Back to Journals » Clinical Interventions in Aging » Volume 17
Intraoperative Glycemic Variability and Mean Glucose are Predictors for Postoperative Delirium After Cardiac Surgery: A Retrospective Cohort Study
Authors Choi H , Park CS, Huh J, Koo J , Jeon J, Kim E, Jung S, Kim HW, Lim JY, Hwang W
Received 10 September 2021
Accepted for publication 20 December 2021
Published 5 February 2022 Volume 2022:17 Pages 79—95
DOI https://doi.org/10.2147/CIA.S338712
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 6
Editor who approved publication: Prof. Dr. Nandu Goswami
Hoon Choi,1 Chul Soo Park,1 Jaewon Huh,1 Jungmin Koo,1 Joonpyo Jeon,2 Eunsung Kim,1 Sangmin Jung,1 Hwan Wook Kim,3 Ju Yong Lim,3 Wonjung Hwang1
1Department of Anesthesia and Pain Medicine, Seoul St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea; 2Department of Anesthesia and Pain Medicine, Eunpyeong St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea; 3Department of Thoracic and Cardiovascular Surgery, Seoul St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea
Correspondence: Wonjung Hwang
Department of Anesthesia and Pain Medicine, Seoul St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, 222, Banpo-daero, Seocho-gu, Seoul, 06591, Republic of Korea
, Tel +82-2-22586162
, Fax +82-2-5371951
, Email [email protected]
Purpose: Postoperative delirium (POD) is a common but serious complication after cardiac surgery and is associated with various short- and long-term outcomes. In this study, we investigated the effects of intraoperative glycemic variability (GV) and other glycemic variables on POD after cardiac surgery.
Patients and Methods: A retrospective single-center cohort analysis was conducted using data from electronic medical record from 2018 to 2020. A total of 705 patients undergoing coronary artery bypass graft surgery and/or valve surgery, and/or aortic replacement surgery were included in the analysis. Intraoperative GV was assessed with a coefficient of variation (CV), which was defined as the standard deviation of five intraoperative blood glucose measurements divided by the mean. POD assessment was performed three times a day in the ICU and twice a day in the ward until discharge by trained medical staff. POD was diagnosed if any of the Confusion Assessment Method for the Intensive Care Unit was positive in the ICU, and the Confusion Assessment Method was positive in the ward. Multivariable logistic regression was used to identify associations between intraoperative GV and POD.
Results: POD occurred in 306 (43.4%) patients. When intraoperative glycemic CV was compared as a continuous variable, the delirium group had higher intraoperative glycemic CV than the non-delirium group (22.59 [17.09, 29.68] vs 18.19 [13.00, 23.35], p < 0.001), and when intraoperative glycemic CV was classified as quartiles, the incidence of POD increased as intraoperative glycemic CV quartiles increased (first quartile 29.89%; second quartile 36.67%; third quartile 44.63%; and fourth quartile 62.64%, p < 0.001). In the multivariable logistic regression model, patients in the third quartile of intraoperative glycemic CV were 1.833 times (OR 1.833, 95% CI: 1.132– 2.967, p = 0.014), and patients in the fourth quartile of intraoperative glycemic CV were 3.645 times (OR 3.645, 95% CI: 2.235– 5.944, p < 0.001) more likely to develop POD than those in the first quartile of intraoperative glycemic CV.
Conclusion: Intraoperative blood glucose fluctuation, manifested by intraoperative GV, is associated with POD after cardiac surgery. Patients with a higher intraoperative GV have an increased risk of POD.
Keywords: postoperative delirium, cardiac surgery, glycemic variability, coefficient of variation
Introduction
Postoperative delirium (POD) is an acute and fluctuating alteration of the mental state that occurs after surgery, characterized by an altered consciousness with reduced ability to focus, sustain, or shift attention.1 The incidence of POD after cardiac surgery ranges from 4.1% to 54.9% when validated assessment tools are used.2,3 Despite efforts on prevention and treatment, POD remains one of the most common complications after cardiac surgery.4 POD after cardiac surgery is associated with both short-term and long-term consequences, including increased anxiety, depression, cognitive decline, length of stay in an intensive care unit (ICU) and hospital, in-hospital complications, and mortality.5–7
Extensive research has been conducted in this field considering the high incidence and adverse outcomes of POD after cardiac surgery. The prevention and identification of the risk factors for POD are of utmost importance because therapeutic interventions for POD have little effect on outcomes once they are developed.8 A recent meta-analysis suggested several risk factors for POD, including age, diabetes, preoperative depression, mild cognitive impairment, carotid artery stenosis, New York Heart Association (NYHA) functional class III or IV, duration of mechanical ventilation, and length of ICU stay.2 However, these risk factors are either preoperatively (age and underlying conditions) and/or postoperatively (duration of mechanical ventilation and length of ICU stay) non-modifiable factors. Therefore, these risk factors are of little value in preventing POD. Identifying modifiable risk factors that occur before the development of POD (preoperative and intraoperative, not postoperative) should be paramount in the management of POD.8–11 For example, risk factors such as preoperative physical activity measured by Life Space Assessment, are likely to be modified before surgery.12
Although the exact mechanism of POD is not well understood and is considered multifactorial, blood glucose has been proposed to play a key role in predisposing or directly causing POD.13 Moreover, glucose tolerance is often impaired during cardiac surgery due to the stress response associated with cardiopulmonary bypass pump (CPB), as well as from insulin resistance induced by hypothermia.14 Glucose homeostasis and inflammation are interlinked,15,16 and neuroinflammatory response as a result of systemic inflammation is thought to be a crucial mechanism in the development of POD.17 Preoperative hemoglobin A1c (HbA1c), perioperative hyperglycemia, and hypoglycemia are all associated with POD after cardiac surgery.18–20 Recently, glycemic variability (GV) has been suggested as a novel measure of glycemic control.21 GV is a measure of fluctuations in blood glucose levels.22 Increased GV is known to cause more harm than chronic hyperglycemia.23 In cardiac surgery, increased postoperative GV was associated with major adverse events, including postoperative cardiac arrest, myocardial infarction, pneumonia, renal failure, stroke, sepsis, deep sternal infection, reoperation, and 30-day mortality.24,25 However, studies on the association between GV and POD in cardiac surgery are rare. Although one study examined the association between postoperative GV and POD in patients with acute aortic dissection,26 no studies have been conducted on intraoperative GV, the period during which glucose tolerance is altered the most.
The current study aimed to determine the effect of intraoperative GV on POD after cardiac surgery in mixed cardiac surgery patients. The effects of other intraoperative glycemic variables, including intraoperative mean glucose, were also investigated in relation to the development of POD. We hypothesized that increased intraoperative GV would be associated with POD after cardiac surgery.
