Back to Journals » Drug Design, Development and Therapy » Volume 13
Intravitreal corticosteroid implant vs intravitreal ranibizumab for the treatment of macular edema: a meta-analysis of randomized controlled trials
Authors Wei Q, Chen R, Lou Q, Yu J
Received 19 August 2018
Accepted for publication 19 December 2018
Published 11 January 2019 Volume 2019:13 Pages 301—307
DOI https://doi.org/10.2147/DDDT.S184520
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Qiongyu Guo
Qingquan Wei,1,2,* Rui Chen,1,2,* Qiyang Lou,1,2 Jing Yu1,2
1Department of Ophthalmology, Ninghai First Hospital, Zhejiang 315600, People’s Republic of China; 2Department of Ophthalmology, Shanghai Tenth People’s Hospital Affiliated with Tongji University, Shanghai 200072, People’s Republic of China
*These authors contributed equally to this work
Purpose: The purpose of this meta-analysis was to compare the efficacy and safety of corticosteroid implant and intravitreal ranibizumab for the treatment of macular edema (ME).
Materials and methods: PubMed, Embase, Web of Science, and the Cochrane Central Register of Controlled Trials were comprehensively searched for studies comparing dexamethasone implant with ranibizumab in patients with ME. Best-corrected visual acuity (BCVA), central subfield thickness (CST), and adverse events were extracted from the final eligible studies. RevMan 5.3 software was used to analyze the data, and the modified Jadad assessment tool was used to access the quality of outcomes.
Results: Three randomized controlled trials (RCTs) were included in our analysis. The types of causes of ME include central retinal vein occlusion (CRVO), branch retinal vein occlusion (BRVO), and diabetic retinopathy (DR). The ranibizumab treatment group had significantly better BCVA compared with the corticosteroid treatment group (standard mean difference [SMD] -0.80; 95% CI -1.08, -0.53; P<0.00001). The ranibizumab treatment group also had higher CST reduction compared with the corticosteroid treatment group, and there was a significant difference (weighted mean difference [WMD] 167.58; 95% CI 125.21–209.95; P<0.00001). There was no significant difference in serious adverse effects between the two groups (SMD 1.67; 95% CI 0.69, 4.05; P=0.26). However, the use of corticosteroid implant had a higher risk of intraocular pressure (IOP) (OR 6.88; 95% CI 4.53–10.44; P<0.00001) elevation and cataract (OR 3.98; 95% CI 1.89–8.37; P=0.0003) than ranibizumab treatment and fewer injections.
Conclusions: Compared with ranibizumab, corticosteroid implant did not have greater improved BCVA, but corticosteroid implant had less CST reduction. The advantages of corticosteroids are fewer injections, while the advantages of ranibizumab include fewer side effects.
Keywords: macular edema, dexamethasone implant, ranibizumab, meta-analysis
Introduction
Macular edema (ME) is a common phenomenon in various retinal diseases in which fluid and protein accumulate in the extracellular space within the retina.1,2 ME is associated with several disorders including retinal vein occlusion (RVO) and diabetic retinopathy (DR) and can lead to causes of vision loss in patients.3 Several processes have been implicated in the breakdown of the blood–retinal barrier (BRB) that leads to ME. These processes include production of pro-inflammatory mediators, such as prostaglandins and IL-6, and increased amounts of vascular permeability factors such as VEGF and loss of endothelial tight junction integrity.4 Reduction in ME may be associated with improved vision.
Currently, there are many treatments available for ME due to central retinal vein occlusion (CRVO), branch retinal vein occlusion (BRVO), and DR, but the most common treatments are intravitreal anti-VEGF agents and intravitreal steroids.5 Both ranibizumab and corticosteroid implant separately have been found to be effective in improving visual acuity and reducing ME in patients with RVO and DR.6–19 Specifically, ranibizumab, a humanized monoclonal antibody fragment that neutralizes all isoforms of VEGF-A, was approved by the US Food and Drug Administration (FDA) for the treatment of ME secondary to CRVO, BRVO, and DR and showed resolution of ME and improvement in visual acuity in these patients in a long-term follow-up.6–9,16,18 Ranibizumab has been effective in improving visual acuity in patients with ME; however, a mean of at least seven injections may be required during the first year, together with monthly follow-up.20–23
Recently, anti-inflammatory and antiangiogenic strategies have been used to target vascular permeability and leakage to reduce ME and improve vision.24 Corticosteroids have anti-inflammatory, antiangiogenic, and antipermeability properties that make them an attractive therapeutic option for several posterior segment diseases.10,25–27 Dexamethasone or triamcinolone intravitreal implant is a biodegradable implant that provides sustained release of the corticosteroid dexamethasone into the vitreous humor for up to 6 months.19,28,29 However, long-acting steroid preparations have potential side effects including cataract and glaucoma, which are more likely to occur when treatment exceeds 6 months and may result in additional intraocular surgery, which can be considered a drawback of treatment.30
To date, no systematic review has discussed the therapeutic effect and safety of intravitreal ranibizumab vs corticosteroid implant in ME. We performed a meta-analysis to quantify the efficacy and safety of these two treatments.
