Back to Journals » Nature and Science of Sleep » Volume 12
Inspiratory Muscle Training in the Severity of Obstructive Sleep Apnea, Sleep Quality and Excessive Daytime Sleepiness: A Placebo-Controlled, Randomized Trial
Authors Nóbrega-Júnior JCN , Dornelas de Andrade A, Andrade EAM, Andrade MA, Ribeiro ASV, Pedrosa RP, Ferreira APL , Lima AMJ
Received 27 June 2020
Accepted for publication 1 September 2020
Published 2 December 2020 Volume 2020:12 Pages 1105—1113
DOI https://doi.org/10.2147/NSS.S269360
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Steven A Shea
José Carlos Nogueira Nóbrega-Júnior,1 Armèle Dornelas de Andrade,2 Erika Alves Marinho de Andrade,2 Maria do Amparo Andrade,2 Alice Santana Valadares Ribeiro,3 Rodrigo Pinto Pedrosa,4 Ana Paula de Lima Ferreira,2 Anna Myrna Jaguaribe de Lima2,5
1Department of Physical Therapy, Facottur Faculty, Olinda, Pernambuco, Brazil; 2Department of Physical Therapy Federal University of Pernambuco, Recife, Pernambuco, Brazil; 3Department of Rehabilitation, Hospital Otávio de Freitas, Recife, Pernambuco, Brazil; 4Sleep and Heart Laboratory, Pronto Socorro Cardiológico de Pernambuco (PROCAPE)- University of Pernambuco, Recife, Pernambuco, Brazil; 5Department of Morphology and Animal Physiology, Federal Rural University of Pernambuco, Recife, Pernambuco, Brazil
Correspondence: Anna Myrna Jaguaribe de Lima
Federal Rural University of Pernambuco, Rua Manoel de Medeiros, s/n - Dois Irmãos, Recife, PE 52171-900, Brazil
+55 81 3320-6475
Email [email protected]
Purpose: Exercise programs have been considered as an adjuvant treatment in obstructive sleep apnea (OSA). However, few studies have focused on the effects of the inspiratory muscle training (IMT) in reducing the severity and the symptoms of OSA.
Patients and Methods: A randomized controlled trial was conducted and approved by the local Ethics Committee. All subjects signed the informed consent form and were randomized into 2 groups: a) IMT group (n = 8), 8 weeks of IMT with 75% of maximal inspiratory pressure (MIP) and b) placebo group (n = 8): subjects performed IMT without load.
Results: IMT group showed reduction in the apnea–hypopnea index (AHI) (p = 0.01), in the Berlin questionnaire score (p = 0.001) and an increase in inspiratory muscle strength (p = 0.018). IMT group demonstrated a reduction in the AHI (31.7 ± 15.9 events/h vs 29.9 ± 15.8 events/h; p < 0.001), in the Berlin questionnaire scores (2.6 ± 0.5 vs 1.2 ± 0.5; p = 0.016), Pittsburgh Sleep Quality Index (PSQI) score (7.2 ± 3.6 vs 3.7 ± 1.3; p = 0.008), in the Epworth Sleepiness Scale (ESS) (12.5 ± 4.0 vs 7.7 ± 3.0; p = 0.008) and increase in MIP (83.6 ± 26.5 cmH2O and 127.9 ± 32.5 cmH2O; p = 0.010).
Conclusion: The IMT promotes discrete changes in the AHI and improves sleep quality and excessive daytime sleepiness in OSA. Moreover, IMT is a cheap, useful and simple home-based training program and can be considered as an adjunct therapy for OSA patients.
Keywords: adjuvant therapy, respiratory exercises, obstructive sleep apnea symptoms, exercise training
Introduction
Obstructive sleep apnea (OSA) is a common sleep disorder characterized by recurrent upper airway obstructive events which result in hypoxia/reoxygenation episodes during sleep.1 The current “gold standard” treatment for OSA is continuous positive airway pressure (CPAP), considered the first choice in moderate or severe cases, but it has high cost and controversial adherence. In addition, other treatment forms have proven to be effective in mild to moderate OSA, such as intraoral and nasal valve devices, hypoglossal nerve stimulation and the performance of specific exercises for orofacial and oropharyngeal muscles.2–4
In this context, exercise has become an effective low-cost strategy that is easily applicable and has good adherence.5 Furthermore, a recent meta-analysis of eight studies revealed that exercise training was able to reduce the severity of OSA.6 However, there is still a significant gap to be filled regarding inspiratory muscle training (IMT) in OSA. The IMT is a specific type of exercise training to improve the strength and performance of the respiratory muscles in healthy individuals and athletes, as well as in treating cardiorespiratory diseases.7–9 Concerning OSA, only three studies have reported the exclusive use of IMT in these patients.10–12
Therefore, the aim of this study was to verify the effectiveness of 8 weeks of inspiratory muscle training on the severity and symptoms of OSA in subjects diagnosed with moderate or severe OSA.
