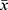Back to Journals » Drug Design, Development and Therapy » Volume 14
Inhibitory Effect of Curcumin on Artery Restenosis Following Carotid Endarterectomy and Its Associated Mechanism in vitro and in vivo
Authors Zhang D, Yang Y, Li Y, Zhang G, Cheng Z
Received 2 September 2019
Accepted for publication 10 February 2020
Published 26 February 2020 Volume 2020:14 Pages 855—866
DOI https://doi.org/10.2147/DDDT.S229607
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Yan Zhu
Dapeng Zhang,1 Yanhui Yang,2 Yuanchao Li,1 Guodong Zhang,1 Zhenguo Cheng1
1Neurosurgery Department, Xinxiang Central Hospital, Xinxiang, Henan Province 453000, People’s Republic of China; 2Color Ultrasonic Room, People’s Hospital of Huixian, Xinxiang, Henan Province 453600, People’s Republic of China
Correspondence: Zhenguo Cheng
Neurosurgery Department, Xinxiang Central Hospital, No. 56 Jinsui Street, Xinxiang, Henan Province 453000, People’s Republic of China
Tel +86 156 3735 9907
Email [email protected]
Objective: The present study aimed to assess the effect of curcumin (Cur) on carotid artery restenosis following carotid endarterectomy (CEA) and its associated mechanism in vivo and in vitro.
Methods: Ang II was used to induce excessive proliferation of rabbit aortic smooth muscle cells (CCC-SMC-1) in order to establish a hemadostenosis cell model. Similarly, the animal model of carotid artery restenosis was established by carotid artery gas drying injury combined with high-fat feed prior to CEA. CCC-SMC-1 cells and animals were treated by Cur and its effects on neointimal hyperplasia, inflammation and oxidative stress were detected and observed. The proteins that were associated with the Raf/MEK/ERK pathway were detected in cells and rabbit carotid artery tissues.
Results: Cur inhibited the proliferation of smooth muscle cells and neointimal formation and reduced the inflammation and oxidative stress indices. Concomitantly, Cur reduced the phosphorylation of the Raf/MEK/ERK pathway proteins.
Conclusion: Cur could inhibit carotid restenosis following CEA by inhibiting the activation of the Raf/MEK/ERK pathway.
Keywords: curcumin, carotid endarterectomy, restenosis, vascular smooth muscle cells, Raf/MEK/ERK
Introduction
Stroke is one of the major causes of adult mortality and disability worldwide.1 Carotid atherosclerosis combined with stenosis is one of the most common causes of ischemic stroke, which is accompanied by high morbidity, high mortality, disability and recurrence rate.2 At present, carotid endarterectomy (CEA) has become a safe and effective method to treat severe and symptomatic moderate carotid artery stenosis.3 However, carotid restenosis following CEA is one of the most important complications affecting the curative effect of surgery. Previous studies have shown that approximately 5–15% of patients with carotid artery stenosis experience restenosis following CEA.4,5 Although the detailed mechanisms are yet to be determined, it is generally believed that the pathological proliferation and inflammatory responses of vascular smooth muscle cells (VSMCs) can potentially contribute to atherosclerosis and arterial restenosis.6,7 Therefore, the inhibition of the proliferation of VSMCs and the reduction of the occurrence of restenosis following CEA has become a major research focus.
Curcumin (Cur) is a natural phenolic substance extracted from the rhizome of the plant Curcuma longa. Turmeric is commonly present in ordinary diets and its traditional use for the treatment of various diseases in China originates from several eons ago.8 Extensive investigations indicate that Cur has diverse pharmacological effects, such as antiinflammatory, antioxidant, antiproliferative and anticarcinogenic, whereas it also protects against the development of cerebrovascular diseases.9,10 Cur provides these beneficial effects due to its ability to regulate certain molecular targets, including adhesion molecules, growth factors, transcription factors, and factors involved in proliferation and apoptosis that are associated with several different cell types.11,12 Certain studies have shown that Cur can inhibit the proliferation of VSMCs and prevent stent restenosis following angioplasty.13,14 However, the anti-restenosis effect of Cur and its molecular mechanism following CEA are still unclear. The present study aimed to investigate the effect of Cur on the formation of vascular restenosis following CEA and its mechanism of action by specific in vitro and in vivo experiments.