Materials and methods
Literature search
A systematic English language search of PubMed, Embase, Web of Science, and the Cochrane Central Register of Controlled Trials for human studies published up to June 2018 was conducted with language restrictions. Key terms included the following: “macular edema/oedema”, “ranibizumab”, “corticosteroid intravitreal implants”, “intravitreal dexamethasone drug delivery system”, “steroid implants”, “dexamethasone insert”, “Ozurdex”, “fluocinolone acetonide insert”, “Retisert”, “intravitreal triamcinolone acetonide,” and “Iluvien”. We manually searched the reference lists of all original studies and review articles identified by the electronic search to identify other potentially eligible articles.
Inclusion criteria
We selected the following studies: 1) types of ME include RVO and DR, 2) studies featuring a randomized controlled trial (RCT) design comparing the combination of intravitreal dexamethasone implant and intravitreal ranibizumab, 3) studies measuring at least one outcome of interest, 4) 6 months or more follow-up period.
Exclusion criteria
The following studies were excluded: 1) studies that were not RCTs, 2) studies with inconsistent or erroneous data, 3) studies that had the follow-up period of <6 months.
Data extraction and quality assessment
Titles and abstracts were reviewed by two reviewers using the abovementioned selection criteria. Disagreements were resolved via consensus after discussion. Full-text versions of all relevant studies were obtained for detailed evaluations. Data extraction and quality assessments were conducted using the modified Jadad assessment tool.31 The following data were extracted from each study: the name of the first author, the study design, and the major inclusion and exclusion criteria as well as information regarding study population characteristics (age, sex, and number of eyes in the study), intervention groups, injection intervals and outcome variables, and duration of follow-up.
Statistical analyses
The meta-analysis was conducted using RevMan5.3 software. Risk ratios (RRs) were measured using 95% CIs for dichotomous data, while weighted mean differences (WMDs) were measured using 95% CIs for continuous data. Standard mean differences (SMDs) were used when all of the trials assessed the same outcomes in several ways. The Q test or I2 test was used to evaluate heterogeneity. Both fixed-effects and random-effects models were used to obtain summary ORs, WMDs, or SMDs. In the absence of heterogeneity between groups, the fixed-effects model and random-effects model yielded concordant results. When the heterogeneity was significant, the random-effects model was employed. An I2 value of >50% accompanied by a P-value of <0.05 for the Q test was determined to indicate the presence of significant heterogeneity. Potential publication bias was estimated using Egger’s test and by visually evaluating a funnel plot.
Results
Literature search
Figure 1 shows the flowchart of the selection process used to identify eligible studies. A total of 236 articles were initially identified. We screened the titles and abstracts of these articles and identified 15 eligible studies. During examination of these full-length articles, eight were excluded from the present meta-analysis, because the treatment was combined with laser therapy. One study was ruled out because bevacizumab was first used. Ultimately, three published16–18 articles were eligible for analysis.
  | Figure 1 Study selection. |
Included studies
The basic characteristics of the three included studies are summarized in Table 1. Sample sizes ranged from 243 to 363 eyes. Mean patient age ranged from 63.4 to 66.9 years. The dose of ranibizumab was 0.5 mg in the ranibizumab groups of the included studies. The dose of dexamethasone was 0.7 mg in the dexamethasone groups of the included studies. Moreover, the duration of follow-up varied from 6 to 12 months among the studies. The three studies were assessed regarding methodological quality according to the Jadad score and were determined to be of high quality.