Patients and Methods
Design
A randomized, controlled, and double-blind clinical trial was conducted with patients diagnosed with moderate or severe OSA and approved by the Human Research Ethics Committee of the Federal University of Pernambuco (UFPE). All procedures were in accordance with the ethical standards of the Human Research Ethics Committee of the UFPE and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The research was registered in Clinical Trials and can be accessed by the code NCT02963844. The sample size calculation was performed by Gpower software using the severity of OSA as the primary outcome measure, with an effect size of 0.35 (f), a significance level of 95% and power of 80%, giving a total sample size of 16 individuals (8 in each group).
Randomization
Selected patients were randomized through software randomization.com. A third person not involved in the research carried out the allocation of numbers in black and opaque sealed envelopes for the patients and the first evaluator was blind.
Inclusion/Exclusion Criteria
Inclusion criteria were subjects of both genders, aged between 30 and 65 years, sedentary lifestyle, with a diagnosis of moderate or severe obstructive sleep apnea (AHI≥15 events/hour) conducted by polygraphy and 18≥BMI≤ 39.9Kg/m2.
Measures
OSA severity was measured before and after 8 weeks of IMT. The apnea–hypopnea index (AHI) was obtained by an Embletta portable polygraphy system (Embla, Embletta® Gold, USA).
The Pittsburgh sleep quality index (PSQI) was used to assess the subjective quality of sleep. The overall score is given by the sum of scores obtained in seven components to classify the quality of sleep as good (0 to 4 points) or bad (>5 points).13 The Epworth sleepiness scale (ESS) was used to evaluate excessive daytime sleepiness. It is a subjective self-assessment scale on the possibility to rest during common daily activities. An overall score ≥ 10 reflects excessive daytime sleepiness.14
The International Physical Activity Questionnaire (IPAQ - short form) was used to ensure that all participants were classified as sedentary or insufficiently active. This instrument enables estimating the amount of time spent during the week doing physical activities with moderate and strong intensity, in different day-to-day contexts.15
The Berlin questionnaire is a useful tool to screen for OSA and can also be used to assess the presence of snoring. This instrument is composed of self-assessment questions divided into three categories. The presence of snoring, daytime sleepiness and sleep during daily activities, history of hypertension (HTN) and calculation of body mass index (BMI).16
The corresponding values for maximal respiratory pressures were obtained by a manometer (MVD 300, Globalmed, Brazil). The system considered three valid maneuvers (coefficient of variation below 10%).17 The highest obtained value was used for the analysis, considering the reference values for the adult Brazilian population.18
Finally, a spirometer (MicroLoop®, Micro Medical, UK) was used to evaluate lung function. The following variables were obtained: FEV1 (forced expiratory volume in 1 s), FVC (forced vital capacity), PEF (peak expiratory flow) and FEV1/FVC ratio. The test was performed at least three times until the system considers the best maneuver as reproducible and acceptable and the reference values for the Brazilian adult population were considered.19
Intervention
Powerbreath® ClassicLight equipment was used for implementing the inspiratory muscle training (IMT). The IMT group performed the training with progressive load during 8 weeks (50% maximal inspiratory pressure (MIP) in the first 2 weeks of training, 60% MIP in the third and fourth week and 75% MIP in the last 4 weeks). Load adjustments were performed by a second evaluator weekly in face-to-face meetings in the laboratory. The apparatus resistance was removed for the placebo group and remained so during the 8 weeks. All participants were instructed to perform three cycles of 30 breaths, with an interval of 1 min between the series, twice per day, 7 days per week.11
Statistical Analysis
The statistical analysis was performed with GraphPad Prism 4.0 (GraphPad Software Inc., USA) and SigmaPlot 12.0 (Systat Software Inc., Germany). Continuous variables were expressed as mean and standard deviation, mean difference and a confidence interval of 95%, or median and inter-quartile interval. The categorical variables were expressed as number of cases and frequency. The Shapiro–Wilk normality test was performed for the data distribution analysis. Two-way ANOVA with post-test was performed for multiple Holm-Sidak comparisons which were used for comparison between the interventions (IMT and placebo) and comparing the effect of time within each intervention (pre- and post-intervention). We used the Friedman test with the Tukey’s test for non-parametric data. We used the Student’s t-test for independent samples for a comparison of variation among the groups. The Chi-squared test or Fisher’s Exact test was performed to compare the categorical variables. Values were considered significant at p < 0.05.