Materials and Methods
Materials and Animals
Cur (purity over 98%) was purchased from Sigma (St Louis, MO, USA), whereas the rabbit aortic smooth muscle cell CCC-SMC-1 was purchased from ATCC (Manassas, VA, USA). 40 healthy male New Zealand rabbits aged 3 months, weighing 2.0–2.5 kg were purchased from the Guangdong Medical Laboratory Animal Center and were fed high-fat or normal diets.
Cell Grouping
CCC-SMC-1 cells were incubated in DMEA containing 10% fetal bovine serum (FBS) in a humidified atmosphere of 5% CO2 at 37°C. The cells that were from the 4th passage or from a higher passage number were used for the experiments. The cells were divided into the following groups: control group, Ang II group, Cur (10, 20 and 40 μmol/l) groups and GW5074 (Raf-1 inhibitor) group. The control group was incubated in DMEA containing 3% FBS, whereas the Ang II group was incubated in DMEA containing 107mol/l Ang II and 3% FBS. The Cur groups were incubated with different concentrations of Cur (10, 20 and 40 μmol/l) in DMEA containing 107mol/l Ang II and 3% FBS, whereas the GW5074 (inhibitor of Raf) group was incubated with 10 μM GW5074 in DMEA containing 107mol/l Ang II and 3% FBS.
MTT Assay-Detection of Cell Viability
The cells were seeded in 96-well plates (1×104 cells/well) and allowed to adapt overnight. Following an additional 24 h of serum starvation, the cells were incubated with different concentrations of Cur (0, 10, 20 and 40 μmol/l) in DMEA containing 3% FBS, or incubated according to the aforementioned experimental groups. A total of 20 μL MTT stock solution (5 mg/mL) was added to the wells at 48 h and incubated for 4 h at 37°C. A total of 200 μL DMSO was added to the medium to solubilize the crystals. The optical density (OD) value was measured using a microplate reader (Bio-Rad, USA) at a wavelength of 490 nm. The cell viability (%) was estimated as follows = (experimental group OD)/(control group OD) × 100%.
Cell Apoptosis and Cell Cycle Detection by Flow Cytometry
The cells were seeded in 96-well plates (1×106 cells/well) and allowed to grow overnight. Following an additional 24 h of serum starvation, the cells were incubated according to the above experimental groups. The cells were left to grow for another 48 h and were collected by centrifugation. The induction of cell apoptosis and the cell cycle distribution were measured by flow cytometry (FCM).
Determination of Cellular Inflammation and Oxidative Stress Indices
The contents of cellular monocyte chemotactic protein 1 (MCP-1) and interleukin 6 (IL-6) were determined by an ELISA kit (Beijing Solarbio science-technology, China). The content of malondialdehyde (MDA) and the activity of superoxide dismutase (SOD) in the cell culture medium were determined by a biochemical method. All operations were carried out in strict accordance with the operating instructions of the kit.
Animal Grouping and Treatment
A total of 40 New Zealand rabbits were fed in the room at a constant temperature of 22 to 25°C and divided into the control group, the model group (100 mg/kg Cur) and the Cur group (200 mg/kg Cur). A total of 10 animals were used in each group. The control group was fed with standard rabbit diet and the other groups were fed with a high-fat diet (2% cholesterol, 6% peanut oil and 92% standard rabbit diet). The model group and Cur groups had their carotid atherosclerotic stenosis surgically established by gas drying injury combined with high fat diet supplementation for 4 weeks. CEA was performed subsequently. The 100 mg/kg Cur and 200 mg/kg Cur groups were provided Cur at 100 or 200 mg/kg/d by intragastric administration following CEA, while the model group was administered normal saline of the same volume. All animals were allowed free access to water.
Animal Model of Carotid Atherosclerotic Stenosis
The model and Cur groups were fed with standard rabbit diet for one week and subsequently fed with a high-fat diet (2% cholesterol, 6% peanut oil and 92% standard rabbit diet) for 1 week. Subsequently, carotid artery gas drying injury was carried out as follows: The animals were restricted to water access for 6 h and 30 mg/kg pentobarbital sodium was injected along the ear margin vein for anesthesia. A vertical incision was made on the anterior-middle line of the carotid artery. The skin, superficial fascia, superficial cervical fascia and superficial organ fascia were successively incised. The right common carotid artery was dissociated horizontally along with the thyroid cartilage and subsequently the blood flow was blocked with an arterial clip at the two ends. The scalp was punctured with a needle parallel to the longitudinal axis of the blood vessel following rinsing with saline. The blood flow was then connected and remained continuous at a rate of 150 mL/min for 10 min in order to cause vascular intimal damage. The area was rinsed with saline and the clip was released to restore blood flow and to suture the wound. The animals were subcutaneously injected with heparin (150 U/kg/d) postoperatively for 7 days and intramuscularly by penicillin sodium (100,000 U/kg/d) for 3 days.
