Back to Journals » International Journal of Women's Health » Volume 14
Iron Deficiency Anemia and Associated Factors Among Adolescent Girls and Women in a Rural Area of Jatinangor, Indonesia
Authors Sari P , Judistiani RTD , Herawati DMD, Dhamayanti M , Hilmanto D
Received 24 May 2022
Accepted for publication 3 August 2022
Published 23 August 2022 Volume 2022:14 Pages 1137—1147
DOI https://doi.org/10.2147/IJWH.S376023
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Elie Al-Chaer
Puspa Sari,1 Raden Tina Dewi Judistiani,1 Dewi Marhaeni Diah Herawati,1 Meita Dhamayanti,2 Dany Hilmanto2
1Department of Public Health, Faculty of Medicine, Padjadjaran University, Sumedang, West Java, Indonesia; 2Department of Child Health, Hasan Sadikin Hospital, and Faculty of Medicine, Padjadjaran University, Sumedang, West Java, Indonesia
Correspondence: Puspa Sari, Department of Public Health, Faculty of Medicine, Padjadjaran University, Jalan Ir. Soekarno KM. 21, Jatinangor, Sumedang, 45363, Indonesia, Tel +62 022 84288888, Email [email protected]
Background: Iron deficiency anemia is a common problem among adolescent girls and women, with significant consequences on personal health. One of the causes of iron deficiency anemia is inadequate nutritional intake. This study explores iron-deficiency anemia and associated factors among adolescent girls and women in a rural area of Jatinangor, Indonesia.
Methods: A cross-sectional study was conducted with 95 adolescent girls and 85 women between April and November 2018. Cluster random sampling was used to select the participants from seven villages in the Jatinangor district. After obtaining informed consent, we collected sociodemographic data, menstrual histories, and related data, including nutritional intake using 24-hour dietary recall. Anthropometrics were gathered to determine the body mass Index (BMI), and venous blood samples were analyzed for complete blood count and hemoglobin levels. Descriptive statistics followed by bivariate and multivariable logistic regression were used to identify anemia-associated factors.
Results: The prevalence of iron deficiency anemia among the girls was 21.1% and 9.4% among women, with an average hemoglobin level in adolescents of 10.75 g/dL (± 0.79) and in adults 11.20 g/dL (± 0.61), whereas MCV was 74.49± 8.22 fL in adolescents and 7.61± 8.62 fL in adults. The majority of our samples were not stunted in growth and were also within a normal weight range. Multivariate logistic regression analysis showed that protein intake (OR=0.25; 95% CI 0.11– 0.58) was a positively associated factor with anemia.
Conclusion: The prevalence of iron-deficiency anemia in this study represents a mild public health problem in the study sample. Based on the hemoglobin level, anemia, can be classified as moderate in adolescents and mild in adults. Low levels of MCH indicate iron-deficiency anemia. Sufficient protein intake did not prevent anemia due to macronutrient and micronutrient intake.
Keywords: hematological profile, iron deficiency anemia, nutrient intake
Introduction
Anemia is a global public health concern affecting both developing and developed countries.1 It defines a condition in which the number of red blood cells (RBCs) and their oxygen-carrying capacity are insufficient to meet the body’s physiological needs. A diagnosis of anemia is made when the red blood cell (RBC) count is <4.2 million/µL, or hemoglobin (Hb) is <12 g/dL.2 The most significant cause of anemia is iron deficiency, which accounts for at least 50% of cases of anemia.3–7 Low levels of MCH can be used as a standard for the diagnosis of iron-deficiency anemia (IDA).8
Iron is a micronutrient that is essential for hemoglobin production and enzyme function. IDA is particularly high among pregnant women, children under five, adolescent girls, and women due to their higher nutritional requirements for iron.9–11 Iron is required in menstruation, physical growth, reproductive maturation, and for cognitive growth and activity.10,12–15 Iron deficiency generally occurs when iron intake from foods cannot meet the required needs to support its physiological needs and the iron supplied in the body is thus depleted. Adolescents with anemia tend to also experience anemia during pregnancy, which results in various adverse effects on pregnancy and childbirth. Some other studies have reported that anemic adolescent girls exhibit stunting.16 Anemia can also be caused by other factors, including vitamin deficiencies, malaria, schistosomiasis, and hookworm.9–11,14,17–20 Iron deficiency anemia is predicted to be the crucial nutritional problem.21 According to Indonesia Baseline Health Research report in 2018, as many as 96.8% of adolescents in Indonesia aged 10–14 years and 96.4% of adolescents aged 15–19 years do not consume vegetables or fruit, which, as is known, are foods rich in essential micronutrients.22 Protein is contained in vegetables and, lack of vegetable consumption can cause protein deficiency. The nutritional intake problems including nutritional deficiencies is one of the causes of the double burden of the disease. Adequate nutrition is the foundation for a healthy lifestyle and appropriate development. One of the United Nation’s Sustainable Development Goals is to address the nutritional needs of adolescent girls.23