Results
Figure 1 shows the process of capturing, randomization and allocation of the participants.
 |
Figure 1 Flow diagram of patient recruitment and progress through the randomized controlled trial. |
Anthropometric, clinical, and variables related to the pulmonary function and respiratory muscle strength of the sample are shown in Table 1. FEV1 and FVC were greater than or equal to 80% of the value predicted for the adult Brazilian population and the MIP presented higher values than 60cmH2O.
 |
Table 1 Characteristics of the Sample |
The baseline data of the sample regarding the sleep parameters are demonstrated in Table 2.
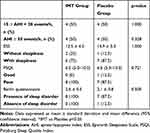 |
Table 2 Baseline Sleep Parameters Data of the Sample |
The comparison between pre and post-intervention data related to sleep and inspiratory muscle strength variables in IMT and placebo groups is shown in Table 3. The IMT group presented a reduction on the AHI (31.7 ± 15.9 events/h vs 29.9 ± 15.8 events/h; p<0.001), in the Berlin questionnaire scores (2.6 ± 0.5 vs 1.2 ± 0.5; p=0.016), the Pittsburgh Sleep Quality Index (PSQI) (7.2 ± 3.6 vs 3.7 ± 1.3; p=0.008) and the SDE score (12.5 ± 4.0 vs 7.7 ± 3.0; p=0.008), and an increase in the inspiratory muscle strength (83.6 ± 26.5 cmH2O vs 127.9 ± 32.5 cmH2O; p=0.010) in relation to baseline values. There was a reduction in the PSQI (p=0.010) in the IMT group in the intergroup comparison.
 |
Table 3 Sleep Parameters and Inspiratory Muscle Strength in IMT and Placebo Groups |
AHI, PSQI, Berlin questionnaire, EDS and MIP changes (post–pre) in the IMT and placebo groups are presented in Figure 2. The IMT group demonstrated a reduction on the AHI (p=0.01) and the Berlin questionnaire score (p=0.001) and an increase in MIP (p=0.018) compared to the placebo group.
Figure 3 illustrates the individual pre-post intervention changes in the apnea–hypopnea index (AHI), Pittsburgh Sleep Quality Index (PSQI), Berlin questionnaire, Excessive Daytime Sleepiness (EDS) and Maximum Inspiratory Pressure (MIP) in IMT and placebo group.
Discussion
To our knowledge, this is the first study to evaluate the effectiveness of 8 weeks of inspiratory muscle training in patients with moderate and severe OSA on the severity of the disease, the quality of sleep and excessive daytime sleepiness. A reduction in AHI and in the Berlin questionnaire score was observed for the intervention group when compared to the placebo group in the present study. An improvement was verified regarding sleep quality and excessive daytime sleepiness after 8 weeks of IMT for the experimental group when compared to baseline values.