CEA
High-fat feeding continued for 4 weeks following modeling and subsequently the animals were administered CEA. The animals were restricted access to water for 6 h and 30 mg/kg pentobarbital sodium was injected along the ear margin vein for anesthesia. The tissue layer was reopened following the previous incision and the right common carotid artery was separated and blocked at the same place. The sheath of the artery was cut open to separate the carotid artery from the vagus nerve under the microscope. The wall of the diseased artery was visible and was subsequently dissociated and clipped with an arterial clip. The longitudinal incision was made and the boundary between the atherosclerotic plaque and the vascular medium was separated by fiber tweezers. Finally, the whole plaque was separated and stripped. The lumen of the surgical area was irrigated and the carotid artery incision was sutured. The artery clip was opened in order to restore the blood flow and suture the incision following hemostasis. Postoperative continuous intramuscular injection of penicillin sodium (100,000 U/kg/d) was performed for 3 days.
Determination of Blood Lipid, Inflammation, and Oxidative Stress Indices
A total of 2 mL of periauricular vein blood was collected 4 weeks following CEA and the serum was centrifuged at 3000 rpm for 15 min and frozen at −80°C. The levels of total cholesterol (TC), high-density lipoprotein (HDL), low-density lipoprotein (LDL) and triglycerides (TGs) were measured by an automatic biochemical analyzer (Beckman Coulter, USA). The contents of MCP-1 and IL-6 were determined by an ELISA kit, whereas the content of serum MDA was determined by the thiobarbituric acid method and the activity of serum SOD was measured by the xanthine oxidase method. All operations were carried out in strict accordance with the operating instructions of the kit.
Morphological Changes of the Carotid Artery by Hematoxylin and Eosin (H&E) Staining
The rabbits in each group were sacrificed by excessive anesthesia at 4 weeks following CEA. The tested carotid artery was fixed in 10% formaldehyde solution for 48 h. Following paraffin embedding, the carotid arteries were sectioned continuously by a slicer (4 μm). Subsequently, they were dewaxed, hydrated by gradient alcohol, stained with hematoxylin solution for 15 min, restained with eosin solution for 5 min, dehydrated by gradient alcohol and sealed in a transparent slide. Optical microscopy (Leica, DM3000, Germany) was used to observe the changes of the neointima. The Image-Pro Plus (Media Cybernetics, Silver Spring, MD, USA) was used to measure the area of the neointima and of the medium and to calculate the neointima-to-media area ratio.
Western Blot Analysis
The total protein derived from each group of cells and from the rabbit carotid artery tissues was extracted by a protein extraction kit (Beijing solarbio science﹠technology, China). The process was conducted according to the kit instructions. The BCA (Bi Yuntian, China) protein assay kit was used to determine the protein concentration. A total of 40 μg protein samples were separated by polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride (PVDF) membranes. The membranes were blocked at room temperature with skimmed milk powder for 2 h and subsequently incubated with p-Raf-1, p-MEK, p-ERK1/2, Raf-1, MEK and ERK1/2 antibodies (Abcam, UK) at 4°C overnight. Subsequently, the membranes were incubated with the corresponding secondary antibody IgG (1:2000) for 1 h at room temperature.
Statistical Analysis
Numerical data were presented as mean ± standard deviation (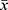 ±s). The differences among multiple groups were analyzed by one-way ANOVA followed by the Tukey Post Hoc tests for multiple comparisons. A P value lower than 0.05 (P<0.05) was considered for significant differences. All statistical analyses were performed using the SPSS19.0 software.
±s). The differences among multiple groups were analyzed by one-way ANOVA followed by the Tukey Post Hoc tests for multiple comparisons. A P value lower than 0.05 (P<0.05) was considered for significant differences. All statistical analyses were performed using the SPSS19.0 software.
