauma. 2018:
10. Freire R, D’Adda DFF, Wu L, et al. Cleavage of the bloom’s syndrome gene product during apoptosis by caspase-3 results in an impaired interaction with topoisomerase IIIalpha. Nucleic Acids Res. 2017;29(15):3172.
11. Guang-qiang Y-J, Xiao-qing B-Q. Inhibition of cerebral ischemia/reperfusion injury-induced apoptosis: nicotiflorin and JAK2/STAT3 pathway. Research CJNR. 2017;12(1):96–102.
12. Ravindran S, Hussain SJ, Boovarahan SR, Kurian GAJC-BI. Sodium thiosulfate post-conditioning protects rat hearts against ischemia reperfusion injury via reduction of apoptosis and oxidative stress. Chem Biol Interact. 2017;274:24–34.
13. Jafari M, Ghadami E, Dadkhah T. PI3k/AKT signaling pathway: erythropoiesis and beyond. J Cell Physiol. 2019;234:3.
14. Wu J, Liu H, Chu T, Jiang P, Li ST. Neuregulin-1β attenuates sepsis-induced diaphragm atrophy by activating the PI3K/Akt signaling pathway. J Muscle Res Cell Motil. 2019;1–9.
15. Lalhminghlui K, Jagetia GC. Evaluation of the free-radical scavenging and antioxidant activities of Chilauni, Schima wallichiiKorthin vitro. Future Sci OA. 2018;4(2):FSO272.
16. Barliana MI, Suradji EW, Abdulah R, et al. Antiplasmodial properties of kaempferol-3-O-rhamnoside isolated from the leaves of Schima wallichii against chloroquine-resistant Plasmodium falciparum. Biomed Rep. 2014;2(4):579–583.
17. Thapa LB, Kaewchumnong K, Sinkkonen A, Sridith KJWR. Plant invasiveness and target plant density: high densities of native Schima wallichii seedlings reduce negative effects of invasive Ageratina adenophora. Weed Res. 2017;57(2):72–80.
18. Diantini A, Subarnas A, Lestari K, et al. Kaempferol-3-O-rhamnoside isolated from the leaves of Schima wallichii Korth. inhibits MCF-7 breast cancer cell proliferation through activation of the caspase cascade pathway. Oncol Lett. 2012;3(5):1069–1072.
19. Mishra VK, Passari AK, Singh BP. In Vitro Antimycotic and Biosynthetic Potential of Fungal Endophytes Associated with Schima Wallichii. In Kumar P, Gupta V, Tiwari A, Kamle M, editors. Current Trends in Plant Disease Diagnostics and Management Practices. Springer, Cham, 2016;367–381.
20. Irungu BN, Adipo N, Orwa JA, et al. Antiplasmodial and cytotoxic activities of the constituents of Turraea robusta and Turraea nilotica. J Ethnopharmacol. 2015;174(Complete):419–425.
21. Kocacaliskan U, Yilmaz I, Taslidere A, Yalcin FN, Aka C, Sekeroglu N. Cuscuta arvensisBeyr “Dodder”: inVivoHepatoprotective effects against acetaminophen-induced hepatotoxicity in rats. J Med Food. 2018;21(6):625–631.
22. Chung MJ, Pandey RP, Choi JW, Sohng JK, Choi DJ, Park YIJII. Inhibitory effects of kaempferol-3-O-rhamnoside on ovalbumin-induced lung inflammation in a mouse model of allergic asthma. Int Immunopharmacol. 2015;25(2):302–310.
23. Zhang X, Deguchi K, Yamashita T, et al. Temporal and spatial differences of multiple protein expression in the ischemic penumbra after transient MCAO in rats. Brain Res. 2010;1343(2):143–152. doi:10.1016/j.brainres.2010.04.027
24. Chen L, Xiang Y, Kong L, et al. Hydroxysafflor yellow A protects against cerebral ischemia–reperfusion injury by anti-apoptotic effect through PI3K/Akt/GSK3β pathway in rat. Neurochem Res. 2013;38(11):2268–2275. doi:10.1007/s11064-013-1135-8
25. Sporns PB, Hälker L, Heindel W, et al. Impact of anamnestic information and neurological deficits on the detection rate of secondary headaches. Radiologe. 2018;58:1–5.