The World Health Organization (WHO) defines adolescence as the period from 10 to 19 years of age.24 The worldwide prevalence of anemia among adolescents is 27% in developing countries and 6% in developed countries.25 In Indonesia, anemia is a considerable health concern, with a prevalence of 22.7% in women of childbearing age, 37.1% in pregnant women, and 30.0–46.6% in female workers.26,27 According to the WHO, anemia in Indonesia among women of reproductive age (15–49 years) has increased from 21.6% in 2018 to 22.3% in 2019.28 National data on the prevalence of anemia in Indonesia are not reported every year. In addition, anemia trends have not been published. For example, in the National Basic Health Research Survey by the National Institute of Health Research and Development, Ministry of Health conducted in 2013, the prevalence of anemia was estimated in children 0 up to 15 years, pregnant women, non-pregnant women, and men.29 Moreover, in 2018, only anemia rates in pregnant women are reported. In terms of public health importance, the WHO has stated that a prevalence of anemia ≤4.9% is not a public health problem, 5.0–19.9% is a mild public health problem, 20.0–39.9% is a moderate public health problem, and ≥40% is a severe public health problem.12
Anemia reduces physical and mental capacity, work productivity, cognitive performance, resistance to infection, reproductive health, and quality of life.9,11,25,30–33 Expansion of iron supplementation programs, food fortification, nutritional education, screening, and other strategies to improve iron intake may reduce the prevalence of anemia, and causes of anemia other than iron deficiency should also be investigated, while intestinal parasites should be treated as soon as possible.11,13,34 Additionally, attention to adolescents and adult women is not as sufficient as the attention placed on pregnant women and toddlers. Therefore, effective measures must be taken to eradicate this nutritional problem especially for increasing the quality of life.7,15,35,36
Although the program administering iron supplements has been in place for a long time in Indonesia, the prevalence of iron deficiency anemia is still high, especially in rural areas. Nonetheless, there have been limited anemia studies specifically focused on the prevalence and causes of anemia among women and adolescent girls in this region. Therefore, this study aimed to explore iron deficiency anemia and associated factors among this demographic in a rural area of Jatinangor, Indonesia.
Materials and Methods
Study Design
A cross-sectional study was conducted between April and November 2018 to assess iron deficiency anemia and associated factors among women and adolescent girls in a rural area of Jatinangor, Indonesia. The study was conducted following the guidelines of the Declaration of Helsinki. The study was approved by the Ethical Committee of the Faculty of Medicine, Universitas Padjadjaran, approval number 321/UN6.KEP/EC/2018. The study involved an approval from the district health offices, the primary health care units, and at the subdistrict, and village level. Participants were informed about the study method and asked to provide written informed consent. The parents or legal guardians of the adolescent girls approved the informed consent. The study was conducted in seven villages in the Jatinangor Primary Health Care district.
Sample Size and Sampling Procedure
Adolescent girls (10–19 years) and women (20–35 years) who were healthy, had lived in the study area for greater than 1 year, nonpregnant and nonlactating during the study period were included in the study. In addition, subjects with a history of any form of thalassemia, were excluded. A cluster random sampling technique was used to select study participants. This study divided the population of adolescent girls and women into clusters based on the primary health care working area village, namely Cilayung, Cileles, Cipacing, Mekargalih, Cikeruh, Sayang, and Hegarmanah, then selected participants with a simple random sampling technique. Sample size for this study was calculated with G*Power 3.1.9.4 for Windows, with means difference between two groups statistical test, the assumptions of: 95% confidence level (CI), 5% margin of error, the minimum sample size was 176. We achieved 180 sample size for this study (95 adolescent girls and 85 women).
Sociodemographic Data
A structured questionnaire was used to obtain information from participants. Data collected included education, occupation, marital status, menstruation history, and macronutrient and micronutrient intake over 24 hours, these data were analyzed by NutriSurvey (EBISpro) 2007.