Results
Effect of Cur on Cell Viability of CCC-SMC-1 Cells
The MTT results indicated no significant difference in the cell viability of CCC-SMC-1 cells treated with different concentrations of Cur, suggesting that 0–40 μmol/l Cur exhibited no significant effects on cell viability of VSMCs (P<0.05, Figure 1A). The cell viability of CCC-SMC-1 cells in the Ang II group was significantly higher than that of the control group (P<0.05, Figure 1B), suggesting that Ang II could promote the viability of these cells. The cell viability of the Cur groups was significantly reduced compared with that of the Ang II group (P<0.05, Figure 1B), indicating that Cur could inhibit the cell viability of CCC-SMC-1 cells in a similar way as GW5074.
Effect of Cur on Apoptosis and Cell Cycle of CCC-SMC-1 Cells
FCM was used to detect the induction of apoptosis and the cell cycle arrest of the cells treated with Cur. The results indicated no significant difference in the apoptotic rate between the control and the Ang II groups (P>0.05, Figure 2A and B). However, the apoptotic rate of the Cur group was increased gradually following the increase of the dose compared with that of the Ang II group (P<0.05, Figure 2A and B), suggesting that Cur could promote the induction of apoptosis in CCC-SMC-1 cells. The proportion of G0/G1 phase cells in the Ang II group was significantly lower than that of the control group, while the proportion of S and G2/M phase cells was significantly higher than that of the control group (P<0.05, Figure 2C), indicating that Ang II could affect the distribution of the cell cycle. The proportion of G0/G1 phase cells in the Cur group was significantly higher than that of the Ang II group, while the proportion of S and G2/M phase cells was significantly lower than that of the Ang II group (P<0.05, Figure 2C).
Effect of Cur on Inflammation and Oxidative Stress of CCC-SMC-1 Cells
The contents of MCP-1, IL-6, and MDA in the Ang II group were significantly higher than those of the control group, whereas the activity of SOD was significantly lower than that of the control group (P<0.05). The contents of MCP-1, IL-6, and MDA in the Cur group were significantly lower than those of the Ang II group, whereas the activity of SOD was significantly higher than that of the Ang II group. (P<0.05, Table 1). These effects were noted in a dose-dependent manner. The results suggested that Cur could inhibit the cellular inflammation and oxidative stress response induced by Ang II.
 |
Table 1 Effect of Cur on Cellular Inflammation and Oxidative Stress ( |
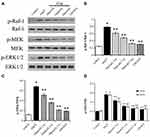
Effect of Cur on the Raf-MEK-ERK Signaling Pathway in vitro
Western blot analysis indicated that the expression levels of p-Raf-1, p-MEK, and p-ERK1/2 in the Ang II group were significantly higher than those of the control group (P<0.05), while the expression levels of p-Raf-1, p-MEK and p-ERK1/2 in the Cur group were significantly lower than those of the Ang II group (P<0.05, Figure 3), indicating that Cur could inhibit the activation of the Raf-MEK-ERK signaling pathway.
Effects of Cur on Neointimal Formation in Rabbits Following CEA
4 weeks after CEA, the carotid artery was taken for HE staining (Figure 4). Those results showed that the neointimal area and the neointima-to-media area ratio of the model group were more significantly larger than the control group, while that was decreased to various degrees in two Cur groups (P<0.05, Figure 5). The neointimal area and the neointima-to-media area ratio of in the Cur groups were significantly lower than the model group (P<0.05, Figure 5), suggesting that Cur intervention can effectively inhibit the carotid neointimal formation and vascular restenosis.
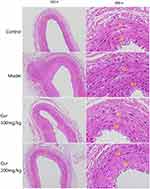 |
Figure 4 Observation of rabbit carotid vessel by HE staining (100×and 400×). Abbreviations: IN, intima; M, medium. |
Effect of Cur on Blood Lipid of Restenosis Rabbits
The levels of TC, LDL and TG in the model group were significantly higher than those of the control group, while the HDL levels were significantly lower than those of the control group (P<0.05). The levels of TC, LDL and TG in the Cur groups were significantly lower than those of the model group (P<0.05, Table 2), suggesting that Cur could regulate blood lipid levels in atherosclerosis.
 |
Table 2 Comparison of Cur on Serum Lipid Levels of Restenosis Rabbits After CEA (n=10, |
Effect of Cur on Inflammation and Oxidative Stress in Restenosis-Induced Rabbits
The serum levels of MCP-1, IL-6, and MDA in the model group were significantly higher, while the activity of SOD was significantly lower compared with those of the control group (P<0.05, Table 3). The serum levels of MCP-1, IL-6, and MDA in the Cur groups were significantly lower, while the activity of SOD was significantly higher (P<0.05) compared with those of the model group, suggesting that Cur could reduce inflammation and oxidative stress in carotid restenosis.