Estimation of outcomes
Improvement in best-corrected visual acuity (BCVA)
In our included studies, only one study at some point reported about BCVA, another study about visual gain, so we converted the data into visual gain. Visual acuity was the most important outcome measure with respect to treatment efficacy. Figure 2 shows the results regarding changes in mean BCVA. The pooled results revealed that the ranibizumab treatment group had a better visual acuity compared with the corticosteroid treatment group, and there was a significant difference (SMD −0.80; 95% CI −1.08, −0.53; P<0.00001). The random-effects model was used due to the high heterogeneity of the effect size (I2=56%, P=0.13).
  | Figure 2 Forest plot showing the changes in mean BCVA compared with baseline. |
Mean change in central subfield thickness (CST)
Figure 3 shows the effect of intravitreal corticosteroid or ranibizumab implant on CST. The pooled results indicate that the ranibizumab treatment group had higher CST reduction compared with the corticosteroid treatment group, and there was a significant difference (WMD 167.58; 95% CI 125.21–209.95; P<0.00001) without heterogeneity (I2=49%, P=0.16).
  | Figure 3 Forest plot showing the changes in mean CST compared with baseline. |
Total serious adverse events
We compared the numbers of serious adverse events, such as cardiac disorders, congenital disorders, familial and genetic disorders, and so on, in the corticosteroid group with those in the ranibizumab group, and heterogeneity was detected between studies (I2=68%, P=0.04). Analysis using a random-effects model noted that there were no significant differences between the two intervention groups (OR 1.67; 95% CI 0.69; 4.05, P=0.26; Figure 4).
  | Figure 4 Forest plot showing the number of serious adverse events. |
Elevation of intraocular pressure (IOP)
All cases demonstrated increased IOP after injection of corticosteroid or ranibizumab. The pooled results demonstrated a significant difference between corticosteroid and ranibizumab treatment (OR 6.88; 95% CI 4.53–10.44; P<0.00001) without heterogeneity (P=0.66, I2=0%; Figure 5).
  | Figure 5 Forest plot showing the elevation of intraocular pressure. |
Adverse events: cataract
A significant difference was found between the corticosteroid and ranibizumab groups (OR 3.98; 95% CI 1.89–8.37; P=0.0003) without heterogeneity (P=0.99, I2=0%; Figure 6).
  | Figure 6 Forest plot showing the adverse events: cataract. |
Mean number of intravitreal injections
In all studies presented, more injections were required in the ranibizumab group. In addition, the mean number of intravitreal injections between corticosteroid and ranibizumab treatment is summarized in Table 2.
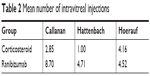  | Table 2 Mean number of intravitreal injections |
Heterogeneity, sensitivity analysis, and publication bias
A sensitivity analysis was conducted to assess the stability of the results by sequential removal of individual studies. When the analysis result was high heterogeneity, we used a random-effects model. These sensitivity analyses indicated that our conclusions were generally robust. Funnel plots and Egger’s test were not used, because there were less than ten studies for each comparison.
Discussion
In this meta-analysis, we assessed three RCTs including 875 patients (433 patients in the corticosteroid group and 442 patients in the ranibizumab group). We found that both corticosteroid implant and ranibizumab could achieve significant functional and anatomical improvement during early treatment. Furthermore, the ranibizumab group revealed significant improvement compared with the corticosteroid implant group in BCVA. The CST results showed that the ranibizumab implant treatment group had higher CST reduction compared with the corticosteroid treatment group. The BCVA and CST results are in accordance with previous studies, which have proven the clinical efficacy of ranibizumab and corticosteroids for RVO therapy.29,32,33
Although ranibizumab has good treatment effects, repeated injections carry increased risks, such as infectious endophthalmitis, intraocular inflammation, and even stroke or myocardial infarction.34 In this meta-analysis, we noted that the average number of treatments in the corticosteroid group was lower than that in the ranibizumab group. Therefore, ranibizumab treatment may not be a good therapy for all patients. Corticosteroids have been shown to have a complex effect on diseases; in addition to reduce the expression of VEGF and leukostasis, reducing inflammatory cytokines may be another function for these treatment-naive patients.35 Diabetics are found to have higher concentrations of pro-inflammatory cytokines, such as tumor necrosis factor α (TNF-α), IL-1β, IL-6, and intracellular adhesion molecule (ICAM)-1. These cytokines induce persistent chronic inflammation in the retina, which leads to leukostasis, increased vascular permeability, and dysfunction of the BRB.36,37 In addition, according to a study, diabetic macular edema (DME) was shown to be related to elevated cytokines in the aqueous or vitreous humor, such as TGF-β, IL-6, ICAM-1, and monocyte chemotactic protein 1 (MCP-1).38 Therefore, treatment principally aims to deal with the effect of these two pathogenic pathways. Intravitreal corticosteroids have also been approved for ME secondary to RVO based on the results of the GENEVA study in which one injection of corticosteroid implant showed significant improvement in visual acuity compared with sham injections in patients with RVO10 with a favorable efficacy and safety profile over 12 months as well.11 Above all, corticosteroid implant could be a better alternative for ME.