Anthropometric Measurements
Weight was measured using an Omron scale to the nearest 0.1 kilogram. Height was measured with a Seca stadiometer to the nearest 0.1 centimeter. The body mass index (BMI) was determined by calculating weight (kg)/height (m). Participants were classified as underweight, normal, overweight, or obese based on the WHO BMI charts.12
Hemoglobin
Anemia was the outcome variable of the study. If the hemoglobin level was <12 g/dL for a participant, it indicated the presence of anemia. Based on WHO classifications, hemoglobin levels below 8 g/dL were classified as severe anemia, 8–10.99 g/dL as moderate anemia, and 11–11.9 g/dL as mild anemia. Hb of 12 g/dL and above were classified as normal.37 MCV can also be used as a measure to diagnose anemia, at the same time, normal MCV is between 80 to 100 fL.
Data Processing and Analysis
Data were checked, cleaned, and coded using the IBM SPSS version 27 statistical software. Descriptive statistics such as frequencies, proportions, and standard deviations were calculated. Bivariate logistic regression analyses were then performed to look for correlations between each independent variable and the outcome variable (anemia). All variables with a p-value <0.5 in the bivariate analysis were entered in a multivariable logistic regression analysis. Odds ratios (OR) with 95% confidence intervals (CI) were used to identify the independent predictors of anemia.
Results
A total of 95 adolescent girls and 85 women participated in this study (Table 1). The majority of the participants adolescent girls (95.79%) and women (54.12%) had completed secondary school education. Most participants, (98.9%) adolescent girls and women (90.6%) were unmarried. About (75.8%) adolescent girls and (75.3%) women had a regular menstruation cycle. Most participants (83.2%) adolescent girls and (91.8%) had ≥6 days of bleeding in each menses. Most of participants required ≥3 sanitary pad usage per day.
 |
Table 1 Sociodemographic Data and Menstruation History |
Approximately 21.1% adolescent girls and 9.4% women were classified as have no anemia (Table 2). Most adolescent girls (89.5%) and women (96.5%) were not stunted in growth. Most adolescent girls (59%) and women (77.7%) were of normal weight. The prevalence of anemia and the weight classification between adolescent girls and women were significantly different (P<0.005). Both adolescent girls and women had a normal BMI value (Table 3). According to the Dietary Reference Intake guidelines, both adolescent girls and women consumed inadequate dietary fat, folic acid, vitamin C, and iron.38,39 In addition not, only weight, height, fat, and folic acid intake, but also MCH presented significant differences between adolescent girls and women.
 |
Table 2 Prevalence and Classification of Anemia, Stunting, and Weight Classification |
 |
Table 3 Anthropometric Measures, Nutritional Intake, and Hematological Characteristics |
Most adolescent girls (82.67%) had ≥6 days of bleeding at each menses, with a regular menstruation cycle (74.67%), normal weight (76.4%), not stunted (89.33%), but had a low macronutrient and micronutrient intake according to Dietary Reference Intake (Table 4). Hematology parameters including hemoglobin, hematocrit, thrombocytes, MCV, MCH, and MCHC levels between anemia and no-anemia adolescent girls presented significantly differences with p<0.05.
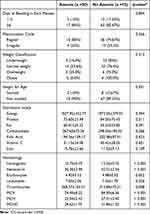 |
Table 4 Menstruation History, Height and Weight Classification, Nutritional Intake, and Hematological Characteristics Among Adolescent Girls |
Most women (92.21%) had ≥6 days of bleeding at each menses, with regular menstruation cycle (76.62%), normal weight (96.11%), no stunted growth (96.11%), but had a low macronutrient and micronutrient intake according to the Dietary Reference Intake guidelines (Table 5). Similar to the results shown for adolescent girls, hematology parameters including hemoglobin, hematocrit, thrombocytes, MCV, MCH, and MCHC levels between anemia and no-anemia women presented significant differences with p<0.05. Multivariate analyses revealed that the factor that significantly associated with anemia among adolescent girls and women was protein intake with p<0.001 (Table 6).