 |
Table 3 The Levels of MCP-1, MDA and SOD and IL-6 in Each Group (n=10, |
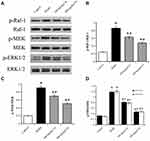
Effect of Cur on the Raf-MEK-ERK Signaling Pathway
Western blot results indicated that the expression levels of p-Raf-1, p-MEK and p-ERK1/2 in the model group were significantly higher than those of the control group, while the expression levels of p-Raf-1, p-MEK1/2, and p-ERK1/2 in the Cur groups were significantly lower than those of the model group (P<0.05, Figure 6). The data indicated that the Raf-MEK-ERK signaling pathway may be involved in the mechanism of restenosis following CEA and that the effect of Cur on restenosis may be associated with this signaling pathway.
Discussion
CEA has been recognized as an effective treatment for extracranial ischemic stroke. However, the problem of carotid restenosis following CEA remains a major challenge. Previous studies have shown that approximately 10% of patients develop restenosis at the intimal exfoliation site due to the re-formation of the atherosclerotic plaques in the blood vessels.15 The main pathological types of carotid restenosis following CEA include neointimal formation of the wound surface following exfoliation of the carotid intimal plaque. Atherosclerosis, thrombosis and lumen stenosis were caused by direct suture of the carotid artery incision.16–18 Cur is a plant polyphenol with a wide range of biological activities. Extensive investigations have shown that Cur exhibits antiproliferative, antimigratory, antioxidant and antiinflammatory effects. This compound has been used for the treatment of idiopathic inflammatory orbital pseudotumors, chronic anterior uveitis, as well as various types of cancer. However, the effect and mechanism of Cur on postoperative restenosis of CEA remain unclear.
Several studies have shown that the mechanism of carotid restenosis was closely associated with the excessive proliferation of VSMCs,19 which was accompanied with apoptosis reduction.20 The induction of cell apoptosis can reduce the number of VSMCs on the injured arterial wall and retain a dynamic balance on cell proliferation. Excessive proliferation and low apoptotic rate can thicken the blood vessel’s intima and cause hemadostenosis.21 In addition, the apoptotic endothelial cells can enhance platelet adhesion, promote cell inflammation and lipid infiltration and induce VSMC proliferation, migration, and thrombosis.22 A previous study has shown that Cur could inhibit intimal hyperplasia and VSMC proliferation following vascular injury.23 Moreover, it could induce VSMC apoptosis by regulating inflammatory-associated signaling pathways.23 Chen et al demonstrated that Cur could accelerate reendothelialization and ameliorate intimal hyperplasia of rat carotid artery tissues by promoting endothelial cell autophagy.24 In addition, a previous study has shown that inflammatory cells can participate in intimal restenosis by secreting inflammatory factors to stimulate the increase of VSMCs and their migration to the subintima.25 The proliferation of VSMCs and the carotid artery restenosis following vascular injury are associated with the increase in the levels of reactive oxygen species (ROS).26 ROS has been recognized as an important signaling pathway in the proliferation of VSMCs, which is closely associated with the pathophysiological processes of atherosclerosis and restenosis.27,28 The results of the present study indicated that Cur could inhibit cell viability, inflammatory and oxidative stress responses and induce apoptosis of CCC-SMC-1 cells treated with Ang II, which was consistent with previous studies. In addition, Jang HS et al demonstrated that Cur may be a safer and more effective method to prevent restenosis in a hypercholesterolemic rabbit Iliac artery stent model.13 Therefore, in the present study, rabbits were used to prepare the restenosis model following CEA and the results indicated that Cur could reduce the serum levels of MCP-1, IL-6, MDA, the neointimal area and the neointima-to-media area ratio of the carotid arteries following CEA, as well as enhance the activity of SOD in the restenosis rabbit model. The data suggested that Cur could effectively alleviate the intimal hyperplasia and prevent vascular restenosis following CEA by inhibiting vascular endothelial cell proliferation and alleviating inflammation and oxidative stress.