Materials and Methods
Study Population
This study was a retrospective single-center cohort analysis. The study protocol was approved by the Institutional Review Board (IRB) of Seoul St. Mary’s Hospital, the Catholic University of Korea (approval number: KC21RISI0488, approval date: July 9, 2021). The requirement for written informed consent was waived due to the retrospective nature of the study and the lack of interaction with the patients. After obtaining approval from the IRB, we evaluated patients aged over 18 years who underwent coronary artery bypass graft surgery (CABG) and/or valve surgery, and/or aortic replacement surgery at Seoul St. Mary’s Hospital, the Catholic University of Korea between January 2018 and December 2020 from our electronic medical records. The following patients were excluded: 1) patients who underwent off-pump CABG, 2) patients with altered mental status before the surgery (Glasgow Coma Scale less than 13), 3) patients who remained in a coma and/or died within 24 h after the surgery, and 4) patients with incomplete intraoperative glucose measurements. Patients who underwent off-pump CABG were excluded because the use of CPB was associated with profound alterations in glucose tolerance and cerebral perfusion. Patients with altered mental status before surgery and those who remained in a coma and/or died within 24 h after the surgery were excluded because the assessment of POD was not possible. The study was conducted in accordance with the Declaration of Helsinki. The study was reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.27
Delirium Assessment
The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, fifth edition (DSM-5), lists five key features that characterize delirium: a disturbance in attention and awareness; a disturbance that develops over a short period, represents a change from baseline, and tends to fluctuate during the course of the day; an additional disturbance in cognition; a disturbance that is not better explained by another preexisting, evolving, or established neurocognitive disorder, and does not occur in the context of a severely reduced level of arousal, such as coma; and evidence from the history, physical examination, or laboratory findings that the disturbance is caused by a medical condition, substance intoxication or withdrawal, or medication side effects.28 POD assessment was performed in every cardiac surgery patient as part of our institutional cardiac surgery and anesthesia protocol. It was performed three times a day in the ICU and twice a day in the ward until discharge by trained medical staff. POD was diagnosed if any of the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU) was positive in the ICU, and the Confusion Assessment Method (CAM) was positive in the ward.29,30 We considered POD occurring only within the first 24 h as positive.
Measurement of Glycemic Variables and Glycemic Control Protocol
Preoperative HbA1c and fasting serum glucose values were collected within a day before surgery. Intraoperative glycemic management was standardized as part of our institutional cardiac surgery and anesthesia protocol during the study period.14 Intraoperative blood glucose was measured using a point-of-care blood gas analyzer (ABL800 FLEX blood gas analyzer, Radiometer Medical ApS, Brønshøj, Denmark) with arterial blood. It was measured at a minimum of five defined periods: 1) at the induction of anesthesia, 2) 5 min before the initiation of CPB, 3) 30 min after the initiation of CPB, 4) 5 minutes after the termination of CPB, and 5) at the end of the surgery. If multiple blood glucose measurements were performed between the defined periods, blood glucose measurements were averaged into a single value. Five different intraoperative glycemic variables were calculated. Intraoperative mean glucose was the average of five intraoperative blood glucose measurements, and intraoperative peak glucose was the highest of the five. A hyperglycemic episode was defined as any intraoperative blood glucose level > 200 mg/dL, and a hypoglycemic episode was defined as any intraoperative blood glucose level < 60 mg/dL. Intraoperative GV was assessed with a coefficient of variation (CV), which was defined as the standard deviation of five intraoperative blood glucose measurements divided by the mean.21,22,31
Blood glucose levels were targeted below 180 mg/dL throughout the surgery.14 When blood glucose exceeded 180 mg/dL, four units of regular insulin were administered intravenously for blood glucose between 180 and 200 mg/dL, and eight units of regular insulin were administered intravenously for blood glucose between 200 and 250 mg/dL. When blood glucose exceeded 250 mg/dL or remained above 180 mg/dL after two boluses of regular insulin, continuous infusion of regular insulin was commenced at a rate of 5 units/h and was adjusted as follows: 1. blood glucose < 120 mg/dL, stop insulin, recheck in 30 min, and when > 150 mg/dL, restart with rate 50% of previous rate; 2. blood glucose 120–180 mg/dL, no action; 3. blood glucose 180–200 mg/dL, bolus 3 units and infusion 3 units/h; 4. blood glucose 200–225 mg/dL, bolus 4 units and infusion 4 units/h; 5. blood glucose 225–250, bolus 5 units and infusion 5 units/h; and 6. blood glucose > 250 mg/dL, bolus dose (blood glucose/50) and infusion 6 units/h. When blood glucose fell below 70 mg/dL, 20 mL of 50% dextrose in water was administered. Blood glucose was re-measured 30 min after any intervention. No glucose-containing solutions were used throughout the surgery.
Anesthesia and Surgery
Standard anesthesia and surgical protocols were used during the study period. Anesthesia was induced with intravenous propofol, etomidate, rocuronium, and remifentanil. Anesthesia was maintained with sevoflurane, and a continuous infusion of dexmedetomidine and remifentanil to maintain a bispectral index (BIS; A-2000TM SP, Aspect Medical Systems, Norwood, MA, USA) value between 40 and 60. Skeletal muscle relaxation was maintained with a continuous infusion of rocuronium. No glucocorticoids were used throughout the surgery.
All surgeries were performed via a median sternotomy. Anticoagulation was achieved with the administration of 300 IU/kg of heparin to target an activated clotting time of more than 480 s. A non-pulsatile CPB was used with standard hypothermic and hyperkalemic cardioplegia. Alpha-stat pH management was used during CPB. Mild hypothermia (32–35 °C) or deep hypothermia (< 28 °C) was used depending on the type of surgery and the need for circulatory arrest. The mean arterial pressure was targeted at 50–80 mmHg. The arterial partial pressure of oxygen was maintained at 150–250 mmHg, and alpha-stat management of pH was used to maintain partial pressure of carbon dioxide at 35–45 mmHg. After the termination of CPB, intravenous protamine sulfate was administered to reverse the effects of heparin. Immediately after the surgery, the patients were transferred to the ICU being intubated. Sedation with a continuous infusion of dexmedetomidine and remifentanil was continued until the patient was extubated. The patients were extubated after confirming successful ventilator weaning and hemodynamic stability.
Data Collection
After obtaining approval from the IRB, we collected baseline characteristics and perioperative findings associated with POD after cardiac surgery.2,32–35 Baseline characteristics were as follows: age, sex, body mass index (BMI), history of smoking, history of alcohol abuse, and final education level of the patient.
Preoperative variables were the following: hypertension, diabetes, dyslipidemia, chronic kidney disease (dialysis-dependent), chronic lung disease (FEV1 < 75%, or on chronic inhaled or oral bronchodilator therapy), liver disease (history of hepatitis B, hepatitis C, drug-induced hepatitis, autoimmune hepatitis, cirrhosis, portal hypertension, esophageal varix, liver transplant, or congestive hepatopathy), peripheral vascular disease, cerebrovascular disease (history of stroke, transient ischemic accident, or ≥ 50% stenosis of any of the major extracranial or intracranial vessels to the brain), neuropsychiatric disease (major depressive disorder, bipolar disorder, schizophrenia, or cognitive impairment), immunocompromised-state, endocarditis, history of atrial fibrillation, type of hypoglycemic agents, delirium-related medication use, polypharmacy, history of prior cardiac surgery, emergency, NYHA classification, European System for Cardiac Operative Risk Evaluation (EuroSCORE) II, ejection fraction on preoperative echocardiogram, and laboratory findings including hematocrit, glomerular filtration rate (GFR), albumin, C-reactive protein (CRP) and sodium disturbance. Delirium-related medication use was defined as the use of any medication known to cause delirium according to the American Geriatric Society Beers Criteria.36 Polypharmacy was defined as the daily use of five or more medications.37 Emergency was defined as operation before the beginning of the next working day after the decision to operate. Sodium disturbance was defined as an abnormal serum sodium level (< 136 mmol/L or > 145 mmol/L).