Different types of exercise training can improve quality of life, sleep quality, excessive daytime sleepiness, and reduce the AHI.5,6,20–22 Exercise benefits for OSA occur by reducing the nightly displacement of the rostral fluid to the thorax and neck. Therefore, the exercise acts by optimizing the circulatory system transport, activating the peripheral muscle pump and increasing venous return. In turn, fluid accumulation in the intravascular and interstitial spaces during the day is minimized which consequently reduces liquid displacement during sleep.5 Furthermore, another possible cause for decreasing the severity of the disease associated with exercise can be explained by increasing the upper airway (UA) muscle tonus, making them more resistant to collapse. Exercise promotes an increase in the duration of slow-wave sleep and therefore increased motor activity of the VAS musculature, mainly of the genioglossus muscle, giving greater strength and stability to this musculature, and thereby reducing airway collapsibility.23
In this context, IMT has been considered as a form of exercise to be used in treating OSA. There are only three studies that address the exclusive use of IMT in OSA.10–12 The first study evaluated the effectiveness of a six-week IMT protocol on the plasma catecholamine levels, OSA severity and sleep variables in patients with mild to moderate symptoms.10 Souza et al (2018)11 examined the effectiveness of 12 weeks of IMT on the functional capacity of exercise, sleep quality and excessive daytime sleepiness. Lin et al (2020)11 investigated the effect of 12 weeks of IMT training on OSA severity, daytime sleepiness, and pulmonary function and identified the IMT responsiveness predictors. Another paper examined the effects of 4 weeks of respiratory muscle training (inspiratory/expiratory) in AHI, sleep quality, excessive daytime sleepiness and quality of life in eutrophic patients with mild to moderate OSA.24
The present study showed that the IMT group presented a reduction in OSA severity and the Berlin questionnaire score. In our study, 8 weeks of IMT promoted a reduction (5.7%) in the AHI. Lin et al (2020)12 have also reported similar results, a decrease in the AHI (6%) in OSA patients after 12 weeks of IMT. Despite a statistically significant reduction in the AHI, these findings AHI could not be clinically relevant. The difference in the AHI could be explained by the night variability in the OSA severity. Sforza et al (2019)25 analyzed patients referred for probable OSA and related that AHI internight variability was associated to sleep structure, fragmentation, and position. However, the changes were larger in patients with mild OSA.
Other studies related no improvement in OSA severity after 6 weeks of IMT10 and after 4 weeks of respiratory muscle training (RMT).24 The results of these studies are conflicting and might be explained by the sample size and the different protocols used for the IMT. Although our study and two others are randomized clinical trials,10,12 they have a small sample size, ranging from 8 to 12 patients being exercised. Moreover, only few studies exist that assess the effect of IMT on OSA severity, with different durations of IMT intervention, resulting in heterogeneity among studies.10,11,24 Thus, more studies are needed to clarify the hypothesis.
In the analysis of sleep quality, the present study showed improvement in overall PSQI scores of the IMT group when compared with baseline values, thereby demonstrating good sleep quality (PSQI scores < 5). Similarly, a significant improvement in the overall score of the PSQI was observed after 12 weeks of IMT with moderate load (50–60% of MIP), specifically in the components related to sleep quality and sleep duration, indicating an overall change for good sleep quality.11
Our results corroborate the findings obtained in the study by Vranish & Bailey.10 These authors observed improvement in the PSQI scores after an intervention of 6 weeks of IMT (75% of MIP) when compared to the placebo group. Despite reductions not being found in AHI for any group, the individuals of the IMT group recorded a smaller number of awakenings.
The respiratory events are responsible for hypoxemia and hypercapnia during sleep. These gasometric changes stimulate the central and peripheral chemoreceptors, triggering the increased activity of the sympathetic nervous system and consequently causing sudden awakenings and arousals to restore ventilation. In another way, hypoxemia causes an increase in metabolic residues and with it afferent metabolic receptors (which are located between the skeletal muscle fibers) will be encouraged, promoting an increase in sympathetic stimulation to restore perfusion and the supply of O2 and remove the waste accumulated in ischemic tissues.26 This compensatory response results in sleep fragmentation and consequently a decrease in sleep quality.27
The sample in the present study was composed of subjects with sedentary behavior and who were insufficiently active. The fact that both groups have been subjected to a planned activity, even though it was insufficient to cause training in the case of the placebo group, generated an increase in the physical activity level which may have caused the improvement in the excessive daytime sleepiness (EDS) in both groups. A study conducted an epidemiological study in France and showed a negative correlation between the levels of physical activity and the presence of EDS.28
On the other hand, Souza et al11 and Herkenrath et al24 reported no changes in EDS after inspiratory and respiratory muscle training, respectively. Permanent EDS after respiratory muscle training should be due to a lower baroreflex sensitivity and greater activation of the sympathetic nervous system associated with sleep arousals.24
This study presents some limitations despite being a randomized clinical and controlled, double-blind trial and meeting the calculated sample size. However, the small sample size could be impacted by the internight variability of the AHI and overestimated the severity of OSA. Moreover, quantifying the apnea/hypopnea duration before and after the intervention could also help to clarify the effects of the IMT in the severity of OSA. Regarding the sleep quality measure which was performed using the PSQI and is validated and has a strong correlation with the gold standard method, it is a subjective measure and assesses the perception that the individual has on their quality of sleep. An analysis of the polysomnography parameters would complete the analysis of the PSQI with more reliable and objective data related to the sleep quality of these patients.