The Raf-MEK-ERK signaling pathway is one of the MAPK signaling pathways, which is closely associated with the physiological processes of cell growth, development, proliferation, and differentiation.29,30 The Raf-MEK-ERK signaling pathway plays an important role in the pathophysiological process of atherosclerosis stenosis. Zhang et al31 suggested that the downregulation of the Raf-MEK-ERK signaling pathway could inhibit the proliferation of endothelial progenitor cells. Pintus et al reported that activation of the PKC/Raf/MEK/ERK pathway could regulate blood lipid levels and increase the risk of atherosclerosis by increasing the expression of native LDL and E2F-1.32 The activation of ERK1/2 plays an important role in this pathway33,34 and inhibition of ERK1/2 activation can inhibit the phosphorylation of MAPK, thereby regulating cell proliferation and migration. In addition, the activation of ERK1/2 can increase the expression of the downstream nuclear transcription factor NF-κB and the cell cycle proteins, such as Cyclin D1, which leads to a change in cell proliferation.35,36 It has been shown that the application of ERK1/2 inhibitors or the induction of ERK1/2 gene knockout can inhibit the proliferation of VSMCs and the formation of neointima following injury.37 Certain reports have been performed regarding the inhibition of Cur on the Raf/MEK/ERK pathway.38 However, it is not clear whether Cur can inhibit carotid stenosis by interfering with the Raf-MEK-ERK signaling pathway. In the present study, the results indicated that Cur could inhibit the phosphorylation levels of Raf1, MEK and ERK1/2 in CCC-SMC-1 cells. The effect was noted following treatment of the cells with the Raf-1 inhibitor GW504. Moreover, the phosphorylation levels of Raf-1, MEK and ERK1/2 in the Cur groups were significantly lower than those in the model group of the rabbit carotid artery tissue. These results suggested that Cur may inhibit the excessive proliferation of VSMCs and the neointimal formation by inhibiting the activation of the Raf-MEK-ERK signaling pathway. It is worth noting that the change in the expression levels of the proliferation-associated transcription factors induced by ERK1/2 following its nuclear translocation is still unclear. Therefore, additional studies should be conducted in the future to examine these interactions.
In conclusion, Cur can inhibit the excessive proliferation of VSMCs in the neointima and alleviate the formation of intima following CEA. The mechanism of action may be associated with the inhibition of inflammation and oxidative stress, the induction of cell apoptosis and the inhibition of the activation of the Raf-MEK-ERK signaling pathway. The present study may provide a new experimental basis for expanding the understanding of the effect of Cur intervention on carotid restenosis.
Ethical Statement
All experiments procedures meet the ethical standards of the United Nations Convention on Biodiversity, the treatment and welfare of the laboratory animals followed the ARRIVE (Animal Research: Reporting of in vivo Experiments) guidelines and this study has been approved by the Ethics Committee of the Xinxiang Central Hospital.
Author Contributions
ZC made substantial contributions to conception and design of this study, DZ and YY contribute to the conduction of experiments and acquisition, analysis of and interpretation of the experimental data; YL drafted the article and GZ took part in revising and translation work. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
This study was support by the key scientific and technological research projects of Henan province (Grant No. 0623060800), China.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Wang W, Jiang B, Sun H, et al. Prevalence, incidence, and mortality of stroke in China: results from a Nationwide Population-Based Survey of 480 687 adults. Circulation. 2017;8:759–771. doi:10.1161/CIRCULATIONAHA.116.025250
2. Wang YJ, Zhang SM, Zhang L, et al. Chinese guidelines for the secondary prevention of ischemic stroke and transient ischemic attack 2010. CNS Neurosci Ther. 2012;2:93–101. doi:10.1111/j.1755-5949.2011.00290.x
3. Gabriel RA, Lemay A, Beutler SS, et al. Practice variations in anesthesia for carotid endarterectomies and associated outcomes. J Cardiothorac Vasc Anesth. 2016;1:23–29. doi:10.1053/j.jvca.2015.06.014
4. Yang YG, Chen Z, Kou L, et al. Analysis of risk factors for cerebrovascular complications after carotid endarterectomy. Zhonghua Yi Xue Za Zhi. 2019;21:1636–1640.
5. Okazaki T, Kanematsu Y, Shimada K, et al. A single-center retrospective study with 5- and 10-year follow-up of carotid endarterectomy with patch graft. Neurol Med Chir (Tokyo). 2019;6:231–237. doi:10.2176/nmc.oa.2018-0309
6. Jiang D, Yang Y, Li D. Lipopolysaccharide induced vascular smooth muscle cells proliferation: a new potential therapeutic target for proliferative vascular diseases. Cell Prolif. 2017;2.