Intraoperative variables were the following: type of surgery, duration of surgery, duration of CPB, use of total circulatory arrest (TCA), total fluid administration, packed red blood cell (PRC) transfusion, urine output, mean BIS value, mean cerebral oximeter value, highest lactic acid value, and vasoactive-inotropic score (VIS)38 at the end of the surgery. The type of surgery was categorized into three: 1) isolated CABG, 2) valve surgery (isolated valve surgery and CABG combined with valve surgery), and 3) aortic surgery (isolated aortic surgery, CABG, and/or valve surgery combined with aortic surgery). These were categorized by the need for an open chamber procedure (ie valve surgery) and arterial cannulation at the axillary artery (ie surgery on the ascending aorta) because they have the potential for embolic events and/or compromised cerebral perfusion.4 The mean BIS and cerebral oximeter values were defined as the average of all intraoperative values, which were recorded every 15 min during surgery.
Additional postoperative outcome variables were the duration of mechanical ventilation, duration of ICU stay, duration of hospital stay, major adverse events (MAE), and 30-day mortality. MAE was defined as a composite outcome of mortality, permanent stroke, pneumonia, renal failure, prolonged ventilation, deep sternal wound infection, and reoperation.
Statistical Analysis
The patients were divided into delirium and non-delirium groups. Continuous variables were tested for normality using the Kolmogorov–Smirnov test and Q-Q plot, and none of the continuous variables were normally distributed. Continuous variables of the two groups were compared using the Mann–Whitney U-test and are presented as median (IQR). Categorical variables of the two groups were compared using Pearson chi-square or Fisher’s exact test, as appropriate, and are presented as the number of patients (percentage).
To assess the association between intraoperative glycemic variables and POD after cardiac surgery, multivariable logistic regression analysis was performed. Separate models were constructed for two different intraoperative glycemic variables: intraoperative glycemic CV and intraoperative mean glucose. Preoperative and intraoperative variables with a p-value < 0.1 on univariable analysis were considered significant predictors for multivariable analysis. All continuous variables, except intraoperative glycemic CV, were dichotomized by the median or appropriate clinical cut-off; intraoperative glycemic CV was classified into quartiles (first quartile < 15%; second quartile 15–20%; third quartile 20–25%; fourth quartile > 25%). Multivariable logistic regression models were constructed by selecting variables kept in a backward stepwise selection. Multicollinearity was assessed using the variance inflation factor (VIF), and all predictors were less than 10. The variables that ended up in the models were age, final education level, GFR, type of surgery, duration of CPB, mean BIS value and VIS at the end of the surgery. The model fit was evaluated using the Hosmer-Lemeshow test. The predictive accuracy of the final models after multivariable analysis was assessed using the area under the receiver operator characteristic (AUROC) statistics and Brier score. Delong’s test was used to compare the models. Further analysis was performed adjusting for three preoperative variables that might affect intraoperative blood glucose levels: diabetes, preoperative HbA1c, and the type of hypoglycemic agents. Three models were constructed for each of the three variables forced into the model, and another model was constructed forcing all three factors into the model. A subgroup analysis was performed classifying patients according to the type of surgery: isolated CABG, valve surgery, and aortic surgery. Statistical significance was set at p < 0.05. Statistical analysis was performed using SPSS (version 25.0; IBM SPSS Inc., Chicago, IL, USA) and MedCalc (version 19.5; MedCalc Software Ltd., Ostend, Belgium).
Results
During the study period, 782 patients underwent CABG, valve surgery, and/or aortic replacement surgery. In total, the excluded patients were 77, and 12 of them were excluded due to incomplete intraoperative glucose measurements. A total of 705 patients were included in the analysis. A flow diagram of the patients included in this study is shown in Figure 1.
 |
Figure 1 Flow diagram of patients included in the study. |
The baseline characteristics of the patients are shown in Table 1. Of the 705 patients included in the analysis, 306 (43.4%) were diagnosed with POD after cardiac surgery. The delirium group had a higher age and BMI than the non-delirium group. Female patients and patients with a final education below high school were more likely to develop POD.
 |
Table 1 Baseline Characteristics of the Patients |
A comparison of preoperative variables between the two groups is shown in Table 2. Patients with hypertension and a history of atrial fibrillation were more likely to develop POD. The incidence of diabetes did not differ between the two groups. Variables regarding the patients’ medications did not differ between the two groups. Patients who underwent emergency surgery and were classified as NYHA classification III or IV were more likely to develop POD. The delirium group had higher EuroSCORE II and lower hematocrit, GFR, albumin, and CRP values than the non-delirium group. Patients with sodium disturbance were more likely to develop POD.
 |
Table 2 Comparison of Preoperative Variables Between the Delirium Group and the Non-Delirium Group |
A comparison of intraoperative variables between the two groups is shown in Table 3. Patients who underwent aortic surgery showed the highest incidence of POD, whereas patients who underwent isolated CABG showed the lowest incidence of POD. The delirium group received a longer duration of surgery and CPB and were more likely to undergo TCA than the non-delirium group. The delirium group received more PRC transfusions and had less urine output than the non-delirium group. In addition, the delirium group had smaller mean BIS and cerebral oximeter values than the non-delirium group. Patients with a higher lactic acid value and VIS at end of surgery were more likely to develop POD.
 |
Table 3 Comparison of Intraoperative Variables Between the Delirium Group and the Non-Delirium Group |
Preoperative and intraoperative glycemic variables were the main predictors of interest. The median preoperative HbA1c level was 5.8%, and the median glucose level at admission was 103 mg/dL. The median values of intraoperative mean glucose and peak glucose were 134 mg/dL and 167 mg/dL, respectively. The average blood glucose values at five intraoperative defined periods are shown in Figure 2. The percentage of patients with hyperglycemic episodes was 27.09%, and the median intraoperative glycemic CV was 19.97%.
Intergroup comparisons of preoperative and intraoperative glycemic variables are shown in Table 4. The preoperative HbA1c and glucose values at admission did not differ between the two groups. The delirium group had higher intraoperative mean glucose and peak glucose than the non-delirium group (142 [126, 163] vs 130 [119, 146], p < 0.001, and 186 [155, 219] vs 158 [141, 189], p < 0.001, respectively). Patients with intraoperative hyperglycemic episodes were more likely to develop POD (38.24% vs 18.55%, p < 0.001). When intraoperative glycemic CV was compared as a continuous variable, the delirium group had higher intraoperative glycemic CV than the non-delirium group (22.59 [17.09, 29.68] vs 18.19 [13.00, 23.35], p < 0.001), and when intraoperative glycemic CV was classified as quartiles, the incidence of POD increased as intraoperative glycemic CV quartiles increased (first quartile 29.89%; second quartile 36.67%; third quartile 44.63%; and fourth quartile 62.64%, p < 0.001).
 |
Table 4 Intergroup Comparison of Preoperative and Intraoperative Glycemic Variables |
The two separate models constructed with two different intraoperative glycemic variables (intraoperative glycemic CV and mean glucose) were statistically significant (p < 0.001 for both models). The Hosmer-Lemeshow test was not statistically significant in either model (intraoperative glycemic CV, p = 0.368; mean glucose, p = 0.256). Eight independent variables were associated with POD after cardiac surgery in the intraoperative glycemic CV model. Patients in the third quartile of intraoperative glycemic CV had 1.833 times (OR 1.833, 95% CI: 1.132–2.967, p = 0.014), and patients in the fourth quartile of intraoperative glycemic CV had 3.645 times (OR 3.645, 95% CI: 2.235–5.944, p < 0.001) higher odds of POD than patients in the first quartile of intraoperative glycemic CV after adjusting for age, education level, GFR, type of surgery, duration of CPB, mean BIS, and mean VIS at the end of the surgery (Table 5). A summary of the two models is presented in Table 6. The model with intraoperative glycemic CV explained 27.2% (NagelKerke R2) of the variance in POD and correctly classified 70.2% of cases. The sensitivity and specificity were 60.5% and 77.7%, respectively. The AUROC curve statistics for the two models were both statistically significant (p < 0.001 for both models). In the model with intraoperative glycemic CV, the AUROC curve statistic was not different from the model with intraoperative mean glucose, (0.766, 95% CI: 0.732–0.796 vs 0.758, 95% CI: 0.725–0.789, respectively; Figure 3), and the Brier score was not different from the model with intraoperative mean glucose (0.194 vs 0.198, respectively). However, the differences were not statistically significant (p = 0.507).