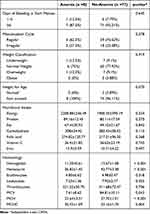 |
Table 5 Menstruation History, Height and Weight Classification, Nutritional Intake, and Hematological Characteristics Among Women |
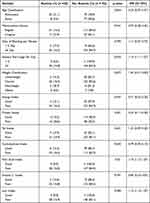 |
Table 6 Factors Associated with Iron-Deficiency Anemia Among Women and Adolescent Girls in Jatinangor, Indonesia |
Discussion
The prevalence of iron deficiency anemia in Jatinangor, Indonesia, was found to be 21.1% in adolescent girls and 9.4% in women. The anemia prevalence among women was smaller than the national averages of 22.7% but higher among adolescent girls, which is 15% in the national average.3–5 According to the WHO, this was classified as a mild public health problem. The prevalence of anemia in this study was lower than that found in similar studies conducted in Tasikmalaya, Indonesia, which was 22.2%.2 This might be due to differences in sociodemographic and eating habits. The hemoglobin level observed in this study can be classified as moderate anemia in adolescents and mild anemia in adults. In addition, we found there was a difference in the average hemoglobin of adolescents and adults; the possible justification is that adolescents are in a period of rapid growth and development, and they require more iron. Moreover, this variation may be due to the education level of participants and also affects the prevalence of anemia. The highest level of education of women in this study was mainly secondary school and university. Previous studies have found significant associations between education and knowledge particularly relating to anemia.6,9,10 This result is consistent with studies conducted in Sub-Saharan Africa indicating that education decrease the prevalence of anemia.40 Education influences a participant’s knowledge, especially about anemia.17 The possible justification is that someone who already understands anemia prevention will make efforts to prevent anemia.
The number of adolescent girls enrolled was more than the number of women, because this study was conducted in villages around the Padjadjaran University campus, where most of the population are students; moreover, residents’ houses are rented out as boarding houses. Furthermore, participants with the most anemia in this study were single. Other studies have revealed that marital status and contraceptives significantly increased the prevalence of anemia.40 In contrast, we found that the prevalence of anemia in women was less than in adolescents. A compelling reason is that among the participants who were married in this study, most had a secondary education level and above, so that their ability to improve literacy regarding efforts to prevent anemia may be higher. However, this study has a limitation that it did not explore the contraceptive history of adult women. Furthermore, we found that the menstrual cycle, length of day blood flow in each menses, and sanitary pad usage per day were not significantly associated with anemia. This result is different from that of studies conducted in Ethiopia, indicating that blood loss during menstruation was correlated with anemia.11 Menstruation is not the only cause of anemia, and we found that level of education influences the prevalence of anemia in this study.
Previous research has found a significant association between anemia and BMI, with participants who had a low BMI being 3.2 times more likely to be anemic.9 Other studies have reported that a higher BMI was significantly associated with the prevalence of anemia.7 However, our study found no correlation. Most of our participants had a normal BMI. Additionally, anemia can be caused by other factors, including parasitic infections of malaria, schistosomiasis, and hookworm.11,12,14,24–27,41 The limitation of this study is no laboratory tests were carried out for thalassemia. Other studies have found that testing to diagnose IDA or anemia would be helpful as a thalassemia carrier is needed.42
Our study shows that most participants was not stunted in growth. Adolescents with anemia tend to experience anemia during pregnancy, which results in various adverse effects during pregnancy and childbirth, including stunting growth.16 There is a difference between weight and height among adolescent girls and women, this is due to the age difference. We found that the average body weight of adolescent girls is smaller than that of adult women. The possible reason is because, in general, adolescents desire to have an ideal body weight as a positive self-image.43 This is inversely proportional to the macronutrient intake results, where adolescents consume more fat than women. In accordance with Indonesian national data, adolescents consume more junk food and fatty instant foods than vegetables and fruit.44 Moreover, adolescents and adult women have the habit of snacking without regard to the nutrients they eat. Another study states that adolescent have a tendency to consume foods containing sugar.45 Our research also shows that the average folic acid intake of adolescents is lower than that of women, this is in accordance with other studies which state that adolescents have a reduced intake of micronutrients.45,46 In the study reported by Rahfiludin, IDA was associated with less animal-based foods characterized by iron with higher bioavailability.47 This finding can also explain why anemia in this study is more common in adolescents than in adults. One factor that causes anemia is an inadequate nutritional intake. These nutritional intake problems include nutritional deficiencies and excesses, such as iron deficiency and nutritional excess, giving rise to a double burden of malnutrition. In fact, several programs and policies are already in place aimed at improving adolescent nutrition, including iron supplementation and obesity prevention programs in schools. Nonetheless, adolescent nutrition has not become a priority in the national development agenda.36 Therefore, nutrition education is important to solve this problem.48
We determined there were differences in hematological parameters including hemoglobin, MCH, MCV, and MCHC levels in our study population. The MCV and MCH values can identify iron deficiency in the body.49 This study shows that the average of MCV, MCH and MCHC values among respondents were below the standard levels. Low levels of MCH in this study could indicate iron deficiency anemia. MCV defines the size of the RBCs and is expressed as femtoliters (10–15; fL) or as cubic microns (µm3). The normal values for MCV are 87±7 fL. Additionally, MCH quantifies the amount of hemoglobin per RBC. The normal values for MCH are 29±2 picograms (pg) per cell, moreover MCHC indicates the amount of hemoglobin per unit volume. The normal values for MCHC are 34±2 g/dL.8
In our study, we found that protein intake was significantly associated with anemia. Adequate protein intake may be related to the prevalence of IDA observed in this study. The possible reason could be that proteins contribute to hemoglobin synthesis. This finding was in agreement with the finding which is reported by a study done in Japan that protein intake may reduce iron deficiency among females.50 This result is supported by Indonesia Baseline Health Research, that adolescents in Indonesia consume fewer vegetables, where vegetables are a source of protein.