Conclusion
We conclude that the inspiratory muscle training with moderate load performed during 8 weeks promotes discrete changes in the AHI and improves the sleep quality and excessive daytime sleepiness in OSA. Thus, the IMT may be used by complementing other treatment forms for OSA, as a supportive therapy used associated with CPAP to provide a better compliance rate. Furthermore, IMT can be performed in a home environment, has a low cost and easy implementation, enabling its insertion in patient’s daily routines.
There are potential benefits of conducting follow-up studies with larger sample sizes to elucidate the effects of IMT in OSA severity. Furthermore, studies will be required to investigate parameters for IMT prescription such as duration, intensity, and frequency, thus providing an optimization of physiological adaptations afforded by IMT in OSA patients.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001.
Disclosure
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; or expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
References
1. Younes M. Pathogenesis of obstructive sleep apnea. Clin Chest Med. 2019;40:317–330. doi:10.1016/j.ccm.2019.02.008
2. van Zeller M, Severo M, Santos AC, Drummond M. 5-years APAP adherence in OSA patients–do first impressions matter? Respir Med. 2013;107(12):2046–2052. doi:10.1016/j.rmed.2013.10.011
3. Iftikhar IH, Bittencourt L, Youngstedt SD, et al. Comparative efficacy of CPAP, MADs, exercise-training, and dietary weight loss for sleep apnea: a network meta-analysis. Sleep Med. 2017;30:7–14. doi:10.1016/j.sleep.2016.06.001
4. de Felício CM, da Silva Dias FV, Trawitzki LVV. Obstructive sleep apnea: focus on myofunctional therapy. Nat Sci Sleep. 2018;10:271–286. doi:10.2147/NSS.S141132
5. Mendelson M, Bailly S, Marillier M, et al. Obstructive sleep apnea syndrome, objectively measured physical activity and exercise training interventions: a systematic review and meta-analysis. Front Neurol. 2018;9–73.
6. Aiello KD, Caughey WG, Nelluri B, Sharma A, Mookadam F, Mookadam M. Effect of exercise training on sleep apnea: a systematic review and meta-analysis. Respir Med. 2016;116:85–92. doi:10.1016/j.rmed.2016.05.015
7. Illi SK, Held U, Frank I, Spengler CM. Effect of respiratory muscle training on exercise performance in healthy individuals. Sports Med. 2012;42:707–724. doi:10.1007/BF03262290
8. Plentz RDM, Sbruzzi G, Ribeiro RA, Ferreira JB, Dal Lago P. Treinamento muscular inspiratório em pacientes com insuficiência cardíaca: metanálise de estudos randomizados. Arq Bras Cardiol. 2012;99:762–771. doi:10.1590/S0066-782X2012001100011
9. Hill K, Cecins NM, Eastwood PR, Jenkins SC. Inspiratory muscle training for patients with chronic obstructive pulmonary disease: a practical guide for clinicians. Arch Phys Med Rehab. 2010;91:1466–1470. doi:10.1016/j.apmr.2010.06.010
10. Vranish JR, Bailey EF. Inspiratory muscle training improves sleep and mitigates cardiovascular dysfunction in obstructive sleep apnea. Sleep. 2016;39:1179–1185. doi:10.5665/sleep.5826
11. Souza AKF, Dornelas de Andrade A, de Medeiros AIC, et al. Effectiveness of inspiratory muscle training on sleep and functional capacity to exercise in obstructive sleep apnea: a randomized controlled trial. Sleep Breath. 2018;22:631–639. doi:10.1007/s11325-017-1591-5
12. Lin HC, Chiang LL, Ong JH, Tsai KL, Hung CH, Lin CY. The effects of threshold inspiratory muscle training in patients with obstructive sleep apnea: a randomized experimental study. Sleep Breath. 2020;24(1):201–209. doi:10.1007/s11325-019-01862-y
13. Bertolazi AN, Fagondes SC, Hoff LS, et al. Validation of the Brazilian Portuguese version of the Pittsburgh Sleep Quality Index. Sleep Med. 2011;12:70–75. doi:10.1016/j.sleep.2010.04.020
14. Bertolazi AN, Fagondes SC, Hoff LS, Pedro VD, Menna Barreto SS, Johns MW. Validação da escala de sonolência de Epworth em português para uso no Brasil. J Bras Pneumol. 2009;35:877–883. doi:10.1590/S1806-37132009000900009
15. Matsudo S, Araújo T, Matsudo V, et al. Questionário Internacional De Atividade Física (IPAQ): estudo de validade e reprodutibilidade no Brasil. Rev Bras Ativ Fis Saúde. 2001;6:5–18.