 |
Table 5 Logistic Regression Models for Delirium After Cardiac Surgery |
 |
Table 6 Model Summary |
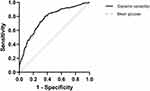 |
Figure 3 The receiver operator characteristic curves of the models with intraoperative glycemic variability and mean glucose. |
Further analysis of three preoperative variables that might affect intraoperative blood glucose levels showed that patients in the third quartile of intraoperative glycemic CV had 1.837 times (OR 1.837, 95% CI: 1.134–2.976, p = 0.013), and patients in the fourth quartile of intraoperative glycemic CV had 3.666 times (OR 3.666, 95% CI: 2.247–5.980, p < 0.001) higher odds of POD than patients in the first quartile of intraoperative glycemic CV after adjusting for diabetes and the seven variables in the previous model. After adjusting for preoperative HbA1c, patients in the third and fourth quartile had 1.832 times and 3.625 times higher odds of POD than the first quartile (OR 1.832, 95% CI: 1.131–2.967, p = 0.014 and OR 3.625, 95% CI: 2.223–5.911, p < 0.001, respectively). After adjusting for the type of hypoglycemic agents used, patients in the third and fourth quartile had 1.827 times and 3.653 times higher odds of POD than the first quartile (OR 1.827, 95% CI: 1.127–2.960, p = 0.014 and OR 3.653, 95% CI: 2.239–5.960, p < 0.001, respectively). The model adjusted for all previous seven statistically significant variables and three additional variables showed that patients in the third quartile and fourth quartile had 1.829 times and 3.643 times higher odds of POD than the first quartile (OR 1.829, 95% CI: 1.128–2.966, p = 0.014 and OR 3.643, 95% CI: 2.232–5.946, p < 0.001, respectively). Detailed results are presented in Supplement Table 1.
The subgroup analysis of the patients according to the type of surgery (isolated CABG, valve surgery, and aortic surgery) showed that the baseline characteristics, preoperative and intraoperative variables were different among the three groups (Supplement Tables 2–4). The incidence of POD was significantly different in the three groups (isolated CABG 61 (29.33%) vs valve surgery 143 (43.87%) vs aortic surgery 102 (59.65%), p < 0.001). The comparison of intraoperative glycemic variables in the three groups showed that intraoperative glycemic CV was significantly different (isolated CABG 18.15 [12.94, 23.90] vs valve surgery 20.55 [15.81, 25.90] vs aortic surgery 21.69 [15.92, 28.04], p = 0.001; Supplement Table 5). In the isolated CABG group, patients in the fourth quartile of intraoperative glycemic CV had 4.675 times higher odds of POD than patients in the first quartile of intraoperative glycemic CV (OR 4.675, 95% CI: 1.761, 12.407, p = 0.002). In the valve surgery group and the aortic surgery group, patients in the fourth quartile had 3.780 times and 2.743 times higher odds of POD than patients in the first quartile (OR 3.780, 95% CI: 1.838–7.777, p < 0.001 and OR 2.743, 95% CI: 1.005–7.484, p = 0.049, respectively; Supplement Table 6). Detailed results are presented in Supplement Tables 2–6.
The postoperative effects of intraoperative glycemic CV are shown in Table 7. Patients with high intraoperative glycemic CV had a longer duration of mechanical ventilation, ICU stay, and hospital stay than patients with low intraoperative glycemic CV. The incidence of MAE was higher in patients with high intraoperative glycemic CV than in those with low intraoperative glycemic CV, and there was no difference in 30 day-mortality between the two groups.
 |
Table 7 Postoperative Outcomes by Intraoperative Glycemic Coefficient of Variation |
Discussion
POD is a common but serious complication after cardiac surgery and is associated with various short- and long-term outcomes. In this study, we investigated the associations of intraoperative GV and other glycemic variables on POD after cardiac surgery. The incidence of POD after cardiac surgery in the present study was 43%. Intraoperative glycemic CV and mean glucose levels were associated with POD after cardiac surgery. The incidence of POD tended to increase as the intraoperative glycemic CV increased. Patients in the third quartile of intraoperative glycemic CV were 1.833 times and patients in the fourth quartile of intraoperative glycemic CV were 3.645 times more likely to develop POD than those in the first quartile of intraoperative glycemic CV.
Although the exact mechanisms are not well understood, glycemic variables have been proposed to play a key role in the development of postoperative neurocognitive disorders including POD.4,13 Glucose is the main source of energy for many cells, including the brain. The brain accounts for 50% of the body’s glucose consumption at any given time and lacks the ability to utilize the energy stored in other forms such as lipids and fatty acids. Therefore, the brain is particularly sensitive to serum glucose concentrations. Glucose-sensing neurons are present in the brain and control the metabolism and homeostasis of blood glucose levels.39 Severe hypoglycemia reduces adenosine triphosphate levels in nerve cells, resulting in neuronal hyperpolarization, which is clinically manifested by seizure activity, alteration in mental status, and delirium.13,40 Hyperglycemia causes oxidative stress, which results in neuronal damage and cognitive impairment.15 Hyperglycemia is both a result and cause of perioperative inflammation,16,41 and the relationship between hyperglycemia and POD is evident considering that neuroinflammation is an important mechanism for the development of POD.4,42 Neuroinflammation can be caused by systemic inflammation,4,17 and systemic inflammation, glucose homeostasis, and oxidative stress are all closely interlinked.15,16,23,42–44 Moreover, hyperglycemia and concomitant inflammation are profound in cardiac surgery due to the use of CPB, administration of heparin, hypothermia, administration of glucose-containing solutions, and the use of inotropes.14 Given that diabetes is linked to Alzheimer’s disease and neurodegeneration, the long-term effects of glucose dysregulation on brain function are clear.45
The association between diabetes, preoperative long-term glucose control, and POD after cardiac surgery is controversial. Diabetes and preoperative long-term glucose control, analyzed by HbA1c, have been identified as predisposing factors for POD after cardiac surgery in several studies,2,19,20,32,33,35 while others did not.34,46 In a study performed in ICU patients, diabetes was not associated with delirium, but glucose dysregulation, including hyperglycemia and hypoglycemia, was associated with delirium.47 The same pattern was demonstrated in our study; diabetes and preoperative HbA1c were not associated with POD after cardiac surgery, but intraoperative glycemic variables were. Moreover, intraoperative GV was associated with POD after adjusting for diabetes, preoperative HbA1c, and the type of hypoglycemic agents used. Although the presence of diabetes and elevated preoperative HbA1c is closely related to perioperative glycemic variables,24,48 the development of POD after cardiac surgery may be attributed to the actual glycemic variables at the time of surgery. This is encouraging because diabetes and preoperative HbA1c are non-modifiable factors, while perioperative glycemic variables can be modified.