26. Walker TL, Kempermann G. One mouse, two cultures: isolation and culture of adult neural stem cells from the two neurogenic zones of individual mice. J Vis Exp. 2014;84:e51225.
27. Causin RL, Souza KBD, Leal LF, Evangelista AF, Matsushita GM. PO-393 Identification of housekeeping genes to quantitative real-time RT-PCR analysis by miRNA expression using liquid-based cervical cytology samples.
28. Swets M, Wouters A, Krijgsman D, et al. HLA-G protein expression in colorectal cancer evaluated by immunohistochemistry and Western blot analysis: its expression characteristics remain enigmatic. Clin Immunol. 2018;194:S152166161730774X.
29. Gao Y, Zhang F, Li C, et al. Optimal strategy of primary percutaneous coronary intervention for acute myocardial infarction due to unprotected left main coronary artery occlusion (OPTIMAL): study protocol for a randomised controlled trial. Trials. 2019;20(1):162.
30. Lekshmi A, Varadarajan SN, Lupitha SS, et al. A quantitative real-time approach for discriminating apoptosis and necrosis. Cell Death Dis. 2017;3:16101.
31. Qiu Y, Yu Q, Shi K, et al. Cell-penetrating peptides induce apoptosis and necrosis through specific mechanism and cause impairment of Na+–K+-ATPase and mitochondria. Amino acids. 2017;49(1):75–88.
32. Gu HZ, Lin RR, Wang HC, Zhu XJ, Yan H, Zheng FYJOL. Effect of Momordica charantia protein on proliferation, apoptosis and the AKT signal transduction pathway in the human endometrial carcinoma Ishikawa H cell line in vitro. Oncol Lett. 2017;13(5):3032–3038.
33. Ghaderi S, Alidadiani N, Dilaver N, et al. Role of glycogen synthase kinase following myocardial infarction and ischemia-reperfusion. Apoptosis. 2017;22(7):1–11.
34. Oktay V, Baydar O, Sinan ÜY, et al. The effect of oxidative stress related with ischemia-reperfusion damage on the pathogenesis of atrial fibrillation developing after coronary artery bypass graft surgery. Arch Turk Soc Cardiol. 2014;42(5):419–425. doi:10.5543/tkda.2014.84032
35. Chen H, Ouyang K, Jiang Y, et al. Constituent analysis of the ethanol extracts of Chimonanthus nitens Oliv. leaves and their inhibitory effect on α-glucosidase activity. Int J Biol Macromol. 2017;98:829–836. doi:10.1016/j.ijbiomac.2017.02.044
36. Xiang HC, Lin LX, Hu XF, et al. AMPK activation attenuates inflammatory pain through inhibiting NF-κB activation and IL-1β expression. J Neuroinflammation. 2019;16:1.
37. Wang W, Xiong Y, Ding X, et al. Cathepsin L activated by mutant p53 and Egr-1 promotes ionizing radiation-induced EMT in human NSCLC. J Exp Clin Cancer Res. 2019;38(1):61.
38. Fu X, Halim A, Tian B, Luo Q, Song G. MT1-MMP downregulation via the PI3K/Akt signaling pathway is required for the mechanical stretching-inhibited invasion of bone-marrow-derived mesenchymal stem cells. J Cell Physiol. 2019;234(8):14133–14144. doi:10.1002/jcp.28105
39. Kim SH, Yi SJ, Lee H, Kim JH, Oh M. β2-Adrenergic receptor (β2-AR) agonist formoterol suppresses differentiation of L6 myogenic cells by blocking PI3K–AKT pathway. Anim Cells Syst. 2019;23(1):18–25.
40. Jiao S, Zhu H, He P, Teng JJB. Betulinic acid protects against cerebral ischemia/reperfusion injury by activating the PI3K/Akt signaling pathway. Biomed Pharmacother. 2016;84:S0753332216315888. doi:10.1016/j.biopha.2016.11.028
41. Farooqui W, Pommergaard HC, Rasmussen AJTR. Remote ischemic preconditioning of transplant recipients to reduce graft ischemia and reperfusion injuries: a systematic review. Transplant Rev. 2017;32(1):S0955470X16301458.
42. Zhang W, Liu Y, Wu M, et al. PI3K inhibition protects mice from NAFLD by down-regulating CMKLR1 and NLRP3 in Kupffer cells. J Physiol Biochem. 2017;73(4):1–12.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.