7. Chung YL, Pan CH, Wang CC, et al. Methyl protodioscin, a steroidal saponin, inhibits neointima formation in vitro and in vivo. J Nat Prod. 2016;6:1635–1644. doi:10.1021/acs.jnatprod.6b00217
8. Tiwari SK, Agarwal S, Tripathi A, et al. Bisphenol-A mediated inhibition of hippocampal neurogenesis attenuated by curcumin via canonical Wnt pathway. Mol Neurobiol. 2016;5:3010–3029. doi:10.1007/s12035-015-9197-z
9. Zeng Z, Zhan L, Liao H, et al. Curcumin improves TNBS-induced colitis in rats by inhibiting IL-27 expression via the TLR4/NF-kappaB signaling pathway. Planta Med. 2013;2:102–109.
10. Akyuz S, Turan F, Gurbuzler L, et al. The anti-inflammatory and antioxidant effects of curcumin in middle ear infection. J Craniofacial Surg. 2016;5:e494–e497. doi:10.1097/SCS.0000000000002810
11. Zhang YP, Li YQ, Lv YT, et al. Effect of curcumin on the proliferation, apoptosis, migration, and invasion of human melanoma A375 cells. Genet Mol Res. 2015;14:1056–1067. doi:10.4238/2015.February.6.9
12. Liu ZC, Yang ZX, Zhou JS, et al. Curcumin regulates hepatoma cell proliferation and apoptosis through the Notch signaling pathway. Int J Clin Exp Med. 2014;7(3):714–718.
13. Jang HS, Nam HY, Kim JM, et al. Effects of curcumin for preventing restenosis in a hypercholesterolemic rabbit iliac artery stent model. Catheter Cardiovasc Interv. 2009;74:881–888. doi:10.1002/ccd.22047.
14. Li HY, Yang M, Li Z, et al. Curcumin inhibits angiotensin II-induced inflammation and proliferation of rat vascular smooth muscle cells by elevating PPAR-gamma activity and reducing oxidative stress. Int J Mol Med. 2017;5:1307–1316. doi:10.3892/ijmm.2017.2924
15. Kumar R, Batchelder A, Saratzis A, et al. Restenosis after carotid interventions and its relationship with recurrent ipsilateral stroke: a systematic review and meta-analysis. Eur J Vasc Endovasc Surg. 2017;6:766–775. doi:10.1016/j.ejvs.2017.02.016
16. Garzon-Muvdi T, Yang W, Rong X, et al. Restenosis after carotid endarterectomy: insight into risk factors and modification of postoperative management. World Neurosurg. 2016;89:159–167. doi:10.1016/j.wneu.2016.01.028
17. Tonolini M, Bareggi E, Salerno R. Endoscopic stenting of malignant, benign and iatrogenic colorectal disorders: a primer for radiologists. Insights Imaging. 2019;1:80. doi:10.1186/s13244-019-0763-1
18. Zhang X, Huang W, Yu H, et al. Study on the patency of the contralateral iliac vein after stenting across the iliocaval confluence with an experimental in vivo model. Vasc Endovascular Surg. 2019;53(8):644–648.
19. Yue Y, Ma K, Li Z, et al. Angiotensin II type 1 receptor-associated protein regulates carotid intimal hyperplasia through controlling apoptosis of vascular smooth muscle cells. Biochem Biophys Res Commun. 2018;2:2030–2037. doi:10.1016/j.bbrc.2017.12.059
20. Ashino T, Yamamoto M, Numazawa S. Nrf2/Keap1 system regulates vascular smooth muscle cell apoptosis for vascular homeostasis: role in neointimal formation after vascular injury. Sci Rep. 2016;26291.
21. Zhang HJ, Wang J, Liu HF, et al. Overexpression of mimecan in human aortic smooth muscle cells inhibits cell proliferation and enhances apoptosis and migration. Exp Ther Med. 2015;1:187–192. doi:10.3892/etm.2015.2444
22. Wang JS, Huang Y, Zhang S, et al. A protective role of paeoniflorin in fluctuant hyperglycemia-induced vascular endothelial injuries through antioxidative and anti-inflammatory effects and reduction of PKCbeta1. Oxid Med Cell Longev. 2019;5647219.