Although studies have examined glycemic variables other than the intraoperative period in the perioperative period, little is known about the association between intraoperative glycemic variables and POD after cardiac surgery. Preoperative fasting glucose,49,50 postoperative glucose,51,52 mean glucose extending to the postoperative period,53 and intraoperative glucose measured only during CPB54 showed conflicting results on POD after cardiac surgery. Compared with the preoperative and postoperative periods, the intraoperative period is the period with the most profound stress and inflammation leading to glucose dysregulation, and thus POD.4,11,14 In addition, given that prevention is the most effective method for managing POD,8,11 postoperative glycemic variables are of little value because it is difficult to accurately assess whether changes in certain postoperative glycemic variables have occurred before and/or after the development of POD.47,55 The impact of intraoperative glucose management on neurologic function has been demonstrated in one randomized controlled trial, in which verbal learning after cardiac surgery was maintained with intraoperative maintenance of normoglycemia.56 Intraoperative glycemic variables, mean glucose, and GV were associated with POD after cardiac surgery in the present study. These results are consistent with those of previous studies in cardiac surgery26,57,58 and surgeries other than cardiac surgery.59,60
GV, a metric for swings in blood glucose levels, is an emerging target in the management of blood glucose.21,22,31 In an observational study of patients undergoing surgery for acute aortic dissection, postoperative GV was associated with POD.26 However, in this study, only postoperative GV was investigated, and it was therefore unclear whether changes in postoperative GV occurred before or after the development of POD. In addition, the measurement of GV used in this study was the standard deviation. Although there are a variety of measurements for GV, and there is not a single universally accepted measurement, CV is considered a preferred measure of GV22 and has been adopted in many studies.24,25,48,61 In this study, we demonstrated that intraoperative GV, presented as CV, is associated with POD after cardiac surgery. Furthermore, the incidence of POD increased as the intraoperative glycemic CV increased. In addition, increased intraoperative GV was related to the duration of ICU and hospital stay, and MAE, which is consistent with previous studies.24,25,48,61
In previous studies, an increase in GV was found to be more harmful than chronic hyperglycemia. In two previous articles reporting cell culture studies, oscillating high glucose caused more oxidative stress and cellular apoptosis in human umbilical vein endothelial cells and coronary artery endothelial cells than persistent high glucose.43,44 These results have been demonstrated in a clinical model with normal and type 2 diabetes patients, with oscillating glucose leading to increased endothelial dysfunction and oxidative stress compared with mean glucose.23 However, the predictive ability of intraoperative GV was not different from that of intraoperative mean glucose in our study. Five intraoperative measurements of blood glucose may not accurately detect all changes in blood glucose levels. Since GV is a more complex measure than mean glucose, future well-designed prospective studies, possibly with continuous glucose monitoring, are needed to justify the use of GV.
Patients who underwent off-pump CABG were excluded from this study because the use of CPB is known to be associated with severe changes in glucose tolerance and cerebral perfusion.62,63 Moreover, large quantities of inflammatory mediators are released during CPB resulting in neuroinflammation, which in turn increases the risk of delirium and neurocognitive dysfunction.64,65 According to recent retrospective analysis, patients receiving off-pump cardiac surgery resulted in a significantly lower incidence of POD than those receiving on-pump cardiac surgery.66 In a clinical trial comparing the effect of off-pump and on-pump CABG on the cognitive outcome, patients who underwent off-pump CABG showed less cognitive dysfunction than those who underwent on-pump CABG.67 In our study population, the incidence of POD in off-pump and on-pump CABG were 16.1% and 29.3%, and intraoperative glycemic CV in off-pump and on-pump CABG was 17.06% and 18.15%. This result is consistent with previous studies.65–67
The three types of cardiac surgery included in this study, CABG, valve surgery, and aortic replacement surgery all pose an inherent risk to the occurrence of POD.4 Coronary artery disease is caused by atherosclerotic plaque formation in the vessel lumen, and diseased vessels are not limited to coronary arteries, but also, the aorta.68 The direct manipulation of the aorta during surgery results in aortic plaque disruption, which can release microemboli that can migrate to the brain.69 In addition, atherosclerosis reduces perfusion to the brain and the muscles, resulting in functional impairment.70 Valve surgery requires the opening of the heart, which frequently results in the formation of gaseous microemboli.71 Complex aortic replacement surgery utilizes TCA, in which cerebral perfusion is impeded for a short period of time.72 Therefore, different risks for POD according to the type of cardiac surgery have been reported in the literature,9,33,73,74 which was also demonstrated in this study.
An important finding in our study was that intraoperative GV varies depending on the type of cardiac surgery. In a previous study that evaluated the effect of different degrees of blood glucose control on clinical outcomes in cardiac surgery, the highest perioperative blood glucose level differed according to the type of cardiac surgery.75 This may be due to differences in patient characteristics and multiple factors leading to different levels of stress.3,4,75 In line with this previous result, intraoperative GV differed depending on the type of cardiac surgery. Nevertheless, intraoperative GV was associated with POD in all types of cardiac surgery analyzed in our study.
Other than intraoperative GV, the predictors that were associated with POD after cardiac surgery were age, educational level, GFR, type of surgery, duration of CPB, mean BIS, and VIS at the end of the surgery. Age is the most important and well-known predisposing risk factor.2,3,9,10,32–35 Final education level is closely linked to cognitive reserve,76 and the cognitive reserve has been identified as a predictor for POD in many studies.3,4,9 The kidney and the brain are known to interact in various mechanisms such as sodium dysregulation, edema formation, extravasation of leukocytes, oxidative stress, and cytokine-induced damage.77 Longer duration of CPB is associated with greater numbers of cerebral microemboli, which is known to be an important factor in the development of POD.78 Low BIS values to anesthetics indicate a higher vulnerability, which may result in an unnecessary increase in anesthesia levels and possible neurotoxic effects.79 Although the association between VIS at the end of the surgery and POD has never been studied in cardiac surgery, high VIS values are known to be associated with adverse outcomes, and the use of beta-adrenergic agents can induce hyperglycemia.38
In the present study, we defined POD as delirium diagnosed using CAM and CAM-ICU within 24 h of surgery. Delirium can be classified into three subtypes: hyperactive, hypoactive, and mixed, and the detection of hypoactive delirium is often difficult in practice.80 Therefore, using a validated tool for the diagnosis of delirium is important for clinical research. CAM and CAM-ICU are validated delirium assessment instruments used in many clinical studies.29,30 Previous studies may have underestimated the incidence by using heterogeneous instruments for the diagnosis of POD after cardiac surgery.2,10,30,32–34
This study has some limitations. First, this was a retrospective study and had the potential for unrecognized confounding factors. However, a rigorous review of previous studies was conducted, and data on all possible confounding factors were collected. The predictors identified in this study, age, educational level, GFR, type of surgery, duration of CPB, mean BIS, and VIS at the end of the surgery, were all supported by previous studies.2−22−35 Second, patients with glucose dysregulation tend to undergo more glucose measurements, resulting in different variances. We tried to overcome this potential bias by averaging multiple blood glucose measurements to a single value within a defined period. Third, inclusion criteria for age in our study was patients aged over 18 years, which resulted in a wide range of age groups. It is well-known that older patients are more susceptible to POD than younger patients. However, the average age of patients in this study was 66 years old, and intraoperative GV was associated with POD after adjusting for age. Fourth, a recent recommendation defines POD as occurring within one week from the procedure,1 but in this study, POD was considered positive to occur only within 24 h because we wanted to limit the concomitant effects of postoperative variables. We found that the association between postoperative factors and POD was difficult to define because the postoperative events may have occurred after the development of POD, which could lead to a bias given the retrospective nature of the study. Moreover, POD after cardiac surgery most often occurs on the initial postoperative day.81 Finally, the use of continuous intraoperative glucose monitoring would have resulted in more accurate intraoperative glycemic variables. Unfortunately, there are currently no continuous glucose monitoring devices approved for use in cardiac surgery.82
Conclusion
Intraoperative blood glucose fluctuation, manifested by intraoperative GV, and a higher mean glucose level are associated with POD after cardiac surgery. Patients with a higher intraoperative GV have an increased risk of POD. Future research is needed to identify other adverse outcomes related to intraoperative GV, and whether optimal intraoperative GV control in patients undergoing cardiac surgery will change outcomes.