Intravitreally administered steroids stabilize visual acuity and reduce ME, but they are also frequently associated with side effects, and the most common side effects are an increase in IOP and progression of cataract. In our study, we found that the most common adverse event was cataract progression, which was higher in the corticosteroid group than in the ranibizumab group, as well as the treatment-related IOP increase in the steroid group. In addition, no new safety risks with either ranibizumab or steroid implant were observed. This observation was consistent with the GENEVA study.11 This finding suggests that doctors should keep an eye out when using steroid implants in patients with high IOP or in young patients with a clear lens.
Because of the abovementioned reasons, corticosteroids are generally used in patients affected by persistent or refractory ME as an alternative to switch between ranibizumab drugs.39,40 Corticosteroids may be recommended as a first choice for the following cases: 1) pseudophakic eyes; 2) patients who are under consideration for cataract surgery in the near future; 3) anti-VEGF-resistant patients;41 4) patients who do not have a higher IOP risk; 5) patients who are reluctant to receive frequent injections during treatment; 6) patients who have a history of cardiovascular and cerebrovascular diseases;41 and 7) patients who have a post-vitrectomy treatment.42 In all of these cases, the IOP should be monitored frequently. Therefore, the corticosteroid implant may not be recommended as a first-line therapy for ME. Corticosteroid implant as a first-line treatment should allow a definite conclusion about whether switching to corticosteroid implant is more beneficial than ranibizumab treatment. We also hope that new treatments are investigated to optimize clinical efficacy and to reduce side effects in the future. This meta-analysis may provide a basis for the clinical treatment of ME.
This meta-analysis had some limitations. At first, we only included three studies assessing a total of 850 eyes. Furthermore, the differences in the durations of the included trials (6 or 12 months) were a potential source of heterogeneity. Finally, the injection interval of dexamethasone in one study16 was not the same as other included studies,17,18 which may have been heterogeneous. To reinforce the validity of our meta-analysis, clinical trials comparing the different corticosteroid agents with the ranibizumab implant as well as extended follow-up trials should be conducted in the future.
Conclusion
This meta-analysis of data revealed that the ranibizumab group had a significant improvement in vision and had higher CST reduction compared with the corticosteroid implant group. The corticosteroid implant group also had more ocular adverse events, but it required fewer injections.
Acknowledgments
This work was supported by the National Natural Science Foundation of China in 2014 (project number: 81470648) and the Fundamental Research Funds for the Central Universities. The authors thank the researchers of the original studies included in this meta-analysis. The authors also thank American Journal Experts for English language editing.
Disclosure
The authors report no conflicts of interest in this work.