Study Limitations
One limitation of this study is that no laboratory tests were conducted for thalassemia, which is one of the causes of microcytic anemia. In addition, we did not explore the contraceptive history of adult women.
Conclusion
According to the WHO, the prevalence of iron deficiency anemia in this study represents a mild public health problem. Based on the hemoglobin level, it can be classified as moderate anemia in adolescents and mild anemia in adults. The difference in average hemoglobin in adults and adolescents is that the during the adolescent growth period, the body requires sufficient iron, and the adult education level is quite good. Low levels of MCH indicate iron deficiency anemia. Most participants were not stunted in growth and had average weight. Sufficient protein intake did not prevent anemia due to macronutrient and micronutrient intake. We recommend that nutrition education is a crucial preventative strategy for IDA.
Acknowledgments
We are thankful to Universitas Padjadjaran for the research fund, and the girls, women, and midwives in the Primary Health Care Facility.
Funding
This research and publication were supported by Universitas Padjadjaran.
Disclosure
The authors report no conflicts of interest in this work.
References
1. McLean E, Cogswell M, Egli I, et al. Worldwide prevalence of anaemia, WHO vitamin and mineral nutrition information system, 1993–2005. Public Health Nutr. 2009;12(4):444–454. doi:10.1017/S1368980008002401
2. World Health Organization (WHO). Global Nutrition Targets 2025: anemia Policy Brief; 2014. Available from: https://www.who.int/nutrition/publication/globaltargets2025%20policybrief%20anemia/en/.
3. Chandrakumari AS, Sinha P, Singaravelu S, et al. Prevalence of anemia among adolescent girls in a rural area of Tamil Nadu, India. J Family Med Prim Care. 2019;8(4):1414–1417. doi:10.4103/jfmpc.jfmpc_140_19
4. World Health Organization (WHO). Iron deficiency anemia: assessment, prevention, and control: a guide for programme managers; 2011.
5. Kassebaum NJ; Collaborators GBDA. The global burden of anemia. Hematol Oncol Clin North Am. 2016;30(2):247–308. doi:10.1016/j.hoc.2015.11.002
6. Kassebaum NJ, Jasrasaria R, Naghavi M, et al. A systematic analysis of global anemia burden from 1990 to 2010. Blood. 2014;123(5):615–624. doi:10.1182/blood-2013-06-508325
7. Woldu B, Enawgaw B, Asrie F, et al. Prevalence and associated factors of anemia among reproductive-aged women in Sayint Adjibar Town, Northeast Ethiopia: community-based cross-sectional study. Anemia. 2020;2020:8683946. doi:10.1155/2020/8683946
8. Sarma PR. Red cell indices. Walker HK, Hall WD, Hurst JW, editors. In: Clinical Methods: The History, Physical, and Laboratory Examinations.