Abbreviations
POD, postoperative delirium; ICU, intensive care unit; NYHA, New York Heart Association; CPB, cardiopulmonary bypass pump; HbA1c, hemoglobin A1c; GV, glycemic variability; IRB, Institutional Review Board; CABG, coronary artery bypass graft surgery; CAM-ICU, Confusion Assessment Method for the Intensive Care Unit; CAM, Confusion Assessment Method; CV, coefficient of variation; BIS, bispectral index; BMI, body mass index; EuroSCORE, European System for Cardiac Operative Risk Evaluation; GFR, glomerular filtration rate; CRP, c-reactive protein; TCA, total circulatory arrest; PRC, packed red blood cell; VIS, vasoactive-inotropic score; MAE, major adverse events; AUROC, area under the receiver operator characteristics.
Data Sharing Statement
The data used and/or analyzed during this study are available from the corresponding author on reasonable request.
Acknowledgments
We would like to thank Editage (www.editage.co.kr) for English language editing.
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT; Ministry of Science and ICT) (No. 2021R1G1A1014702). The funder played no part in the study design; the collection, management, analysis, or interpretation of the data; the writing of the report; or the decision to submit the report for publication.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Evered L, Silbert B, Knopman DS, et al. Recommendations for the nomenclature of cognitive change associated with anaesthesia and surgery—2018. Anesthesiology. 2018;129(5):872–879. doi:10.1097/aln.0000000000002334
2. Chen H, Mo L, Hu H, Ou Y, Luo J. Risk factors of postoperative delirium after cardiac surgery: a meta-analysis. J Cardiothorac Surg. 2021;16(1):113. doi:10.1186/s13019-021-01496-w
3. Brown CH. Delirium in the cardiac surgical ICU. Curr Opin Anaesthesiol. 2014;27(2):117–122. doi:10.1097/aco.0000000000000061
4. Berger M, Terrando N, Smith SK, Browndyke JN, Newman MF, Mathew JP. Neurocognitive function after cardiac surgery. Anesthesiology. 2018;129(4):829–851. doi:10.1097/aln.0000000000002194
5. Crocker E, Beggs T, Hassan A, et al. Long-term effects of postoperative delirium in patients undergoing cardiac operation: a systematic review. Ann Thorac Surg. 2016;102(4):1391–1399. doi:10.1016/j.athoracsur.2016.04.071
6. Brown CH, Probert J, Healy R, et al. Cognitive decline after delirium in patients undergoing cardiac surgery. Anesthesiology. 2018;129(3):406–416. doi:10.1097/aln.0000000000002253
7. Kirfel A, Menzenbach J, Guttenthaler V, et al. Postoperative delirium after cardiac surgery of elderly patients as an independent risk factor for prolonged length of stay in intensive care unit and in hospital. Aging Clin Exp Res. 2021;33(11):3047–3056. doi:10.1007/s40520-021-01842-x
8. Devlin JW, Skrobik Y, Gélinas C, et al. Clinical practice guidelines for the prevention and management of pain, agitation/ sedation, delirium, immobility, and sleep disruption in adult patients in the ICU. Crit Care Med. 2018;46(9):e825–e873. doi:10.1097/ccm.0000000000003299
9. Hollinger A, Siegemund M, Goettel N, Steiner LA. Postoperative delirium in cardiac surgery: an unavoidable menace? J Cardiothorac Vasc Anesth. 2015;29(6):1677–1687. doi:10.1053/j.jvca.2014.08.021
10. O’Neal JB, Shaw AD. Predicting, preventing, and identifying delirium after cardiac surgery. Perioper Med. 2016;5(1). doi:10.1186/s13741-016-0032-5
11. Aldecoa C, Bettelli G, Bilotta F, et al. European Society of Anaesthesiology evidence-based and consensus-based guideline on postoperative delirium. Eur J Anaesthesiol. 2017;34(4):192–214. doi:10.1097/eja.0000000000000594
12. Ogawa M, Izawa KP, Kitamura A, et al. Preoperative physical activity in relation to postoperative delirium in elective cardiac surgery patients. Int J Cardiol. 2015;201:154–156. doi:10.1016/j.ijcard.2015.06.154
13. Sanford AM, Flaherty JH. Do nutrients play a role in delirium?. Curr Opin Clin Nutr Metab Care. 2014;17(1):45–50. doi:10.1097/mco.0000000000000022
14. Lazar HL, McDonnell M, Chipkin SR, et al. The society of thoracic surgeons practice guideline series: blood glucose management during adult cardiac surgery. Ann Thorac Surg. 2009;87(2):663–669. doi:10.1016/j.athoracsur.2008.11.011
15. Stadler K. Oxidative stress in diabetes. Adv Exp Med Biol. 2012;771:272–287. doi:10.1007/978-1-4614-5441-0_21
16. Rehman K, Akash MSH. Mechanisms of inflammatory responses and development of insulin resistance: how are they interlinked? J Biomed Sci. 2016;23(1). doi:10.1186/s12929-016-0303-y
17. Glumac S, Kardum G, Karanovic N. Postoperative cognitive decline after cardiac surgery: a narrative review of current knowledge in 2019. Med Sci Monitor. 2019;25:3262–3270. doi:10.12659/msm.914435
18. Smulter N, Lingehall HC, Gustafson Y, Olofsson B, Engström KG. Delirium after cardiac surgery: incidence and risk factors. Interact Cardiovasc Thorac Surg. 2013;17(5):790–796. doi:10.1093/icvts/ivt323
19. Kotfis K, Szylińska A, Listewnik M, Brykczyński M, Ely EW, Rotter I. Diabetes and elevated preoperative HbA1c level as risk factors for postoperative delirium after cardiac surgery: an observational cohort study. Neuropsychiatr Dis Treat. 2019;15:511–521. doi:10.2147/ndt.S196973
20. Lechowicz K, Szylińska A, Listewnik M, et al. Cardiac delirium index for predicting the occurrence of postoperative delirium in adult patients after coronary artery bypass grafting. Clin Interv Aging. 2021;16:487–495. doi:10.2147/cia.S302526
21. Service FJ. Glucose variability. Diabetes. 2013;62(5):1398–1404. doi:10.2337/db12-1396
22. Suh S, Kim JH. Glycemic variability: How do we measure it and why is it important?. Diabetes Metab J. 2015;39(4):273–282. doi:10.4093/dmj.2015.39.4.273
23. Ceriello A, Esposito K, Piconi L, et al. Oscillating glucose is more deleterious to endothelial function and oxidative stress than mean glucose in normal and type 2 diabetic patients. Diabetes. 2008;57(5):1349–1354. doi:10.2337/db08-0063
24. Subramaniam B, Lerner A, Novack V, et al. Increased glycemic variability in patients with elevated preoperative HbA1C predicts adverse outcomes following coronary artery bypass grafting surgery. Anesth Analg. 2014;118(2):277–287. doi:10.1213/ane.0000000000000100
25. Clement KC, Suarez-Pierre A, Sebestyen K, et al. Increased glucose variability is associated with major adverse events after coronary artery bypass. Ann Thorac Surg. 2019;108(5):1307–1313. doi:10.1016/j.athoracsur.2019.06.046
26. Lin YJ, Lin LY, Peng YC, et al. Association between glucose variability and postoperative delirium in acute aortic dissection patients: an observational study. J Cardiothorac Surg. 2021;16(1):82. doi:10.1186/s13019-021-01456-4
27. von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Ann Intern Med. 2007;147(8):573–577. doi:10.7326/0003-4819-147-8-200710160-00010