23. Meng Z, Yan C, Deng Q, et al. Curcumin inhibits LPS-induced inflammation in rat vascular smooth muscle cells in vitro via ROS-relative TLR4-MAPK/NF-kappaB pathways. Acta Pharmacol Sin. 2013;7:901–911. doi:10.1038/aps.2013.24
24. Chen D, Tao X, Wang Y, et al. Curcumin accelerates reendothelialization and ameliorates intimal hyperplasia in balloon-injured rat carotid artery via the upregulation of endothelial cell autophagy. Int J Mol Med. 2015;6:1563–1571. doi:10.3892/ijmm.2015.2365
25. Liang JJ, Xue W, Lou LZ, et al. Correlation of restenosis after rabbit carotid endarterectomy and inflammatory cytokines. Asian Pac J Trop Med. 2014;3:231–236. doi:10.1016/S1995-7645(14)60027-4
26. Zhang F, Ren X, Zhao M, et al. Angiotensin-(1-7) abrogates angiotensin II-induced proliferation, migration and inflammation in VSMCs through inactivation of ROS-mediated PI3K/Akt and MAPK/ERK signaling pathways. Sci Rep. 2016;3:34621.
27. Sun HJ, Zhao MX, Ren XS, et al. Salusin-beta promotes vascular smooth muscle cell migration and intimal hyperplasia after vascular injury via ROS/NFkappaB/MMP-9 pathway. Antioxid Redox Signal. 2016;18:1045–1057. doi:10.1089/ars.2015.6475
28. Malekmohammad K, Sewell R, Rafieian-Kopaei M. Antioxidants and atherosclerosis: mechanistic aspects. Biomolecules. 2019;8.
29. Jingshan Z, Qianyu G, Shaohong L, et al. Hydroxysafflor yellow A inhibits rat vascular smooth muscle cells proliferation possibly via blocking signal transduction of MEK-ERK1/2. Zhonghua Xin Xue Guan Bing Za Zhi. 2015;8:728–731.
30. Yang M, Fang J, Liu Q, et al. Role of ROS-TRPM7-ERK1/2 axis in high concentration glucose-mediated proliferation and phenotype switching of rat aortic vascular smooth muscle cells. Biochem Biophys Res Commun. 2017;3–4:526–533. doi:10.1016/j.bbrc.2017.10.122
31. Zhang X, Mao H, Chen JY, et al. Increased expression of microRNA-221 inhibits PAK1 in endothelial progenitor cells and impairs its function via c-Raf/MEK/ERK pathway. Biochem Biophys Res Commun. 2013;431:404–408. doi:10.1016/j.bbrc.2012.12.157
32. Pintus G, Tadolini B, Posadino AM, et al. PKC/Raf/MEK/ERK signaling pathway modulates native-LDL-induced E2F-1 gene expression and endothelial cell proliferation. Cardiovasc Res. 2003;4:934–944. doi:10.1016/S0008-6363(03)00526-1
33. Khalil A, Poelvoorde P, Fayyad-Kazan M, et al. Apoliporotein L3 interferes with endothelial tube formation via regulation of ERK1/2, FAK and Akt signaling pathway. Atherosclerosis. 2018;73–87.
34. Xu S, Zhao J, Liu J, et al. Fibrinopeptide A induces expression of C-reactive protein via the ROS-ERK1/2/P38-NF-kappaB signal pathway in vascular smooth muscle cells. Cell Physiol Biochem. 2018;1:266–278. doi:10.1159/000489805
35. Lopez DVO, Pablos R, Ruiz-Sanz MB, et al. Characterization of the NF-?B signaling triggered by doxorubicin. Free Radic Biol Med. 2014;75:S38. doi:10.1016/j.freeradbiomed.2014.10.784
36. Liu J, Jin P, Lin X, et al. Arsenite increases Cyclin D1 expression through coordinated regulation of the Ca(2+)/NFAT2 and NF-kappaB pathways via ERK/MAPK in a human uroepithelial cell line. Metallomics. 2018;3:486–495. doi:10.1039/C7MT00305F
37. Shi L, Ji Y, Liu D, et al. Sitagliptin attenuates high glucose-induced alterations in migration, proliferation, calcification and apoptosis of vascular smooth muscle cells through ERK1/2 signal pathway. Oncotarget. 2017;44:77168–77180.
38. Guo Y, Shan Q, Gong Y, et al. Curcumin induces apoptosis via simultaneously targeting AKT/mTOR and RAF/MEK/ERK survival signaling pathways in human leukemia THP-1 cells. Pharmazie. 2014;3:229–233.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.