References
Ferris FL, Patz A. Macular edema. A complication of diabetic retinopathy. Surv Ophthalmol. 1984;28 Suppl:452–461. | ||
Patz A, Schatz H, Berkow JW, Gittelsohn AM, Ticho U. Macular edema-an overlooked complication of diabetic retinopathy. Trans Am Acad Ophthalmol Otolaryngol. 1973;77(1):OP34–OP42. | ||
Klein R, Klein BE, Moss SE, Cruickshanks KJ. The Wisconsin Epidemiologic Study of Diabetic Retinopathy. XV. The long-term incidence of macular edema. Ophthalmology. 1995;102(1):7–16. | ||
Antonetti DA, Barber AJ, Khin S, Lieth E, Tarbell JM, Gardner TW. Vascular permeability in experimental diabetes is associated with reduced endothelial occludin content: vascular endothelial growth factor decreases occludin in retinal endothelial cells. Penn State Retina Research Group. Diabetes. 1998;47(12):1953–1959. | ||
Chatziralli I, Nicholson L, Sivaprasad S, Hykin P. Intravitreal steroid and anti-vascular endothelial growth agents for the management of retinal vein occlusion: evidence from randomized trials. Expert Opin Biol Ther. 2015;15(12):1685–1697. | ||
Campochiaro PA, Brown DM, Awh CC, et al. Sustained benefits from ranibizumab for macular edema following central retinal vein occlusion: twelve-month outcomes of a phase III study. Ophthalmology. 2011;118(10):2041–2049. | ||
Heier JS, Campochiaro PA, Yau L, et al. Ranibizumab for macular edema due to retinal vein occlusions: long-term follow-up in the HORIZON trial. Ophthalmology. 2012;119(4):802–809. | ||
Campochiaro PA, Sophie R, Pearlman J, et al. Long-term outcomes in patients with retinal vein occlusion treated with ranibizumab: the RETAIN study. Ophthalmology. 2014;121(1):209–219. | ||
Farinha C, Marques JP, Almeida E, et al. Treatment of Retinal Vein Occlusion with Ranibizumab in Clinical Practice: Longer-Term Results and Predictive Factors of Functional Outcome. Ophthalmic Res. 2015;55(1):10–18. | ||
Haller JA, Bandello F, Belfort R, et al. Randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with macular edema due to retinal vein occlusion. Ophthalmology. 2010;117(6):1134–1146. | ||
Haller JA, Bandello F, Belfort R, et al. Dexamethasone intravitreal implant in patients with macular edema related to branch or central retinal vein occlusion twelve-month study results. Ophthalmology. 2011;118(12):2453–2460. | ||
Maggio E, Polito A, Guerriero M, Pertile G. Intravitreal dexamethasone implant for macular edema secondary to retinal vein occlusion: 12-month follow-up and prognostic factors. Ophthalmologica. 2014;232(4):207–215. | ||
Ferrini W, Ambresin A. Intravitreal dexamethasone implant for the treatment of macular edema after retinal vein occlusion in a clinical setting. Klin Monbl Augenheilkd. 2013;230(4):423–426. | ||
Bezatis A, Spital G, Höhn F, et al. Functional and anatomical results after a single intravitreal Ozurdex injection in retinal vein occlusion: a 6-month follow-up – the SOLO study. Acta Ophthalmol. 2013;91(5):e340–e347. | ||
Chatziralli I, Theodossiadis G, Kabanarou SA, et al. Ranibizumab versus dexamethasone implant for central retinal vein occlusion: the RANIDEX study. Graefes Arch Clin Exp Ophthalmol. 2017;255(10):1899–1905. | ||
Callanan DG, Loewenstein A, Patel SS, et al. A multicenter, 12-month randomized study comparing dexamethasone intravitreal implant with ranibizumab in patients with diabetic macular edema. Graefes Arch Clin Exp Ophthalmol. 2017;255(3):463–473. | ||
Hattenbach LO, Feltgen N, Bertelmann T, et al. Head-to-head comparison of ranibizumab PRN versus single-dose dexamethasone for branch retinal vein occlusion (COMRADE-B). Acta Ophthalmol. 2018;96(1):e10–e18. | ||
Hoerauf H, Feltgen N, Weiss C, et al. Clinical Efficacy and Safety of Ranibizumab Versus Dexamethasone for Central Retinal Vein Occlusion (COMRADE C): A European Label Study. Am J Ophthalmol. 2016;169:258–267. | ||
Karst SG, Lammer J, Mitsch C, et al. Detailed analysis of retinal morphology in patients with diabetic macular edema (DME) randomized to ranibizumab or triamcinolone treatment. Graefes Arch Clin Exp Ophthalmol. 2018;256(1):49–58. | ||