16. Vaz AP, Drummond M, Mota PC, Severo M, Almeida J, Winck JC. Translation of Berlin Questionnaire to Portuguese language and its application in OSA identification in a sleep disordered breathing clinic. Rev Port Pneumol. 2011;17:59–65. doi:10.1016/S0873-2159(11)70015-0
17. American Thoracic Society/European Respiratory Society. ATS/ERS statement on respiratory muscle testing. Am J Resp Crit Care. 2002;166:518–624.
18. Neder JA, Andreoni S, Lerario MC, Nery LE. Reference values for lung function tests. II. Maximal respiratory pressures and voluntary ventilation. Braz J Med Biol Res. 1999;32:719–727. doi:10.1590/S0100-879X1999000600007
19. Pereira CA, Duarte AA, Gimenez A, Soares MR. Comparison between reference values for FVC, FEV1, and FEV1/FVC ratio in White adults in Brazil and those suggested by the Global Lung Function Initiative 2012. J Bras Pneumol. 2014;40:397–402. doi:10.1590/S1806-37132014000400007
20. Iftikhar IH, Kline CE, Youngstedt SD. Effects of exercise training on sleep apnea: a meta-analysis. Lung. 2014;192:175–184. doi:10.1007/s00408-013-9511-3
21. Awad KM, Malhotra A, Barnet JH, Quan SF, Peppard PE. Exercise is associated with a reduced incidence of sleep-disordered breathing. Am J Med. 2012;125:485–490. doi:10.1016/j.amjmed.2011.11.025
22. Peppard PE, Young T. Exercise and sleep-disordered breathing: an association independent of body habitus. Sleep. 2004;27:480–484. doi:10.1093/sleep/27.3.480
23. McSharry DG, Saboisky JP, Deyoung P, et al. A mechanism for upper airway stability during slow wave sleep. Sleep. 2013;36:555–563. doi:10.5665/sleep.2544
24. Herkenrath SD, Treml M, Priegnitz C, Galetke W, Randerath WJ. Effects of respiratory muscle training (RMT) in patients with mild to moderate obstructive sleep apnea (OSA). Sleep Breath. 2018;(2018)(22):323–328. doi:10.1007/s11325-017-1582-6
25. Sforza E, Roche F, Chapelle C, Pichot V. Internight variability of apnea-hypopnea index in obstructive sleep apnea using ambulatory polysomnography. Front Physiol. 2019;10:849. doi:10.3389/fphys.2019.00849
26. Belli JFC, Bacal F, Bocchi EA, Guimarães GV. Comportamento do ergorreflexo na insuficiência cardíaca. Arq Bras Cardiol. 2011;97:171–178. doi:10.1590/S0066-782X2011005000072
27. Cheng L, Ivanova O, Fan HH, Khoo MC. An integrative model of respiratory and cardiovascular control in sleep-disordered breathing. Respir Physiol Neurobiol. 2010;174:4–28. doi:10.1016/j.resp.2010.06.001
28. Andrianasolo RM, Menai M, Galan P, et al. Leisure-time physical activity and sedentary behavior and their cross-sectional associations with excessive daytime sleepiness in the french SU.VI.MAX-2 Study. Int J Behav Med. 2016;23:143–152. doi:10.1007/s12529-015-9501-3
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