9. Gebreyesus SH, Endris BS, Beyene GT, et al. Anaemia among adolescent girls in three districts in Ethiopia. BMC Public Health. 2019;19(1):92. doi:10.1186/s12889-019-6422-0
10. Habib N, Abbasi SS, Aziz W. An Analysis of societal determinant of anemia among adolescent girls in Azad Jammu and Kashmir, Pakistan. Anemia. 2020;2020:1628357. doi:10.1155/2020/1628357
11. Mengistu G, Azage M, Gutema H. Iron deficiency anemia among in-school adolescent girls in rural area of Bahir Dar City Administration, North West Ethiopia. Anemia. 2019;2019:1097547. doi:10.1155/2019/1097547
12. World Health Organization (WHO). The global prevalence of anaemia in 2011; 2015. Available from: https://apps.who.int/iris/bitstream/handle/10665/177094/9789241564960_eng.pdf.
13. World Health Organization (WHO). Prevention of iron deficiency anaemia in adolescents; 2011.
14. Engidaw MT, Wassie MM, Teferra AS. Anemia and associated factors among adolescent girls living in Aw-Barre refugee camp, Somali regional state, Southeast Ethiopia. PLoS One. 2018;13(10):e0205381. doi:10.1371/journal.pone.0205381
15. Haas JD, Brownlie T. Iron deficiency and reduced work capacity: a critical review of the research to determine a causal relationship. J Nutr. 2001;131(2S–2):
16. Tura MR, Egata G, Fage SG, et al. Prevalence of Anemia and Its Associated Factors Among Female Adolescents in Ambo Town, West Shewa, Ethiopia. J Blood Med. 2020;11:279–287. doi:10.2147/JBM.S263327
17. Young people’s health–a challenge for society. Report of a WHO Study Group on young people and “Health for All by the Year 2000. World Health Organ Tech Rep Ser. 1986;731:1–117.
18. Chami GF, Fenwick A, Bulte E, et al. Influence of Schistosoma mansoni and Hookworm infection intensities on anaemia in Ugandan villages. PLoS Negl Trop Dis. 2015;9(10):e0004193. doi:10.1371/journal.pntd.0004193
19. Moreno LA, Gonzalez-Gross M, Kersting M, et al. Assessing, understanding and modifying nutritional status, eating habits and physical activity in European adolescents: the HELENA (Healthy Lifestyle in Europe by Nutrition in Adolescence) Study. Public Health Nutr. 2008;11(3):288–299. doi:10.1017/S1368980007000535
20. Zimmermann MB, Hurrell RF. Nutritional iron deficiency. Lancet. 2007;370(9586):511–520. doi:10.1016/S0140-6736(07)61235-5
21. Chaparro CM; World Health Organization (WHO). Anemia among adolescent and young adult women in Latin America and the Caribbean: a cause for concern. Unit on Child and Adolescent Health. Pan American Health Organization.
22. Baseline Health Research, Indonesia Ministry of Health. Baseline Health Research 2013. Jakarta: Baseline Health Research; 2013.
23. World Health Organization. Global accelerated action for the health of adolescents (AA-HA); 2017. Available from: https://apps.who.int/iris/bitstream/handle/10665/255415/9789241512343-eng.pdf.
24. World Health Organization (WHO). Adolescent Pregnancy. World Health Organization; 2019.
25. Isik Balci Y, Karabulut A, Gurses D, et al. Prevalence and risk factors of anemia among adolescents in Denizli, Turkey. Iran J Pediatr. 2012;22(1):77–81.
26. Ministry of Health, the Republic of Indonesia. National Basic Health Research (Riset Kesehatan Dasar) 2013”. Agency for Health Research and Development. Jakarta: Indonesia; 2013.
27. UNICEF. The Situation of Children and Women in Indonesia 2000–2010: Working Towards Progress with Equity Under decentralisation”UNICEF; 2011. Jakarta: Indonesia: UNICEF; 2011.
28. World Health Organization (WHO). Prevalence of anaemia in women of reproductive age (aged 15–49) (%). Available from: https://www.who.int/data/gho/data/indicators/indicator-details/GHO/prevalence-of-anaemia-in-women-of-reproductive-age.