Kiss S, Liu Y, Brown J, et al. Clinical utilization of anti-vascular endothelial growth-factor agents and patient monitoring in retinal vein occlusion and diabetic macular edema. Clin Ophthalmol. 2014;8:1611–1621. | ||
Nguyen QD, Brown DM, Marcus DM, et al. Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology. 2012;119(4):789–801. | ||
Mitchell P, Bandello F, Schmidt-Erfurth U, et al. The RESTORE study: ranibizumab monotherapy or combined with laser versus laser monotherapy for diabetic macular edema. Ophthalmology. 2011;118(4):615–625. | ||
Massin P, Bandello F, Garweg JG, et al. Safety and efficacy of ranibizumab in diabetic macular edema (RESOLVE Study): a 12-month, randomized, controlled, double-masked, multicenter phase II study. Diabetes Care. 2010;33(11):2399–2405. | ||
Cabrera M, Yeh S, Albini TA. Sustained-Release Corticosteroid Options. J Ophthalmol. 2014;2014(3):1–5. | ||
Kuppermann BD, Blumenkranz MS, Haller JA, et al. Randomized controlled study of an intravitreous dexamethasone drug delivery system in patients with persistent macular edema. Arch Ophthalmol. 2007;125(3):309–317. | ||
Boyer DS, Yoon YH, Belfort R, et al. Three-year, randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with diabetic macular edema. Ophthalmology. 2014;121(10):1904–1914. | ||
Campochiaro PA, Brown DM, Pearson A, et al. Long-term benefit of sustained-delivery fluocinolone acetonide vitreous inserts for diabetic macular edema. Ophthalmology. 2011;118(4):626–635. | ||
Chang-Lin JE, Attar M, Acheampong AA, et al. Pharmacokinetics and pharmacodynamics of a sustained-release dexamethasone intravitreal implant. Invest Ophthalmol Vis Sci. 2011;52(1):80–86. | ||
Yumusak E, Buyuktortop N, Ornek K. Early results of dexamethasone implant, ranibizumab, and triamcinolone in macular edema due to branch retinal vein occlusion. Eur J Ophthalmol. 2016;26(1):54–59. | ||
Comyn O, Lightman SL, Hykin PG. Corticosteroid intravitreal implants vs. ranibizumab for the treatment of vitreoretinal disease. Curr Opin Ophthalmol. 2013;24(3):248–254. | ||
Jadad AR, Moore RA, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17(1):1–12. | ||
Thom HH, Capkun G, Nixon RM, Ferreira A. Indirect comparisons of ranibizumab and dexamethasone in macular oedema secondary to retinal vein occlusion. BMC Med Res Methodol. 2014;14:140. | ||
Gu X, Yu X, Song S, Dai H. Intravitreal dexamethasone implant versus intravitreal ranibizumab for the treatment of macular edema secondary to retinal vein occlusion in a Chinese population. Ophthalmic Res. 2017;58(1):8–14. | ||
Wells JA, Glassman AR, Ayala AR, et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema: two-year results from a comparative effectiveness randomized clinical trial. Ophthalmology. 2016;123(6):1351–1359. | ||
Tamura H, Miyamoto K, Kiryu J, et al. Intravitreal injection of corticosteroid attenuates leukostasis and vascular leakage in experimental diabetic retina. Invest Ophthalmol Vis Sci. 2005;46(4):1440–1444. | ||
Tang J, Kern TS. Inflammation in diabetic retinopathy. Prog Retin Eye Res. 2011;30(5):343–358. | ||
Adamis AP, Berman AJ. Immunological mechanisms in the pathogenesis of diabetic retinopathy. Semin Immunopathol. 2008;30(2):65–84. | ||
Jonas JB, Jonas RA, Neumaier M, Findeisen P. Cytokine concentration in aqueous humor of eyes with diabetic macular edema. Retina. 2012;32(10):2150–2157. | ||
Lazic R, Lukic M, Boras I, et al. Treatment of anti-vascular endothelial growth factor-resistant diabetic macular edema with dexamethasone intravitreal implant. Retina. 2014;34(4):719–724. | ||
Thomas BJ, Yonekawa Y, Wolfe JD, Hassan TS. Contralateral eye-to-eye comparison of intravitreal ranibizumab and a sustained-release dexamethasone intravitreal implant in recalcitrant diabetic macular edema. Clin Ophthalmol. 2016;10:1679–1684. | ||
Schmidt-Erfurth U, Garcia-Arumi J, Bandello F, et al. Guidelines for the management of diabetic macular edema by the European Society of Retina Specialists (EURETINA). Ophthalmologica. 2017;237(4):185–222. | ||
Boyer DS, Faber D, Gupta S, et al. Dexamethasone intravitreal implant for treatment of diabetic macular edema in vitrectomized patients. Retina. 2011;31(5):915–923. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