28. Diagnostic and statistical manual of mental disorders: DSM-5. American Psychiatric Association; 2013.
29. Ely EW, Inouye SK, Bernard GR, et al. Delirium in mechanically ventilated patients: validity and reliability of the confusion assessment method for the intensive care unit (CAM-ICU). JAMA. 2001;286(21):2703–2710. doi:10.1001/jama.286.21.2703
30. Smulter N, Lingehall HC, Gustafson Y, Olofsson B, Engström KG. Validation of the confusion assessment method in detecting postoperative delirium in cardiac surgery patients. Am J Crit Care. 2015;24(6):480–487. doi:10.4037/ajcc2015551
31. Devries JH. Glucose variability: where it is important and how to measure it. Diabetes. 2013;62(5):1405–1408. doi:10.2337/db12-1610
32. Koster S, Hensens AG, Schuurmans MJ, Van Der Palen J. Risk factors of delirium after cardiac surgery: a systematic review. Eur J Cardiovasc Nurs. 2011;10(4):197–204. doi:10.1016/j.ejcnurse.2010.09.001
33. Lin Y, Chen J, Wang Z. Meta-analysis of factors which influence delirium following cardiac surgery. J Card Surg. 2012;27(4):481–492. doi:10.1111/j.1540-8191.2012.01472.x
34. Gosselt AN, Slooter AJ, Boere PR, Zaal IJ. Risk factors for delirium after on-pump cardiac surgery: a systematic review. Critical Care. 2015;19(1). doi:10.1186/s13054-015-1060-0
35. Greaves D, Psaltis PJ, Davis DHJ, et al. Risk factors for delirium and cognitive decline following coronary artery bypass grafting surgery: a systematic review and meta‐analysis. J Am Heart Assoc. 2020;9(22). doi:10.1161/jaha.120.017275
36. American Geriatrics Society. 2019 updated AGS beers criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2019;67(4):674–694. doi:10.1111/jgs.15767
37. Tse L, Schwarz SB, Bowering J, et al. Pharmacological risk factors for delirium after cardiac surgery: a review. Curr Neuropharmacol. 2012;10(3):181–196. doi:10.2174/157015912803217332
38. Belletti A, Lerose CC, Zangrillo A, Landoni G. Vasoactive-inotropic score: evolution, clinical utility, and pitfalls. J Cardiothorac Vasc Anesth. 2020. doi:10.1053/j.jvca.2020.09.117
39. Scheen AJ. Central nervous system: a conductor orchestrating metabolic regulations harmed by both hyperglycaemia and hypoglycaemia. Diabetes Metab. 2010;36(Suppl 3):S31–8. doi:10.1016/s1262-3636(10)70464-x
40. Bazzigaluppi P, Ebrahim Amini A, Weisspapir I, Stefanovic B, Carlen PL. Hungry neurons: metabolic insights on seizure dynamics. Int J Mol Sci. 2017;18(11):2269. doi:10.3390/ijms18112269
41. Tsalamandris S, Antonopoulos AS, Oikonomou E, et al. The role of inflammation in diabetes: current concepts and future perspectives. Eur Cardiol. 2019;14(1):50–59. doi:10.15420/ecr.2018.33.1
42. Cerejeira J, Firmino H, Vaz-Serra A, Mukaetova-Ladinska EB. The neuroinflammatory hypothesis of delirium. Acta Neuropathol. 2010;119(6):737–754. doi:10.1007/s00401-010-0674-1
43. Quagliaro L, Piconi L, Assaloni R, Martinelli L, Motz E, Ceriello A. Intermittent high glucose enhances apoptosis related to oxidative stress in human umbilical vein endothelial cells: the role of protein kinase C and NAD(P) H-oxidase activation. Diabetes. 2003;52(11):2795–2804. doi:10.2337/diabetes.52.11.2795
44. Liu TS, Pei YH, Peng YP, Chen J, Jiang SS, Gong JB. Oscillating high glucose enhances oxidative stress and apoptosis in human coronary artery endothelial cells. J Endocrinol Invest. 2014;37(7):645–651. doi:10.1007/s40618-014-0086-5
45. Shinohara M, Sato N. Bidirectional interactions between diabetes and Alzheimer’s disease. Neurochem Int. 2017;108:296–302. doi:10.1016/j.neuint.2017.04.020
46. Itagaki A, Sakurada K, Matsuhama M, Yajima J, Yamashita T, Kohzuki M. Impact of frailty and mild cognitive impairment on delirium after cardiac surgery in older patients. J Cardiol. 2020;76(2):147–153. doi:10.1016/j.jjcc.2020.02.007
47. van Keulen K, Knol W, Belitser SV, et al. Diabetes and glucose dysregulation and transition to delirium in ICU patients. Crit Care Med. 2018;46(9):1444–1449. doi:10.1097/ccm.0000000000003285
48. Bardia A, Khabbaz K, Mueller A, et al. The association between preoperative hemoglobin A1C and postoperative glycemic variability on 30-day major adverse outcomes following isolated cardiac valvular surgery. Anesth Analg. 2017;124(1):16–22. doi:10.1213/ane.0000000000001715
49. Krzych ŁJ, Wybraniec MT, Krupka-Matuszczyk I, et al. Complex assessment of the incidence and risk factors of delirium in a large cohort of cardiac surgery patients: a single-center 6-year experience. Biomed Res Int. 2013;2013:1–9. doi:10.1155/2013/835850
50. Krzych ŁJ, Wybraniec MT, Krupka-Matuszczyk I, Skrzypek M, Bochenek AA. Delirium Screening in Cardiac Surgery (DESCARD): a useful tool for nonpsychiatrists. Can J Cardiol. 2014;30(8):932–939. doi:10.1016/j.cjca.2014.04.020
51. Shi Q, Mu X, Zhang C, Wang S, Hong L, Chen X. Risk factors for postoperative delirium in type a aortic dissection patients: a retrospective study. Med Sci Monitor. 2019;25:3692–3699. doi:10.12659/msm.913774
52. Kazmierski J, Kowman M, Banach M, et al. Incidence and predictors of delirium after cardiac surgery: results from The IPDACS Study. J Psychosom Res. 2010;69(2):179–185. doi:10.1016/j.jpsychores.2010.02.009
53. Andrejaitiene J, Sirvinskas E. Early post-cardiac surgery delirium risk factors. Perfusion. 2012;27(2):105–112. doi:10.1177/0267659111425621
54. Butterworth J, Wagenknecht LE, Legault C, et al. Attempted control of hyperglycemia during cardiopulmonary bypass fails to improve neurologic or neurobehavioral outcomes in patients without diabetes mellitus undergoing coronary artery bypass grafting. J Thorac Cardiovasc Surg. 2005;130(5):
55. Van Keulen K, Knol W, Belitser SV, et al. Glucose variability during delirium in diabetic and non-diabetic intensive care unit patients: a prospective cohort study. PLoS One. 2018;13(11):e0205637. doi:10.1371/journal.pone.0205637