29. Barkley JS, Kendrick KL, Codling K, et al. Anaemia prevalence over time in Indonesia: estimates from the 1997, 2000, and 2008 indonesia family life surveys. Asia Pac J Clin Nutr. 2015;24(3):452–455. doi:10.6133/apjcn.2015.24.3.22
30. Halterman JS, Kaczorowski JM, Aligne CA, et al. Iron deficiency and cognitive achievement among school-aged children and adolescents in the United States. Pediatrics. 2001;107(6):1381–1386. doi:10.1542/peds.107.6.1381
31. Shaka MF, Wondimagegne YA. Anemia, a moderate public health concern among adolescents in South Ethiopia. PLoS One. 2018;13(7):e0191467. doi:10.1371/journal.pone.0191467
32. Tesfaye M, Yemane T, Adisu W, et al. Anemia and iron deficiency among school adolescents: burden, severity, and determinant factors in southwest Ethiopia. Adolesc Health Med Ther. 2015;6:189–196. doi:10.2147/AHMT.S94865
33. Khani Jeihooni A, Hoshyar S, Afzali Harsini P, et al. The effect of nutrition education based on PRECEDE model on iron deficiency anemia among female students. BMC Women’s Health. 2021;21(1):256. doi:10.1186/s12905-021-01394-2
34. Campbell RK, Aguayo VM, Kang Y, et al. Epidemiology of anaemia in children, adolescent girls, and women in Bhutan. Matern Child Nutr. 2018;14(Suppl):e12740. doi:10.1111/mcn.12740
35. Tandoh MA, Appiah AO, Edusei AK. Prevalence of anemia and undernutrition of adolescent females in selected schools in Ghana. J Nutr Metab. 2021;2021:6684839. doi:10.1155/2021/6684839
36. Soekarjo DD, Roshita A, Thow AM, et al. Strengthening nutrition-specific policies for adolescents in Indonesia: a qualitative policy analysis. Food Nutr Bull. 2018;39(3):475–486. doi:10.1177/0379572118785054
37. World Health Organization (WHO). Nutritional Anaemias: Tools for Effective Prevention and Control. Geneva, Switzerland; 2017.
38. National Institutes of Health. Nutrient Recommendations: dietary Reference Intakes (DRI). Available from: https://ods.od.nih.gov/HealthInformation/Dietary_Reference_Intakes.aspx.
39. Dietary Reference Intake; 1997. Available from: https://en.wikipedia.org/wiki/Dietary_Reference_Intake.
40. Nti J, Afagbedzi S, da-Costa Vroom FB, Ibrahim NA, Guure C. Variations and determinants of anemia among reproductive age women in five Sub-Saharan Africa Countries. Biomed Res Int. 2021;2021:9957160. doi:10.1155/2021/9957160
41. World Health Organization (WHO). Prevention of Iron deficiency anemia in adolescents. Role of Weekly Iron and Folic Acid Supplementation; 2011.
42. Susanti AI, Sahiratmadja E, Winarno G, et al. Low hemoglobin among pregnant women in midwives practice of primary Health Care, Jatinangor, Indonesia: iron deficiency anemia or β -thalassemia trait? Anemia. 2017;2017:6935648. doi:10.1155/2017/6935648
43. Mueller AS, Pearson J, Muller C, Frank K, Turner A, Mueller AS, Pearson J, Muller C, et al. Sizing up peers: adolescent girls’ weight control and social comparison in the school context. J Health Soc Behav. 2010;51(1):64–78. doi:10.1177/0022146509361191
44. Indonesia Basic Health Research (RISKESDAS); 2018.
45. Bates B, Cox L, Nicholson S, et al. National diet and nutrition survey results from years 5 and 6 (combined) of the Rolling Programme (2012/2013–2013/2014); 2016. Available at: https://www.gov.uk/government/statistics/ndns-results-from-years-5-and-6-combined.
46. Bush LA, Hutchinson J, Hooson J, et al. Measuring energy, macro and micronutrient intake in UK children and adolescents: a comparison of validated dietary assessment tools. BMC Nutr. 2019;5:53. doi:10.1186/s40795-019-0312-9
47. Rahfiludin MZ, Arso SP, Joko T, et al. Plant-based diet and iron deficiency anemia in Sundanese Adolescent Girls at Islamic boarding schools in Indonesia. J Nutr Metab. 2021;2021:6469883. doi:10.1155/2021/6469883
48. Burrows T, Golley RK, Khambalia A, et al. The quality of dietary intake methodology and reporting in child and adolescent obesity intervention trials: a systematic review. Obes Rev. 2012;13(12):1125–1138. doi:10.1111/j.1467-789X.2012.01022.x
49. World Health Organization (WHO). Nutritional anaemias: tools for effective prevention and control. 2017. Available at: https://www.who.int/publications/i/item/9789241513067.
50. Kokubo Y, Kisara K, Yokoyama Y, et al. Habitual dietary protein intake affects body iron status in Japanese female college rhythmic gymnasts: a follow-up study. Springerplus. 2016;5(1):862. doi:10.1186/s40064-016-2569-7
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