56. Schricker T, Sato H, Beaudry T, Codere T, Hatzakorzian R, Pruessner JC. Intraoperative maintenance of normoglycemia with insulin and glucose preserves verbal learning after cardiac surgery. PLoS One. 2014;9(6):e99661. doi:10.1371/journal.pone.0099661
57. Gandhi GY, Nuttall GA, Abel MD, et al. Intraoperative hyperglycemia and perioperative outcomes in cardiac surgery patients. Mayo Clin Proc. 2005;80(7):862–866. doi:10.4065/80.7.862
58. Puskas F, Grocott HP, White WD, Mathew JP, Newman MF, Bar-Yosef S. Intraoperative hyperglycemia and cognitive decline after CABG. Ann Thorac Surg. 2007;84(5):1467–1473. doi:10.1016/j.athoracsur.2007.06.023
59. Hermanides J, Qeva E, Preckel B, Bilotta F. Perioperative hyperglycemia and neurocognitive outcome after surgery: a systematic review. Minerva Anestesiol. 2018;84(10):1178–1188. doi:10.23736/s0375-9393.18.12400-x
60. Windmann V, Spies C, Knaak C, et al. Intraoperative hyperglycemia increases the incidence of postoperative delirium. Minerva Anestesiol. 2019;85(11):1201–1210. doi:10.23736/s0375-9393.19.13748-0
61. Duncan AE, Abd-Elsayed A, Maheshwari A, Xu M, Soltesz E, Koch CG. Role of intraoperative and postoperative blood glucose concentrations in predicting outcomes after cardiac surgery. Anesthesiology. 2010;112(4):860–871. doi:10.1097/ALN.0b013e3181d3d4b4
62. Anderson RE, Brismar K, Barr G, Ivert T. Effects of cardiopulmonary bypass on glucose homeostasis after coronary artery bypass surgery. Eur J Cardiothorac Surg. 2005;28(3):425–430. doi:10.1016/j.ejcts.2005.05.025
63. Kosir G, Tetickovic E. Intraoperative transcranial Doppler ultrasonography monitoring of cerebral blood flow during coronary artery bypass grafting. Acta Clin Croat. 2011;50(1):5–11.
64. Ramlawi B, Rudolph JL, Mieno S, et al. Serologic markers of brain injury and cognitive function after cardiopulmonary bypass. Ann Surg. 2006;244(4):593–601. doi:10.1097/01.sla.0000239087.00826.b4
65. Dominici C, Salsano A, Nenna A, et al. Neurological outcomes after on-pump vs off-pump CABG in patients with cerebrovascular disease. J Card Surg. 2019;34(10):941–947. doi:10.1111/jocs.14158
66. Bucerius J, Gummert JF, Borger MA, et al. Predictors of delirium after cardiac surgery delirium: effect of beating-heart (off-pump) surgery. J Thorac Cardiovasc Surg. 2004;127(1):57–64. doi:10.1016/s0022-5223(03)01281-9
67. Van Dijk D, Jansen EW, Hijman R, et al. Cognitive outcome after off-pump and on-pump coronary artery bypass graft surgery: a randomized trial. JAMA. 2002;287(11):1405–1412. doi:10.1001/jama.287.11.1405
68. Fazio GP, Redberg RF, Winslow T, Schiller NB. Transesophageal echocardiographically detected atherosclerotic aortic plaque is a marker for coronary artery disease. J Am Coll Cardiol. 1993;21(1):144–150. doi:10.1016/0735-1097(93)90729-k
69. Evered LA, Silbert BS, Scott DA. Postoperative cognitive dysfunction and aortic atheroma. Ann Thorac Surg. 2010;89(4):1091–1097. doi:10.1016/j.athoracsur.2009.11.056
70. Maloberti A, Fribbi F, Motto E, et al. Ankle-brachial index is a predictor of in-hospital functional status but not of complications in hospitalized elderly patients. Gerontology. 2021:1–7. doi:10.1159/000514450
71. Chaudhuri K, Marasco SF. The effect of carbon dioxide insufflation on cognitive function during cardiac surgery. J Card Surg. 2011;26(2):189–196. doi:10.1111/j.1540-8191.2011.01217.x
72. Misfeld M, Leontyev S, Borger MA, et al. What is the best strategy for brain protection in patients undergoing aortic arch surgery? A single center experience of 636 patients. Ann Thorac Surg. 2012;93(5):1502–1508. doi:10.1016/j.athoracsur.2012.01.106
73. Tse L, Schwarz SKW, Bowering JB, Moore RL, Barr AM. Incidence of and risk factors for delirium after cardiac surgery at a quaternary care center: a retrospective cohort study. J Cardiothorac Vasc Anesth. 2015;29(6):1472–1479. doi:10.1053/j.jvca.2015.06.018
74. Kotfis K, Szylińska A, Listewnik M, et al. Early delirium after cardiac surgery: an analysis of incidence and risk factors in elderly (≥65 years) and very elderly (≥80 years) patients. Clin Interv Aging. 2018;13:1061–1070. doi:10.2147/cia.s166909
75. Ascione R, Rogers CA, Rajakaruna C, Angelini GD. Inadequate blood glucose control is associated with in-hospital mortality and morbidity in diabetic and nondiabetic patients undergoing cardiac surgery. Circulation. 2008;118(2):113–123. doi:10.1161/circulationaha.107.706416
76. Thow ME, Summers MJ, Saunders NL, Summers JJ, Ritchie K, Vickers JC. Further education improves cognitive reserve and triggers improvement in selective cognitive functions in older adults: The Tasmanian healthy brain project. Alzheimers Dement. 2018;10:22–30. doi:10.1016/j.dadm.2017.08.004
77. Lu R, Kiernan MC, Murray A, Rosner MH, Ronco C. Kidney-brain crosstalk in the acute and chronic setting. Nat Rev Nephrol. 2015;11(12):707–719. doi:10.1038/nrneph.2015.131
78. Brown WR, Moody DM, Challa VR, Stump DA, Hammon JW. Longer duration of cardiopulmonary bypass is associated with greater numbers of cerebral microemboli. Stroke. 2000;31(3):707–713. doi:10.1161/01.str.31.3.707
79. Fedorow C, Grocott HP. Cerebral monitoring to optimize outcomes after cardiac surgery. Curr Opin Anaesthesiol. 2010;23(1):89–94. doi:10.1097/ACO.0b013e3283346d10
80. Peritogiannis V, Bolosi M, Lixouriotis C, Rizos DV. Recent insights on prevalence and correlations of hypoactive delirium. Behav Neurol. 2015;2015:416792. doi:10.1155/2015/416792
81. Hamadnalla H, Sessler DI, Troianos CA, et al. Optimal interval and duration of CAM-ICU assessments for delirium detection after cardiac surgery. J Clin Anesth. 2021;71:110233. doi:10.1016/j.jclinane.2021.110233
82. Krinsley JS, Chase JG, Gunst J, et al. Continuous glucose monitoring in the ICU: clinical considerations and consensus. Crit Care. 2017;21(1):197. doi:10.1186/s13054-017-1784-0
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

